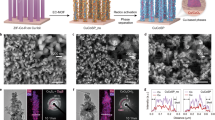
Abstract
The electrocatalytic conversion of nitrate (NO3−) in NO3−-rich wastewater streams to ammonia (NH3) can promote reactive nitrogen recovery and decentralized energy storage. However, it remains challenging to efficiently produce NH3 from NO3−. Here we designed a high-performance CuPd bimetallic catalyst with abundant Cu–Pd hydride interfaces under electrochemical NO3− reduction reaction conditions. The NH3 production rate in a membrane electrode assembly electrolyser reached ∼19.9 mmol h−1 cm−2 with a current density of 5.0 A cm−2 at ∼2.56 V, and the catalyst remained stable at 2.0 A cm−2 with an NH3 Faradaic efficiency of ∼86.8% for 1,000 h. Mechanistic studies attribute the high performance to the Cu–Pd hydride interface structure that facilitates *NO hydrogenation and *NH3 desorption. Furthermore, we successfully extended the high performance to an electrolyser stack with five 100-cm2 membrane electrode assemblies. The demonstrated scalability and long-term robustness underscore the industrial applicability of this approach for upstream integration with NOx sources.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper and the Supplementary Information. Source data are provided with this paper.
Change history
04 December 2025
A Correction to this paper has been published: https://doi.org/10.1038/s44160-025-00969-3
References
MacFarlane, D. R. et al. A roadmap to the ammonia economy. Joule 4, 1186–1205 (2020).
Smith, C., Hill, A. K. & Torrente-Murciano, L. Current and future role of Haber–Bosch ammonia in a carbon-free energy landscape. Energy Environ. Sci. 13, 331–344 (2020).
Soloveichik, G. Electrochemical synthesis of ammonia as a potential alternative to the Haber–Bosch process. Nat. Catal. 2, 377–380 (2019).
Chen, J. G. et al. Beyond fossil fuel-driven nitrogen transformations. Science 360, 873 (2018).
Chen, F.-Y. et al. Electrochemical nitrate reduction to ammonia with cation shuttling in a solid electrolyte reactor. Nat. Catal. https://doi.org/10.1038/s41929-024-01200-w (2024).
Zhang, G. et al. Ammonia recovery from nitrate-rich wastewater using a membrane-free electrochemical system. Nat. Sustain. https://doi.org/10.1038/s41893-024-01406-7 (2024).
Nguyen, T. T. P. et al. Selectiveness of copper and polypyrrole modified copper electrodes for nitrate electroreduction: a comparative study and application in ground water. ECS Trans. 53, 41–52 (2013).
Chauhan, R. & Srivastava, V. C. Electrochemical denitrification of highly contaminated actual nitrate wastewater by Ti/RuO2 anode and iron cathode. Chem. Eng. J. 386, 122065 (2020).
Katsounaros, I., Dortsiou, M. & Kyriacou, G. Electrochemical reduction of nitrate and nitrite in simulated liquid nuclear wastes. J. Hazard. Mater. 171, 323–327 (2009).
John, J., MacFarlane, D. R. & Simonov, A. N. The why and how of NOx electroreduction to ammonia. Nat. Catal. 6, 1125–1130 (2023).
Liu, W. et al. Efficient ammonia synthesis from the air using tandem non-thermal plasma and electrocatalysis at ambient conditions. Nat. Commun. 15, 3524 (2024).
Chen, G.-F. et al. Electrochemical reduction of nitrate to ammonia via direct eight-electron transfer using a copper–molecular solid catalyst. Nat. Energy 5, 605–613 (2020).
Wang, Y., Zhou, W., Jia, R., Yu, Y. & Zhang, B. Unveiling the activity origin of a copper-based electrocatalyst for selective nitrate reduction to ammonia. Angew. Chem. Int. Ed. 59, 5350–5354 (2020).
Hu, Q. et al. Reaction intermediate-mediated electrocatalyst synthesis favors specified facet and defect exposure for efficient nitrate–ammonia conversion. Energy Environ. Sci. 14, 4989–4997 (2021).
Li, P. et al. Pulsed nitrate-to-ammonia electroreduction facilitated by tandem catalysis of nitrite intermediates. J. Am. Chem. Soc. 145, 6471–6479 (2023).
Bai, L. et al. Electrocatalytic nitrate and nitrite reduction toward ammonia using Cu2O nanocubes: active species and reaction mechanisms. J. Am. Chem. Soc. 146, 9665–9678 (2024).
Hao, D. et al. Emerging alternative for artificial ammonia synthesis through catalytic nitrate reduction. J. Mater. Sci. Technol. 77, 163–168 (2021).
Liu, J.-X., Richards, D., Singh, N. & Goldsmith, B. R. Activity and selectivity trends in electrocatalytic nitrate reduction on transition metals. ACS Catal. 9, 7052–7064 (2019).
Chen, F. Y. et al. Efficient conversion of low-concentration nitrate sources into ammonia on a Ru-dispersed Cu nanowire electrocatalyst. Nat. Nanotechnol. 17, 759–767 (2022).
Gao, Q. et al. Breaking adsorption-energy scaling limitations of electrocatalytic nitrate reduction on intermetallic CuPd nanocubes by machine-learned insights. Nat. Commun. 13, 2338 (2022).
Liu, H. et al. Efficient electrochemical nitrate reduction to ammonia with copper-supported rhodium cluster and single-atom catalysts. Angew. Chem. Int. Ed. 61, e202202556 (2022).
Zhang, S. et al. Fe/Cu diatomic catalysts for electrochemical nitrate reduction to ammonia. Nat. Commun. 14, 3634 (2023).
Fang, J. Y. et al. Ampere-level current density ammonia electrochemical synthesis using CuCo nanosheets simulating nitrite reductase bifunctional nature. Nat. Commun. 13, 7899 (2022).
Wang, Y. et al. Enhanced nitrate-to-ammonia activity on copper–nickel alloys via tuning of intermediate adsorption. J. Am. Chem. Soc. 142, 5702–5708 (2020).
Wu, L. et al. Boosting electrocatalytic nitrate-to-ammonia via tuning of N-intermediate adsorption on a Zn–Cu catalyst. Angew. Chem. Int. Ed. 62, e202307952 (2023).
Johnson, N. J. J. et al. Facets and vertices regulate hydrogen uptake and release in palladium nanocrystals. Nat. Mater. 18, 454–458 (2019).
Xiao, L. et al. Pd-intercalated black phosphorus: an efficient electrocatalyst for CO2 reduction. Sci. Adv. 10, eadn2707 (2024).
Gao, D. et al. Switchable CO2 electroreduction via engineering active phases of Pd nanoparticles. Nano Res 10, 2181–2191 (2017).
Chen, C. et al. Coupling N2 and CO2 in H2O to synthesize urea under ambient conditions. Nat. Chem. 12, 717–724 (2020).
Du, C. et al. Selectively reducing nitrate into NH3 in neutral media by PdCu single-atom alloy electrocatalysis. ACS Catal. 13, 10560–10569 (2023).
Xu, Y. et al. Electronic metal-support interaction triggering interfacial charge polarization over CuPd/N-doped-C nanohybrids drives selectively electrocatalytic conversion of nitrate to ammonia. Small 18, e2203335 (2022).
Zhou, Y. et al. Optimizing intermediate adsorption over PdM (M = Fe, Co, Ni, Cu) bimetallene for boosted nitrate electroreduction to ammonia. Angew. Chem. Int. Ed. 63, e202319029 (2024).
Teschner, D. et al. The roles of subsurface carbon and hydrogen in palladium-catalyzed alkyne hydrogenation. Science 320, 86–89 (2008).
Lin, C.-M. et al. Size-dependent lattice structure of palladium studied by X-ray absorption spectroscopy. Phys. Rev. B 75, 125426 (2007).
Liu, G. et al. Hydrogen-intercalation-induced lattice expansion of Pd@Pt core–shell nanoparticles for highly efficient electrocatalytic alcohol oxidation. J. Am. Chem. Soc. 143, 11262–11270 (2021).
Li, H. et al. Oxidative stability matters: a case study of palladium hydride nanosheets for alkaline fuel cells. J. Am. Chem. Soc. 144, 8106–8114 (2022).
Kabiraz, M. K. et al. Ligand effect of shape-controlled β-palladium hydride nanocrystals on liquid-fuel oxidation reactions. Chem. Mater. 31, 5663–5673 (2019).
Bugaev, A. L. et al. Temperature- and pressure-dependent hydrogen concentration in supported PdHx nanoparticles by Pd K-edge X-ray absorption spectroscopy. J. Phys. Chem. C 118, 10416–10423 (2014).
Schnitkey, G., Paulson, N., Zulauf, C. & Baltz, J. Fertilizer prices and company profits going into spring 2023. Farmdoc Daily https://farmdocdaily.illinois.edu/2023/02/fertilizer-prices-and-company-profits-going-into-spring-2023.html (2023).
Majhi, K. C. et al. In-tandem electrochemical reduction of nitrate to ammonia on ultrathin-sheet-assembled iron-nickel alloy nanoflowers. Angew. Chem. Int. Ed. 64, e202500167 (2025).
Zhang, L.-H. et al. Dopant-induced electronic states regulation boosting electroreduction of dilute nitrate to ammonium. Angew. Chem. Int. Ed. 62, e202303483 (2023).
Krzywda, P. M., Paradelo Rodríguez, A., Benes, N. E., Mei, B. T. & Mul, G. Carbon–nitrogen bond formation on Cu electrodes during CO2 reduction in NO3− solution. Appl. Catal. B Environ. 316, 121512 (2022).
Henson, M. et al. Resonance Raman investigation of equatorial ligand donor effects on the Cu2O22+ core in end-on and side-on μ-peroxo-dicopper(II) and bis-μ-oxo-dicopper(III) complexes. J. Am. Chem. Soc. 125, 5186–5192 (2003).
Fujisawa, K., Lehnert, N., Ishikawa, Y. & Okamoto, K.-i. Diazene complexes of copper: synthesis, spectroscopic analysis, and electronic structure. Angew. Chem. Int. Ed. 116, 5052–5055 (2004).
Torreggiani, A., Esposti, A. D., Tamba, M., Marconi, G. & Fini, G. Experimental and theoretical Raman investigation on interactions of Cu(II) with histamine. J. Raman Spectrosc. 37, 291–298 (2006).
Wang, Y. et al. Wide-pH-range adaptable ammonia electrosynthesis from nitrate on Cu–Pd interfaces. Sci. China Chem. 66, 913–922 (2023).
Butcher, D. P. & Gewirth, A. A. Nitrate reduction pathways on Cu single crystal surfaces: effect of oxide and Cl−. Nano Energy 29, 457–465 (2016).
Liu, S. et al. Proton-filtering covalent organic frameworks with superior nitrogen penetration flux promote ambient ammonia synthesis. Nat. Catal. 4, 322–331 (2021).
Wang, C. et al. In situ loading of Cu2O active sites on island-like copper for efficient electrochemical reduction of nitrate to ammonia. ACS Appl. Mater. Interfaces 14, 6680–6688 (2022).
Chang, Z. et al. Dual-site W–O–CoP catalysts for active and selective nitrate conversion to ammonia in a broad concentration window. Adv. Mater. 35, e2304508 (2023).
Yao, Y., Zhu, S., Wang, H., Li, H. & Shao, M. A spectroscopic study on the nitrogen electrochemical reduction reaction on gold and platinum surfaces. J. Am. Chem. Soc. 140, 1496–1501 (2018).
Yao, Y., Zhu, S., Wang, H., Li, H. & Shao, M. Regulating active hydrogen adsorbed on grain boundary defects of nano-nickel for boosting ammonia electrosynthesis from nitrate. Energy Environ. Sci. 16, 2611–2620 (2023).
Zhang, B. et al. Defect-induced triple synergisticmodulation in copper for superior electrochemical ammonia production across broad nitrate concentrations. Nat. Commun. 15, 2816 (2024).
Hafner, J. Ab-initio simulations of materials using VASP: density-functional theory and beyond. J. Comput. Chem. 29, 2044–2078 (2008).
Perdew, J., Burke, K. & Ernzerhof, F. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 27, 1787–1799 (2006).
Yang, J. et al. Dynamic behavior of single-atom catalysts in electrocatalysis: identification of Cu–N3 as an active site for the oxygen reduction reaction. J. Am. Chem. Soc. 143, 14530–14539 (2021).
Bader, R. F. W. Atoms in Molecules: A Quantum Theory (Oxford Univ. Press, 1990).
Nørskov, J. K. et al. Trends in the exchange current for hydrogen evolution. J. Electrochem. Soc. 152, J23–J26 (2005).
Acknowledgements
This work was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB0600200, G.W.), the National Natural Science Foundation of China (22125205, G.W.; 22302204, P.W.; 22321002, G.W.; 22350710789, G.W.), the Natural Science Foundation of Shanghai (25DZ3000400, G.W.), the Liaoning Binhai Laboratory (LBLF-2023-02, G.W.), the Fundamental Research Funds for the Central Universities (20720220008, G.W.) and the Photon Science Center for Carbon Neutrality (JZHKYPT-2021-07, G.W.). We thank the staff of the Shanghai Synchrotron Radiation Facility beamlines BL11B and BL14W1 for assistance with XAS measurements. We thank M. Gong and S. Cha for assistance with the ATR-SEIRAS measurements. We thank F. Yang and S. Lou for assistance with the ICP-OES measurements.
Author information
Authors and Affiliations
Contributions
G.W. and X.B. supervised the project and conceived of the idea. Y.F. designed and synthesized the materials, and conducted the electrochemical and structural characterizations. S.W. performed the theoretical calculation study. P.W. performed in situ XRD analyses and the scale-up demonstration. R.L. and Q.F. performed quasi in situ XPS analyses. Y.F., S.W., P.W., Y.W., R.L., J.S., D.G., Q.F, G.W. and X.B. discussed the experimental results. Y.F., S.W., P.W., D.G., Q.F., G.W. and X.B. wrote and edited the paper with input from all authors. All authors discussed the results and assisted during the preparation of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Ramendra Sundar Dey, Geun Ho Gu and Jason Chun-Ho Lam for their contribution to the peer review of this work. Primary Handling Editor: Alexandra Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Figs. 1–61, Tables 1–18 and References 1–31.
Supplementary Data 1 (download XLSX )
Source data for Supplementary Figs. 1–61.
Source data
Source Data 1–5 (download XLSX )
Source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fu, Y., Wang, S., Wei, P. et al. Copper–palladium hydride interfaces promote electrochemical ammonia synthesis. Nat. Synth 5, 433–444 (2026). https://doi.org/10.1038/s44160-025-00941-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-025-00941-1