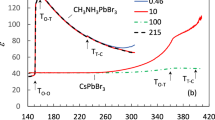
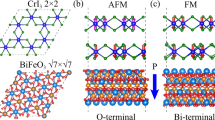
Abstract
The mixed tetrahedral–octahedral (MTO) architecture, a characteristic structural motif of brownmillerite and spinel oxides, underpins the multiferroic behaviour and magnetoelectric coupling exhibited by these oxides. Yet, this structural motif remains unexplored in two-dimensional hybrid organic–inorganic perovskites (HOIPs) that typically adopt the octahedral framework. Here we demonstrate that incorporating sterically demanding organic ligands into copper halide HOIPs stabilizes the MTO phase over the Ruddlesden–Popper phase during single-crystal growth. This approach yields a family of MTO perovskites exhibiting structural asymmetry and diverse magnetic orderings. Interestingly, (4,4-difluoropiperidinium)2CuBr4 displays concurrent ferroelectricity and intralayer antiferromagnetism. By monitoring strain-mediated phase evolution, we examine the reversible transition between the MTO and Ruddlesden–Popper phases. Our work broadens the structural diversity of 2D HOIP beyond conventional octahedral frameworks, establishing a framework for exploring emergent multiferroicity and magnetoelectric phenomena.

This is a preview of subscription content, access via your institution
Access options





Similar content being viewed by others
Data availability
All data supporting the findings of this study are available in the article and its Supplementary Information. Source data are provided with this paper. The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers DFPR2CuBr4 (CCDC 2350516, 2454190, 2428613), DFPP2CuBr4 (CCDC 2350517), DFAZ2CuBr4·0.5H2O (CCDC 2375732), DFAT2CuBr4 (CCDC 2350515), DFPP2CuCl4 (CCDC 2428842) and (R-3FP)2CuCl4 (CCDC 2429039, 2429040). These data can be obtained free of charge via The Cambridge Crystallographic Data Centre at www.ccdc.cam.ac.uk/data_request/cif.
References
Xing, Y. et al. Monitoring the formation of infinite-layer transition metal oxides through in situ atomic-resolution electron microscopy. Nat. Chem. 17, 66–73 (2025).
Zhang, J. et al. A correlated ferromagnetic polar metal by design. Nat. Mater. 23, 912–919 (2024).
Kang, K. T. et al. A room-temperature ferroelectric ferromagnet in a 1D tetrahedral chain network. Adv. Mater. 31, 1808104 (2019).
Kim, W. J. et al. Geometric frustration of Jahn–Teller order in the infinite-layer lattice. Nature 615, 237–243 (2023).
Jeen, H. et al. Topotactic phase transformation of the brownmillerite SrCoO2.5 to the perovskite SrCoO3–δ. Adv. Mater. 25, 3651–3656 (2013).
Emori, S. et al. Coexistence of low damping and strong magnetoelastic coupling in epitaxial spinel ferrite thin films. Adv. Mater. 29, 1701130 (2017).
Burdett, J. K., Price, G. D. & Price, S. L. Role of the crystal-field theory in determining the structures of spinels. J. Am. Chem. Soc. 104, 92–95 (1982).
Zhang, J. et al. Brownmillerite Ca2Co2O5: synthesis, stability, and re-entrant single crystal to single crystal structural transitions. Chem. Mater. 26, 7172–7182 (2014).
Chen, X. et al. Theoretical investigation of magnetic anisotropy at the La0.5Sr0.5MnO3/LaCoO2.5 interface. Phys. Rev. B 100, 144413 (2019).
Yamasaki, Y. et al. Magnetic reversal of the ferroelectric polarization in a multiferroic spinel oxide. Phys. Rev. Lett. 96, 207204 (2006).
Hemberger, J. et al. Relaxor ferroelectricity and colossal magnetocapacitive coupling in ferromagnetic CdCr2S4. Nature 434, 364–367 (2005).
Sandemann, J. R. et al. Direct visualization of magnetic correlations in frustrated spinel ZnFe2O4. Adv. Mater. 35, 2207152 (2023).
Xing, Y. et al. Atomic-scale operando observation of oxygen diffusion during topotactic phase transition of a perovskite oxide. Matter 5, 3009–3022 (2022).
Khare, A. et al. Topotactic metal–insulator transition in epitaxial SrFeOx thin films. Adv. Mater. 29, 1606566 (2017).
Li, J. et al. Topotactic phase transformations by concerted dual-ion migration of B-site cation and oxygen in multivalent cobaltite La–Sr–Co–Ox films. Nano Energy 78, 105215 (2020).
Choi, H. S. et al. Molecularly thin, two-dimensional all-organic perovskites. Science 384, 60–66 (2024).
Aubrey, M. L. et al. Directed assembly of layered perovskite heterostructures as single crystals. Nature 597, 355–359 (2021).
Mao, L., Stoumpos, C. C. & Kanatzidis, M. G. Two-dimensional hybrid halide perovskites: principles and promises. J. Am. Chem. Soc. 141, 1171–1190 (2019).
Sun, J. et al. Emerging two-dimensional organic semiconductor-incorporated perovskites─a fascinating family of hybrid electronic materials. J. Am. Chem. Soc. 145, 20694–20715 (2023).
Morteza Najarian, A. et al. Homomeric chains of intermolecular bonds scaffold octahedral germanium perovskites. Nature 620, 328–335 (2023).
Gilley, I. W. et al. Combining organic cations of different sizes grants improved control over perovskitoid dimensionality and bandgap. J. Am. Chem. Soc. 147, 7777–7787 (2025).
Abdelwahab, I. et al. Two-dimensional chiral perovskites with large spin Hall angle and collinear spin Hall conductivity. Science 385, 311–317 (2024).
Kim, K. Y. et al. Intrinsic magnetic order of chemically exfoliated 2D Ruddlesden–Popper organic–inorganic halide perovskite ultrathin films. Small 16, 2005445 (2020).
Walsh, K. M., Smith, R. T. & Gamelin, D. R. Anion exchange and lateral heterostructure formation in ferromagnetic PEA2Cr(Cl,Br)4 two-dimensional perovskites. J. Am. Chem. Soc. 146, 29159–29168 (2024).
Nakayama, Y., Nishihara, S., Inoue, K., Suzuki, T. & Kurmoo, M. Coupling of magnetic and elastic domains in the organic–inorganic layered perovskite-like (C6H5C2H4NH3)2FeIICl4 crystal. Angew. Chem. Int. Ed. 56, 9367–9370 (2017).
Zheng, H. et al. Chiral multiferroicity in two-dimensional hybrid organic–inorganic perovskites. Nat. Commun. 15, 5556 (2024).
Gu, H. et al. Phase-pure two-dimensional layered perovskite thin films. Nat. Rev. Mater. 8, 533–551 (2023).
Hao, J. et al. Direct detection of circularly polarized light using chiral copper chloride–carbon nanotube heterostructures. ACS Nano 15, 7608–7617 (2021).
Mao, L., Chen, J., Vishnoi, P. & Cheetham, A. K. The renaissance of functional hybrid transition-metal halides. Acc. Mater. Res. 3, 439–448 (2022).
Daub, M., Natalukha, D. & Hillebrecht, H. Crystal structures of the perovskite-related system A/Rb/Cu(II)/Br (A = BA, Gu, PEA, 5-AVA, H2en) with winners, losers and compromises—versatility from 0D to 3D on different levels. Eur. J. Inorg. Chem. 2022, e202200136 (2022).
Li, X. et al. (C6H5CH2NH3)2CuBr4: a lead-free, highly stable two-dimensional perovskite for solar cell applications. ACS Appl. Energy Mater. 1, 2709–2716 (2018).
Zhan, X. et al. Enhanced structural stability and pressure-induced photoconductivity in two-dimensional hybrid perovskite (C6H5CH2NH3)2CuBr4. Angew. Chem. Int. Ed. 61, e202205491 (2022).
Harper, K. C., Bess, E. N. & Sigman, M. S. Multidimensional steric parameters in the analysis of asymmetric catalytic reactions. Nat. Chem. 4, 366–374 (2012).
Brethomé, A. V., Fletcher, S. P. & Paton, R. S. Conformational effects on physical-organic descriptors: the case of sterimol steric parameters. ACS Catal. 9, 2313–2323 (2019).
Howard, J. R., Bhakare, A., Akhtar, Z., Wolf, C. & Anslyn, E. V. Data-driven prediction of circular dichroism-based calibration curves for the rapid screening of chiral primary amine enantiomeric excess values. J. Am. Chem. Soc. 144, 17269–17276 (2022).
Ai, Y., Lv, H.-P., Wang, Z.-X., Liao, W.-Q. & Xiong, R.-G. H/F substitution for advanced molecular ferroelectrics. Trends Chem. 3, 1088–1099 (2021).
Liu, H.-Y., Zhang, H.-Y., Chen, X.-G. & Xiong, R.-G. Molecular design principles for ferroelectrics: ferroelectrochemistry. J. Am. Chem. Soc. 142, 15205–15218 (2020).
Sun, B. et al. Intrinsic ferromagnetic semiconductors with high saturation magnetization from hybrid perovskites. Adv. Mater. 35, 2303945 (2023).
Sheldrick, G. Crystal structure refinement with SHELXL. Acta Crystallogr. C 71, 3–8 (2015).
Chen, Z., Xue, J., Wang, Z. & Lu, H. Magnetic hybrid transition metal halides. Mater. Chem. Front. 8, 210–227 (2024).
Zhang, H.-Y. et al. Observation of vortex domains in a two-dimensional lead iodide perovskite ferroelectric. J. Am. Chem. Soc. 142, 4925–4931 (2020).
Wang, C.-F. et al. Centimeter-sized single crystals of two-dimensional hybrid iodide double perovskite (4,4-Difluoropiperidinium)4AgBiI8 for high-temperature ferroelectricity and efficient X-Ray detection. Adv. Funct. Mater. 31, 2009457 (2021).
Comstock, A. H. et al. Hybrid magnonics in hybrid perovskite antiferromagnets. Nat. Commun. 14, 1834 (2023).
Wong, R. J. H., Willett, R. D. & Drumheller, J. E. An EPR study of interlayer exchange coupling in the quasi-two-dimensional salts, (CnH2n+1NH3)2CuCl4, with n = 1, 2, and 3. J. Chem. Phys. 74, 6018–6021 (1981).
Gong, C. et al. Discovery of intrinsic ferromagnetism in two-dimensional van der Waals crystals. Nature 546, 265–269 (2017).
Huang, B. et al. Layer-dependent ferromagnetism in a van der Waals crystal down to the monolayer limit. Nature 546, 270–273 (2017).
Taniguchi, K. et al. Magneto-electric directional anisotropy in polar soft ferromagnets of two-dimensional organic–inorganic hybrid perovskites. Angew. Chem. Int. Ed. 60, 14350–14354 (2021).
Zhao, T. et al. Electrical control of antiferromagnetic domains in multiferroic BiFeO3 films at room temperature. Nat. Mater. 5, 823–829 (2006).
Hu, Q. et al. Ferrielectricity controlled widely-tunable magnetoelectric coupling in van der Waals multiferroics. Nat. Commun. 15, 3029 (2024).
Sun, M.-E. et al. Chirality-dependent structural transformation in chiral 2D perovskites under high pressure. J. Am. Chem. Soc. 145, 8908–8916 (2023).
Stepakova, L. V. et al. Vibrational spectroscopic and force field studies of copper(II) chloride and bromide compounds, and crystal structure of KCuBr3. J. Raman Spectrosc. 39, 16–31 (2008).
Jaffe, A. et al. High compression-induced conductivity in a layered Cu–Br perovskite. Angew. Chem. Int. Ed. 59, 4017–4022 (2020).
Mao, H. K., Xu, J. & Bell, P. M. Calibration of the ruby pressure gauge to 800 kbar under quasi-hydrostatic conditions. J. Geophys. Res. 91, 4673–4676 (1986).
Wang, L., Maxisch, T. & Ceder, G. Oxidation energies of transition metal oxides within the GGA+ U framework. Phys. Rev. B 73, 195107 (2006).
Tsirlin, A. A., Abakumov, A. M., Ritter, C. & Rosner, H. (CuCl)LaTa2O7 and quantum phase transition in the (CuX)LaM2O7 family (X= Cl, Br; M= Nb, Ta). Phys. Rev. B 86, 064440 (2012).
Zheng, H. et al. Strain-driven solid–solid crystal conversion in chiral hybrid pseudo-perovskites with paramagnetic-to-ferromagnetic transition. J. Am. Chem. Soc. 145, 3569–3576 (2023).
Acknowledgements
K.P.L. acknowledges funding support from Hong Kong’s Research Grants Council GRF project ID P0046892 (funding reference 1530223) and HK PolyU’s grant no. P0043087, as well as Singapore’s Ministry of Education Tier 1 Grant ‘A new class of 2D metal free dielectric’ grant no. A80026710000. W.H. acknowledges the support by the National Key R&D Program (2022YFB3603800, 2022YFA1204401, 2021YFB3600700), the National Natural Science Foundation of China (52121002, U21A6002). High-pressure XRD data were collected at beamline station BL15U1 (31124.02.SSRF.BL15U1) of Shanghai Synchrotron Radiation Facility (SSRF, 31124.02.SSRF), Shanghai, China.
Author information
Authors and Affiliations
Contributions
The work was conceived by M.W., S.Y., D.J., W.H. and K.P.L. with input from all authors. M.W. carried out the synthesis, single-crystal XRD, PFM and P–E curve characterizations. S.Y. performed DFT calculations and analysed the results. D.J. performed the DAC experiment under the supervision of Y.W. W.P.D.W. and H.Y. analysed the single-crystal XRD data. H.S.C. performed variable-temperature XRD. C.Z. conducted non-contact atomic force microscope. X.Z. helped to measure the magnetic properties. W.H. and K.P.L. supervised this project. M.W. and K.P.L. cowrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Linn Leppert and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alexandra Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Materials and conventional methods; Supplementary Notes 1 and 2, Figs. 1–28 and Tables 1–6; and X-ray structures.
Supplementary Data 1
Crystallographic data for compound DFPR2CuBr4 at room temperature, CCDC 2350516.
Supplementary Data 2
Crystallographic data for compound DFPR2CuBr4 at 240 K, CCDC 2454190.
Supplementary Data 3
Crystallographic data for compound DFPR2CuBr4 at 100 K, CCDC 2428613.
Supplementary Data 4
Crystallographic data for compound DFPP2CuBr4 at room temperature, CCDC 2350517.
Supplementary Data 5
Crystallographic data for compound DFAZ2CuBr4·0.5H2O at room temperature, CCDC 2375732.
Supplementary Data 6
Crystallographic data for compound DFAT2CuBr4 at room temperature, CCDC 2350515.
Supplementary Data 7
Crystallographic data for compound DFPP2CuCl4 at room temperature, CCDC 2428842.
Supplementary Data 8
Crystallographic data for compound (R-3FP)2CuCl4 at room temperature, CCDC 2429039.
Supplementary Data 9
Crystallographic data for compound (R-3FP)2CuCl4 at 340 K, CCDC 2429040.
Source data
Source Data Fig. 3 (download XLSX )
Statistical source data for Fig. 3d,f,g.
Source Data Fig. 4 (download XLSX )
Statistical source data for Fig. 4a–e.
Source Data Fig. 5 (download XLSX )
Statistical source data for Fig. 5c–f,i.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, M., Yuan, S., Jiang, D. et al. Two-dimensional mixed tetrahedral–octahedral hybrid perovskites with coexisting ferroelectricity and intralayer antiferromagnetism. Nat. Synth 5, 445–454 (2026). https://doi.org/10.1038/s44160-025-00942-0
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44160-025-00942-0