Abstract
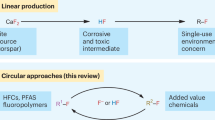
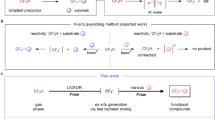
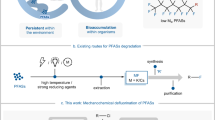
The valorization of fluorinated waste for the synthesis of value-added non-per- and polyfluoroalkyl substances (PFASs) presents a substantial challenge. Fluoroform (HFC-23), an extremely potent waste greenhouse gas produced in multi-tonne quantities, has traditionally been incinerated or utilized as a C1 building block. Here we report the application of fluoroform to the synthesis of trifluorovinylamine, an original C2 building block. Trifluorovinylamine exhibits unique reactivity and functions as a difluoroacylium surrogate. This distinctive reactivity facilitates the efficient formation of α,α-difluoromethyl ketones via electrophilic aromatic substitution and enables the development of a hydrodifluoroacetylation reaction. This class of reagent not only introduces alternative chemical reactivity but also offers an innovative pathway for the valorization of HFC-23, advancing the synthesis of non-PFAS compounds.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper and its Supplementary Information.
References
Groult, H. et al. (eds) Modern Synthesis Processes and Reactivity of Fluorinated Compounds, Progress in Fluorine Science (Elsevier, 2016).
Britton, R. et al. Contemporary synthetic strategies in organofluorine chemistry. Nat. Rev. Methods Primers 1, 47 (2021).
O’Hagan, D. Understanding organofluorine chemistry. An introduction to the C–F bond. Chem. Soc. Rev. 37, 308–319 (2008).
Wang, Z. et al. A new OECD definition for per- and polyfluoroalkyl substances. Environ. Sci. Technol. 55, 15575–15578 (2021).
Evich, M. G. et al. Per- and polyfluoroalkyl substances in the environment. Science 375, eabg9065 (2022).
Cousins, I. T. et al. The high persistence of PFAS is sufficient for their management as a chemical class. Environ. Sci. Process. Impacts 22, 2307–2312 (2020).
Freeling, F. & Björnsdotter, M. K. Assessing the environmental occurrence of the anthropogenic contaminant trifluoroacetic acid (TFA). Curr. Opin. Green Sustain. Chem. 41, 100807 (2023).
Solomon, K. R. et al. Sources, fates, toxicity, and risks of trifluoroacetic acid and its salts: relevance to substances regulated under the Montreal and Kyoto Protocols. J. Toxicol. Environ. Health B Crit. Rev. 19, 289–304 (2016).
Cahard, D. & Ma, J.-A. (eds) Emerging Fluorinated Motifs (Wiley-VCH, 2020)
Spennacchio, M. et al. A unified flow strategy for the preparation and use of trifluoromethyl-heteroatom anions. Science 385, 991–996 (2024).
Tlili, A., Toulgoat, F. & Billard, T. Synthetic approaches to trifluoromethoxy-substituted compounds. Angew. Chem. Int. Ed. 55, 11726–11735 (2016).
Besset, T., Jubault, P., Pannecoucke, X. & Poisson, T. New entries toward the synthesis of OCF3-containing molecules. Org. Chem. Front. 3, 1004–1010 (2016).
Crousse, B. Recent advances in the syntheses of N-CF3 scaffolds up to their valorization. Chem. Rec. 23, e202300011 (2023).
Milcent, T. & Crousse, B. in Emerging Fluorinated Motifs (eds Cahard, D. and Ma, J.-A.) 723–761 (Wiley-VCH, 2020).
Scattolin, T., Bouayad-Gervais, S. & Schoenebeck, F. Straightforward access to N-trifluoromethyl amides, carbamates, thiocarbamates and ureas. Nature 573, 102–107 (2019).
Savoie, P. R. & Welch, J. T. Preparation and utility of organic pentafluorosulfanyl-containing compounds. Chem. Rev. 115, 1130–1190 (2015).
Xiong, H.-Y., Pannecoucke, X. & Besset, T. Recent advances in the synthesis of SCF2H, SCF2FG-containing molecules. Chem. Eur. J. 22, 16734–16749 (2016).
Sessler, C. D. et al. CF2H, a hydrogen bond donor. J. Am. Chem. Soc. 139, 9325–9332 (2017).
Zafrani, Y. et al. Difluoromethyl bioisostere: examining the “lipophilic hydrogen bond donor” concept. J. Med. Chem. 60, 797–804 (2017).
Reconciling Terminology of the Universe of Per- and Polyfluoroalkyl Substances: Recommendations and Practical Guidance (OECD, 2021); https://doi.org/10.1787/e458e796-en
Ruyet, L. & Besset, T. Copper-based fluorinated reagents for the synthesis of CF2R-containing molecules (R ≠ F). Beilstein J. Org. Chem. 16, 1051–1065 (2020).
Li, X. & Song, Q. Introduction of difluoromethyl through radical pathways. Eur. J. Org. Chem. 27, e202400423 (2024).
Downing, F. B., Benning, A. F. & McHarness, R. C. Pyrolysis of chloro-fluoro alkanes. US patent 2,551,573 (1945).
Stanley, K. M. et al. Increase in global emissions of HFC-23 despite near-total expected reductions. Nat. Commun. 11, 397 (2020).
Rust, D. et al. Effective realization of abatement measures can reduce HFC-23 emissions. Nature 633, 96–100 (2024).
Grushin, V. V. Fluoroform as a feedstock for high-value fluorochemicals: novel trends and recent developments. Chim. Oggi. Chem. Today 32, 81–88 (2014).
Prakash, G. K. S., Jog, P. V., Batamack, P. T. D. & Olah, G. A. Taming of fluoroform: direct nucleophilic trifluoromethylation of Si, B, S, and C centers. Science 338, 1324–1327 (2012).
Zanardi, A., Novikov, M. A., Martin, E., Benet-Buchholz, J. & Grushin, V. V. Direct cupration of fluoroform. J. Am. Chem. Soc. 133, 20901–20913 (2011).
Novak, P., Lishchynskyi, A. & Grushin, V. V. Highly enantioselective copper-catalyzed electrophilic trifluoromethylation of β-ketoesters. J. Am. Chem. Soc. 134, 16167–16170 (2012).
Kawai, H., Yuan, Z., Tokonaga, E. & Shibata, N. A sterically demanding organo-superbase avoids decomposition of a naked trifluoromethyl carbanion directly generated from fluoroform. Org. Biomol. Chem. 11, 1446–1450 (2013).
Musio, B., Gala, E. & Ley, S. V. Real-time spectroscopic analysis enabling quantitative and safe consumption of fluoroform during nucleophilic trifluoromethylation in flow. ACS Sust. Chem. Eng. 6, 1489–1495 (2018).
Yang, X. & Tsui, G. C. Copper-mediated 1,2-bis(trifluoromethylation) of arynes. Chem. Sci. 9, 8871–8875 (2018).
Xiang, J.-X., Ouyang, Y., Xu, X.-H. & Qing, F.-L. Argentination of fluoroform: preparation of a stable AgCF3 solution with diverse reactivities. Angew. Chem. Int. Ed. 58, 10320–10324 (2019).
Fu, W. C., MacQueen, P. M. & Jamison, T. F. Continuous flow strategies for using fluorinated greenhouse gases in fluoroalkylations. Chem. Soc. Rev. 50, 7378–7394 (2021).
Lu, Z., Wang, L., Hughes, M., Smith, S. & Shen, Q. nBu4N+[AgI(CF3)2]−: trifluoromethylated argentate derived from fluoroform and its reaction with (hetero)aryl diazonium salts. Org. Lett. 26, 2773–2777 (2023).
Thomoson, C. S., Wang, L. & Dolbier, W. R. Use of fluoroform as a source of difluorocarbene in the synthesis of N-CF2H heterocycles and difluoromethoxypyridines. J. Fluorine Chem. 168, 34–39 (2014).
Aikawa, K., Maruyama, K., Nitta, J., Hashimoto, R. & Mikami, K. Siladifluoromethylation and difluoromethylation onto C(sp3), C(sp2),and C(sp) centers using Ruppert–Prakash reagent and fluoroform. Org. Lett. 18, 3354–3357 (2016).
Okusu, S., Tokunaga, E. & Shibata, N. Difluoromethylation of terminal alkynes by fluoroform. Org. Lett. 17, 3802–3805 (2015).
Ida, T., Hashimoto, R., Aikawa, K., Ito, S. & Mikami, K. Umpolung of fluoroform by C–F bond activation: direct difluoromethylation of lithium enolates. Angew. Chem. Int. Ed. 51, 9535–9538 (2012).
Aikawa, K., Maruyama, K., Honda, K. & Mikami, K. α-Difluoromethylation on sp3 carbon of nitriles using fluoroform and Ruppert–Prakash reagent. Org. Lett. 17, 4882–4885 (2015).
Köckinger, M. et al. Utilization of fluoroform for difluoromethylation in continuous flow: a concise synthesis of α-difluoromethyl-amino acids. Green Chem. 20, 108–112 (2018).
Yang, Z.-Y., Wiemers, D. M. & Burton, D. J. Trifluoromethylcopper: a useful difluoromethylene transfer reagent: a novel double insertion of difluoromethylene into pentafluorophenylcopper. J. Am. Chem. Soc. 114, 4402–4403 (1992).
Li, L. et al. TMSCF3 as a convenient source of CF2=CF2 for pentafluoroethylation, (aryloxy)tetrafluoroethylation, and tetrafluoroethylation. Angew. Chem. Int. Ed. 56, 9971–9975 (2017).
Han, W. et al. Experimental and chemical kinetic study of the pyrolysis of trifluoroethane and the reaction of trifluoromethane with methane. J. Fluorine Chem. 131, 751–760 (2010).
Hauptschein, M. & Fainberg, A. H. Production of fluorinated compounds. US patent 3,009,966 (1960).
Thomoson, C. S. & Dolbier, W. R. Use of fluoroform as a source of difluorocarbene in the synthesis of difluoromethoxy- and difluorothiomethoxyarenes. J. Org. Chem. 78, 8904–8908 (2013).
Goetz, A. E. et al. Large-scale cyclopropanation of butyl acrylate with difluorocarbene and classical resolution of a key fluorinated building block. Org. Process. Dev. 26, 683–697 (2022).
Yoshida, J.-I., Takahashi, Y. & Nagaki, A. Flash chemistry: flow chemistry that cannot be done in batch. Chem. Commun. 49, 9896–9904 (2013).
Sergeev, A. P., Novozhilova, T. I. & Yakubovich, A. Y. Direct fluoroalkenylation reactions. VI. Improvement of the synthesis method of a fluoroalkenylamine. Zh. Org. Khim. 6, 747–754 (1970).
Yakubovich, A. Y., Sergeev, A. P. & Novozhilova, T. I. Direct fluoroalkenylation reactions. IX. Some reactions to fluoroalkenylamines. Zh. Org. Khim. 6, 2192–2196 (1970).
Yakubovich, A. Y. & Sergeev, A. P. Direct fluoroalkenylation reactions. IV. Kinetics of hydrolysis of α-fluoro and α-bromovinylamines. Zh. Obshch. Khim. 37, 842–846 (1967).
Sergeev, A. P. & Yakubovich, A. Y. Direct fluoroalkenylation reactions. II. Synthesis of α-brominated fluorovinylamines. Zh. Obshch. Khim. 36, 1801–1807 (1966).
Belyaeva, I. N., Smolyanitskaya, V. V., Gitel, P. O., Sergeev, A. P. & Yakubovich, A. Y. Direct fluoroalkenylation reactions. III. Synthesis of N-fluorovinylcarbazoles. Zh. Obshch. Khim. 36, 1793–1797 (1966).
Yakubovich, A. Y., Sergeev, A. P. & Belyaeva, I. N. Direct fluorovinylation reaction. Dokl. Akad. Nauk SSSR 161, 1362–1364 (1965).
Voropaeva, A. V. & Fel’dman, I. K. Synthesis in the pyridine series. VII. Quaternary salts of some β-aminoalkyl 2-pyridyl sulfides. Chem. Heterocycl. Compd. 1, 177–179 (1965).
Anastassiou, A. G., Elliott, R. L. & Reichmanis, E. Effect of heteroatom electronegativity on the development of diatropic character in cis,trans,cis,trans,cis,trans-aza[13]annulenes. J. Am. Chem. Soc. 96, 7823–7825 (1974).
Evano, G., Leconte, M., Thilmany, P. & Theunissen, C. Keteniminium ions: unique and versatile reactive intermediates for chemical synthesis. Synthesis 49, 3183–3214 (2017).
Madelaine, C., Valerio, V. & Maulide, N. Revisiting keteniminium salts: more than the nitrogen analogs of ketenes. Chem. Asian J. 6, 2224–2239 (2011).
Capaldo, L., Wen, Z. & Noël, T. A field guide to flow chemistry for synthetic organic chemists. Chem. Sci. 14, 4230–4247 (2023).
Alfano, A. I., Garcia-Lacuna, J., Griffiths, O. M., Ley, S. V. & Baumann, M. Continuous flow synthesis enabling reaction discovery. Chem. Sci. 15, 4618–4630 (2024).
Box, J. R., Atkins, A. P. & Lennox, A. J. J. Direct electrochemical hydrodefluorination of trifluoromethylketones enabled by non-protic conditions. Chem. Sci. 12, 10252–10258 (2021).
Ispizua-Rodriguez, X., Munoz, S. B., Krishamurti, V., Mathew, T. & Prakash, G. K. S. Direct synthesis of tri-/difluoromethyl ketones from carboxylic acids by cross-coupling with acyloxyphosphonium ions. Chem. Eur. J. 27, 15908–15913 (2021).
Acknowledgements
J.D., T.P. and P.J. acknowledge Normandy Valorisation (FLUORO project) for funding. J.D., I.M. and T.P. acknowledge the RIN ZIRCO_PHOTO_COP programme (Normandy Region, ERDF/FSE 2021–2027, no. 21E05645) for funding. I.M. and T.P. acknowledge the PHOTODERACS programme (Normandy Region, no. 0015464) for funding. P.I., T.P. and P.J. acknowledge the RIN CYLIA programme (Normandy Region ERDF/FSE 2021–2027, no. 22E02979) for funding. We acknowledge the API programme and NFC (NormandyFlowChem platform) supported by the European Union through the operational programme (Normandy Region, ERDF/FSE 2014–2020, no. 20E04976) and FNADT-DRACCARE (programme 2020–2023 (N)15179) for funding. Normandie Université (NU), Région Normandie, CNRS, Université Rouen Normandie, INSA Rouen Normandie, Labex SynOrg (ANR-11-LABX0029), the graduate school for research XL–Chem (ANR-18-EURE-0020 XL CHEM) and Innovation Chimie Carnot (I2C) are acknowledged for support.
Author information
Authors and Affiliations
Contributions
P.I., J.D., T.P. and P.J. conceived the project. P.I., J.D. and I.M. performed the synthetic work. M.H. performed the NMR spectroscopy studies under the supervision of H.O. Funding was secured by T.P. and P.J. who also supervised the research and wrote the paper with contributions from all authors.
Corresponding authors
Ethics declarations
Competing interests
P.I., J.D., T.P. and P.J. are inventors on an international patent application PCT 2024/051749, held by INSA Rouen Normandie, Université Rouen Normandie and CNRS and submitted by Normandie Valorisation, which covers the synthesis of the TFVA reagents and their reactivity. The other authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Gavin Tsui and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Peter Seavill, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–38, Tables 1–4, Schemes 1–3 and experimental details.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ivashkin, P., Decaens, J., Marchand, I. et al. Fluoroform upcycling to trifluorovinylamine as a C2 reagent to prepare difluoromethylated molecules. Nat. Synth (2026). https://doi.org/10.1038/s44160-026-01001-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-026-01001-y