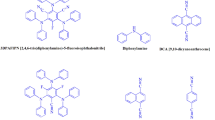
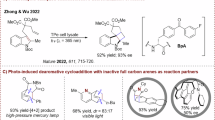
Abstract
The skeletal remodelling of benzene has been widely acknowledged as an attractive yet challenging approach for utilizing the extensive array of aromatic compounds. A primary challenge lies not only in overcoming the high activation barrier associated with the dearomatization step but also in achieving site-selective skeletal modifications within a single operational sequence. Here we present a photothermal cascade activation mode that unlocks the photoactivation reactivity of the transiently generated light-absorbing intermediate formed during interrupted Fischer indolization. Complementary to arene ring expansion methodologies, this protocol offers a practical ring contraction approach for the modular synthesis of fused pyridines with good functional group tolerance and predictable regioselectivity. This strategy is expected to broaden the chemical landscape for the design of innovative photochemical reactions.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Crystallographic data for 3n reported in this article have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition number 2385332. Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. All other data supporting the findings of this study are available within this article and its Supplementary Information. The experimental procedures and characterization of all new compounds are provided in the Supplementary Information.
References
Hui, C., Craggs, L. & Antonchick, A. P. Ring contraction in synthesis of functionalized carbocycles. Chem. Soc. Rev. 51, 8652–8675 (2022).
Hu, S., Shima, T. & Hou, Z. Carbon–carbon bond cleavage and rearrangement of benzene by a trinuclear titanium hydride. Nature 512, 413–415 (2014).
Jakoobi, M. & Sergeev, A. G. Transition-metal-mediated cleavage of C−C bonds in aromatic rings. Chem. Asian J. 14, 2181–2192 (2019).
Qiu, X. et al. Cleaving arene rings for acyclic alkenylnitrile synthesis. Nature 597, 64–69 (2021).
Hutskalova, V. & Sparr, C. Aromatic ring-opening metathesis. Nature 638, 697–703 (2025).
Tsien, J., Hu, C., Merchant, R. R. & Qin, T. Three-dimensional saturated C(sp3)-rich bioisosteres for benzene. Nat. Rev. Chem. 8, 605–627 (2024).
Patel, S. C. & Burns, N. Z. Conversion of aryl azides to aminopyridines. J. Am. Chem. Soc. 144, 17797–17802 (2022).
Pearson, T. J. et al. Aromatic nitrogen scanning by ipso-selective nitrene internalization. Science 381, 1474–1479 (2023).
Woo, J., Stein, C., Christian, A. H. & Levin, M. D. Carbon-to-nitrogen single-atom transmutation of azaarenes. Nature 623, 77–82 (2023).
Li, L. et al. Diversification of naphthol skeletons triggered by aminative dearomatization. Org. Lett. 26, 4910–4915 (2024).
Liu, W. et al. Rare-earth metal-enabled ring-opening metathesis of benzene. J. Am. Chem. Soc. 147, 1300–1306 (2025).
Benitez, V. M., Grau, J. M., Yori, J. C., Pieck, C. L. & Vera, C. R. Hydroisomerization of benzene-containing paraffinic feedstocks over Pt/WO3−ZrO2 catalysts. Energy Fuels 20, 1791–1798 (2006).
Slanina, T. et al. Impact of excited-state antiaromaticity relief in a fundamental benzene photoreaction leading to substituted bicyclo[3.1.0]hexenes. J. Am. Chem. Soc. 142, 10942–10954 (2020).
Guo, Y.-Y. et al. Unlocking mild-condition benzene ring contraction using nonheme diiron N-oxygenase. Chem. Sci. 14, 11907–11913 (2023).
Kang, X. et al. Mechanistic insights into ring cleavage and contraction of benzene over a titanium hydride cluster. J. Am. Chem. Soc. 138, 11550–11559 (2016).
Prier, C. K., Rankic, D. A. & MacMillan, D. W. C. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013).
Yu, X.-Y., Chen, J.-R. & Xiao, W.-J. Visible light-driven radical-mediated C–C bond cleavage/functionalization in organic synthesis. Chem. Rev. 121, 506–561 (2021).
Kärkäs, M. D., Porco, J. A. Jr. & Stephenson, C. R. J. Photochemical approaches to complex chemotypes: applications in natural product synthesis. Chem. Rev. 116, 9683–9747 (2016).
Buzzetti, L., Crisenza, G. E. M. & Melchiorre, P. Mechanistic studies in photocatalysis. Angew. Chem. Int. Ed. 58, 3730–3747 (2019).
Silvi, M., Arceo, E., Jurberg, I. D., Cassani, C. & Melchiorre, P. Enantioselective organocatalytic alkylation of aldehydes and enals driven by the direct photoexcitation of enamines. J. Am. Chem. Soc. 137, 6120–6123 (2015).
Silvi, M., Verrier, C., Rey, Y. P., Buzzetti, L. & Melchiorre, P. Visible-light excitation of iminium ions enables the enantioselective catalytic β-alkylation of enals. Nat. Chem. 9, 868–873 (2017).
Brenninger, C., Jolliffe, J. D. & Bach, T. Chromophore activation of α,β-unsaturated carbonyl compounds and its application to enantioselective photochemical reactions. Angew. Chem. Int. Ed. 57, 14338–14349 (2018).
Bonilla, P., Rey, Y. P., Holden, C. M. & Melchiorre, P. Photo-organocatalytic enantioselective radical cascade reactions of unactivated olefins. Angew. Chem. Int. Ed. 57, 12819–12823 (2018).
Woźniak, Ł, Magagnano, G. & Melchiorre, P. Enantioselective photochemical organocascade catalysis. Angew. Chem. Int. Ed. 57, 1068–1072 (2018).
Huang, X. et al. Photoenzymatic enantioselective intermolecular radical hydroalkylation. Nature 584, 69–74 (2020).
Peng, Q. et al. Photochemical phosphorus-enabled scaffold remodeling of carboxylic acids. Science 385, 1471–1477 (2024).
Xu, Z. et al. Photoinduced formal [4 + 2] cycloaddition of two electron-deficient olefins and its application to the synthesis of lucidumone. Nat. Commun. 15, 9748 (2024).
Zhang, Y., Wang, J., Guo, Y., Liu, S. & Shen, X. Carbonyl olefin metathesis and dehydrogenative cyclization of aromatic ketones and gem-difluoroalkenes. Angew. Chem. Int. Ed. 63, e202315269 (2024).
Yan, M., Lo, J. C., Edwards, J. T. & Baran, P. S. Radicals: reactive intermediates with translational potential. J. Am. Chem. Soc. 138, 12692–12714 (2016).
Fischer, E. & Jourdan, F. Ueber die hydrazine der brenztraubensäure. Ber. Dtsch. Chem. Ges. 16, 2241–2245 (1883).
Boal, B. W., Schammel, A. W. & Garg, N. K. An interrupted Fischer indolization approach toward fused indoline-containing natural products. Org. Lett. 11, 3458–3461 (2009).
Nazaré, M., Schneider, C., Lindenschmidt, A. & Will, D. W. A Flexible, palladium-catalyzed indole and azaindole synthesis by direct annulation of chloroanilines and chloroaminopyridines with ketones. Angew. Chem. Int. Ed. 43, 4526–4528 (2004).
Simmons, B. J. et al. Understanding and interrupting the Fischer azaindolization reaction. J. Am. Chem. Soc. 139, 14833–14836 (2017).
Zhan, F. & Liang, G. Formation of enehydrazine intermediates through coupling of phenylhydrazines with vinyl halides: entry into the Fischer indole synthesis. Angew. Chem. Int. Ed. 52, 1266–1269 (2013).
Mo, Y., Zhao, J., Chen, W. & Wang, Q. Recent advance of the application of interrupted Fischer indolization toward bioactive indoline alkaloids. Res. Chem. Intermed. 41, 5869–5877 (2015).
Wagaw, S., Yang, B. H. & Buchwald, S. L. A palladium-catalyzed method for the preparation of indoles via the Fischer indole synthesis. J. Am. Chem. Soc. 121, 10251–10263 (1999).
Kulagowski, J. J., Moody, C. J. & Rees, C. W. Generation and rearrangement of 4aH-carbazoles. J. Chem. Soc. Chem. Commun. https://doi.org/10.1039/C39820000548 (1982).
Kulagowski, J. J., Moody, C. J. & Rees, C. W. Generation and rearrangement of 4aH-carbazoles. J. Chem. Soc. Perkin Trans. 1, 2725–2732 (1985).
Kulagowski, J. J., Mitchell, G., Moody, C. J. & Rees, C. W. Preparation and rearrangement of 6a-methyl-6aH-benzo[a]carbazole and 11b-methyl-11bH-benzo[c]carbazole. J. Chem. Soc. Chem. Commun. https://doi.org/10.1039/C39850000650 (1985).
Bhutani, P. et al. US. FDA approved drugs from 2015–June 2020: a perspective. J. Med. Chem. 64, 2339–2381 (2021).
Marshall, C. M., Federice, J. G., Bell, C. N., Cox, P. B. & Njardarson, J. T. An update on the nitrogen heterocycle compositions and properties of US FDA-approved pharmaceuticals (2013–2023). J. Med. Chem. 67, 11622–11655 (2024).
Dwivedi, A. R. et al. A decade of pyridine-containing heterocycles in US FDA approved drugs: a medicinal chemistry-based analysis. RSC Med. Chem. 16, 12–36 (2025).
Müller, S., Webber, M. J. & List, B. The catalytic asymmetric Fischer indolization. J. Am. Chem. Soc. 133, 18534–18537 (2011).
Huang, S., Kötzner, L., De, C. K. & List, B. Catalytic asymmetric dearomatizing redox cross coupling of ketones with aryl hydrazines giving 1,4-diketones. J. Am. Chem. Soc. 137, 3446–3449 (2015).
Hughes, D. L. & Zhao, D. Mechanistic studies of the Fischer indole reaction. J. Org. Chem. 58, 228–233 (1993).
Armesto, D., Martin, J. A. F., Pérez-Ossorio, R. & Horspool, W. M. A novel aza-di-π-methane rearrangement the photoreaction of 4,4-dimethyl-1,6,6-triphenyl-2-aza-hexa-2,5-diene. Tetrahedron Lett. 23, 2149–2152 (1982).
Giam, C. S. & Ambrozich, D. A new approach to the preparation of 1,6-and 1,7-naphthyidines. J. Chem. Soc. Chem. Commun. https://doi.org/10.1039/C39840000265 (1984).
Liu, S. & Cheng, X. Insertion of ammonia into alkenes to build aromatic N-heterocycles. Nat. Commun. 13, 425 (2022).
Wu, F.-P. et al. Ring expansion of indene by photoredox-enabled functionalized carbon-atom insertion. Nat. Catal. 7, 242–251 (2024).
Lyle, M. P. A. & Wilson, P. D. Synthesis of a new chiral nonracemic C2-symmetric 2,2′-bipyridyl ligand and its application in copper(I)-catalyzed enantioselective cyclopropanation reactions. Org. Lett. 6, 855–857 (2004).
Acknowledgements
We are grateful for financial support from the National Natural Science Foundation of China (grant numbers 22571130, 22071086 and 21801165 to A.H., and grant numbers 22201113 and 22371106 to J.-J.G.), the Science and Technology Major Program of Gansu Province of China (22ZD6FA006, 23ZDFA015) and the Fundamental Research Funds for the Central Universities (lzujbky-2020-ct01, lzujbky-2023-ct02). We thank Z. Zuo, Y. Li and Y.-T. Ding for helpful discussions and advice during the preparation of this paper. We also thank the Supercomputing Center of Lanzhou University for providing the computing time.
Author information
Authors and Affiliations
Contributions
J.-J.G. and A.H. designed and conceived the project. K.L., Y.Z., K.-H.L., Y.Y., J.S., H.G. and Y.T. conducted all the synthetic reactions. K.L., Y.Z., K.-H.L., Y.Y. and A.H. analysed and interpreted the experiment data. K.-H.L., Y.Y., J.-J.G. and A.H. designed and performed the mechanistic experiments. K.L. performed the high-resolution mass spectrometry experiments and analysed the data. J.-J.G. performed the DFT calculations. A.H. prepared the paper. K.-H.L., Y.Y., J.-J.G. and A.H. prepared the Supplementary Information.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Daniel Werz and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor Thomas West, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
–Supplementary Sections 1–9, Figs. 1–22 and Tables 1–4.
Supplementary Data 1
Crystallographic data for 3n. CCDC 2385332.
Supplementary Data 2 (download ZIP )
Source data for Supplementary Figs. S8–S11.
Supplementary Data 3 (download ZIP )
Raw NMR data for compounds 1b to 1s.
Supplementary Data 4 (download ZIP )
Raw NMR data for compounds 2 and 6 to 15.
Supplementary Data 5 (download ZIP )
Raw NMR data for compounds 3a to 3r.
Supplementary Data 6 (download ZIP )
Raw NMR data for compounds 3aa to 3aq.
Supplementary Data 7 (download ZIP )
Raw NMR data for compounds 3ar to 3bj.
Supplementary Data 8 (download ZIP )
Raw NMR data for compound 3bd (1).
Supplementary Data 9 (download ZIP )
Raw NMR data for compound 3bd (2).
Supplementary Data 10 (download ZIP )
Raw NMR data for compounds 3s to 3z.
Supplementary Data 11 (download ZIP )
Raw NMR data for compound 4 (1).
Supplementary Data 12 (download ZIP )
Raw NMR data for compound 4 (2).
Supplementary Data 13 (download ZIP )
Raw NMR data for compound 4HCl (1).
Supplementary Data 14 (download ZIP )
Raw NMR data for compound 4HCl (2).
Supplementary Data 15 (download ZIP )
Raw NMR data for compound 4HCl (3).
Supplementary Data 16 (download ZIP )
Raw NMR data for compound 10 (1).
Supplementary Data 17 (download ZIP )
Raw NMR data for compound 10 (2).
Supplementary Data 18 (download ZIP )
Raw NMR data for demethoxysitamaquine and onychine.
Supplementary Data 19 (download ZIP )
Raw NMR data for compounds in Figs. S12 to S17.
Supplementary Data 20 (download ZIP )
Raw NMR data for compounds S1 to S16.
Source data
Source Data Fig. 4 (download XLS )
Statistical source data for Fig. 4.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, K., Zeng, Y., Li, KH. et al. Photoinduced benzene ring contraction of arylhydrazines for the synthesis of fused pyridines. Nat. Synth (2026). https://doi.org/10.1038/s44160-025-00976-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-025-00976-4