Abstract
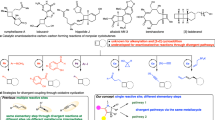
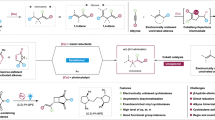
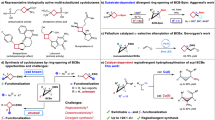
The configuration of cyclobutane-based drug molecules plays a critical role in their biological and pharmacological properties. Although substantial progress has been made in the chemoselective and diastereoselective construction of substituted cyclobutanes, achieving efficient diastereodivergent catalysis from the same set of starting materials remains challenging. In this work, an electrochemical cobalt-catalysed method for the reductive coupling of alkynes and cyclobutenes is reported, enabling the synthesis of a range of substituted cyclobutane diastereoisomers. Proton sources are used to generate cobalt–hydride intermediates through a sequence of cathodic reduction and protonation. By modulating the steric and electronic properties of the proton sources, the trans/cis ratio can be altered, offering a different approach for the efficient synthesis of two diastereoisomers of the desired cyclobutane product. Mechanistic studies support a Co(II)–H reaction pathway and suggest that diastereodivergence arises from distinct distortions of the proton source and differential interactions between the cyclobutyl cobalt intermediate and the respective proton sources.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data are available in the main text or the Supplementary Information. The crystallographic data for the structures reported in this paper are freely available from the Cambridge Crystallographic Data Centre under the associated CCDC codes: 2401812 (rac-38), 2401813 (33-cis), 2401814 (38) and 2401815 (33-cis (S,R)). Copies of the data can be obtained free of charge from the CCDC via https://www.ccdc.cam.ac.uk/structures/.
References
Yang, P., Jia, Q., Song, S. & Huang, X. [2+2]-Cycloaddition-derived cyclobutane natural products: structural diversity, sources, bioactivities, and biomimetic syntheses. Nat. Prod. Rep. 40, 1094–1129 (2023).
Dembitsky, V. M. Naturally occurring bioactive cyclobutane-containing (CBC) alkaloids in fungi, fungal endophytes, and plants. Phytomedicine 21, 1559–1581 (2014).
Van der Kolk, M. R., Janssen, M. A. C. H., Rutjes, F. P. J. T. & Blanco-Ania, D. Cyclobutanes in small-molecule drug candidates. ChemMedChem 17, e202200020 (2022).
Genzink, M. J., Rossler, M. D., Recendiz, H. & Yoon, T. P. A general strategy for the synthesis of truxinate natural products enabled by enantioselective [2 + 2] photocycloadditions. J. Am. Chem. Soc. 145, 19182–19188 (2023).
Xu, Y., Conner, M. L. & Brown, M. K. Cyclobutane and cyclobutene synthesis: catalytic enantioselective [2 + 2] cycloadditions. Angew. Chem. Int. Ed. 54, 11918–11928 (2015).
Poplata, S., Tröster, A., Zou, Y.-Q. & Bach, T. Recent advances in the synthesis of cyclobutanes by olefin [2 + 2] photocycloaddition reactions. Chem. Rev. 116, 9748–9815 (2016).
Xiong, P., Ivlev, S. I. & Meggers, E. Photoelectrochemical asymmetric dehydrogenative [2 + 2] cycloaddition between C–C single and double bonds via the activation of two C(sp3)–H bonds. Nat. Catal. 6, 1186–1193 (2023).
Das, S. et al. Asymmetric counteranion-directed photoredox catalysis. Science 379, 494–499 (2023).
Guo, J. et al. Visible light-mediated intermolecular crossed [2 + 2] cycloadditions using a MOF-supported copper triplet photosensitizer. Nat. Catal. 7, 307–320 (2024).
Goetzke, F. W., Hell, A. M. L., van Dijk, L. & Fletcher, S. P. A catalytic asymmetric cross-coupling approach to the synthesis of cyclobutanes. Nat. Chem. 13, 880–886 (2021).
Liang, Z. et al. Cobalt-catalyzed diastereo- and enantioselective carbon–carbon bond forming reactions of cyclobutenes. J. Am. Chem. Soc. 145, 3588–3598 (2023).
Girvin, Z. C. et al. Asymmetric photochemical [2 + 2]-cycloaddition of acyclic vinylpyridines through ternary complex formation and an uncontrolled sensitization mechanism. J. Am. Chem. Soc. 144, 20109–20117 (2022).
Hu, L. et al. PdII-catalyzed enantioselective C(sp3)−H activation/cross-coupling reactions of free carboxylic acids. Angew. Chem. Int. Ed. 58, 2134–2138 (2019).
Xiao, L.-J. et al. PdII-catalyzed enantioselective C(sp3)–H arylation of cyclobutyl ketones using a chiral transient directing group. Angew. Chem. Int. Ed. 59, 9594–9600 (2020).
Lan, S. et al. Asymmetric transfer hydrogenation of cyclobutenediones. J. Am. Chem. Soc. 146, 4942–4957 (2024).
Wang, Y.-M., Bruno, N. C., Placeres, ÁL., Zhu, S. & Buchwald, S. L. Enantioselective synthesis of carbo- and heterocycles through a CuH-catalyzed hydroalkylation approach. J. Am. Chem. Soc. 137, 10524–10527 (2015).
Kim, D. K., Riedel, J., Kim, R. S. & Dong, V. M. Cobalt catalysis for enantioselective cyclobutanone construction. J. Am. Chem. Soc. 139, 10208–10211 (2017).
Chen, J., Zhou, Q., Fang, H. & Lu, P. Dancing on ropes—enantioselective functionalization of preformed four-membered carbocycles. Chin. J. Chem. 40, 1346–1358 (2022).
Yasukawa, T., Gilles, P., Martin, J., Boutet, J. & Cossy, J. Enantioselective reduction of cyclobutenones using ene-reductases. Adv. Synth. Catal. 366, 3257–3261 (2024).
Wang, Z., Zhu, J., Wang, M. & Lu, P. Palladium-catalyzed divergent enantioselective functionalization of cyclobutenes. J. Am. Chem. Soc. 146, 12691–12701 (2024).
Du, J., Skubi, K. L., Schultz, D. M. & Yoon, T. P. A dual-catalysis approach to enantioselective [2 + 2] photocycloadditions using visible light. Science 344, 392–396 (2014).
Jiang, Y., Wang, C., Rogers, C. R., Kodaimati, M. S. & Weiss, E. A. Regio- and diastereoselective intermolecular [2 + 2] cycloadditions photocatalysed by quantum dots. Nat. Chem. 11, 1034–1040 (2019).
Sherbrook, E. M., Jung, H., Cho, D., Baik, M.-H. & Yoon, T. P. Brønsted acid catalysis of photosensitized cycloadditions. Chem. Sci. 11, 856–861 (2020).
Sherbrook, E. M. et al. Chiral Brønsted acid-controlled intermolecular asymmetric [2 + 2] photocycloadditions. Nat. Commun. 12, 5735 (2021).
Bower, J. F., Kim, I. S., Patman, R. L. & Krische, M. J. Catalytic carbonyl addition through transfer hydrogenation: a departure from preformed organometallic reagents. Angew. Chem. Int. Ed. 48, 34–46 (2009).
Holmes, M., Schwartz, L. A. & Krische, M. J. Intermolecular metal-catalyzed reductive coupling of dienes, allenes, and enynes with carbonyl compounds and imines. Chem. Rev. 118, 6026–6052 (2018).
Doerksen, R. S. et al. Ruthenium-catalyzed cycloadditions to form five-, six-, and seven-membered rings. Chem. Rev. 121, 4045–4083 (2021).
Gandeepan, P. & Cheng, C.-H. Cobalt catalysis involving π components in organic synthesis. Acc. Chem. Res. 48, 1194–1206 (2015).
Kong, J.-R., Ngai, M.-Y. & Krische, M. J. Highly enantioselective direct reductive coupling of conjugated alkynes and α-ketoesters via rhodium-catalyzed asymmetric hydrogenation. J. Am. Chem. Soc. 128, 718–719 (2006).
Zbieg, J. R., Yamaguchi, E., McInturff, E. L. & Krische, M. J. Enantioselective C–H crotylation of primary alcohols via hydrohydroxyalkylation of butadiene. Science 336, 324–327 (2012).
Saxena, A., Choi, B. & Lam, H. W. Enantioselective copper-catalyzed reductive coupling of alkenylazaarenes with ketones. J. Am. Chem. Soc. 134, 8428–8431 (2012).
Wei, C.-H., Mannathan, S. & Cheng, C.-H. Regio- and enantioselective cobalt-catalyzed reductive [3 + 2] cycloaddition reaction of alkynes with cyclic enones: a route to bicyclic tertiary alcohols. Angew. Chem. Int. Ed. 51, 10592–10595 (2012).
Bishop, H. D., Zhao, Q. & Uyeda, C. Catalytic asymmetric synthesis of zinc metallacycles. J. Am. Chem. Soc. 145, 20152–20157 (2023).
Liu, R. Y., Yang, Y. & Buchwald, S. L. Regiodivergent and diastereoselective CuH-catalyzed allylation of imines with terminal allenes. Angew. Chem. Int. Ed. 55, 14077–14080 (2016).
Köpfer, A., Sam, B., Breit, B. & Krische, M. J. Regiodivergent reductive coupling of 2-substituted dienes to formaldehyde employing ruthenium or nickel catalyst: hydrohydroxymethylation via transfer hydrogenation. Chem. Sci. 4, 1876–1880 (2013).
Geary, L. M., Woo, S. K., Leung, J. C. & Krische, M. J. Diastereo- and enantioselective iridium-catalyzed carbonyl propargylation from the alcohol or aldehyde oxidation level: 1,3-enynes as allenylmetal equivalents. Angew. Chem. Int. Ed. 51, 2972–2976 (2012).
Sam, B., Breit, B. & Krische, M. J. Paraformaldehyde and methanol as C1 feedstocks in metal-catalyzed C−C couplings of π-unsaturated reactants: beyond hydroformylation. Angew. Chem. Int. Ed. 54, 3267–3274 (2015).
Oda, S., Sam, B. & Krische, M. J. Hydroaminomethylation beyond carbonylation: allene–imine reductive coupling by ruthenium-catalyzed transfer hydrogenation. Angew. Chem. Int. Ed. 54, 8525–8528 (2015).
Ortiz, E., Evarts, M. M., Strong, Z. H., Shezaf, J. Z. & Krische, M. J. Ruthenium-catalyzed C−C coupling of terminal alkynes with primary alcohols or aldehydes: α,β-acetylenic ketones (ynones) via oxidative alkynylation. Angew. Chem. Int. Ed. 62, e202303345 (2023).
Cui, K., Li, Y.-L., Li, G. & Xia, J.-B. Regio- and stereoselective reductive coupling of alkynes and crotononitrile. J. Am. Chem. Soc. 144, 23001–23009 (2022).
Gu, Z.-Y., Li, W.-D., Li, Y.-L., Cui, K. & Xia, J.-B. Selective reductive coupling of vinyl azaarenes and alkynes via photoredox cobalt dual catalysis. Angew. Chem. Int. Ed. 62, e202213281 (2023).
Cheng, X. et al. Recent applications of homogeneous catalysis in electrochemical organic synthesis. CCS Chem 4, 1120–1152 (2022).
Malapit, C. A. et al. Advances on the merger of electrochemistry and transition metal catalysis for organic synthesis. Chem. Rev. 122, 3180–3218 (2022).
Leech, M. C. & Lam, K. A practical guide to electrosynthesis. Nat. Rev. Chem. 6, 275–286 (2022).
Yan, M., Kawamata, Y. & Baran, P. S. Synthetic organic electrochemical methods since 2000: on the verge of a renaissance. Chem. Rev. 117, 13230–13319 (2017).
Li, P., Wang, Y., Zhao, H. & Qiu, Y. Electroreductive cross-coupling reactions: carboxylation, deuteration, and alkylation. Acc. Chem. Res. 58, 113–129 (2025).
Zhu, C., Chen, H., Yue, H. & Rueping, M. Electrochemical chemo- and regioselective arylalkylation, dialkylation and hydro(deutero)alkylation of 1,3-enynes. Nat. Synth. 2, 1068–1081 (2023).
Montgomery, C. L., Amtawong, J., Jordan, A. M., Kurtz, D. A. & Dempsey, J. L. Proton transfer kinetics of transition metal hydride complexes and implications for fuel-forming reactions. Chem. Soc. Rev. 52, 7137–7169 (2023).
Gnaim, S. et al. Cobalt-electrocatalytic HAT for functionalization of unsaturated C–C bonds. Nature 605, 687–695 (2022).
Derosa, J., Garrido-Barros, P., Li, M. & Peters, J. C. Use of a PCET mediator enables a Ni-HER electrocatalyst to act as a hydride delivery agent. J. Am. Chem. Soc. 144, 20118–20125 (2022).
Wang, T., He, F., Jiang, W. & Liu, J. Electrohydrogenation of nitriles with amines by cobalt catalysis. Angew. Chem. Int. Ed. 63, e202316140 (2024).
Gao, S., Wang, C., Yang, J. & Zhang, J. Cobalt-catalyzed enantioselective intramolecular reductive cyclization via electrochemistry. Nat. Commun. 14, 1301 (2023).
Ai, W., Zhong, R., Liu, X. & Liu, Q. Hydride transfer reactions catalyzed by cobalt complexes. Chem. Rev. 119, 2876–2953 (2019).
Zhong, R., Wei, Z., Zhang, W., Liu, S. & Liu, Q. A practical and stereoselective in situ NHC–cobalt catalytic system for hydrogenation of ketones and aldehydes. Chem 5, 1552–1566 (2019).
Liu, X., Rong, X., Liu, S., Lan, Y. & Liu, Q. Cobalt-catalyzed desymmetric isomerization of exocyclic olefins. J. Am. Chem. Soc. 143, 20633–20639 (2021).
Liu, B. et al. Ligand-controlled stereoselective synthesis of 2-deoxy-β-C-glycosides by cobalt catalysis. Angew. Chem. Int. Ed. 62, e202218544 (2023).
Rong, X., Yang, J., Liu, S., Lan, Y. & Liu, Q. Remote stereocontrol of all-carbon quaternary centers via cobalt-catalyzed asymmetric olefin isomerization. CCS Chem 5, 1293–1300 (2023).
Akutagawa, T., Takeda, S., Hasegawa, T. & Nakamura, T. Proton transfer and a dielectric phase transition in the molecular conductor (HDABCO+)2(TCNQ)3. J. Am. Chem. Soc. 126, 291–294 (2004).
Ess, D. H. & Houk, K. N. Distortion/interaction energy control of 1,3-dipolar cycloaddition reactivity. J. Am. Chem. Soc. 129, 10646–10647 (2007).
Ess, D. H. & Houk, K. N. Theory of 1,3-dipolar cycloadditions: distortion/interaction and frontier molecular orbital models. J. Am. Chem. Soc. 130, 10187–10198 (2008).
Acknowledgements
We thank S. Liu for helpful discussions. Financial support from the National Natural Science Foundation of China (22225103) and the Tsinghua University Initiative Scientific Research Program are greatly appreciated.
Author information
Authors and Affiliations
Contributions
J.Y. and Q.L. conceptualized the study. J.Y. contributed to the methodology. J.Y. and Q.W. did the investigation. H.L and Q.W. performed the DFT calculations. Q.L. was involved in funding acquisition, did the project administration and supervised the work. J.Y. and Q.L wrote the original draft. J.Y. and Q.L contributed to the writing, review and editing of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Jolene Reid, Wen-Jing Xiao, Junliang Zhang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Thomas West, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Experimental details, Supplementary Figs. 1–56 and Tables 1–26.
Supplementary Data 1
Crystallographic data for compound rac-38; CCDC reference 2401812.
Supplementary Data 2
Crystallographic data for compound 33-cis; CCDC reference 2401813.
Supplementary Data 3
Crystallographic data for compound 38; CCDC reference 2401814.
Supplementary Data 4
Crystallographic data for compound 33-cis (S,R); CCDC reference 2401815.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, J., Li, H., Wang, Q. et al. Diastereodivergent construction of disubstituted cyclobutanes via electrochemical cobalt-catalysed reductive coupling. Nat. Synth (2026). https://doi.org/10.1038/s44160-025-00947-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-025-00947-9