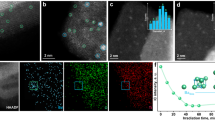
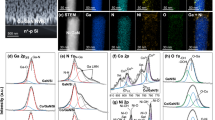
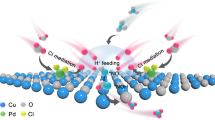
Abstract
Despite the development of the Haber–Bosch process, ammonia synthesis under mild conditions remains challenging due to the high bond energy (945 kJ mol⁻1) of the N≡N triple bond. Both thermal- and photocatalytic processes often suffer from the intrinsic scaling relationship between N2 activation and NH3 desorption efficiencies. Here we report that the photocatalytic process over an AlFe nanoalloy catalyst provides a promising solution through a photoinduced nitrogen spillover reaction mechanism. Fe acts as the primary active site for N2 adsorption and dissociation. The transferred photoexcited electrons from Al to Fe enhance N2 activation. Al serves as a secondary active site, facilitating N spillover from Fe to Al sites under photoexcitation, promoting NH3 desorption. This dual-site strategy enables an ammonia synthesis rate of 8.6 mmol gcat−1 h−1 at 4.28 W cm−2 without additional thermal input under ambient pressure. The performance surpasses that of conventional industrial Fe catalysts under thermocatalytic conditions. This study proposes a photoassisted active site modulation strategy for efficient ammonia synthesis catalyst to circumvent scaling relationships.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the article and its Supplementary Information. Source data are provided with this paper.
References
Fu, X. et al. Comtinuous-flow electrosynthesis of ammonia by nitrogen reduction and hydrogen oxidation. Science 379, 707–712 (2023).
Ye, T. N. et al. Vacancy-enabled N2 activation for ammonia synthesis on an Ni-loaded catalyst. Nature 583, 391–395 (2020).
Li, W. Q., Xu, M., Chen, J. S. & Ye, T. N. Enabling sustainable ammonia Synthesis: from nitrogen activation strategies to emerging materials. Adv. Mater. 36, 2408434 (2024).
Kitano, M. et al. Ammonia synthesis using a stable electride as an electron donor and reversible hydrogen store. Nat. Chem. 4, 934–940 (2012).
Kobayashi, Y. et al. Titanium-based hydrides as heterogeneous catalysts for ammonia synthesis. J. Am. Chem. Soc. 139, 18240–18246 (2017).
Ye, T. N. et al. Contribution of nitrogen vacancies to ammonia synthesis over metal ntride catalysts. J. Am. Chem. Soc. 142, 14374–14383 (2020).
Chang, F., Gao, W., Guo, J. & Chen, P. Emerging materials and methods toward ammonia-based energy storage and conversion. Adv. Mater. 33, 2005721 (2021).
Wang, T. & Abild-Pedersen, F. Achieving industrial ammonia synthesis rates at near-ambient conditions through modified scaling relations on a confined dual site. Proc. Natl Acad. Sci. USA 118, 2106527118 (2021).
Gong, Y. et al. Ternary intermetallic LaCoSi as a catalyst for N2 activation. Nat. Catal. 1, 178–185 (2018).
Shi, Y. et al. Engineering photocatalytic ammonia synthesis. Chem. Soc. Rev. 52, 6938–6956 (2023).
Hattori, M., Miyashita, K., Nagasawa, Y., Suzuki, R. & Hara, M. Ammonia synthesis over an iron catalyst with an inverse structure. Adv. Sci. 12, 2410313 (2025).
Al Maksoud, W. et al. Silicon clathrate-supported catalysts with low work functions for ammonia synthesis. Adv. Mater. 36, 2406944 (2024).
Zhang, Z. et al. Anion vacancies activate N2 to ammonia on Ba–Si orthosilicate oxynitride-hydride. Nat. Chem. 17, 679–687 (2025).
Zhang, Y. et al. Fullerene on non-iron cluster-matrix co-catalysts promotes collaborative H2 and N2 activation for ammonia synthesis. Nat. Chem. 16, 1781–1787 (2024).
Jiang, Y. et al. Single-atom Fe catalysts with improved metal loading for efficient ammonia synthesis under mild conditions. Angew. Chem. Int. Ed. 64, e202501190 (2025).
Sfeir, A. et al. Unlocking the potential of MXene in catalysis: decorated Mo2CTx catalyst for ammonia synthesis under mild conditions. J. Am. Chem. Soc. 146, 20033–20044 (2024).
Chang, F. et al. Potassium hydride-intercalated graphite as an efficient heterogeneous catalyst for ammonia synthesis. Nat. Catal. 5, 222–230 (2022).
Kim, J.-H. et al. Achieving volatile potassium promoted ammonia synthesis via mechanochemistry. Nat. Commun. 14, 2319 (2023).
Han, G.-F. et al. Mechanochemistry for ammonia synthesis under mild conditions. Nat. Nanotechnol. 16, 325–330 (2020).
Monyoncho, E. A. & Dasog, M. Photocatalytic plasmon-enhanced nitrogen reduction to ammonia. Adv. Energy Sustain. Res. 2, 2000055 (2021).
Puértolas, B., Comesaña-Hermo, M., Besteiro, L. V., Vázquez-González, M. & Correa-Duarte, M. A. Challenges and opportunities for renewable ammonia production via plasmon-assisted photocatalysis. Adv. Energy Mater. 12, 2103909 (2022).
Wang, S. et al. Light-switchable oxygen vacancies in ultrafine Bi5O7Br nanotubes for boosting solar-driven nitrogen fixation in pure water. Adv. Mater. 29, 1701774 (2017).
Hu, C. et al. Surface plasmon enabling nitrogen fixation in pure water through a dissociative mechanism under mild conditions. J. Am. Chem. Soc. 141, 7807–7814 (2019).
Jia, H. et al. Site-selective growth of crystalline ceria with oxygen vacancies on gold nanocrystals for near-infrared nitrogen photofixation. J. Am. Chem. Soc. 141, 5083–5086 (2019).
Yang, J. et al. High-efficiency ‘working-in-tandem’ nitrogen photofixation achieved by assembling plasmonic gold nanocrystals on ultrathin titania Nanosheets. J. Am. Chem. Soc. 140, 8497–8508 (2018).
Guan, Y. et al. Light-driven ammonia synthesis under mild conditions using lithium hydride. Nat. Chem. 16, 373–379 (2024).
Swearer, D. F., Knowles, N. R., Everitt, H. O. & Halas, N. J. Light-driven chemical looping for ammonia synthesis. ACS Energy Lett. 4, 1505–1512 (2019).
Mao, C. et al. Photochemical acceleration of ammonia production by Pt1–Ptn–TiN reduction and N2 activation. J. Am. Chem. Soc. 145, 13134–13146 (2023).
Zhou, L. et al. Quantifying hot carrier and thermal contributions in plasmonic photocatalysis. Science 362, 69–72 (2018).
Collado, L. et al. Light-driven nitrogen fixation routes for green ammonia production. Chem. Soc. Rev. 53, 11334–11389 (2024).
Mao, C. et al. Beyond the thermal equilibrium limit of ammonia synthesis with dual temperature zone catalyst powered by solar light. Chem 5, 2702–2717 (2019).
Yang, Y. et al. Regulating the scaling relations in ammonia synthesis through a light-driven bendable seesaw effect on tailored iron catalyst. Angew. Chem. Int. Ed. 136, 202408309 (2024).
Nørskov, J. K., Bligaard, T., Rossmeisl, J. & Christensen, C. H. Towards the computational design of solid catalysts. Nat. Chem. 1, 37–46 (2009).
Samanta, B., Sengupta, T. & Pal, S. Specificity of amino acid–aluminum cluster interaction and subsequent oxygen activation by the above complex. J. Phys. Chem. C 122, 28310–28323 (2018).
Song, B. et al. Irreversible denaturation of proteins through aluminum-induced formation of backbone ring structures. Angew. Chem. Int. Ed. 53, 6358–6363 (2014).
Dhindsa, P. et al. Light-driven dehydrogenation of propane using plasmonic Al@TiO2 core–shell nanoparticles with Pt single atoms and clusters. ACS Energy Lett. 9, 6047–6054 (2024).
Eelman, D. A., Dahn, J. R., MacKay, G. R. & Dunlap, R. A. An investigation of mechanically alloyed Fe–Al. J. Alloys Compd. 266, 234–240 (1998).
Bilovol, V. et al. Al-doped BaFe12O19 hexaferrites: dopant distribution in the crystal lattice and its effect on the magnetic order. J. Magn. Magn. Mater. 596, 171913 (2024).
Zitolo, A. et al. Identification of catalytic sites for oxygen reduction in iron- and nitrogen-doped Graphene Materials. Nat. Mater. 14, 937–942 (2015).
Chmielewski, T. et al. Phase structure evolution of the Fe–Al Arc-sprayed coating stimulated by annealing. Materials 14, 3210 (2021).
Yuan, Y. et al. Steam methane reforming using a regenerable antenna–reactor plasmonic photocatalyst. Nat. Catal. 7, 1339–1349 (2024).
Robatjazi, H. et al. Plasmon-driven carbon–fluorine (C(sp3)–F) bond activation with mechanistic insights into hot-carrier-mediated pathways. Nat. Catal. 3, 564–573 (2020).
Yuan, Y. et al. Earth-abundant photocatalyst for H2 generation from NH3 white light-emitting diode illumination. Science 378, 889–893 (2022).
Li, H., Shang, J., Ai, Z. & Zhang, L. Efficient visible light nitrogen fixation with BiOBr nanosheets of oxygen vacancies on the exposed {001} facets. J. Am. Chem. Soc. 137, 6393–6399 (2015).
Mateo, D., Cerrillo, J. L., Durini, S. & Gascon, J. Fundamentals and applications of photo-thermal catalysis. Chem. Soc. Rev. 50, 2173–2210 (2021).
Zhou, L. et al. Aluminum nanocrystals as a plasmonic photocatalyst for hydrogen dissociation. Nano Lett. 16, 1478–1484 (2016).
Bian, X. et al. Quantifying the contribution of hot electrons in photothermal catalysis: a case study of ammonia synthesis over carbon-supported Ru catalyst. Angew. Chem. Int. Ed. 62, 202304452 (2023).
Aika, K. I. & Ozaki, A. Kinetics and isotope effect of ammonia synthesis over a singly-promoted iron catalyst. J. Catal. 19, 350–352 (1970).
Hagen, S. Ammonia synthesis with barium-promoted iron–cobalt alloys supported on carbon. J. Catal. 214, 327–335 (2003).
Zhang, Z.-Q. et al. Nano-island-encapsulated cobalt single-atom catalysts for breaking activity-stability trade-off in Fenton-like reactions. Nat. Commun. 16, 115 (2025).
Illas, F. Fundamental concepts in heterogeneous catalysis. Angew. Chem. Int. Ed. 54, 10404–10405 (2015).
Zhang, L. et al. Oxophilicity-controlled CO2 electroreduction to C2+ alcohols over lewis acid metal-doped Cuδ+ Catalysts. J. Am. Chem. Soc. 145, 21945–21954 (2023).
Herran, M. et al. Plasmonic bimetallic two-dimensional supercrystals for H2 generation. Nat. Catal. 6, 1205–1214 (2023).
Li, J. et al. Utilizing full-spectrum sunlight for ammonia decomposition to hydrogen over GaN nanowires-supported Ru nanoparticles on silicon. Nat. Commun. 15, 7393 (2024).
Winter, L. R., Ashford, B., Hong, J., Murphy, A. B. & Chen, J. G. Identifying surface reaction intermediates in plasma catalytic ammonia synthesis. ACS Catal. 10, 14763–14774 (2020).
Collado, L. et al. Unravelling the effect of charge dynamics at the plasmonic metal/semiconductor interface for CO2 photoreduction. Nat. Commun. 9, 4986 (2018).
Goodwin, C. M. et al. Operando probing of the surface chemistry during the Haber–Bosch process. Nature 625, 282–286 (2024).
You, D. et al. Interface control for pure ultraviolet electroluminescence from nano-ZnO-based heterojunction devices. Sci. Bull. 63, 38–45 (2018).
Kuball, M. Raman spectroscopy of GaN, AlGaN and AlN for process and growth monitoring/control. Surf. Interface Anal. 31, 987–999 (2001).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Kresse, G. & Hafner, J. Ab initiomolecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Henkelman, G. & Jónsson, H. Improved tangent estimate in the nudged elastic band method for finding minimum energy paths and saddle points. J. Chem. Phys. 113, 9978–9985 (2000).
Henkelman, G., Uberuaga, B. P. & Jónsson, H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 113, 9901–9904 (2000).
Dronskowski, R. & Blöchl, P. E. Crystal orbital Hamilton populations. Energy-resolved visualization of chemical bonding in solids based on density-functional calculations. J. Phys. Chem. 97, 8617–8624 (1993).
Maintz, S., Deringer, V. L., Tchougréeff, A. L. & Dronskowski, R. Analytic projection from plane-wave and PAW wavefunctions and application to chemical-bonding analysis in solids. J. Comput. Chem. 34, 2557–2567 (2013).
Maintz, S., Deringer, V. L., Tchougréeff, A. L. & Dronskowski, R. LOBSTER: a tool to extract chemical bonding from plane-wave based DFT. J. Comput. Chem. 37, 1030–1035 (2016).
Deringer, V. L., Tchougréeff, A. L. & Dronskowski, R. Crystal orbital Hamilton population (COHP) analysis as projected from plane-wave basis sets. J. Phys. Chem. A 115, 5461–5466 (2011).
Causà, M., D’Amore, M., Gentile, F., Menendez, M. & Calatayud, M. Electron localization function and maximum probability domains analysis of semi-ionic oxides crystals, surfaces and surface defects. Comput. Theor. Chem. 1053, 315–321 (2015).
Becke, A. D. & Edgecombe, K. E. A simple measure of electron localization in atomic and molecular systems. J. Chem. Phys. 92, 5397–5403 (1990).
Momma, K. & Izumi, F. VESTA-3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 44, 1272–1276 (2011).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant nos. 22275121 and 92580124), the National Key R&D Program of China (grant nos. 2023YFA1506300 and 2023YFB4005201), Shanghai Municipal Science and Technology Major Project, Shanghai Municipal Education Commission (grant no. 2024AIYB007), and Fundamental Research Funds for the Central Universities (grant nos. 23×010301599 and 24×010301678). We thank the Shanghai Synchrotron Radiation Facility of BL02B01 (31124.02.SSRF.BL02B01) for the assistance on NAP-XPS measurements.
Author information
Authors and Affiliations
Contributions
T.-N.Y. conceived the idea and directed the project. J.-S.C. supervised the project. W.-Q.L. designed the experiments and conducted most of the synthetic experiments. Y.C., Y.L. and X.-H.L. performed the DFT calculations. X. Lu and J.W. helped conduct in situ FTIR. H.Z., X. Liu and Z. Liu helped perform NAP-XPS measurements. Z. Li provided acquired XPS data. K.Q. and B.D. helped perform the TPD measurements. R.J. and S.Z. provided assistance with XAS data acquisition and analysis. X.Z. provided Rietveld refinement analysis of XRD patterns. Y.Q. helped conduct magnetization measurements. B.Z. provided assistance with the Mössbauer spectra data. T.-N.Y. and W.-Q.L. cowrote the paper. All authors revised and approved the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Laura Collado, Zhenyu Sun and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alexandra Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–34 and Tables 1–8.
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, WQ., Chen, Y., Lu, X. et al. Photoinduced nitrogen spillover enables ammonia synthesis on iron–aluminium dual-site catalysts. Nat. Synth (2026). https://doi.org/10.1038/s44160-026-01013-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-026-01013-8