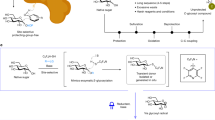
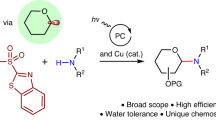
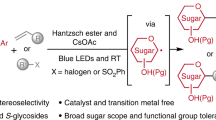
Abstract
C-heteroaryl glycosides, predominantly in unprotected forms, are common entities in bioactive molecules and have extensive applications in chemistry and biology. However, the chemical synthesis of these glycosides remains challenging owing to the lack of methods that directly leverage naturally occurring (native) sugars as substrates. Here we show that fully unprotected native sugars, capped as redox-active glycosyl sulfide donors, can be merged with N-heteroarenes in the presence of triethylamine and a photocatalyst under mild visible-light irradiation. The C–C coupling transformation proceeds with control over chemo-, site- and stereoselectivities and is compatible with a diverse range of N-heteroarenes bearing acidic and basic functional groups. The utility of this method is highlighted by the glycosylation of nucleosides, as well as by the direct coupling of d-mannose with pentoxifylline to generate a compound exhibiting glycogen-metabolism-inhibitory properties. In contrast to previously established mechanisms, the photocatalytic species is found to trigger the in situ generation of a thiyl radical that promotes hydrogen atom transfer to afford the target product, with triethylamine serving as a reductant through photoinduced charge-transfer complexation with the glycosyl sulfide.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Crystallographic data are available free of charge from the Cambridge Crystallographic Data Centre under deposition numbers CCDC-2449025 (46) and CCDC-2449026 (31). All other data are available in the Article or its Supplementary Information.
References
Bokor, E. et al. C-glycopyranosyl arenes and hetarenes: Synthetic methods and bioactivity focused on antidiabetic potential. Chem. Rev. 117, 1687–1764 (2017).
Yang, Y. & Yu, B. Recent advances in the chemical synthesis of C-glycosides. Chem. Rev. 117, 12281–12356 (2017).
Báti, G., He, J.-X., Pal, K. B. & Liu, X.-W. Stereo- and regioselective glycosylation with protection-less sugar derivatives: an alluring strategy to access glycans and natural products. Chem. Soc. Rev. 48, 4006–4018 (2019).
Štambaský, J., Hocek, M. & Kočovský, P. C-nucleosides: synthetic strategies and biological applications. Chem. Rev. 109, 6729–6764 (2009).
Villadsen, K., Martos-Maldonado, M. C., Jensen, K. J. & Thygesen, M. B. Chemoselective reactions for the synthesis of glycoconjugates from unprotected carbohydrates. ChemBioChem 18, 574–612 (2017).
Cao, X. et al. Carbohydrate-based drugs launched during 2000-2021. Acta. Pharm. Sin. B 12, 3783–3821 (2022).
Warren, T. K. et al. Therapeutic efficacy of the small molecule GS-5734 against ebola virus in rhesus monkeys. Nature 531, 381–385 (2016).
Kun, S. et al. New synthesis of 3-(β-d-glucopyranosyl)-5-substituted-1,2,4-triazoles, nanomolar inhibitors of glycogen phosphorylase. Eur. J. Med. Chem. 76, 567–579 (2014).
Ősz, E. et al. Preparation of 3,5-bis-(β-d-glycopyranosyl)-1,2,4-thiadiazoles from C-(β-d-glycopyranosyl)thioformamides. Tetrahedron. 57, 5429–5434 (2001).
Cohn, W. E. Pseudouridine, a carbon-carbon linked ribonucleoside in ribonucleic acids: isolation, structure, and chemical characteristics. J. Biol. Chem. 235, 1488–1498 (1960).
Wang, M. et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell. Res. 30, 269–271 (2020).
Chen, A., Yang, B., Zhou, Z. & Zhu, F. Recent advances in transition-metal-catalyzed glycosyl cross-coupling reactions. Chem Catal. 2, 3430–3470 (2022).
Xia, L., Fan, W., Yuan, X.-A. & Yu, S. Photoredox-catalyzed stereoselective synthesis of C-nucleoside analogues from glycosyl bromides and heteroarenes. ACS Catal. 11, 9397–9406 (2021).
Wang, Q. et al. Palladium-catalysed C−H glycosylation for synthesis of C-aryl glycosides. Nat. Catal. 2, 793–800 (2019).
Pöhlmann, J., Yuan, B., Purushothaman, R., Wu, J. & Ackermann, L. Photoinduced ruthenium-catalyzed meta-C–H glycosylation. ACS Catal. 15, 10542–10549 (2025).
Xu, S. et al. Generation and use of glycosyl radicals under acidic conditions: glycosyl sulfinates as precursors. Angew. Chem. Int. Ed. 62, e202218303 (2023).
Liu, Y., Wu, J., Zhou, L., Wei, C. & Chen, H. Synthesis of N-heteroaryl C-glycosides and polyhydroxylated alkanes with diaryl groups from unprotected sugars. ACS Omega. 9, 49618–49624 (2024).
Obradors, C. et al. Direct and catalytic C-glycosylation of arenes: Expeditious synthesis of the remdesivir nucleoside. Angew. Chem. Int. Ed. 61, e202114619 (2022).
Jiang, Y. et al. Direct radical functionalization of native sugars. Nature 631, 319–327 (2024).
Cao, S. et al. Regiodivergent functionalization of protected and unprotected carbohydrates using photoactive 4-tetrafluoropyridinylthio fragment as an adaptive activating group. Angew. Chem. Int. Ed. 63, e202412436 (2024).
Li, S. et al. Iridium photoredox-catalyzed stereoselective C-glycosylation with tetrafluoropyridin-4-yl thioglycosides: a facile synthesis of C-α/β-glucogallins and their antioxidant activity. ACS Catal. 14, 17727–17738 (2024).
Dewanji, A. et al. A general arene C–H functionalization strategy via electron donor–acceptor complex photoactivation. Nat. Chem. 15, 43–52 (2023).
Xie, D., Zeng, W., Yang, J. & Ma, X. Visible-light-promoted direct desulfurization of glycosyl thiols to access C-glycosides. Nat. Commun. 15, 9187 (2024).
Wang, Q. et al. Visible light activation enables desulfonylative cross-coupling of glycosyl sulfones. Nat. Synth. 1, 967–974 (2022).
Shang, W. & Niu, D. Radical pathway glycosylation empowered by bench-stable glycosyl donors. Acc. Chem. Res. 56, 2473–2488 (2023).
Xu, L.-Y., Fan, N.-L. & Hu, X.-G. Recent development in the synthesis of C-glycosides involving glycosyl radicals. Org. Biomol. Chem. 18, 5095–5109 (2020).
Chen, A., Cheng, G. & Zhu, F. Recent advances in stereoselective synthesis of non-classical glycosides. Tetrahedron Chem. 9, 100068 (2024).
Jiang, Y.-X. et al. Visible-light-driven synthesis of N-heteroaromatic carboxylic acids by thiolate-catalysed carboxylation of C(sp2)–H bonds using CO2. Nat. Synth. 3, 394–405 (2024).
Capaldo, L., Ravelli, D. & Fagnoni, M. Direct photocatalyzed hydrogen atom transfer (HAT) for aliphatic C–H bonds elaboration. Chem. Rev. 122, 1875–1924 (2022).
Yoshida, N. et al. Direct dehydrative pyridylthio-glycosidation of unprotected sugars in aqueous media using 2-chloro-1,3-dimethylimidazolinium chloride as a condensing agent. Chem. Asian J. 6, 1876–1885 (2011).
Bryden, M. A. & Zysman-Colman, E. Organic thermally activated delayed fluorescence (TADF) compounds used in photocatalysis. Chem. Soc. Rev. 50, 7587–7680 (2021).
Spell, M. L. et al. A visible-light-promoted O-glycosylation with a thioglycoside donor. Angew. Chem. Int. Ed. 55, 6515–6519 (2016).
Abe, H., Shuto, S. & Matsuda, A. Highly α- and β-selective radical C-glycosylation reactions using a controlling anomeric effect based on the conformational restriction strategy. A study on the conformation−anomeric effect−stereoselectivity relationship in anomeric radical reactions. J. Am. Chem. Soc. 123, 11870–11882 (2001).
Giese, B. & Dupuis, J. Diastereoselective syntheses of C-glycopyranosides. Angew. Chem. Int. Ed. Engl. 22, 622–623 (1983).
Proctor, R. S. J., Davis, H. J. & Phipps, R. J. Catalytic enantioselective minisci-type addition to heteroarenes. Science 360, 419–422 (2018).
Fuse, H. et al. Photocatalytic redox-neutral hydroxyalkylation of N-heteroaromatics with aldehydes. Chem. Sci. 11, 12206–12211 (2020).
Jin, J. & MacMillan, D. W. C. Alcohols as alkylating agents in heteroarene C–H functionalization. Nature 525, 87–90 (2015).
Shi, Q. et al. Single-electron oxidation-initiated enantioselective hydrosulfonylation of olefins enabled by photoenzymatic catalysis. J. Am. Chem. Soc. 146, 2748–2756 (2024).
Xuan, L. et al. Remote C(sp3)–H heteroarylation of N-fluorocarboxamides with quinoxalin-2(1H)-ones under visible-light-induced photocatalyst-free conditions. Green Chem. 24, 9475–9481 (2022).
Dong, J. et al. Formyl-selective deuteration of aldehydes with D2O via synergistic organic and photoredox catalysis. Chem. Sci. 11, 1026–1031 (2019).
Lahdenperä, A. S. K. et al. A chiral hydrogen atom abstraction catalyst for the enantioselective epimerization of meso-diols. Science 386, 42–49 (2024).
Guo, S. Y. et al. Visible light-induced chemoselective 1,2-diheteroarylation of alkenes. Nat. Commun. 15, 6102 (2024).
Zhang, C. et al. Visible-light-induced synthesis of heteroaryl C-glycosides via decarboxylative C–H glycosylation. Chem. Commun. 60, 5860–5863 (2024).
Adak, L. et al. Synthesis of aryl C-glycosides via iron-catalyzed cross coupling of halosugars: stereoselective anomeric arylation of glycosyl radicals. J. Am. Chem. Soc. 139, 10693–10701 (2017).
Chen, H., Fan, W., Yuan, X. A. & Yu, S. Site-selective remote C(sp3)–H heteroarylation of amides via organic photoredox catalysis. Nat. Commun. 10, 4743 (2019).
Le Saux, E., Georgiou, E., Dmitriev, I. A., Hartley, W. C. & Melchiorre, P. Photochemical organocatalytic functionalization of pyridines via pyridinyl radicals. J. Am. Chem. Soc. 145, 47–52 (2023).
Van Aerschot, A. A. et al. Antiviral activity of C-alkylated purine nucleosides obtained by cross-coupling with tetraalkyltin reagents. J. Med. Chem. 36, 2938–2942 (1993).
Chang, L. C. W. et al. 2,6-disubstituted and 2,6,8-trisubstituted purines as adenosine receptor antagonists. J. Med. Chem. 49, 2861–2867 (2006).
Gillingham, D., Geigle, S. & Anatole von Lilienfeld, O. Properties and reactivity of nucleic acids relevant to epigenomics, transcriptomics, and therapeutics. Chem. Soc. Rev. 45, 2637–2655 (2016).
Xie, R. et al. Late-stage guanine C8-H alkylation of nucleosides, nucleotides, and oligonucleotides via photo-mediated minisci reaction. Nat. Commun. 15, 2549 (2024).
Chrysina, E. D. et al. Kinetic and crystallographic studies on 2-(β-d-glucopyranosyl)-5-methyl-1, 3, 4-oxadiazole, -benzothiazole, and -benzimidazole, inhibitors of muscle glycogen phosphorylase b. Evidence for a new binding site. Protein Sci. 14, 873–888 (2005).
Acknowledgements
This research is supported by the Ministry of Education of Singapore Academic Research Fund Tier 2: A-8002999-00-00 (M.J.K.), the National Research Foundation, Prime Minister’s Office, Singapore under its Campus for Research Excellence and Technological Enterprise (CREATE) SM3 programme and A*STAR under its Manufacturing, Trade and Connectivity (MTC) Programmatic Fund: M25O1b0015 (M.J.K.). We thank I. I. Roslan (National University of Singapore) for the X-ray crystallographic measurements.
Author information
Authors and Affiliations
Contributions
M.J.K. and Q.-Y.Z. conceived the work. Q.-Y.Z. conducted the optimization and reaction scope studies. Q.-Y.Z., J.W. and S.L. conducted the mechanistic studies. D.Z.W.N. and W.L.L. conducted the biological studies under the direction of E.C.Y.C. M.J.K. directed the research. All authors contributed to the writing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Yong-Min Liang, Feng Zhu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Joel Cejas-Sánchez, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Sections 1–11, including Supplementary Figs. 1–13, Discussion and Tables 1–9.
Supplementary Crystal 1
Crystallographic data for compound 31, CCDC 2449026.
Supplementary Data 2
Crystallographic data for compound 46, CCDC 2449025.
Source Data Supplementary Fig. 11 (download XLSX )
The raw data of cyclic voltammograms of Supplementary Fig. 11.
Source Data Supplementary Fig. 13 (download ZIP )
The raw data of bioactivity investigation of Supplementary Fig. 13.
Source Data Supplementary Fig. 2 (download ZIP )
The raw data of UV–vis spectroscopy studies of Supplementary Fig. 2.
Source Data Supplementary Fig. 5 (download XLSX )
The raw data of the quenching ability of NEt3 of Supplementary Fig. 5.
Source Data Supplementary Fig. 7 (download XLSX )
The raw data of the quenching ability of isoquinoline of Supplementary Fig. 7.
Source Data Supplementary Fig. 4 (download XLSX )
The raw data of the quenching ability of the glycosyl donor of Supplementary Fig. 4.
Source Data Supplementary Fig. 6 (download XLSX )
The raw data of the quenching ability of NEt3 and thiol of Supplementary Fig. 6.
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data for Fig. 2b.
Source Data Fig. 3 (download XLSX )
Statistical source data for Fig. 3c.
Source Data Fig. 6 (download XLSX )
Statistical source data for Fig. 6b.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, QY., Ng, D.Z.W., Wu, J. et al. Photocatalytic coupling of unprotected sugars and N-heteroarenes. Nat. Synth (2026). https://doi.org/10.1038/s44160-025-00980-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-025-00980-8