Abstract
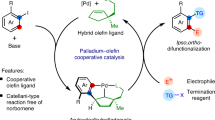
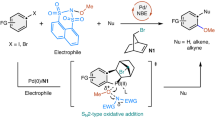
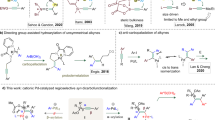
Direct access to multifunctionalized arenes through regioselective vicinal difunctionalization of aryl substrates is a challenging yet highly sought-after process. Palladium–norbornene catalysis enables the synthesis of ipso,ortho-difunctionalized products from haloarenes; however, selective mono-ortho-alkylative vicinal difunctionalization reactions of para-substituted haloarene substrates have remained elusive. Here we report the use of a methyl-modified thio-cycloolefin ligand for the palladium-catalysed ortho-alkylative vicinal difunctionalization of para-substituted iodoarenes. This catalytic system demonstrates compatibility with a variety of para-substituted iodoarenes, alkyl iodides and termination reagents, facilitating ortho-alkylative vicinal difunctionalization unattainable by conventional palladium–norbornene catalysis. In addition, the catalytic system can be used for the selective mono-ortho-arylation of para-substituted iodoarenes, enabling the synthesis of triphenylenes. Mechanistic studies reveal the origins of site selectivity within the catalytic process, through isolation of key intermediates and examination of their stoichiometric reactivity. This work highlights the versatility of palladium–olefin catalysis in addressing the complexities associated with constructing multifunctionalized aromatic frameworks through rational molecular design.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the article and its Supplementary Information. Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers CCDC 2393060 (Int-8•PPh3), 2393061 (Int-5•PPh3), 2393062 (Int-6•PPh3), 2393063 (Int-3•PPh3), 2393064 (Int-4•PPh3), 2409485 (Int-7•PPh3), 2409488 (Int-5) and 2409562 (Int-3). These data can be obtained free of charge from the CCDC (http://www.ccdc.cam.ac.uk/data_request/cif).
References
Nilova, A., Campeau, L. C., Sherer, E. C. & Stuart, D. R. Analysis of benzenoid substitution patterns in small molecule active pharmaceutical ingredients. J. Med. Chem. 63, 13389–13396 (2020).
Dupommier, D. & Besset, T. New trends for transition metal-catalyzed ortho/ipso difunctionalizations of arenes. Chem 10, 2651–2665 (2024).
Catellani, M., Motti, E. & Della Ca, N. Catalytic sequential reactions involving palladacycle-directed aryl coupling steps. Acc. Chem. Res. 41, 1512–1522 (2008).
Martins, A., Mariampillai, B. & Lautens, M. Synthesis in the key of Catellani: norbornene-mediated ortho C–H functionalization. Top. Curr. Chem. 292, 1–33 (2010).
Ye, J. & Lautens, M. Palladium-catalyzed norbornene-mediated C–H functionalization of arenes. Nat. Chem. 7, 863–870 (2015).
Wang, J. & Dong, G. Palladium/norbornene cooperative catalysis. Chem. Rev. 119, 7478–7528 (2019).
Catellani, M., Frignani, F. & Rangoni, A. A complex catalytic cycle leading to a regioselective synthesis of o,o′-disubstituted vinylarenes. Angew. Chem., Int. Ed. Engl. 36, 119–122 (1997).
Lautens, M. & Piguel, S. A New route to fused aromatic compounds by using a palladium-catalyzed alkylation-alkenylation sequence. Angew. Chem. Int. Ed. 39, 1045–1046 (2000).
Bressy, C., Alberico, D. & Lautens, M. A route to annulated indoles via a palladium-catalyzed tandem alkylation/direct arylation reaction. J. Am. Chem. Soc. 127, 13148–13149 (2005).
Candito, D. A. & Lautens, M. Palladium-catalyzed domino direct arylation/N-arylation: convenient synthesis of phenanthridines. Angew. Chem. Int. Ed. 48, 6713–6716 (2009).
Dong, Z. & Dong, G.-B. Ortho vs ipso: site-selective Pd and norbornene-catalyzed arene C–H amination using aryl halides. J. Am. Chem. Soc. 135, 18350–18353 (2013).
Huang, Y., Zhu, R., Zhao, K. & Gu, Z. Palladium-catalyzed Catellani ortho-acylation reaction: an efficient and regiospecific synthesis of diaryl ketones. Angew. Chem. Int. Ed. 54, 12669–12672 (2015).
Sun, F.-G. et al. Cleavage of the C(O)−S bond of thioesters by palladium/norbornene/copper cooperative catalysis: an efficient synthesis of 2-(arylthio)aryl ketones. J. Am. Chem. Soc. 138, 7456–7459 (2016).
Liu, C. et al. Synthesis of indolines via a palladium/norbornene-catalyzed reaction of aziridines with aryl iodides. Chem. Commun. 54, 3407–3410 (2018).
Li, R. & Dong, G. Direct annulation between aryl iodides and epoxides through palladium/norbornene cooperative catalysis. Angew. Chem. Int. Ed. 57, 1697–1701 (2018).
Cheng, H.-G. et al. Epoxides as alkylating reagents for the catellani reaction. Angew. Chem. Int. Ed. 57, 3444–3448 (2018).
Lv, W., Chen, Y., Wen, S., Ba, D. & Cheng, G. Modular and stereoselective synthesis of C-aryl glycosides via Catellani reaction. J. Am. Chem. Soc. 142, 14864–14870 (2020).
Liu, X., Fu, Y., Chen, Z., Liu, P. & Dong, G. Ortho-C–H methoxylation of aryl halides enabled by a polarity-reversed N–O reagent. Nat. Chem. 15, 1391–1399 (2023).
Bai, L. et al. Palladium/norbornene-catalyzed C–H alkylation/alkyne insertion/indole dearomatization domino reaction: assembly of spiroindolenine-containing pentacyclic frameworks. Angew. Chem. Int. Ed. 57, 5151–5155 (2018).
Wang, J. & Gu, Z. Synthesis of 2-(1-Alkoxyvinyl)anilines by Palladium/norbornene-catalyzed amination followed by termination with vinyl ethers. Adv. Synth. Catal. 358, 2990–2995 (2016).
Botla, V. et al. Closing the cycle as it begins: synthesis of ortho-iodobiaryls via Catellani reaction. Angew. Chem. Int. Ed. 62, e202218928 (2023).
Jiang, W.-T., Xu, M.-Y., Yang, S., Xie, X.-Y. & Xiao, B. Alkylation-terminated Catellani reactions using alkyl carbagermatranes. Angew. Chem. Int. Ed. 59, 20450–20454 (2020).
Ye, J.-X. et al. Triple cross-electrophile coupling enabled by palladium/norbornene cooperative catalysis. Sci. Adv. 11, eadu4573 (2025).
Dong, Z., Lu, G., Wang, J. C., Liu, P. & Dong, G. Modular ipso/ortho difunctionalization of aryl bromides via palladium/norbornene cooperative catalysis. J. Am. Chem. Soc. 140, 8551–8562 (2018).
Ye, R., Liu, X. & Dong, G. Enabling aryl chloride-mediated palladium/norbornene cooperative catalysis. Angew. Chem. Int. Ed. 64, e202500897 (2025).
Wang, J.-S. et al. ortho C–H alkylation of aryl chlorides by a Catellani strategy. Angew. Chem. Int. Ed. 64, e202509300 (2025).
Han, M.-L. et al. Palladium/norbornene-catalyzed decarbonylative difunctionalization of thioesters. JACS Au 1, 1877–1884 (2021).
Li, H. et al. Transformations of aryl ketones via ligand-promoted C–C bond activation. Angew. Chem. Int. Ed. 59, 14388–14393 (2020).
Tao, K. L. et al. Multisite modifications of arenes using ketones as removable handles enabled by Pd and norbornene cooperative catalysis. Nat. Synth. 4, 209–218 (2025).
Xie, Y.-H., Zang, L. & Ritter, T. Late-stage cross-electrophile coupling of arylthianthrenium salts with (hetero)aryl (pseudo)halides via palladium catalysis. Angew. Chem. Int. Ed. 64, e202502441 (2025).
Wang, J. et al. Modular and regioselective synthesis of all-carbon tetrasubstituted olefins enabled by an alkenyl Catellani reaction. Nat. Chem. 11, 1106–1112 (2019).
Jiao, L. & Bach, T. Palladium-catalyzed direct 2-alkylation of indoles by norbornene-mediated regioselective cascade C–H activation. J. Am. Chem. Soc. 133, 12990–12993 (2011).
Jiao, L., Herdtweck, E. & Bach, T. Pd(II)-catalyzed regioselective 2-alkylation of indoles via a norbornene-mediated C–H activation: mechanism and applications. J. Am. Chem. Soc. 134, 14563–14572 (2012).
Wang, X.-C. et al. Ligand-enabled meta-C–H activation using a transient mediator. Nature 519, 334–338 (2015).
Shi, G., Shao, C., Ma, X., Gu, Y. & Zhang, Y. Pd(II)-catalyzed Catellani-type domino reaction utilizing arylboronic acids as substrates. ACS Catal. 8, 3775–3779 (2018).
Chen, S. et al. The discovery of a palladium(II)-initiated borono-Catellani reaction. Angew. Chem. Int. Ed. 57, 7161–7165 (2018).
Li, R.-H., Liu, F.-P. & Dong, G.-B. Redox-neutral ortho functionalization of aryl boroxines via palladium/norbornene cooperative catalysis. Chem 5, 929–939 (2019).
Li, R. & Dong, G. Structurally modified norbornenes: a key factor to modulate reaction selectivity in the palladium/norbornene cooperative catalysis. J. Am. Chem. Soc. 142, 17859–17875 (2020).
Shen, P.-X., Wang, X.-C., Wang, P., Zhu, R.-Y. & Yu, J.-Q. Ligand-enabled meta-C–H alkylation and arylation using a modified norbornene. J. Am. Chem. Soc. 137, 11574–11577 (2015).
Dong, Z., Wang, J., Ren, Z. & Dong, G. Ortho C–H acylation of aryl iodides by palladium/norbornene catalysis. Angew. Chem. Int. Ed. 54, 12664–12668 (2015).
Shi, H., Herron, A. N., Shao, Y., Shao, Q. & Yu, J.-Q. Enantioselective remote meta-C–H arylation and alkylation via a chiral transient mediator. Nature 558, 581–585 (2018).
Wang, J., Li, R., Dong, Z., Liu, P. & Dong, G. Complementary site-selectivity in arene functionalization enabled by overcoming the ortho constraint in palladium/norbornene catalysis. Nat. Chem. 10, 866–872 (2018).
Chen, S.-Q. et al. Construction of axial chirality via palladium/chiral norbornene cooperative catalysis. Nat. Catal. 3, 727–733 (2020).
Wang, J., Zhou, Y., Xu, X., Liu, P. & Dong, G. Entry to 1,2,3,4-tetrasubstituted arenes through addressing the “meta constraint” in the palladium/norbornene catalysis. J. Am. Chem. Soc. 142, 3050–3059 (2020).
Liu, X., Wang, J. & Dong, G. Modular entry to functionalized tetrahydrobenzo(β)azepines via the palladium/norbornene cooperative catalysis enabled by a C7-modified norbornene. J. Am. Chem. Soc. 143, 9991–10004 (2021).
Sukowski, V., van Borselen, M., Mathew, S. & Fernández-Ibáñez, M. A. S. O-ligand promoted meta-C–H arylation of anisole derivatives via palladium/norbornene catalysis. Angew. Chem. Int. Ed. 61, e202201750 (2022).
Zheng, Y.-X. & Jiao, L. Hybrid cycloolefin ligands for palladium–olefin cooperative catalysis. Nat. Synth. 1, 180–187 (2022).
Wang, F.-Y., Li, Y.-X. & Jiao, L. Functionalized cycloolefin ligand as a solution to ortho-constraint in the Catellani-type reaction. J. Am. Chem. Soc. 145, 4871–4881 (2023).
Wang, X.-X. & Jiao, L. Dual ligand enabled Pd-catalyzed ortho-alkylation of iodoarenes. J. Am. Chem. Soc. 146, 25552–25561 (2024).
Qureshi, Z., Schlundt, W. & Lautens, M. Introduction of hindered electrophiles via C–H functionalization in a palladium-catalyzed multicomponent domino reaction. Synthesis 47, 2446–2456 (2015).
Gao, Q. et al. Catalytic synthesis of atropisomeric o-terphenyls with 1,2-diaxes via axial-to-axial diastereoinduction. J. Am. Chem. Soc. 143, 7253–7260 (2021).
Stambuli, J. P., Bühl, M. & Hartwig, J. F. Synthesis, characterization, and reactivity of monomeric, arylpalladium halide complexes with a hindered phosphine as the only dative ligand. J. Am. Chem. Soc. 124, 9346–9347 (2002).
Sukowski, V., van Borselen, M., Mathew, S., de Bruin, B. & Fernández-Ibáñez, M. A. Meta-C−H arylation of aniline derivatives via palladium/S,O-ligand/norbornene cooperative catalysis. Angew. Chem. Int. Ed. 63, e202317741 (2024).
Shin, S. S. et al. Preparation of sulfonylaminophenylalkylphenylacrylamide derivatives for use as vanilloid receptor antagonists. WO2009096701 A2 (World Intellectual Property Organization, 2009).
Mehnaz, S. et al. Lahorenoic acids A–C, ortho-dialkyl-substituted aromatic acids from the biocontrol strain Pseudomonas aurantiaca PB-St2. J. Nat. Prod. 76, 135–141 (2013).
Mariampillai, B., Alliot, J., Li, M. & Lautens, M. A convergent synthesis of polysubstituted aromatic nitriles via palladium-catalyzed C–H functionalization. J. Am. Chem. Soc. 129, 15372–15379 (2007).
Liu, Z.-S. et al. Palladium/norbornene cooperative catalysis to access tetrahydronaphthalenes and indanes with a quaternary center. ACS Catal 8, 4783–4788 (2018).
Pozo, I., Guitián, E., Pérez, D. & Peña, D. Synthesis of nanographenes, starphenes, and sterically congested polyarenes by aryne cyclotrimerization. Acc. Chem. Res. 52, 2472–2481 (2019).
Yang, Y. et al. Palladium-catalyzed synthesis of triphenylenes via sequential C–H activation and decarboxylation. Org. Lett. 20, 5402–5405 (2018).
Li, S. et al. Palladium-catalyzed annulation reaction of bay-diiodinated arenes with o-chloroaromatic carboxylic acids to access polycyclic aromatic compounds. J. Org. Chem. 89, 15665–15677 (2024).
Acknowledgements
We are grateful to the National Natural Science Foundation of China (grant no. 21933007 to L.J.) and the Tsinghua Xuetang Program for financial support. The technology platform of CBMS is acknowledged for providing instrumentation.
Author information
Authors and Affiliations
Contributions
Y.-X.L. and C.-H.Z. performed ligand synthesis and developed the reaction methods. Y.-X.L. performed mechanistic studies. Y.-X.L. and Y.M. explored the substrate scope. Y.-X.L. and L.J. wrote the paper. L.J. directed the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Huixiong Dai, Qianghui Zhou and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Thomas West, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Experimental details, Supplementary Sections 1–4 and Figs. 1–4.
Supplementary Data 1
Crystallographic data for complex Int-8•PPh3, CCDC 2393060.
Supplementary Data 2
Crystallographic data for complex Int-5•PPh3, CCDC 2393061.
Supplementary Data 3
Crystallographic data for complex Int-6•PPh3, CCDC 2393062.
Supplementary Data 4
Crystallographic data for complex Int-3•PPh3, CCDC 2393063.
Supplementary Data 5
Crystallographic data for complex Int-4•PPh3, CCDC 2393064.
Supplementary Data 6
Crystallographic data for complex Int-7•PPh3, CCDC 2409485.
Supplementary Data 7
Crystallographic data for complex Int-5, CCDC 2409488.
Supplementary Data 8
Crystallographic data for complex Int-3, CCDC 2409562.
Source data
Source Data Fig. 3 (download XLSX )
Source data for the kinetic plots in Fig. 3.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, YX., Zong, CH., Meng, Y. et al. Complementary site selectivity in ortho-alkylative vicinal difunctionalization reactions of iodoarenes enabled by palladium–olefin catalysis. Nat. Synth 5, 240–250 (2026). https://doi.org/10.1038/s44160-025-00920-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-025-00920-6