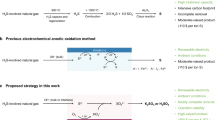
Abstract
Sulfur dioxide (SO2) is a widespread industrial pollutant from fossil fuel combustion and metal smelting that causes serious environmental and health concerns. Converting SO2 into valuable chemicals provides a sustainable solution for emission mitigation and resource use. Here we show a paired electrolysis strategy that directly transforms SO2 into cyclic sulfite esters—high-value organosulfur intermediates widely used in organic synthesis and as precursors for functional materials—under mild conditions. SO2 is reduced at the cathode to elemental sulfur, which then undergoes anodic oxidation and couples with alcohols to form five-membered, six-membered and seven-membered cyclic sulfite esters. Mechanistic studies reveal key sulfur-containing intermediates and elucidate the critical redox pathways. This method efficiently converts even low concentrations of SO2, including simulated industrial flue gas, demonstrating practical applicability. The strategy provides a versatile and environmentally friendly platform for green organosulfur synthesis and pollutant valorization, opening new avenues for sustainable chemical manufacturing.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All experimental and spectroscopic data are included in Supplementary Information. Source data are provided with this paper.
Change history
04 February 2026
A Correction to this paper has been published: https://doi.org/10.1038/s44160-026-01011-w
References
Zeng, W. & Qiu, Y. Electrochemical conversion of organic compounds and inorganic small molecules. Sci. China Chem. 67, 3223–3246 (2024).
Sun, G.-Q., Liao, L.-L., Ran, C.-K., Ye, J.-H. & Yu, D.-G. Recent advances in electrochemical carboxylation with CO2. Acc. Chem. Res. 57, 2728–2745 (2024).
Ran, C.-K. et al. Transition-metal-free synthesis of thiazolidin-2-ones and 1,3-thiazinan-2-ones from arylamines, elemental sulfur and CO2. Green Chem. 23, 274–279 (2021).
Katzenstein, A. W. Sulfur dioxide emissions. Science 228, 390–391 (1985).
Grennfelt, P. et al. Acid rain and air pollution: 50 years of progress in environmental science and policy. Ambio 49, 849–864 (2020).
Dubey, S. Acid rain-the major cause of pollution: its causes, effects and solution. Int. J. Sci. Eng. Technol. 2, 772–775 (2013).
Abelson, P. H. Air pollution and acid rain. Science 230, 617 (1985).
Chen, T.-M., Kuschner, W. G., Gokhale, J. & Shofer, S. Outdoor air pollution: nitrogen dioxide, sulfur dioxide, and carbon monoxide health effects. Am. J. Med. Sci. 333, 249–256 (2007).
Khan, M. I., Amin, A., Khan, M. T., Jabeen, H. & Chaudhry, S. R. Unveiling the hidden hazards of smog: health implications and antibiotic resistance in perspective. Aerobiologia 40, 353–372 (2024).
Ng, K. H., Lai, S. Y., Jamaludin, N. F. M. & Mohamed, A. R. A review on dry-based and wet-based catalytic sulphur dioxide (SO2) reduction technologies. J. Hazard. Mater. 423, 127061 (2022).
Li, X., Han, J., Liu, Y., Dou, Z. & Zhang, T. -a Summary of research progress on industrial flue gas desulfurization technology. Sep. Purif. Technol. 281, 119849 (2022).
Zheng, D., Yu, J. & Wu, J. Generation of sulfonyl radicals from aryldiazonium tetrafluoroborates and sulfur dioxide: the synthesis of 3-sulfonated coumarins. Angew. Chem. Int. Ed. 55, 11925–11929 (2016).
Liu, F. et al. Merging [2+ 2] cycloaddition with radical 1,4-addition: metal-free access to functionalized cyclobuta[a]naphthalen-4-ols. Angew. Chem. Int. Ed. 56, 15570–15574 (2017).
Ye, S., Zhou, K., Rojsitthisak, P. & Wu, J. Metal-free insertion of sulfur dioxide with aryl iodides under ultraviolet irradiation: direct access to sulfonated cyclic compounds. Org. Chem. Front. 7, 14–18 (2020).
Deeming, A. S., Russell, C. J. & Willis, M. C. Palladium(II)-catalyzed synthesis of sulfinates from boronic acids and DABSO: a redox-neutral, phosphine-free transformation. Angew. Chem. Int. Ed. 55, 747–750 (2016).
He, F.-S. et al. Photoredox-catalyzed α-sulfonylation of ketones from sulfur dioxide and thianthrenium salts. Org. Lett. 24, 2955–2960 (2022).
de, A. et al. Electrochemical multicomponent synthesis of alkyl alkenesulfonates using styrenes, SO2 and alcohols. Chem. Eur. J. 30, e202400557 (2024).
Blum, S. P., Schollmeyer, D., Turks, M. & Waldvogel, S. R. Metal-and reagent-free electrochemical synthesis of alkyl arylsulfonates in a multi-component reaction. Chem. Eur. J. 26, 8358–8362 (2020).
Lou, T. S. B., Bagley, S. W. & Willis, M. C. Cyclic alkenylsulfonyl fluorides: palladium-catalyzed synthesis and functionalization of compact multifunctional reagents. Angew. Chem. 131, 19035–19039 (2019).
Pedersen, P. S., Blakemore, D. C., Chinigo, G. M., Knauber, T. & MacMillan, D. W. C. One-pot synthesis of sulfonamides from unactivated acids and amines via aromatic decarboxylative halosulfonylation. J. Am. Chem. Soc. 145, 21189–21196 (2023).
Lou, T. S. B. et al. Scalable, chemoselective nickel electrocatalytic sulfinylation of aryl halides with SO2. Angew. Chem. Int. Ed. 61, e202208080 (2022).
Jia, X., Kramer, S., Skrydstrup, T. & Lian, Z. Design and applications of a SO2 surrogate in palladium-catalyzed direct aminosulfonylation between aryl iodides and amines. Angew. Chem. Int. Ed. 60, 7353–7359 (2021).
Blum, S. P., Schäffer, L., Schollmeyer, D. & Waldvogel, S. R. Electrochemical synthesis of sulfamides. Chem. Commun. 57, 4775–4778 (2021).
Blum, S. P., Karakaya, T., Schollmeyer, D., Klapars, A. & Waldvogel, S. R. Metal-free electrochemical synthesis of sulfonamides directly from (hetero)arenes, SO2, and amines. Angew. Chem. Int. Ed. 60, 5056–5062 (2021).
Zheng, D., An, Y., Li, Z. & Wu, J. Metal-free aminosulfonylation of aryldiazonium tetrafluoroborates with DABCO⋅(SO2)2 and hydrazines. Angew. Chem. Int. Ed. 53, 2451–2454 (2014).
Nguyen, B., Emmett, E. J. & Willis, M. C. Palladium-catalyzed aminosulfonylation of aryl halides. J. Am. Chem. Soc. 132, 16372–16373 (2010).
Madec, L. et al. Effect of sulfate electrolyte additives on LiNi1/3Mn1/3Co1/3O2/graphite pouch cell lifetime: correlation between XPS surface studies and electrochemical rest results. J. Phys. Chem. C 118, 29608–29622 (2014).
Shustov, G. V., Kachanov, A. V., Korneev, V. A., Kostyanovsky, R. G. & Rauk, A. Chiroptical properties of C2-symmetric N-haloaziridines. Chiral rules for the N-haloaziridine chromophore. J. Am. Chem. Soc. 115, 10267–10274 (1993).
Marti, C. & Carreira, E. M. Total synthesis of (−)-spirotryprostatin B: synthesis and related studies. J. Am. Chem. Soc. 127, 11505–11515 (2005).
Hu, J. et al. Adduct-catalyzed tandem electro-thermal synthesis of organophosphorus(III) compounds from white phosphorus. Natl Sci. Rev. 12, nwaf008 (2025).
Siu, J. C., Fu, N. & Lin, S. Catalyzing electrosynthesis: a homogeneous electrocatalytic approach to reaction discovery. Acc. Chem. Res. 53, 547–560 (2020).
Xiong, P. & Xu, H.-C. Chemistry with electrochemically generated N-centered radicals. Acc. Chem. Res. 52, 3339–3350 (2019).
Zhang, B. et al. Complex molecule synthesis by electrocatalytic decarboxylative cross-coupling. Nature 623, 745–751 (2023).
Leech, M. C. & Lam, K. Electrosynthesis using carboxylic acid derivatives: new tricks for old reactions. Acc. Chem. Res. 53, 121–134 (2020).
Frontana-Uribe, B. A., Little, R. D., Ibanez, J. G., Palma, A. & Vasquez-Medrano, R. Organic electrosynthesis: a promising green methodology in organic chemistry. Green Chem. 12, 2099–2119 (2010).
Hioki, Y. et al. Overcoming the limitations of Kolbe coupling with waveform-controlled electrosynthesis. Science 380, 81–87 (2023).
Meyer, T. H., Choi, I., Tian, C. & Ackermann, L. Powering the future: how can electrochemistry make a difference in organic synthesis?. Chem 6, 2484–2496 (2020).
Leech, M. C. & Lam, K. A practical guide to electrosynthesis. Nat. Rev. Chem. 6, 275–286 (2022).
Yuan, Y., Yang, J. & Lei, A. Recent advances in electrochemical oxidative cross-coupling with hydrogen evolution involving radicals. Chem. Soc. Rev. 50, 10058–10086 (2021).
Wang, H., Gao, X., Lv, Z., Abdelilah, T. & Lei, A. Recent advances in oxidative R1-H/R2-H cross-coupling with hydrogen evolution via photo-/electrochemistry. Chem. Rev. 119, 6769–6787 (2019).
Huang, C., Tao, Y., Cao, X., Zhou, C. & Lu, Q. Asymmetric paired electrocatalysis: enantioselective olefin–sulfonylimine coupling. J. Am. Chem. Soc. 146, 1984–1991 (2024).
Wood, D. & Lin, S. Deuterodehalogenation under net reductive or redox-neutral conditions enabled by paired electrolysis. Angew. Chem. 135, e202218858 (2023).
Nie, L. et al. Linear paired electrolysis enables redox-neutral (3+ 2) annulation of benzofuran with vinyldiazo compounds. J. Am. Chem. Soc. 146, 31330–31338 (2024).
Dong, X., Roeckl, J. L., Waldvogel, S. R. & Morandi, B. Merging shuttle reactions and paired electrolysis for reversible vicinal dihalogenations. Science 371, 507–514 (2021).
Llorente, M. J., Nguyen, B. H., Kubiak, C. P. & Moeller, K. D. Paired electrolysis in the simultaneous production of synthetic intermediates and substrates. J. Am. Chem. Soc. 138, 15110–15113 (2016).
Liu, D. et al. Paired electrolysis-enabled nickel-catalyzed enantioselective reductive cross-coupling between α-chloroesters and aryl bromides. Nat. Commun. 13, 7318 (2022).
Hilt, G. Recent advances in paired electrolysis and their application in organic electrosynthesis. Curr. Opin. Electrochem. 43, 101425 (2024).
Chen, C. et al. Copper-catalyzed oxidative trifluoromethylthiolation of aryl boronic acids with TMSCF3 and elemental sulfur. Angew. Chem. Int. Ed. 51, 2492–2495 (2012).
Deng, H., Li, Z., Ke, F. & Zhou, X. Cu-catalyzed three-component synthesis of substituted benzothiazoles in water. Chem. Eur. J. 18, 4840–4843 (2012).
Adam, W., Bargon, R. M. & Schenk, W. A. Direct episulfidation of alkenes and allenes with elemental sulfur and thiiranes as sulfur sources, catalyzed by molybdenum oxo complexes. J. Am. Chem. Soc. 125, 3871–3876 (2003).
Nguyen, T. B., Ermolenko, L. & Al-Mourabit, A. Iron sulfide catalyzed redox/condensation cascade reaction between 2-amino/hydroxy nitrobenzenes and activated methyl groups: a straightforward atom economical approach to 2-hetaryl-benzimidazoles and -benzoxazoles. J. Am. Chem. Soc. 135, 118–121 (2013).
Wang, M., Dai, Z. & Jiang, X. Design and application of α-ketothioesters as 1, 2-dicarbonyl-forming reagents. Nat. Commun. 10, 2661 (2019).
Wang, Z., Wang, Y., Zhang, W.-X., Hou, Z. & Xi, Z. Efficient one-pot synthesis of 2, 3-dihydropyrimidinthiones via multicomponent coupling of terminal alkynes, elemental sulfur, and carbodiimides. J. Am. Chem. Soc. 131, 15108–15109 (2009).
Nguyen, T. B., Ermolenko, L., Retailleau, P. & Al-Mourabit, A. Elemental sulfur disproportionation in the redox condensation reaction between o-halonitrobenzenes and benzylamines. Angew. Chem. 126, 14028–14032 (2014).
Jiang, Y. et al. A general and efficient approach to aryl thiols: CuI-catalyzed coupling of aryl iodides with sulfur and subsequent reduction. Org. Lett. 11, 5250–5253 (2009).
Chen, C., Chu, L. & Qing, F.-L. Metal-Free oxidative trifluoromethylthiolation of terminal alkynes with CF3SiMe3 and elemental sulfur. J. Am. Chem. Soc. 134, 12454–12457 (2012).
Huang, Y., Yu, Y., Hu, R. & Tang, B. Z. Multicomponent polymerizations of elemental sulfur, CH2Cl2, and aromatic amines toward chemically recyclable functional aromatic polythioureas. J. Am. Chem. Soc. 146, 14685–14696 (2024).
Jiang, P., Che, X., Liao, Y., Huang, H. & Deng, G.-J. Three-component 2-aryl substituted benzothiophene formation under transition-metal free conditions. RSC Adv. 6, 41751–41754 (2016).
Tang, H. et al. Direct synthesis of thioesters from feedstock chemicals and elemental sulfur. J. Am. Chem. Soc. 145, 5846–5854 (2023).
Chen, J. et al. Four-component approach to N-substituted phenothiazines under transition-metal-free conditions. Org. Lett. 17, 5870–5873 (2015).
Liu, G., Xu, S., Yue, Y., Su, C. & Song, W. Synthesis of thioesters using an electrochemical three-component reaction involving elemental sulfur. Chem. Commun. 60, 6154–6157 (2024).
Chung, W. J. et al. The use of elemental sulfur as an alternative feedstock for polymeric materials. Nat. Chem. 5, 518–524 (2013).
Zeng, J.-L., Niu, L., Wang, Z., Wei, R. & Cahard, D. Monofluoromethylation of cyclic sulfamidates and sulfates with α-fluorocarbanions of fluorobis(phenylsulfonyl)methane and ethyl 2-fluoroacetoacetate. Eur. J. Org. Chem. 26, e202300651 (2023).
Wilckens, K., Uhlemann, M. & Czekelius, C. Gold-catalyzed endo-cyclizations of 1, 4-diynes to seven-membered ring heterocycles. Chem. Eur. J. 15, 13323–13326 (2009).
Carlson, W. W. & Cretcher, L. H. Hydroxyalkylation with cyclic alkylene esters. I. synthesis of hydroxyethylapocupreine. J. Am. Chem. Soc. 69, 1952–1956 (1947).
Acknowledgements
This work was supported by the National Key R&D Program of China (grant nos. 2022YFA1505100 H.Y., 2021YFA1500104 A.L.), the National Natural Science Foundation of China (grant nos. 22031008 A.L., 22522112 H.Y.), the Hubei Natural Science Foundation (grant no. 2024AFB761 H.Z.), Hubei Technological Innovation Program Funding (grant no. 2025BAB025 H.Y.) and the Science Foundation of Wuhan ((grant no. 2020010601012192 A.L.) and the National Natural Science Foundation of China (grant no. 22201222). We thank the support of the Opening Foundation of Xi’an Modern Chemistry Research Institute (grant no. 204-J-2023-2325). Z.W. thanks the support from the National Key R&D Program of China (grant no. 2023YFF0723100) and the National Natural Science Foundation of China (grant no. 22374110). W.L. gratefully acknowledges the support from the National Natural Science Foundation of China (grant no. 212200007). We also thank the support of the Opening Foundation of Xi’an Modern Chemistry Research Institute (grant number 204-J-2023-2325).
Author information
Authors and Affiliations
Contributions
A.L. supervised the project. Jingcheng H., Jiayu H., Y.W., Z.W., H.Y. and A.L. conceived the idea and designed the experiments. Jingcheng H., Jiayu H., Y.W., C.Z. and H.Z. carried out all the experimental work. Jingcheng H., Jiayu H. and W.L. contributed to data analysis and article editing. Z.W., W.L., H.Y. and A.L. cowrote the paper. All authors discussed the results and assisted during the article preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Da-Gang Yu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alexandra Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–21, Tables 1–7 and all experimental and spectroscopic data.
Source data
Source Data Fig. 2 (download XLSX )
Optimization data (Fig. 2a); X-ray photoelectron spectroscopy test data (Fig. 2b); X-ray diffraction test data (Fig. 2c) and cyclic voltammetry test data (Fig. 2d).
Source Data Fig. 4 (download XLSX )
Cyclic voltammetry test data (Fig. 4a); cyclic voltammetry test data (Fig. 4b); cyclic voltammetry test data (Fig. 4c) and monitoring of model reaction data (Fig. 4e).
Source Data Fig. 5 (download XLSX )
In situ EC-MS analysis data (Fig. 5a).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, J., Hu, J., Wang, Y. et al. Parallel paired electrolysis of industrial exhaust SO2 and diols for value-added sulfite esters synthesis. Nat. Synth (2026). https://doi.org/10.1038/s44160-025-00968-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-025-00968-4