Abstract
Heavy water (D2O) is a crucial strategic resource, but its low natural abundance (~0.015%) makes high-purity extraction challenging. The near-identical physicochemical properties, including comparable sizes and chemical characteristics, of H2O and D2O mean traditional separation methods are energy-intensive, underscoring the demand for more efficient alternatives. Porous materials such as metal–organic frameworks and covalent organic frameworks are promising for non-thermal separation. However, the realization of dynamic co-adsorption separation, wherein isotopologue mixtures are propelled through material columns under pressure, has not been achieved despite its desirability for practical applications. Here we report a porous molecularly woven fabric with a three-dimensional weaving network for successful dynamic separation of water isotopologues H2O and D2O. It is constructed through the mortise-and-tenon stacking of two-dimensional molecularly woven layers. Its woven channels provide transport pathways, while its adaptive woven and mortise-and-tenon nodes enhance adaptive adsorption, amplifying the subtle differences between these two isotopologues during the breakthrough process. Consequently, the porous woven network enables efficient dynamic separation of H2O and D2O at 298 K through its topological features.

Similar content being viewed by others
Main
Heavy water (D2O) is a crucial strategic resource widely used in both fundamental scientific research and industrial areas such as nuclear energy and medical applications1,2,3,4,5,6,7. Therefore, obtaining high-purity D2O from natural sources is of paramount importance. However, D2O occurs naturally at an extremely low concentration of approximately 0.015%. More critically, H2O and D2O share very similar physicochemical properties and identical kinetic sizes, making them one of the most challenging isotopologue pairs to separate8. Currently, traditional separation methods based on the subtle kinetic and thermodynamic differences between H2O and D2O, such as distillation, electrolysis and proton exchange, are energy-intensive and inefficient9. As a result, developing inexpensive and efficient methods for separating water isotopologues has been a long-standing goal in this field.
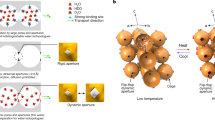
Using porous materials such as carbon materials9,10,11, metal–organic frameworks12 and covalent organic frameworks13,14 for non-thermal, additive-free separation is one of the most promising strategies to achieve this goal. However, designing effective porous materials for water isotopologue separation remains challenging, as these isotopologues possess nearly identical sizes and chemical properties. Consequently, conventional strategies—such as tuning pore size or introducing open adsorption sites—tend to be ineffective. In 2022, Gu and Kitagawa prepared metal–organic frameworks to preferentially adsorb H2O from mixtures of H2O, HDO and D2O, benefitting from the local flexibility of a diffusion-regulatory molecular gate to amplify the diffusion differences of water isotopologues as they hop through the narrow windows8. Currently, progress has been achieved in the static co-adsorption separation of water isotopologues (Fig. 1a), wherein adsorbent materials are immersed directly into water isotopologue mixtures to facilitate enrichment. However, dynamic co-adsorption separation (Fig. 1b), a process involving the flow of water isotopologue mixtures through packed columns under applied pressure15,16, remains a substantial challenge and has not yet been realized. Dynamic co-adsorption-based separation of water isotopologues is highly desirable for practical applications, as highlighted in several recent studies15,16.
a,b, Schematic diagram of static (a) and dynamic (b) co-adsorption separations of water isotopologues8,15,16. c, Top view and side view of one 2D woven layer in the crystal structure of PWPN-1. d, The 3D stacking structure of PWPN-1. Inset: octahedral cavity formed by woven nodes and mortise-and-tenon joints. e, The view along the c-axis direction of the crystal structure showing the woven channels. Here, H atoms and solvent molecules have been omitted for clarity.
To address this challenge, inspired by macroscopic woven materials used in separation processes, where the woven channels can flexibly adapt to the size of the species for efficient separation17,18,19,20, we constructed a porous molecularly woven polymer (PWPN-1) and demonstrated dynamic co-adsorption separation of water isotopologues H2O and D2O at room temperature (RT) (Fig. 1b). This polymer is formed by stacking two-dimensional (2D) woven layers through a mortise-and-tenon arrangement to create a three-dimensional (3D) porous structure. In the structure of PWPN-1, the woven topology not only imparts porosity to the material but also facilitates relative movements of its polymer chains through the cooperative interactions of the woven and mortise-and-tenon nodes, conferring unique adaptive properties to PWPN-1. Therefore, when a mixture of water isotopologues passes through this molecularly woven fabric, the presence of woven pores facilitates the rapid transport of water molecules within the network. Meanwhile, the woven and mortise-and-tenon adaptive nodes spanning the woven channels provide flexibility to the polymeric network, enabling the network structure to adaptively change according to the slight differences (such as hydrogen bonding strength) between H2O and D2O. This highlights the differences in weak interactions of these adaptive nodes with H2O and D2O, thereby regulating the diffusion differences of water isotopologues. Consequently, these subtle diffusion differences accumulate and amplify as H2O and D2O pass through the porous 3D network, ultimately realizing the successful dynamic co-adsorptive separation of H2O and D2O.
Synthesis and structural analysis of PWPN-1
PWPN-1 was synthesized under mild reaction conditions, achieving a 95% yield on a 100-g scale (Supplementary Fig. 1). Specifically, (methanetetrayltetra-4,1-phenylene)-tetrakisboronic acid (TPBA) was mixed with catechol (CA) and 1,2-bis(4-pyridyl)-ethylene (BPE) in benzene. Then, the mixture was heated at 50 °C for 5 h, followed by slow cooling to RT, to give PWPN-1 as yellow crystals21,22,23 (Supplementary Figs. 2–7). To gain a deeper understanding of the woven topological structure in PWPN-1, single-crystal X-ray diffraction was used, which revealed PWPN-1 crystallized in the tetragonal P-421c space group with lattice constants of a = 14.5812(3) Å, b = 14.5812(3) Å and c = 34.9063(6) Å (Supplementary Fig. 8 and Supplementary Table 1).
Through structural analyses, it was discerned that PWPN-1 exhibits a distinct lamellar architecture. Specifically, facilitated by benzene molecules, each tetraborate ester unit is connected to four BPE moieties through dative B−N bonds to form a 2D woven layer consisting of two entangled networks (red and blue) (Supplementary Figs. 9−12). The woven nodes are created by the interlacing of BPE units from each of the two networks. Interestingly, when viewed along the c-axis, regular woven pores are observed, reminiscent of the patterned adaptive pores formed in macroscopic weaving. From the side view along the a-axis, the CA units are distributed on both sides of each 2D woven layer (Fig. 1c). The thickness of a 2D woven layer is measured to be 19.04 Å, with intralayer channels visible. Notably, each 2D woven layer is formed by two networks entangled through a woven topology. As a result, these two networks can undergo local movements along the vertical or horizontal directions, providing these 2D woven layers with substantial structural adaptability.
Subsequently, these 2D woven layers stack into a 3D channel structure through a mortise-and-tenon interlocking pattern (Supplementary Fig. 13 and Supplementary Video 1). The tenons and mortises are formed by the CA units and the woven pores within the layers, respectively. The CA units insert into the woven pores at the woven nodes of the upper and lower woven layers to complete the 3D stacking. It is noteworthy that the incorporation of the CA units does not obstruct the woven pores, and instead they form continuous channels through the 3D stacking. Simultaneously, the insertion of the CA units from the upper and lower layers at the woven nodes of the middle layer creates unique octahedral cavities (Fig. 1d, top right) within the middle layer. These octahedral cavities, functioning as side chambers to the main channels of the woven pores, are interconnected to ensure the continuity of the entire 3D network of channels (Fig. 1e). The synergistic effect of the woven and mortise-and-tenon topologies imparts adaptability to the pore channels of the polymer network. Meanwhile, the abundant oxygen atoms from the CA units within the network pores can form rich hydrogen bonds with water molecules, suggesting that PWPN-1 holds great potential for the separation of water isotopologues.
Adsorption kinetics and breakthrough experiments
To further evaluate the performance of PWPN-1 in water isotopologue separation, it was subjected to activation treatment to remove benzene molecules (yellow and green parts in Fig. 2a) within the octahedral cavities and woven channels of the network. The single-crystal X-ray diffraction results of the activated PWPN-1, namely PWPN-1a, revealed that the overall network structure remained intact, with all benzene molecules completely removed from the woven network (Fig. 2a and Supplementary Fig. 14). Notably, compared with the crystal structure of PWPN-1, the structure of PWPN-1a exhibits no substantial dimensional changes along the a-axes and b-axes, but a marked contraction along the c-axis, from 34.9063 Å to 17.2663 Å (Supplementary Fig. 15 and Supplementary Table 2). This is due to the removal of benzene molecules from the octahedral cavities (at the intersection of the weaving and mortise-and-tenon nodes), which caused reduction of the distances between the woven nodes. As a result, the two networks in each 2D woven layer moved closer, leading to layer compression (Supplementary Fig. 15) and further inducing adaptability changes at the mortise-and-tenon nodes. This suggests that the woven topology can impart adaptive stability to the entire network. When subjected to external stimuli, the polymer structure can undergo adaptive movements while maintaining the overall stability of the network24,25,26.
a, Activation process for PWPN-1 crystals. b, CO2 adsorption isotherm of PWPN-1a at 195 K. c, H2O and D2O vapour sorption curves at 298 K for PWPN-1a at P/Ps from 0 to 0.9. The exposure time for each point is 600 s. d, Time-dependent H2O and D2O adsorption curves at 298 K for PWPN-1a at P/Ps = 0.9. e, Determination of diffusion rates of H2O and D2O in PWPN-1a at 298 K. Here Sqrt time represents the square root of time. The adsorption pressure was set to P/Ps = 0.95. f,g, Experimental breakthrough curves of PWPN-1a for D2O/H2O mixture separation at 298 K and 1 bar: 80/20 (f) and 90/10 (g) volume ratio in the liquid (v/v). h, Comparison of breakthrough times for D2O/H2O mixtures at volume ratios of 80/20 and 90/10 at 298 K. i, Five breakthrough curves of the D2O/H2O mixture at a volume ratio of 90/10 at 298 K. j, Comparison of D2O vapour uptake of PWPN-1a over ten cycles at 298 K for a D2O/H2O mixture (90/10, v/v). k, Powder X-ray diffraction patterns of PWPN-1a before and after ten breakthrough cycling experiments.
The porosity of PWPN-1a was investigated by CO2 gas adsorption at 195 K. The specific surface area was calculated to be 235 m2 g−1 using the Brunauer–Emmett–Teller model, indicating the formation of a woven network exhibiting porosity (Fig. 2b). Subsequently, the sorption isotherm curve of H2O/D2O vapour at 298 K was measured in the range of 0−2.5 kPa. As shown in Fig. 2c, under different pressures, the equilibrium adsorption capacity of the material to D2O is higher than that of H2O. At 2.5 kPa, the adsorption capacities of D2O and H2O are 84.07 and 69.12 ml g−1, respectively. The above results show the feasible equilibrium adsorption separation potential of PWPN-1a for H2O and D2O at 298 K. Then, time-dependent adsorption studies of H2O/D2O on PWPN-1a were further conducted at 283 K, 298 K and 323 K under P/Pₛ = 0.9, to evaluate the differences in their adsorption dynamics (Fig. 2d and Extended Data Fig. 1). Under these three measurement conditions, D2O and H2O reached adsorption equilibrium in PWPN-1a within approximately 80 min. Notably, after 85 min, the dynamic adsorption capacity for D2O reached 329.94 mg g−1 at 298 K, which was higher than the dynamic adsorption capacity of 271.11 mg g−1 observed for H2O. Upon increasing the temperature to 323 K, no substantial difference was observed in the dynamic adsorption capacity or adsorption rate between D2O and H2O. The micropore diffusion of H2O/D2O in PWPN-1a was fitted by the micropore model (see ‘Time-dependent adsorption profiles based on the gravimetric method’ in Supplementary Information). These results show that the adsorption rate of H2O is 4.4 × 10−6 s−1 while that of D2O is 5.2 × 10−6 s−1 at P/Ps = 0.95, and the kinetic selectivity is calculated as 1.2, confirming the material’s potential for dynamic separation of H2O and D2O (Fig. 2e).
To further evaluate the dynamic separation feasibility of PWPN-1a for D2O and H2O, dynamic breakthrough experiments were conducted on a small-scale laboratory bed that mimics the industrial process27 (Supplementary Fig. 16 and Supplementary Video 2). Initial single-component breakthrough experiments were performed under controlled conditions (298 K, 1 bar, 5.0 ml min−1 N2 carrier gas) to eliminate potential hydrogen–deuterium exchange effects. The results revealed distinct retention time of 223 min g−1 for H2O and 686 min g−1 for D2O, clearly demonstrating PWPN-1a’s excellent discrimination capability between water isotopologues (Extended Data Fig. 2). To better simulate practical separation scenarios, we then investigated an 80/20 (v/v) D2O/H2O mixture, where HDO formation is inevitable due to hydrogen–deuterium exchange equilibrium8,28,29,30. Under identical experimental conditions (298 K, 1 bar, 5.0 ml min−1), the mass-spectrometry-corrected breakthrough profiles (Fig. 2f) showed substantial differences in retention times: 280 min g−1 for H2O, 285 min g−1 for HDO and 410 min g−1 for D2O, confirming PWPN-1a’s effectiveness in separating all three water isotopologues. When the D2O concentration was increased to 90/10 (v/v), the separation efficiency was further enhanced, with retention times of 415 min g−1 (H2O), 454 min g−1 (HDO) and 597 min g−1 (D2O) (Fig. 2g,h and Supplementary Figs. 17−20), demonstrating the potential for progressive enrichment of D2O purity through breakthrough separation. Moreover, breakthrough cycling experiments were conducted to evaluate repeatability (Fig. 2i and Extended Data Fig. 3). The separation performance of D2O/H2O was well maintained after ten adsorption–desorption cycles for a D2O/H2O mixture (90/10, v/v). Notably, the breakthrough performance of PWPN-1a is invariable over ten consecutive adsorption–desorption cycles (Fig. 2j). PWPN-1a also retained good crystallinity after ten breakthrough cycling experiments (Fig. 2k). These experimental results demonstrate that PWPN-1a has the capability to achieve dynamic separation of H2O and D2O under RT conditions with good recyclability.
Separation mechanism exploration
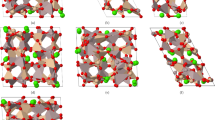
To gain deeper insight into the influence of the woven topological network on the dynamic separation of D2O/H2O mixtures, single crystals H2O@PWPN-1 and D2O@PWPN-1, formed by the adsorptions of H2O and D2O into PWPN-1a at RT, respectively, were analysed (Supplementary Tables 3 and 4 and Supplementary Figs. 21 and 23). By comparing the crystal structures of PWPN-1a, H2O@PWPN-1 and D2O@PWPN-1, it was found that in H2O@PWPN-1, water molecules preferentially anchor at sites within the octahedral cavities, whereas in D2O@PWPN-1, D2O molecules have anchoring sites in both the octahedral cavities and the woven channels (Fig. 3a–c and Supplementary Figs. 24–31). Notably, the woven channels undergo clear expansion upon absorbing H2O, as indicated by the quadrilateral shape formed by the four oxygen atoms near the inner side of a woven channel, with its edge length increasing from 5.15 Å to 5.31 Å (Fig. 3d,e). When PWPN-1a was immersed in D2O, the woven channels underwent an adaptive expansion to facilitate the adsorption of D2O molecules, with the edge length of the quadrilateral increasing from 5.15 Å to 5.60 Å (Fig. 3f). This suggests that the woven network absorbs water molecules through adaptive motion, and its self-adjusting expansion enables the selective distinction between H2O and D2O, primarily due to differences in kinetic size and weak interactions within the channels, allowing D2O to anchor while H2O remains unanchored.
a–c, Crystal structures: after activation (PWPN-1a) (a); after H2O adsorption (H2O@PWPN-1) (b); after D2O adsorption (D2O@PWPN-1) (c). d–f, Woven channels in crystal structures: PWPN-1a (d); H2O@PWPN-1 (e); D2O@PWPN-1 (f). g–i, Octahedral cavities in crystal structures: PWPN-1a (g); H2O@PWPN-1 (h); D2O@PWPN-1 (i).
Moreover, the octahedral cavity, formed by woven and mortise-and-tenon nodes, is rich in oxygen atoms. When H2O and D2O molecules enter the cavity, they form weak interactions, predominantly hydrogen bonds, with the oxygen atoms on its walls. As the cavity is constructed from these woven and mortise-tenon nodes, it undergoes expansion deformation, allowing it to capture H2O and D2O molecules more efficiently. Therefore, after adsorbing H2O molecules, the octahedral cavity expands, with its height, the distance between the woven nodes, rising from 5.95 Å in PWPN-1a to 6.31 Å (Fig. 3g,h). After adsorbing D2O molecules, the octahedron further expands, and its height increases to 6.36 Å (Fig. 3i). In summary, the adsorption process of H2O and D2O in the adaptive woven network primarily relies on the structural self-adaptation mechanism, rather than the fixed channel sizes and weak interactions commonly understood. This characteristic is particularly evident in the dynamic separation process: as an H2O/D2O mixture passes through the adaptive sites of the woven network, the network structure spontaneously undergoes adaptive adjustments, preferentially matching with D2O molecules. This unique adaptive response mechanism considerably amplifies the differences (such as hydrogen bonding strength) between H2O and D2O during the transport process, enabling efficient dynamic separation of the mixture.
Computational simulation
To further investigate the dynamic separation mechanism, we computed the binding energies of both D2O and H2O at the two adsorption sites of PWPN-1a. Simulations were performed using the CP2K package31 with the Perdew–Burke–Ernzerhof exchange-correlation functional32 and DFT-D3(BJ) dispersion corrections33. Because single-point energy calculations cannot capture the energy differences caused by the nuclear masses of H and D atoms, zero-point energy was included to account for nuclear quantum effects (NQEs). The binding energies for D2O and H2O were found to be −26.60 kJ mol−1 and −24.82 kJ mol−1, respectively, at Site-1 (within the woven channels; Fig. 4a, dashed red circle), and −18.64 kJ mol−1 and −17.32 kJ mol−1, respectively, at Site-2 (within the octahedral cavity; Fig. 4a, dashed blue circle). D2O exhibited stronger binding at both adsorption sites (Fig. 4a), consistent with our breakthrough results, which demonstrated the material’s energetic preference for D2O adsorption (Supplementary Figs. 32–35 and Supplementary Table 5).
a, Binding energy difference, including zero-point energy, between D2O and H2O at Site-1 (within the woven channels, dashed red circle) and Site-2 (within the octahedral cavity, dashed blue circle). Inset: simplified model illustrating the locations of Site-1 (dashed red circle) and Site-2 (dashed blue circle). b, Free energy surface for H2O in PWPN-1a at 298.15 K, with blue dots (Path-1) and red dots (Path-2) representing the possible transport pathways for water isotopologues. c,d, Free energy profiles for the diffusion of a quantum D2O (red) or H2O (black) molecule along Path-1 (c) or Path-2 (d). e–h, Simulated diffusion paths of water isotopologues in the PWPN-1a structure: Path-1 view along the c-axis (e) and b-axis (f), respectively; Path-2 view along the c-axis (g) and b-axis (h), respectively.
To investigate the dynamic behaviour of D2O and H2O in PWPN-1a, we examined the free energy surface of the water isotopologues using the well-tempered metadynamics method34 over a 10-ns simulation at 298.15 K (Fig. 4b and Supplementary Figs. 36–38). Our analysis revealed that water migration from Site-1 may proceed via two possible minimal-energy pathways: (Path-1) transport to an adjacent Site-1 in the next layer through the same one-dimensional channel or (Path-2) transport across Site-2 to another Site-1 located in an adjacent layer channel in the next layer (Fig. 4b). Details of the metadynamics setup, including the definition of collective variables (CVs), are provided in Supplementary Fig. 37.
Building on the above calculations, we further combined path integral molecular dynamics (PIMD) simulations with umbrella sampling35 to evaluate the diffusion behaviour of H2O and D2O, considering NQEs along the two aforementioned paths. For both paths, the initial positions of H2O and D2O were placed at the centre of each restraint window. PIMD simulations further predicted that the free energy barriers for D2O migration out of Site-1 along both paths were approximately 0.35 kJ mol−1 higher than those for H2O (first peak, Fig. 4c,d). This effect may be attributed to NQEs, which enhance the binding free energy between D2O and Site-1, where four inward-facing oxygen atoms create a strong binding environment. PIMD simulations also revealed that both D2O and H2O preferentially remained at Site-2, the local energy minimum, along both paths, with CV1 values ranging from 0.5 to 0.7. This observation provides a refinement to the previously described Path-1 trajectory obtained from the well-tempered metadynamics method. In addition, the free energy barriers for D2O passing Path-1 and Path-2 were approximately 1.17 kJ mol−1 and 0.76 kJ mol−1 higher than those for H2O, respectively (Fig. 4c,d, second peak). The detailed pathways of Path-1 and Path-2 are shown in Fig. 4e–h. Collectively, these factors contribute to the slower diffusion rate of D2O in PWPN-1a compared with H2O.
Conclusion
In this work, drawing inspiration from macroscopic woven materials used in separation processes, we have designed and synthesized a porous molecularly woven material driven by dative B−N bonds. Leveraging the unique woven channels and abundant adaptive nodes of molecularly woven fabrics, dynamic separation of water isotopologues H2O and D2O has been successfully achieved under mild conditions. This dynamic separation process is well aligned with industrial application requirements, offering an alternative solution to the long-standing technical challenge of water isotopologue separation. This work successfully extends the unique separation properties of woven topologies from the macroscopic to the molecular scale, achieving precise separation of high-value-added chemicals. This transitions molecularly woven materials from structural exploration to practical applications. Particularly in the field of separations, it opens pathways for the efficient separation of chemicals, especially isotopic compounds. The realization of this precise separation relies on the unique structural characteristics and functional advantages of molecularly woven materials.
Methods
Synthesis of PWPN-1
TPBA (14.87 mg, 0.03 mmol), CA (13.21 mg, 0.12 mmol) and BPE (10.93 mg, 0.06 mmol) were added to benzene (15.0 ml) and mixed using ultrasound for 15 min. The mixture was heated at 50 °C for 5 h (Supplementary Scheme 1). The resulting solution was cooled slowly to RT. Yellow single crystals of PWPN-1 were obtained.
Breakthrough experiments and separation factor analysis
Approximately 0.25 g of PWPN-1a was packed into a stainless-steel adsorption column (inner diameter 4.5 mm, length 15 cm). The unfilled portion of the column was packed with asbestos. Fixed-bed breakthrough experiments were conducted using a Breakthrough Analyzer BTA (Micromeritics Instrument). Before the experiment, Ar gas was injected into the breakthrough system pipeline to ensure thorough cleaning. Simultaneously, the sample column was heated at 100 °C for 4 h to remove residual water from the PWPN-1a powder. After cooling the column to RT (298 K), a N2/H2O/D2O mixture was injected at 1 bar and a constant flow rate of 5 ml min−1. The relative humidity of the flow was 92%, corresponding to a ratio of the partial pressure of the H2O/D2O mixture vapour to the saturation pressure of H2O/D2O at the same temperature and pressure (298 K, 1 bar) of approximately 0.92 (Supplementary Fig. 17). The composition of the gas exiting in the adsorption column was monitored using a CirrusTM 3 mass spectrometer (MKS), while the column temperature was maintained at 298 K. A breakthrough experiment was considered complete when the composition of the outgoing gas stabilized. Subsequently, the N2/H2O/D2O mixture flow was stopped, and dry N2 was introduced into the column at a flow rate of 10 ml min−1 for 1 h to purge and desorb adsorbed substances. Following this, the column was rapidly heated to 100 °C (heating rate 15–20 °C min−1) to ensure complete desorption of all adsorbed substances.
Ideal adsorption solution theory calculations of adsorption selectivity
For the binary adsorption of H2O (component 1) and D2O (component 2) mixture vapour, the adsorption selectivity is defined as
where y1 and x1 denote the molar fractions of component 1 in the gas phase and in the adsorbed phase, respectively, and y2 and x2 denote the molar fractions of component 2 in the gas phase and in the adsorbed phase, respectively. The binary adsorption selectivity of PWPN-1a for the H2O/D2O mixture vapour is based on the single-component adsorption isotherm results of H2O/D2O vapour at RT, fitted using the ideal adsorption solution theory Dual-Site Langmuir model.
Data availability
Data supporting the findings of this investigation are available in the article and its Supplementary Information. Crystallographic data for the structures reported have been deposited at the Cambridge Crystallographic Data Centre under deposition numbers CCDC 2376031 (PWPN-1), 2423397 (PWPN-1a), 2423352 (H2O@PWPN-1) and 2423355 (D2O@PWPN-1). Copies of the data can be obtained free of charge at https://www.ccdc.cam.ac.uk/structures/. Source data are provided with this paper.
References
Hillebrand, L., Liang, X. J., Serafim, R. A. M. & Gehringer, M. Emerging and re-emerging warheads for targeted covalent inhibitors: an update. J. Med. Chem. 67, 7668–7758 (2024).
Skalicky, J. J., Sukumaran, D. K., Mills, J. L. & Szyperski, T. Toward structural biology in supercooled water. J. Am. Chem. Soc. 122, 3230–3231 (2000).
Masson, G. R. et al. Recommendations for performing, interpreting and reporting hydrogen deuterium exchange mass spectrometry (HDX-MS) experiments. Nat. Methods 16, 595–602 (2019).
Taylor, R. D., MacCoss, M. & Lawson, A. D. Combining molecular scaffolds from FDA approved drugs: application to drug discovery. J. Med. Chem. 60, 1638–1647 (2017).
Sun, W. et al. Intramolecular arene C(sp2)–H amidation enabled by ferrocenium-mediated decomposition of 1,4,2-dioxazol-5-ones as amidyl radical precursors. ACS Catal. 13, 11389–11398 (2023).
Liu, M. et al. Barely porous organic cages for hydrogen isotope separation. Science 366, 613–620 (2019).
Senga, R. et al. Nanoscale C−H/C−D mapping of organic materials using electron spectroscopy. Nat. Nanotechnol. 20, 740–746 (2025).
Su, Y. et al. Separating water isotopologues using diffusion-regulatory porous materials. Nature 611, 289–294 (2022).
Liang, J. et al. Macroscopic heterostructure membrane of graphene oxide/porous graphene/graphene oxide for selective separation of deuterium water from natural water. Adv. Mater. 34, 2206524 (2022).
Mohammadi, A., Daymond, M. R. & Docoslis, A. Graphene oxide membranes for isotopic water mixture filtration: preparation, physicochemical characterization, and performance assessment. ACS Appl. Mater. Interfaces 12, 34736–34745 (2020).
Ching, K. et al. Liquid-phase water isotope separation using graphene-oxide membranes. Carbon 186, 344–354 (2022).
Paschke, B. et al. Dynamic studies on kinetic H2/D2 quantum sieving in a narrow pore metal–organic framework grown on a sensor chip. Chem. Eur. J. 25, 10766 (2019).
Oh, H., Park, K. S., Kalidindi, S. B., Fischer, R. A. & Hirscher, M. Quantum cryo-sieving for hydrogen isotope separation in microporous frameworks: an experimental study on the correlation between effective quantum sieving and pore size. J. Mater. Chem. A 1, 3244–3248 (2013).
Kim, J. Y., Oh, H. & Moon, H. R. Hydrogen isotope separation in confined nanospaces: carbons, zeolites, metal–organic frameworks, and covalent organic frameworks. Adv. Mater. 31, 1805293 (2019).
Heine, T. & Snurr, R. Q. A molecular flip-flop for separating heavy water. Nature 611, 243–244 (2022).
Shevlin, S. Water discrimination by differing diffusions. Nat. Mater. 22, 406 (2023).
Tarleton, S. & Wakeman, R. Solid/Liquid Separation: Equipment Selection and Process Design (Elsevier, 2006).
Werber, J. R., Osuji, C. O. & Elimelech, M. Materials for next-generation desalination and water purification membranes. Nat. Rev. Mater. 1, 16018 (2016).
Yu, H., Wu, M., Duan, G. & Gong, X. One-step fabrication of eco-friendly superhydrophobic fabrics for high-efficiency oil/water separation and oil spill cleanup. Nanoscale 14, 1296–1309 (2022).
Zhang, H. et al. A crystalline and stable microporous framework based on the dative B←N bonds. Chem 9, 242–252 (2023).
Liu, C.-H. et al. A B←N framework based on 1D dative B←N polymers for exclusive recognition and separation of benzene from its azeotrope. Chem 9, 3532–3543 (2023).
Yuan, F. et al. Synergistic assembly of single-crystal 2D porphyrin-based organic polymers via dative B−N bonds and halogen bonds. Angew. Chem. Int. Ed. 64, e202501875 (2025).
Chen, Y. et al. Transforming membrane distillation to a membraneless fabric distillation for desalination. Nat. Water 2, 52–61 (2024).
Xiao, D. et al. Single crystals of purely organic free-standing two-dimensional woven polymer networks. Nat. Chem. 16, 1906–1914 (2024).
Liu, Y. et al. Weaving of organic threads into a crystalline covalent organic framework. Science 351, 365–369 (2016).
Lewandowska, U. et al. A triaxial supramolecular weave. Nat. Chem. 9, 1068–1072 (2017).
Peng, Y.-L. et al. Efficient propyne/propadiene separation by microporous crystalline physiadsorbents. Nat. Commun. 12, 5768 (2021).
Bigeleisen, J., Lee, M. W. & Mandel, F. Equilibrium isotope effects. Annu. Rev. Phys. Chem. 24, 407–440 (1973).
Hamuro, Y. Tutorial: chemistry of hydrogen/deuterium exchange mass spectrometry. J. Am. Soc. Mass. Spectrom. 32, 133–151 (2021).
Stofella, M. et al. Computational tools for hydrogen–deuterium exchange mass spectrometry data analysis. Chem. Rev. 124, 12242–12263 (2024).
Kühne, T. D. et al. CP2K: An electronic structure and molecular dynamics software package—Quickstep: efficient and accurate electronic structure calculations. J. Chem. Phys. 152, 194103 (2020).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
Barducci, A., Bussi, G. & Parrinello, M. Well-tempered metadynamics: a smoothly converging and tunable free-energy method. Phys. Rev. Lett. 100, 020603 (2008).
Torrie, G. M. & Valleau, J. P. Nonphysical sampling distributions in Monte Carlo free-energy estimation: Umbrella sampling. J. Comput. Phys. 23, 187–199 (1977).
Acknowledgements
We thank the Chemistry Instrumentation Center of Zhejiang University for technical support. This work was supported by the National Key Research and Development Program of China (grant no. 2021YFA0910100 to F.H.), the National Natural Science Foundation of China (grant nos. 22035006 to F.H., 22350007 to F.H., 22205200 to G.L., 22475188 to G.L., 22320102001 to F.H., 22371252 to M.L. and 22405238 to X.Y.), the Zhejiang Provincial Natural Science Foundation of China (grant no. LZ24B040001 to G.L.), the Starry Night Science Fund of Zhejiang University Shanghai Institute for Advanced Study (grant no. SN-ZJU-SIAS-006 to F.H.), the Leading Innovation Team grant from the Department of Science and Technology of Zhejiang Province (grant no. 2022R01005 to M.L.), the ‘Pioneer’ R&D Program of Zhejiang Province (grant no. 2023C01087 to F.H.), the China Postdoctoral Science Foundation (grant no. 2024M762897 to D.X.) and the Postdoctoral Science Preferential Funding of Zhejiang Province (grant no. ZJ2024080 to D.X.).
Author information
Authors and Affiliations
Contributions
F.H., G.L. and M.L. formulated and supervised the project. D.X. and X.Y. performed the synthesis and characterization of the crystals. D.X., D.H. and X.Y. performed the breakthrough experiments. D.X., D.H., X.Y., Z.G., L.C., T.S., R.N., Y.P. and X.M. analysed the data. S.Y. performed the simulation calculations. D.X., D.H., S.Y., X.Y., M.L., G.L. and F.H. wrote the paper. All authors contributed to the data analysis, discussion and paper revision.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Volodymyr Bon, Yue-Biao Zhang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alexandra Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Time-dependent sorption curves of H2O and D2O.
a–f D2O and H2O adsorption curves at 283 K (a, b), 298 K (c, d) and 323 K (e, f) for PWPN-1a. The black line represents the humidity of D2O or H2O containing N2.
Extended Data Fig. 2 Single-component breakthrough experiments.
Experimental breakthrough curves of PWPN-1a for D2O and H2O at 298 K and 1 bar.
Extended Data Fig. 3 Cyclic breakthrough experiments.
Ten breakthrough curves of the D2O/H2O mixture at a volume ratio of 90/10 at 298 K.
Supplementary information
Supplementary Information (download PDF )
Supplementary Scheme 1, Figs. 1–38, discussion and Tables 1–5.
Supplementary Video 1 (download MP4 )
Animation of the assembly of PWPN-1.
Supplementary Video 2 (download MP4 )
Animation of the dynamic separation process.
Supplementary Data 1
Crystallographic data for PWPN-1 (CCDC 2376031).
Supplementary Data 2
Crystallographic data for PWPN-1a (CCDC 2423397).
Supplementary Data 3
Crystallographic data for D2O@PWPN-1 (CCDC 2423355).
Supplementary Data 4
Crystallographic data for H2O@PWPN-1 (CCDC 2423352).
Supplementary Data 5 (download ZIP )
Source data for supplementary figures.
Source data
Source Data Fig. 2 (download XLSX )
Source data for Fig. 2.
Source Data Fig. 4 (download XLSX )
Source data for Fig. 4.
Source Data Extended Data Fig. 1 (download XLSX )
Source data for Extended Data Fig. 1.
Source Data Extended Data Fig. 2 (download XLSX )
Source data for Extended Data Fig. 2.
Source Data Extended Data Fig. 3 (download XLSX )
Source data for Extended Data Fig. 3.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xiao, D., Hu, D., Yang, S. et al. A porous molecularly woven fabric for dynamic separation of water isotopologues. Nat. Synth 5, 180–188 (2026). https://doi.org/10.1038/s44160-025-00913-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-025-00913-5