Abstract
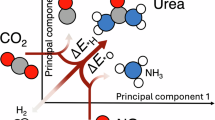
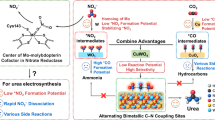
Electrosynthesis of urea at practical relevant current densities remains challenging due to competing side reactions, particularly at the elevated overpotentials required to sustain high currents. Here we propose a catalyst design strategy for selective urea production at practical current densities, emphasizing materials with low activity for competing CO2 reduction and hydrogen evolution, and high activity for nitrate activation under high overpotentials. We develop a cadmium-modified Fe2O3 (Cd–Fe2O3) catalyst composite, achieving a high urea partial current density of approximately 140 mA cm−2 at a modest cathodic potential of −0.5 V versus reversible hydrogen electrode, with an appreciable Faradaic efficiency of 52%. Through detailed kinetics analysis, in situ spectroscopic investigations and density functional theory calculations, we reveal that Cd incorporation into Fe2O3 substantially weakens *CO adsorption by altering the electronic structure and preserving oxidized Fe species. This modification suppresses undesired Volmer-type hydrogen adsorption while promoting *CO2NH2 intermediate protonation, enhancing urea formation. As a result, competing hydrogen evolution is effectively suppressed, and high urea selectivity is maintained at elevated current densities on Cd–Fe2O3.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available in Supplementary Information. Source data are provided with this paper.
References
Martín, A. J., Shinagawa, T. & Pérez-Ramírez, J. Electrocatalytic reduction of nitrogen: from Haber-Bosch to ammonia artificial leaf. Chem 5, 263–283 (2019).
Smith, C., Hill, A. K. & Torrente-Murciano, L. Current and future role of Haber-Bosch ammonia in a carbon-free energy landscape. Energy Environ. Sci. 13, 331–344 (2020).
Hattori, M., Iijima, S., Nakao, T., Hosono, H. & Hara, M. Solid solution for catalytic ammonia synthesis from nitrogen and hydrogen gases at 50 °C. Nat. Commun. 11, 2001 (2020).
Zhou, S. et al. Boron nitride nanotubes for ammonia synthesis: activation by filling transition metals. J. Am. Chem. Soc. 142, 308–317 (2020).
Tang, C. & Qiao, S. Z. How to explore ambient electrocatalytic nitrogen reduction reliably and insightfully. Chem. Soc. Rev. 48, 3166–3180 (2019).
Li, Y. et al. Sequential co-reduction of nitrate and carbon dioxide enables selective urea electrosynthesis. Nat. Commun. 15, 176 (2024).
Luo, Y. et al. Selective electrochemical synthesis of urea from nitrate and CO2 via relay catalysis on hybrid catalysts. Nat. Catal. 6, 939–948 (2023).
Li, J., Zhang, Y., Kuruvinashetti, K. & Kornienko, N. Construction of C–N bonds from small-molecule precursors through heterogeneous electrocatalysis. Nat. Rev. Chem. 6, 303–319 (2022).
Zhao, C. et al. Tailoring activation intermediates of CO2 initiates C–N coupling for highly selective urea electrosynthesis. J. Am. Chem. Soc. 147, 8871–8880 (2025).
Liu, Y. C. et al. Electrosynthesis of pure urea from pretreated flue gas in a proton-limited environment established in a porous solid-state electrolyte electrolyser. Nat. Nanotechnol. 20, 907–913 (2025).
Liu, X., Jiao, Y., Zheng, Y., Jaroniec, M. & Qiao, S. Z. Mechanism of C-N bonds formation in electrocatalytic urea production revealed by ab initio molecular dynamics simulation. Nat. Commun. 13, 5471 (2022).
Huang, D.-S. et al. Electrosynthesis of urea by using Fe2O3 nanoparticles encapsulated in a conductive metal–organic framework. Nat. Synth. 3, 1404–1413 (2024).
Lv, C. et al. Selective electrocatalytic synthesis of urea with nitrate and carbon dioxide. Nat. Sustain. 4, 868–876 (2021).
Hu, Q. et al. Pulsed co-electrolysis of carbon dioxide and nitrate for sustainable urea synthesis. Nat. Sustain 7, 442–451 (2024).
Xu, M. et al. Kinetically matched C–N coupling toward efficient urea electrosynthesis enabled on copper single-atom alloy. Nat. Commun. 14, 6994 (2023).
Qiu, X.-F., Huang, J.-R., Yu, C., Chen, X.-M. & Liao, P.-Q. Highly efficient electrosynthesis of urea from CO2 and nitrate by a metal–organic framework with dual active sites Angew. Chem. Int. Ed. 63, e202410625 (2024).
Geng, J. Ambient electrosynthesis of urea with nitrate and carbon dioxide over iron-based dual-sites. Angew. Chem. Int. Ed. 62, e202210958 (2023).
Cao, N. et al. Oxygen vacancies enhanced cooperative electrocatalytic reduction of carbon dioxide and nitrite ions to urea. J. Colloid Interface Sci. 577, 109–114 (2020).
Feng, Y. et al. Te-doped Pd nanocrystal for electrochemical urea production by efficiently coupling carbon dioxide reduction with nitrite reduction. Nano Lett. 20, 8282–8289 (2020).
Meng, N., Huang, Y., Liu, Y., Yu, Y. & Zhang, B. Electrosynthesis of urea from nitrite and CO2 over oxygen vacancy-rich ZnO porous nanosheets. Cell Rep. Phys. Sci. 2, 100378 (2021).
Yuan, M. et al. Unveiling electrochemical urea synthesis by co-activation of CO2 and N2 with Mott–Schottky heterostructure catalysts. Angew. Chem. Int. Ed. 60, 10910–10918 (2021).
Xing, P. et al. Electrochemical co-reduction of N2 and CO2 to urea using Bi2S3 nanorods anchored to N-doped reduced graphene oxide. ACS Appl. Mater. Interfaces 15, 22101–22111 (2023).
Zhang, X. et al. Electrocatalytic urea synthesis with 63.5 % Faradaic efficiency and 100 % N-selectivity via one-step C−N coupling. Angew. Chem. Int. Ed. 62, e202305447 (2023).
Wu, W. et al. Boosting electrosynthesis of urea from N2 and CO2 by defective Cu-Bi. Chem. Catal. 2, 3225–3238 (2022).
Chen, C. et al. Coupling N2 and CO2 in H2O to synthesize urea under ambient conditions. Nat. Chem. 12, 717–724 (2020).
Saravanakumar, D., Song, J., Lee, S., Hur, N. H. & Shin, W. Electrocatalytic conversion of carbon dioxide and nitrate ions to urea by a titania–Nafion composite electrode. ChemSusChem 10, 3999–4003 (2017).
Zhao, Y. et al. Efficient urea electrosynthesis from carbon dioxide and nitrate via alternating Cu–W bimetallic C–N coupling sites. Nat. Commun. 14, 4491 (2023).
Dai, Z. et al. Surface engineering on bulk Cu2O for efficient electrosynthesis of urea. Nat. Commun. 16, 3271 (2025).
Wei, X. et al. Lattice oxygen-driven co-adsorption of carbon dioxide and nitrate on copper: a pathway to efficient urea electrosynthesis. J. Am. Chem. Soc. 147, 6049–6057 (2025).
Cave, E. R. et al. Trends in the catalytic activity of hydrogen evolution during CO2 electroreduction on transition metals. ACS Catal. 8, 3035–3040 (2018).
Kolb, T., Arif, A. M. & Ernst, R. D. Crystallization and structural characterization of dimeric and trimeric forms of (Neocuproine)CdFe(CO)4. J. Crystallogr. 2014, 704869 (2014).
Ernst, R. D., Marks, T. J. & Ibers, J. A. Metal–metal bond cleavage reactions. The crystal and molecular structure of (2,2′-bipyridyl)cadmium tetracarbonyliron, (bpy)CdFe(CO)4. J. Am. Chem. Soc. 99, 2098–2107 (1977).
Xiao, J. & Frauenheim, T. Activation mechanism of carbon monoxide on α-Fe2O3 (0001) surface studied by using first principle calculations. Appl. Phys. Lett. 101, 041603 (2012).
Amaya-Roncancio, S., Linares, D. H., Duarte, H. A., Lener, G. & Sapag, K. Effect of hydrogen in adsorption and direct dissociation of CO on Fe (100) surface: a DFT study. Am. J. Analyt. Chem. 06, 38–46 (2015).
Hori, Y., Wakebe, H., Tsukamoto, T. & Koga, O. Electrocatalytic process of CO selectivity in electrochemical reduction of CO2 at metal electrodes in aqueous media. Electrochim. Acta 39, 833–1839 (1994).
Raimundo, R. A. et al. High magnetic moment of nanoparticle-sphere-like Co, Fe based composites and alloys prepared by proteic sol-gel synthesis: structure, magnetic study and OER activity. J. Alloys Compd 940, 168783 (2023).
Hai, Y. et al. Ammonia synthesis via electrocatalytic nitrate reduction using NiCoO2 nanoarrays on a copper foam. ACS Appl. Mater. Interfaces 16, 11431–11439 (2024).
Keuleers, R., Desseyn, H. O., Rousseau, B. & Van Alsenoy, C. Vibrational analysis of urea. J. Phys. Chem. A 103, 4621–4630 (1999).
Hammer, B. & Nørskov, J. K. Electronic factors determining the reactivity of metal surfaces. Surf. Sci. 343, 211–220 (1995).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 12, 537–541 (2005).
Chang, X., Malkani, A., Yang, X. & Xu, B. Mechanistic insights into electroreductive C–C coupling between CO and acetaldehyde into multicarbon products. J. Am. Chem. Soc. 142, 2975–2983 (2020).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Hafner, J. Ab initio molecular-dynamics simulation of the liquid-metal–amorphous-semiconductor transition in germanium. Phys. Rev. B 49, 14251–14269 (1994).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Blöchl, P. E., Jepsen, O. & Andersen, O. K. Improved tetrahedron method for Brillouin-zone integrations. Phys. Rev. B 49, 16223–16233 (1994).
Klimeš, J., Bowler, D. R. & Michaelides, A. Van der Waals density functionals applied to solids. Phys. Rev. B 83, 195131 (2011).
Cococcioni, M. & de Gironcoli, S. Linear response approach to the calculation of the effective interaction parameters in the LDA + U method. Phys. Rev. B 71, 35105 (2005).
Dudarev, S. L., Botton, G. A., Savrasov, S. Y., Humphreys, C. J. & Sutton, A. P. Electron-energy-loss spectra and the structural stability of nickel oxide: an LSDA+U study. Phys. Rev. B 57, 1505–1509 (1998).
Wang, L., Maxisch, T. & Ceder, G. Oxidation energies of transition metal oxides within the GGA+U framework. Phys. Rev. B 73, 195107 (2006).
Bengtsson, L. Dipole correction for surface supercell calculations. Phys. Rev. B 59, 12301–12304 (1999).
Carvalho, O. Q. et al. Role of electronic structure on nitrate reduction to ammonium: a periodic journey. J. Am. Chem. Soc. 144, 14809–14818 (2022).
Acknowledgements
We acknowledge the Ministry of Education Singapore for their financial support, through the grant of T2EP50124-0012. We also acknowledge the support of the National Research Foundation (NRF) Singapore, under the NRF Fellowship (NRF-NRFF13-2021-0007) and CRP (NRF-CRP27-2021-0004), as well as the support from the Centre for Hydrogen Innovations at the NUS (CHI-P2024-03). P.O. and Y.L. acknowledge the use of Singapore National Supercomputing Centre high-performance computing facilities, consulting support and/or training services as part of this research. As a National Research Infrastructure funded by the NRF (https://www.nscc.sg), P.O. also acknowledges the support from the National University of Singapore Presidential Young Professorship Start-Up Grant (A-0010024-00-00). We also thank S. Xi at Singapore Synchrotron Light Source for his assistance with XAS characterization.
Author information
Authors and Affiliations
Contributions
L.W. supervised the project. L.W. and B.H. conceived the idea. B.H. and Y.Z. designed and performed the experiments. B.H. and Y.Z. conducted the TEA under L.W.’s supervision. Z.W., Y.S.T. and B.H. conducted the large language model under X.W.’s supervision. Y.L. completed the theoretical calculations under P.O.’s supervision. L.W., P.O., B.H., Y.L., Y.Z., S.Y. and H.S. contributed to the data interpretation and wrote the paper. All the authors contributed to the revision of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Cao-Thang Dinh, Liangzhi Kou and Fusheng Li for their contribution to the peer review of this work. Primary Handling Editor: Alexandra Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–57, Notes 1–8 and Tables 1–3.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data for Fig. 1.
Source Data Fig. 2 (download XLSX )
Statistical source data for Fig. 2.
Source Data Fig. 3 (download XLSX )
Statistical source data for Fig. 3.
Source Data Fig. 4 (download XLSX )
Statistical source data for Fig. 4.
Source Data Fig. 5 (download XLSX )
Statistical source data for Fig. 5.
Source Data Fig. 6 (download XLSX )
Statistical source data for Fig. 6.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, B., Liu, Y., Zhou, Y. et al. Electrosynthesis of urea on cadmium-modified iron oxide. Nat. Synth (2026). https://doi.org/10.1038/s44160-026-00995-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-026-00995-9