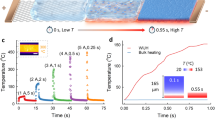
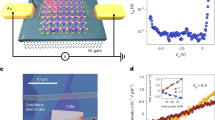
Abstract
Rapid Joule heating (RJH) has emerged as a transformative technique for ultrafast materials synthesis, attributed to extreme thermal conditions, fast reaction speed and high energy efficiency. Here, to improve the controllability and versatility of RJH, we introduce nanocatalysts and establish a catalytic RJH process that combines the precision of vapour–liquid–solid (VLS) growth processes with the ultrafast kinetics of Joule heating, enabling structurally controlled synthesis of one-dimensional (1D) nanomaterials within seconds. We verify the validity of VLS mechanism at temperatures beyond 2,500 °C under RJH, in which the far-from-equilibrium reaction conditions further enhance the growth and diameter control of 1D nanomaterials. Consequently, nanowires of refractory carbides, II–VI/III–V group semiconductors, high-entropy carbides, and multiwalled and single-walled carbon nanotubes are synthesized, showing the generality of the RJH-VLS strategy. With demonstrated scalability to the 10-g scale, low energy consumption on the order of tens of kilojoules per gram, and the ability to precisely control morphology through nanocatalysts, this catalytic RJH strategy shows great promise for the synthesis and production of 1D materials.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data supporting the conclusions of this study are available in the article and its Supplementary Information. Source data are provided with this paper.
References
Yao, Y. et al. Carbothermal shock synthesis of high-entropy-alloy nanoparticles. Science 359, 1489–1494 (2018).
Luong, D. X. et al. Gram-scale bottom-up flash graphene synthesis. Nature 577, 647–651 (2020).
Hu, X. et al. Ultrafast materials synthesis and manufacturing techniques for emerging energy and environmental applications. Chem. Soc. Rev. 52, 1103–1128 (2023).
Han, Y. C., Cao, P. Y. & Tian, Z. Q. Controllable synthesis of solid catalysts by high-temperature pulse. Acc. Mater. Res. 4, 648–654 (2023).
Xie, H. et al. A stable atmospheric-pressure plasma for extreme-temperature synthesis. Nature 623, 964–971 (2023).
Chen, W. et al. Millisecond conversion of metastable 2D materials by flash Joule heating. ACS Nano 15, 1282–1290 (2021).
Shi, W. et al. Transient and general synthesis of high-density and ultrasmall nanoparticles on two-dimensional porous carbon via coordinated carbothermal shock. Nat. Commun. 14, 2294 (2023).
Deng, B. et al. Kinetically controlled synthesis of metallic glass nanoparticles with expanded composition space. Adv. Mater. 36, 2309956 (2024).
Algozeeb, W. A. et al. Flash graphene from plastic waste. ACS Nano 14, 15595–15604 (2020).
Zhu, S. et al. Flash nitrogen-doped graphene for high-rate supercapacitors. ACS Mater. Lett. 4, 1863–1871 (2022).
Eddy, L. et al. Electric field effects in flash Joule heating synthesis. J. Am. Chem. Soc. 146, 16010–16019 (2024).
Liu, S. et al. Extreme environmental thermal shock induced dislocation-rich Pt nanoparticles boosting hydrogen evolution reaction. Adv. Mater. 34, 2106973 (2022).
Liu, S. et al. Dislocation-strained IrNi alloy nanoparticles driven by thermal shock for the hydrogen evolution reaction. Adv. Mater. 32, e2006034 (2020).
Wu, H. et al. Rapid Joule-heating synthesis for manufacturing high-entropy oxides as efficient electrocatalysts. Nano Lett. 22, 6492–6500 (2022).
Yao, Y. et al. Extreme mixing in nanoscale transition metal alloys. Matter 4, 2340–2353 (2021).
Yao, Y. et al. High-entropy nanoparticles: synthesis–structure–property relationships and data-driven discovery. Science 376, eabn3103 (2022).
Choi, C. H. W. et al. Flash-within-flash synthesis of gram-scale solid-state materials. Nat. Chem. 16, 1831–1837 (2024).
Wang, C. W. et al. A general method to synthesize and sinter bulk ceramics in seconds. Science 368, 521–526 (2020).
Deng, B. et al. Phase controlled synthesis of transition metal carbide nanocrystals by ultrafast flash Joule heating. Nat. Commun. 13, 262 (2022).
Han, Y. C. et al. A general method for rapid synthesis of refractory carbides by low-pressure carbothermal shock reduction. Proc. Nat Acad. Sci. USA 119, e2121848119 (2022).
Cheng, Y. et al. Flash upcycling of waste glass fibre-reinforced plastics to silicon carbide. Nat. Sustain. 7, 452–462 (2024).
Chen, C. et al. Catalyst-free in situ carbon nanotube growth in confined space via high temperature gradient. Research 2018, 1793784 (2018).
Wyss, K. M. et al. Upcycling of waste plastic into hybrid carbon nanomaterials. Adv. Mater. 35, 2209621 (2023).
Chen, J. H. et al. Dimension engineering of boron nitride nanostructures through catalytic flash Joule heating. ACS Nano 19, 24904–24911 (2025).
Wang, M. et al. Single-crystal, large-area, fold-free monolayer graphene. Nature 596, 519–524 (2021).
Ma, K. Y. et al. Epitaxial single-crystal hexagonal boron nitride multilayers on Ni (111). Nature 606, 88–93 (2022).
Duan, X. & Lieber, C. M. General synthesis of compound semiconductor nanowires. Adv. Mater. 12, 298–302 (2000).
Seeger, T., Kohler-Redlich, P. & Rühle, M. Synthesis of nanometer-sized sic whiskers in the arc-discharge. Adv. Mater. 12, 279–282 (2000).
Yang, F. et al. Growth modes of single-walled carbon nanotubes on catalysts. Sci. Adv. 8, eabq0794 (2022).
Wu, Y. & Yang, P. Direct observation of vapor−liquid−solid nanowire growth. J. Am. Chem. Soc. 123, 3165–3166 (2001).
Wagner, R. S. & Ellis, W. C. Vapor–liquid–solid mechanism of single crystal growth. Appl. Phys. Lett. 4, 89–90 (1964).
Xia, Y. et al. One-dimensional nanostructures: synthesis, characterization, and applications. Adv. Mater. 15, 353–389 (2003).
Yang, F. et al. Chirality pure carbon nanotubes: growth, sorting, and characterization. Chem. Rev. 120, 2693–2758 (2020).
Wu, L. et al. Vertical graphene on rice-husk-derived SiC/C composite for highly selective photocatalytic CO2 reduction into CO. Carbon 207, 36–48 (2023).
Dubois, G. et al. Molecular network reinforcement of sol–gel glasses. Adv. Mater. 19, 3989–3994 (2007).
Ahn, J. et al. Rapid Joule heating synthesis of oxide-socketed high-entropy alloy nanoparticles as CO2 conversion catalysts. ACS Nano, 12188–12199 (2023).
Dasgupta, N. P. et al. 25th anniversary article: semiconductor nanowires—synthesis, characterization, and applications. Adv. Mater. 26, 2137–2184 (2014).
Su, L. et al. Ultralight, recoverable, and high-temperature-resistant sic nanowire aerogel. ACS Nano 12, 3103–3111 (2018).
Lu, D. et al. Scalable fabrication of resilient sic nanowires aerogels with exceptional high-temperature stability. ACS Appl. Mater. Interfaces 11, 45338–45344 (2019).
Wang, F., Richards, V. N., Shields, S. P. & Buhro, W. E. Kinetics and mechanisms of aggregative nanocrystal growth. Chem. Mater. 26, 5–21 (2014).
Li, X. et al. High temperature resistant polyimide/boron carbide composites for neutron radiation shielding. Composites Part B 159, 355–361 (2019).
Aatthisugan, I., Razal Rose, A. & Selwyn Jebadurai, D. Mechanical and wear behaviour of az91d magnesium matrix hybrid composite reinforced with boron carbide and graphite. J. Magnesium Alloys 5, 20–25 (2017).
Barrigón, E., Heurlin, M., Bi, Z., Monemar, B. & Samuelson, L. Synthesis and applications of III–V nanowires. Chem. Rev. 119, 9170–9220 (2019).
Li, Z., Yan, T. & Fang, X. Low-dimensional wide-bandgap semiconductors for UV photodetectors. Nat. Rev. Mater. 8, 587–603 (2023).
Saito, Y. Nanoparticles and filled nanocapsules. Carbon 33, 979–988 (1995).
Magnin, Y., Amara, H., Ducastelle, F., Loiseau, A. & Bichara, C. Entropy-driven stability of chiral single-walled carbon nanotubes. Science 362, 212–215 (2018).
Dong, Q. et al. Depolymerization of plastics by means of electrified spatiotemporal heating. Nature 616, 488–494 (2023).
Li, M. et al. Metallic catalysts for structure-controlled growth of single-walled carbon nanotubes. Top. Curr. Chem. 375, 25–67 (2017).
Wu, D. N., Sheng, J., Lu, H. G., Li, S. D. & Li, Y. Mass production of graphene using high-power rapid Joule heating method. Chem. Eng. J. 505, 159725 (2025).
Acknowledgements
We gratefully acknowledge R. Wen from Saiyin Materials Co., Ltd. for debugging the RJH equipment. Y.L. acknowledges support from National Natural Science Foundation of China (grant nos. 22120102004, U23A2085 and U21A6004), Shenzhen KQTD Project (grant no. KQTD20180411143400981) and Beijing National Laboratory for Molecular Sciences (grant no. BNLMS-CXTD-202001). This work was supported by the High-Performance Computing Platform of Peking University.
Author information
Authors and Affiliations
Contributions
Y.L., J.S. and Y.X. designed the research. Y.L., J.S., Y.X. and H.-G.L. designed the RJH reactors. H.-G.L. and S.-D.L. designed the RJH control system. J.S. and Y.X. performed the synthesis and characterization of 1D nanomaterials. Z.H., X.Z. and R.L. performed the TEM characterizations. S.S. performed the molecular dynamics simulations. C.X. performed the GC-FID tests. J.S., Y.X. and D.-N.W. performed the amplified synthesis of SiC. All authors discussed the experiments and contributed to the data analysis. Y.L., J.S., Y.X. and S.S. contributed to the writing of the paper. Y.L., J.S. and Y.X. reviewed the paper.
Corresponding author
Ethics declarations
Competing interests
Y.L., J.S., Y.X. and Z.H. are listed as inventors of a patent application filed by Peking University (China National Intellectual Property Administration application no. 202311843212.1). The other authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Feng Ru Fan, Il-Doo Kim and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alexandra Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–7, Figs. 1–42, Tables 1–4 and references.
Source data
Source Data Fig. 1 (download XLSX )
Source data for Fig. 1d
Source Data Fig. 2 (download XLSX )
Source data for Fig. 2a,d,e.
Source Data Fig. 3 (download XLSX )
Source data for Fig. 3b,f,j.
Source Data Fig. 4 (download XLSX )
Source data for Fig. 4a,b,f.
Source Data Fig. 5 (download XLSX )
Source data for Fig. 5e,f,h,i.
Source Data Fig. 6 (download XLSX )
Source data for Fig. 6b–d.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sheng, J., Xu, Y., Han, Z. et al. Catalytic Joule heating synthesis of one-dimensional nanomaterials in seconds. Nat. Synth 5, 367–376 (2026). https://doi.org/10.1038/s44160-025-00933-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-025-00933-1