Abstract
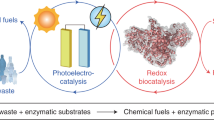
Biophotoelectrocatalysis provides chemo-, regio- and stereoselective routes to chemicals by coupling redox biocatalysis with photoelectrocatalysis. This biomimetic strategy, however, is limited by unwanted photoelectrocatalysis side reactions and the high cost of redox mediators. Here we report a sustainable biosolar platform that uses bicarbonate (HCO3−), a hydrated form of CO2, as a redox mediator to drive oxyfunctionalization of inert C–H bonds. Using molybdenum-doped bismuth vanadate as a model photoelectrode, we accelerate two-electron H2O oxidation for in situ H2O2 production and mitigate enzyme-damaging OH· generation via HCO3− photoredox chemistry. Photoelectrochemical and spectroscopic analyses revealed that HCO3− directs the H2O oxidation pathway towards H2O2 through the formation of a peroxycarbonate intermediate at the photoanode surface. The integration of HCO3− mediation with H2O2-dependent unspecific peroxygenase achieves an exceptional turnover of various enantioselective C–H oxyfunctionalization reactions under ambient conditions. The HCO3−-mediated H2O2 photosynthetic system opens up opportunities for sustainable oxygenative biosynthesis.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of the study are available within the paper and its Supplementary Information.
References
Xue, X.-S., Ji, P., Zhou, B. & Cheng, J.-P. The essential role of bond energetics in C–H activation/functionalization. Chem. Rev. 117, 8622–8648 (2017).
Kamata, K., Yonehara, K., Nakagawa, Y., Uehara, K. & Mizuno, N. Efficient stereo- and regioselective hydroxylation of alkanes catalysed by a bulky polyoxometalate. Nat. Chem. 2, 478–483 (2010).
Zhang, W. et al. Selective aerobic oxidation reactions using a combination of photocatalytic water oxidation and enzymatic oxyfunctionalizations. Nat. Catal. 1, 55–62 (2018).
Sigmund, M.-C. & Poelarends, G. J. Current state and future perspectives of engineered and artificial peroxygenases for the oxyfunctionalization of organic molecules. Nat. Catal. 3, 690–702 (2020).
Freakley, S. J. et al. A chemo-enzymatic oxidation cascade to activate C–H bonds with in situ generated H2O2. Nat. Commun. 10, 4178 (2019).
Kim, J., Nguyen, T. V. T., Kim, Y. H., Hollmann, F. & Park, C. B. Lignin as a multifunctional photocatalyst for solar-powered biocatalytic oxyfunctionalization of C–H bonds. Nat. Synth. 1, 217–226 (2022).
Liu, J., Zou, Y., Jin, B., Zhang, K. & Park, J. H. Hydrogen peroxide production from solar water oxidation. ACS Energy Lett. 4, 3018–3027 (2019).
Zhang, K. et al. Near-complete suppression of oxygen evolution for photoelectrochemical H2O oxidative H2O2 synthesis. J. Am. Chem. Soc. 142, 8641–8648 (2020).
Choi, D. S., Kim, J., Hollmann, F. & Park, C. B. Solar-assisted eBiorefinery: photoelectrochemical pairing of oxyfunctionalization and hydrogenation reactions. Angew. Chem. Int. Ed. 59, 15886–15890 (2020).
Schanz, T., Burek, B. O. & Bloh, J. Z. Fate and reactivity of peroxides formed over BiVO4 anodes in bicarbonate electrolytes. ACS Energy Lett. 8, 1463–1467 (2023).
Lee, C. H., Kim, J. & Park, C. B. Z-Schematic artificial leaf structure for biosolar oxyfunctionalization of hydrocarbons. ACS Energy Lett. 8, 2513–2521 (2023).
Zhang, W. et al. Selective activation of C−H bonds in a cascade process combining photochemistry and biocatalysis. Angew. Chem. Int. Ed. 56, 15451–15455 (2017).
Lee, S. H., Choi, D. S., Kuk, S. K. & Park, C. B. Photobiocatalysis: activating redox enzymes by direct or indirect transfer of photoinduced electrons. Angew. Chem. Int. Ed. 57, 7958–7985 (2018).
Kornienko, N., Zhang, J. Z., Sakimoto, K. K., Yang, P. & Reisner, E. Interfacing nature’s catalytic machinery with synthetic materials for semi-artificial photosynthesis. Nat. Nanotechnol. 13, 890–899 (2018).
Cestellos-Blanco, S., Zhang, H., Kim, J. M., Shen, Y.-x & Yang, P. Photosynthetic semiconductor biohybrids for solar-driven biocatalysis. Nat. Catal. 3, 245–255 (2020).
Kim, J. et al. Robust FeOOH/BiVO4/Cu(In, Ga)Se2 tandem structure for solar-powered biocatalytic CO2 reduction. J. Mater. Chem. A 8, 8496–8502 (2020).
Kim, J. et al. Unbiased photoelectrode interfaces for solar coupling of lignin oxidation with biocatalytic C=C bond hydrogenation. ACS Appl. Mater. Interfaces 14, 11465–11473 (2022).
Kim, J., Jang, J., Hilberath, T., Hollmann, F. & Park, C. B. Photoelectrocatalytic biosynthesis fuelled by microplastics. Nat. Synth. 1, 776–786 (2022).
Kim, J. et al. Biocatalytic C=C bond reduction through carbon nanodot-sensitized regeneration of NADH analogues. Angew. Chem. Int. Ed. 57, 13825–13828 (2018).
Jeon, B. W. et al. Enzymatic conversion of CO2 to formate: the potential of tungsten-containing formate dehydrogenase in flow reactor system. J. CO2 Util. 82, 102754 (2024).
Yun, C.-H., Kim, J., Hollmann, F. & Park, C. B. Light-driven biocatalytic oxidation. Chem. Sci. 13, 12260–12279 (2022).
Fan, L. et al. CO2/carbonate-mediated electrochemical water oxidation to hydrogen peroxide. Nat. Commun. 13, 2668 (2022).
Xia, C. et al. Confined local oxygen gas promotes electrochemical water oxidation to hydrogen peroxide. Nat. Catal. 3, 125–134 (2020).
Li, L. et al. Electrochemical generation of hydrogen peroxide from a zinc gallium oxide anode with dual active sites. Nat. Commun. 14, 1890 (2023).
Gill, T. M., Vallez, L. & Zheng, X. The role of bicarbonate-based electrolytes in H2O2 production through two-electron water oxidation. ACS Energy Lett. 6, 2854–2862 (2021).
Viswanathan, V., Hansen, H. A. & Nørskov, J. K. Selective electrochemical generation of hydrogen peroxide from water oxidation. J. Phys. Chem. Lett. 6, 4224–4228 (2015).
Jiang, Y. et al. Selective electrochemical H2O2 production through two-electron oxygen electrochemistry. Adv. Energy Mater. 8, 1801909 (2018).
Siahrostami, S. et al. Enabling direct H2O2 production through rational electrocatalyst design. Nat. Mater. 12, 1137–1143 (2013).
Perry, S. C. et al. Electrochemical synthesis of hydrogen peroxide from water and oxygen. Nat. Rev. Chem. 3, 442–458 (2019).
Bakhmutova-Albert, E. V., Yao, H., Denevan, D. E. & Richardson, D. E. Kinetics and mechanism of peroxymonocarbonate formation. Inorg. Chem. 49, 11287–11296 (2010).
Wang, L. et al. Suppressing water dissociation via control of intrinsic oxygen defects for awakening solar H2O-to-H2O2 generation. Small 17, 2100400 (2021).
Oxley, J. et al. Raman and infrared fingerprint spectroscopy of peroxide-based explosives. Appl. Spectrosc. 62, 906–915 (2008).
Wang, B. et al. Synthesis and reactivity of a mononuclear non-haem cobalt(IV)-oxo complex. Nat. Commun. 8, 14839 (2017).
Xu, B., Hirsch, A., Kronik, L. & Poduska, K. ristinM. Vibrational properties of isotopically enriched materials: the case of calcite. RSC Adv. 8, 33985–33992 (2018).
Medinas, D. B., Cerchiaro, G., Trindade, D. F. & Augusto, O. The carbonate radical and related oxidants derived from bicarbonate buffer. IUBMB Life 59, 255–262 (2007).
Berge, A. H. et al. Revealing carbon capture chemistry with 17-oxygen NMR spectroscopy. Nat. Commun. 13, 7763 (2022).
Kim, C. H., Kim, J., Hollmann, F. & Park, C. B. Photoelectrocatalytic N2 fixation and C–H oxyfunctionalization driven by H2O oxidation. Appl. Catal. B 336, 122925 (2023).
Ma, Y. et al. A novel unspecific peroxygenase from Galatian marginata for biocatalytic oxyfunctionalization reactions. Mol. Catal. 531, 112707 (2022).
Simon, T. et al. Redox shuttle mechanism enhances photocatalytic H2 generation on Ni-decorated CdS nanorods. Nat. Mater. 13, 1013–1018 (2014).
Shi, X., Zhang, Y., Siahrostami, S. & Zheng, X. Light-driven BiVO4–C fuel cell with simultaneous production of H2O2. Adv. Energy Mater. 8, 1801158 (2018).
Yang, L. et al. Synergetic effects by Co2+ and PO43- on Mo-doped BiVO4 for an improved photoanodic H2O2 evolution. Chem. Eng. Sci. 251, 117435 (2022).
Shi, S. et al. BiVO4-based heterojunction photocathode for high-performance photoelectrochemical hydrogen peroxide production. Nano Lett. 24, 6051–6060 (2024).
Tan, H. L. et al. Solar-driven hydrogen peroxide production via BiVO4-based photocatalysts. Adv. Sci. 12, 2407801 (2025).
Kim, J. & Park, C. B. Collaborative catalysis for solar biosynthesis. Trends Chem. 5, 133–146 (2023).
Yoon, J. et al. Heat-fueled enzymatic cascade for selective oxyfunctionalization of hydrocarbons. Nat. Commun. 13, 3741 (2022).
Gomez de Santos, P. et al. Evolved peroxygenase–aryl alcohol oxidase fusions for self-sufficient oxyfunctionalization reactions. ACS Catal. 10, 13524–13534 (2020).
Kim, J. & Park, C. B. Shedding light on biocatalysis: photoelectrochemical platforms for solar-driven biotransformation. Curr. Opin. Chem. Biol. 49, 122–129 (2019).
Liu, M. et al. Enhanced electrocatalytic CO2 reduction via field-induced reagent concentration. Nature 537, 382–386 (2016).
Acknowledgements
This work was supported by National Research Foundation of Korea (NRF) grants funded by the Ministry of Science and ICT (grant numbers RS-2023-00222078, RS-2024-00440681, RS-2024-00460425). F.H. acknowledges funding by the European Union (ERC, PeroxyZyme, number 101054658).
Author information
Authors and Affiliations
Contributions
C.H.K., C.H.L. and M.L. conceived the project, designed the research, performed experiments, analysed data and wrote the paper. C.B.P. supervised the research. D.O., Y.H. and H.-M.Y. performed experiments and analysed data. C.H.K., C.H.L., M.L., W.J. and C.B.P. discussed the photoelectrocatalysis. F.H. supplied the UPO enzymes. C.H.K., C.H.L., M.L., F.H. and C.B.P. commented on the photoelectrochemical biocatalysis. F.H. and C.B.P. reviewed the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Carla Casadevall, Han Sen Soo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor Thomas West, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Tables 1–4 and Figs. 1–29.
Source data
Source Data Fig. 2 (download XLSX )
Raw data on linear sweep voltammetric curves (Fig. 2b), Nyquist plots (Fig. 2c), and H2O2 production (Fig. 2d).
Source Data Fig. 3 (download XLSX )
Raw data on in situ Raman (Fig. 3a) and electron paramagnetic resonance spectra (Fig. 3c).
Source Data Fig. 4 (download XLSX )
Raw data on time profiles of biophotoelectrocatalytic reactions (Fig. 4a), photoluminescence spectra (Fig. 4b), and control experiments on various substrates (Fig. 4d).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, C.H., Lee, C.H., Lee, M. et al. Hydrated CO2-mediated redox chemistry for biophotoelectrocatalytic oxyfunctionalization of C–H bonds. Nat. Synth (2026). https://doi.org/10.1038/s44160-026-00998-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-026-00998-6