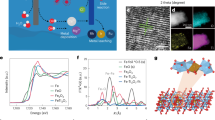
Abstract
Developing active and stable electrocatalysts for the chlorine evolution reaction (CER) is critical for chlor-alkali processes but remains challenging. Here we introduce ultrafine high-entropy alloy nanowires (UF-HEANWs) enriched with atomic steps as efficient CER catalysts for direct chlorine electrosynthesis from seawater. A seawater flow electrolyser equipped with UF-HEANW anodes achieves 98.1% CER selectivity at an industrial-scale current density of 10 kA m−2, maintaining continuous operation for over 5,500 h. Operando studies reveal that atomic steps in lattice-distorted UF-HEANWs create corner-edge electronic heterogeneity, triggering the in situ generation of high-valent Pt–O sites with localized electronic states and unsaturated coordination. These dynamic active structures enhance chloride adsorption and chlorine desorption, leading to improved activity and selectivity during CER. A techno-economic analysis shows that costs are reduced by 32.8% versus the chlor-alkali industry, with 51.3% less electricity used during electrolysis via high-entropy alloy anodes and 83.1% lower feedstock costs from seawater replacement.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the Article and its Supplementary Information. Source data are provided with this paper.
References
Karlsson, R. K. & Cornell, A. Selectivity between oxygen and chlorine evolution in the chlor-alkali and chlorate processes. Chem. Rev. 116, 2982–3028 (2016).
Wang, Y., Liu, Y., Wiley, D., Zhao, S. & Tang, Z. Recent advances in electrocatalytic chloride oxidation for chlorine gas production. J. Mater. Chem. A 9, 18974–18993 (2021).
Yang, J. et al. CO2-mediated organocatalytic chlorine evolution under industrial conditions. Nature 617, 519–523 (2023).
Exner, K. S., Anton, J., Jacob, T. & Over, H. Controlling selectivity in the chlorine evolution reaction over RuO2-based catalysts. Angew. Chem. Int. Ed. 53, 11032–11035 (2014).
Lim, H. W. et al. Rational design of dimensionally stable anodes for active chlorine generation. ACS Catal. 11, 12423–12432 (2021).
Fauvarque, J. The chlorine industry. Pure Appl. Chem. 68, 1713–1720 (1996).
Choi, S. et al. A reflection on sustainable anode materials for electrochemical chloride oxidation. Adv. Mater. 35, 2300429 (2023).
Vos, J. G. et al. Selectivity trends between oxygen evolution and chlorine evolution on iridium-based double perovskites in acidic media. ACS Catal. 9, 8561–8574 (2019).
Exner, K. S., Lim, T. & Joo, S. H. Circumventing the OCl versus OOH scaling relation in the chlorine evolution reaction: Beyond dimensionally stable anodes. Curr. Opin. Electrochem. 34, 100979 (2022).
Xiao, M. et al. Self-adaptive amorphous CoOxCly electrocatalyst for sustainable chlorine evolution in acidic brine. Nat. Commun. 14, 5356 (2023).
Liu, Y. et al. Electrosynthesis of chlorine from seawater-like solution through single-atom catalysts. Nat. Commun. 14, 2475 (2023).
Lim, T. et al. Atomically dispersed Pt–N4 sites as efficient and selective electrocatalysts for the chlorine evolution reaction. Nat. Commun. 11, 412 (2020).
Tong, W. et al. Electrolysis of low-grade and saline surface water. Nat. Energy 5, 367–377 (2020).
Guo, J. et al. Direct seawater electrolysis by adjusting the local reaction environment of a catalyst. Nat. Energy 8, 264–272 (2023).
Yao, Y. et al. Carbothermal shock synthesis of high-entropy-alloy nanoparticles. Science 359, 1489–1494 (2018).
George, E. P., Raabe, D. & Ritchie, R. O. High-entropy alloys. Nat. Rev. Mater. 4, 515–534 (2019).
Li, M. et al. High-entropy alloy electrocatalysts go to (sub-)nanoscale. Sci. Adv. 10, eadn2877 (2024).
Ren, J.-T., Chen, L., Wang, H.-Y. & Yuan, Z.-Y. High-entropy alloys in electrocatalysis: from fundamentals to applications. Chem. Soc. Rev. 52, 8319–8373 (2023).
Sun, Y. & Dai, S. High-entropy materials for catalysis: a new frontier. Sci. Adv. 7, eabg1600 (2021).
Yao, Y. et al. High-entropy nanoparticles: synthesis–structure–property relationships and data-driven discovery. Science 376, eabn3103 (2022).
Han, X. et al. Nanoscale high-entropy alloy for electrocatalysis. Matter 6, 1717–1751 (2023).
Li, H. et al. Fast site-to-site electron transfer of high-entropy alloy nanocatalyst driving redox electrocatalysis. Nat. Commun. 11, 5437 (2020).
Hao, J. et al. Unraveling the electronegativity-dominated intermediate adsorption on high-entropy alloy electrocatalysts. Nat. Commun. 13, 2662 (2022).
Zhao, H. et al. Strong transboundary electron transfer of high-entropy quantum-dots driving rapid hydrogen evolution kinetics. Energy Environ. Sci. 17, 6594–6605 (2024).
Wang, B. et al. General synthesis of high-entropy alloy and ceramic nanoparticles in nanoseconds. Nat. Synth. 1, 138–146 (2022).
He, L. et al. Single-atom Mo-tailored high-entropy-alloy ultrathin nanosheets with intrinsic tensile strain enhance electrocatalysis. Nat. Commun. 15, 2290 (2024).
Chen, Z. W. et al. Unusual Sabatier principle on high entropy alloy catalysts for hydrogen evolution reactions. Nat. Commun. 15, 359 (2024).
Chida, Y. et al. Experimental study platform for electrocatalysis of atomic-level controlled high-entropy alloy surfaces. Nat. Commun. 14, 4492 (2023).
Gan, L. et al. Element-specific anisotropic growth of shaped platinum alloy nanocrystals. Science 346, 1502–1506 (2014).
Chen, W. et al. A map of single-phase high-entropy alloys. Nat. Commun. 14, 2856 (2023).
Doan-Nguyen, V. V. et al. Bulk metallic glass-like scattering signal in small metallic nanoparticles. ACS Nano 8, 6163–6170 (2014).
Moreno-Hernandez, I. A., Brunschwig, B. S. & Lewis, N. S. Crystalline nickel, cobalt, and manganese antimonates as electrocatalysts for the chlorine evolution reaction. Energy Environ. Sci. 12, 1241–1248 (2019).
The Electrolysis Process and the Real Costs of Production (Euro Chlor Communications, 2018); http://www.eurochlor.org/
Kermeli, K. & Worrell, E. Energy Efficiency and Cost-saving Opportunities for the Chlor-Alkali Industry. EPA-430-R-24-008 (US Environmental Protection Agency, 2025).
Zhao, S., Yang, Y. & Tang, Z. Insight into structural evolution, active sites, and stability of heterogeneous electrocatalysts. Angew. Chem. Int. Ed. 61, e202110186 (2022).
Deeley, C. M. & Mills, I. M. Vibration-rotation spectra and the harmonic force field of HOCl. J. Mol. Spectrosc. 114, 368–376 (1985).
Li, S., Guo, X., Liu, X. & Shui, J. Atomic single-layer Ir clusters enabling 100% selective chlorine evolution reaction. ACS Catal. 14, 1962–1969 (2024).
Mei, Y. et al. MoZn-based high entropy alloy catalysts enabled dual activation and stabilization in alkaline oxygen evolution. Sci. Adv. 10, eadq6758 (2024).
Huang, J. et al. Experimental Sabatier plot for predictive design of active and stable Pt-alloy oxygen reduction reaction catalysts. Nat. Catal. 5, 513–523 (2022).
Escudero-Escribano, M. et al. Tuning the activity of Pt alloy electrocatalysts by means of the lanthanide contraction. Science 352, 73–76 (2016).
Jiao, S., Fu, X. & Huang, H. Descriptors for the evaluation of electrocatalytic reactions: d-band theory and beyond. Adv. Funct. Mater. 32, 2107651 (2022).
Juhás, P., Davis, T., Farrow, C. L. & Billinge, S. J. PDFgetX3: a rapid and highly automatable program for processing powder diffraction data into total scattering pair distribution functions. J. Appl. Cryst. 46, 560–566 (2013).
Wu, Z.-P. et al. Alloying–realloying enabled high durability for Pt–Pd-3d-transition metal nanoparticle fuel cell catalysts. Nat. Commun. 12, 859 (2021).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Rad. 12, 537–541 (2005).
Jain, A. et al. Commentary: The Materials Project: a materials genome approach to accelerating materials innovation. APL Mater. 1, 011002 (2013).
Ofuchi, H., Matsumoto, T. & Honma, T. Construction of XAFS standard sample database at BL14B2 in SPring-8. Radiat. Phys. Chem. 218, 111581 (2024).
Yuwono, J. A. et al. A computational approach for mapping electrochemical activity of multi-principal element alloys. npj Mater. Degrad. 7, 87 (2023).
Doležal, T. D. & Samin, A. J. Adsorption of oxygen to high entropy alloy surfaces for up to 2 ML coverage using density functional theory and Monte Carlo calculations. Langmuir 38, 3158–3169 (2022).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Kresse, G. & Furthmuller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H–Pu. J. Chem. Phys. 132, 154104 (2010).
Acknowledgements
S.Z. is thankful for support from the National Key R&D Program of China (grant no. 2024YFA1211100) and the National Natural Science Foundation of China (grant no. 22373027). Yongchao Yang is thankful for financial support from the FH Loxton Fellowship. J.A.Y. acknowledges financial support from the Australian Research Council (grant no. DE250101071) and high-performance computational support from the National Computing Infrastructure Australia. R.D.T. would like to acknowledge support from the Australian Research Council (grant no. CE230100032). We acknowledge the European Synchrotron Radiation Facility for the provision of synchrotron radiation facilities for HE-XRD/PDF measurements at the ID11 beamline. XAS measurements were performed at the XAS and SXR beamlines of the Australian Synchrotron, part of ANSTO. HAXPES survey was carried out on the Spectroscopy Soft and Tender 2 (SST-2) beamline at the National Synchrotron Light Source II. We acknowledge the Mark Wainwright Analytical Centre and Microscopy Australia for support of facilities at the Electron Microscope Unit (EMU) at UNSW. The scientific and technical support from EMU at UNSW in Sydney is appreciated.
Author information
Authors and Affiliations
Contributions
S.Z. proposed the research direction and supervised the research project. Yongchao Yang performed the material design, synthesis and electrochemical experiments, and drafted and revised the whole manuscript. S.C. and R.D.T. performed HRTEM, HAADF-STEM and EDS characterizations. Yuwei Yang and N.M.B. performed XAS, HE-XRD and HAXPES measurements. Yongchao Yang and X.S. conducted the techno-economic analysis. T.Z. conducted in situ FTIR characterization. J.A.Y. conducted the theoretical calculations. Yongchao Yang and S.Z. discussed the research results. All authors assisted during the preparation of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Kai S. Exner and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alexandra Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Text, Figs. 1–67 and Tables 1–19.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, Y., Yang, Y., Yuwono, J.A. et al. High-entropy alloy nanowires for direct electrosynthesis of chlorine from seawater. Nat. Synth (2026). https://doi.org/10.1038/s44160-026-00999-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-026-00999-5