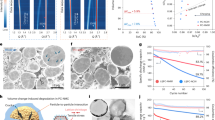
Abstract
Transition-metal nanocrystal (TMN) catalysts are essential for energy conversion and storage, but their facile and controllable synthesis remains challenging. Here we report a general precursor-valence-gating strategy to prepare various monocrystalline and polycrystalline TMNs (MC/PC-TMNs, M = Co, Ni or Cu). Unlike complex regulation of traditional experimental parameters, we discover that the solid-state precursor’s oxidation state—an intrinsic parameter—governs the nucleation rate and ultimately dictates atomic crystallization periodicity. Consequently, the controllable preparation of MC/PC-TMNs was readily achieved through designing the valence diversity of the corresponding precursors. Furthermore, taking MC/PC-Co catalysts as examples, we investigate the correlation between atomic arrangements and catalytic dynamics and reveal that the PC-Co catalyst with the lower-coordination environment, compared with MC-Co, substantially promotes the conversion of reaction intermediates in the hydrazine oxidation reaction. As a result, PC-Co exhibits an ultralow Tafel slope of 14 mV dec−1, and an assembled hydrazine oxidation reaction-based battery demonstrates excellent stability for 1,200 h at 10 mA cm−2. This work paves the way for controllable synthesis of MC/PC-TMNs and fundamentally advances the understanding of precursor-guided crystal growth kinetics.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data that support the findings of this study are available in the article and its Supplementary Information. Source data are provided with this paper.
References
Liang, J. et al. Atomic arrangement engineering of metallic nanocrystals for energy-conversion electrocatalysis. Joule 3, 956–991 (2019).
Lin, F., Li, M., Zeng, L., Luo, M. & Guo, S. Intermetallic nanocrystals for fuel-cells-based electrocatalysis. Chem. Rev. 123, 12507–12593 (2023).
Xia, Y., Xiong, Y., Lim, B. & Skrabalak, S. E. Shape-Controlled synthesis of metal nanocrystals: simple chemistry meets complex physics?. Angew. Chem. Int. Ed. 48, 60–103 (2008).
Zhao, K., Luo, M., Zhang, Y., Chang, X. & Xu, B. Coupled cation–electron transfer at the Pt(111)/perfluoro-sulfonic acid ionomer interface and its impact on the oxygen reduction reaction kinetics. Nat. Catal. 8, 46–57 (2025).
Kim, S.-J., Maligal-Ganesh, R. V., Mahmood, J., Babar, P. & Yavuz, C. T. Structural control over single-crystalline oxides for heterogeneous catalysis. Nat. Rev. Chem. 9, 397–414 (2025).
Yang, Y. et al. Operando studies reveal active Cu nanograins for CO2 electroreduction. Nature 614, 262–269 (2023).
Geng, X. et al. Grain boundary engineering for efficient and durable electrocatalysis. Nat. Commun. 15, 8534 (2024).
Tao, J. et al. Synthesis of noble metal nanoarrays via agglomeration and metallurgy for acidic water electrolysis. Nat. Commun. 16, 4996 (2025).
Huang, L. L. et al. Shape regulation of high-index facet nanoparticles by dealloying. Science 365, 1159–1163 (2019).
Zhao, H. et al. Atomic-scale structure dynamics of nanocrystals revealed by in situ and environmental transmission electron microscopy. Adv. Mater. 35, 2206911 (2023).
Wu, K.-J., Tse, E. C. M., Shang, C. & Guo, Z. Nucleation and growth in solution synthesis of nanostructures—from fundamentals to advanced applications. Prog. Mater. Sci. 123, 100821 (2022).
Karthika, S., Radhakrishnan, T. K. & Kalaichelvi, P. A review of classical and nonclassical nucleation theories. Cryst. Growth Des. 16, 6663–6681 (2016).
Chen, G. et al. Direct synthesis of hydrazine by efficient electrochemical ruthenium-catalysed ammonia oxidation. Nat. Catal. 6, 949–958 (2023).
Chai, Y. X. et al. Environmental and economic assessment of advanced oxidation for the treatment of unsymmetrical dimethylhydrazine wastewater from a life cycle perspective. Sci. Total Environ. 873, 162264 (2023).
Sun, F. et al. Energy-saving hydrogen production by chlorine-free hybrid seawater splitting coupling hydrazine degradation. Nat. Commun. 12, 4182 (2021).
Zhu, L. et al. Active site recovery and N-N bond breakage during hydrazine oxidation boosting the electrochemical hydrogen production. Nat. Commun. 14, 1997 (2023).
Feng, Y. et al. Decoupled electrochemical hydrazine ‘splitting’ via a rechargeable Zn–hydrazine battery. Adv. Mater. 34, 2207747 (2022).
Liu, Q. et al. Low-coordinated cobalt arrays for efficient hydrazine electrooxidation. Energy Environ. Sci. 15, 3246–3256 (2022).
Zhu, W. et al. A hydrazine-nitrate flow battery catalyzed by a bimetallic RuCo precatalyst for wastewater purification along with simultaneous generation of ammonia and electricity. Angew. Chem. Int. Ed. 62, e202300390 (2023).
Fei, H. et al. General synthesis and definitive structural identification of MN4C4 single-atom catalysts with tunable electrocatalytic activities. Nat. Catal. 1, 63–72 (2018).
Tang, B. et al. A Janus dual-atom catalyst for electrocatalytic oxygen reduction and evolution. Nat. Synth. 3, 878–890 (2024).
Xiao, X. et al. Rational design of flexible Zn-based batteries for wearable electronic devices. ACS Nano 17, 1764–1802 (2023).
Wang, B. et al. Sulfion oxidation assisting self-powered hydrogen production system based on efficient catalysts from spent lithium-ion batteries. Proc. Natl Acad. Sci. USA 120, e2317174120 (2023).
Yang, Y. et al. Hierarchical Ohmic contact interface engineering for efficient hydrazine-assisted hydrogen evolution reaction. Adv. Mater. 36, 2307979 (2023).
Feng, C. et al. Lattice strain engineering of Ni2P enables efficient catalytic hydrazine oxidation-assisted hydrogen production. Adv. Mater. 35, 2305598 (2023).
Pang, K. et al. Redirecting configuration of atomically dispersed selenium catalytic sites for efficient hydrazine oxidation. Matter 7, 655–667 (2024).
Fu, H. Q. et al. Hydrogen spillover-bridged Volmer/Tafel processes enabling ampere-level current density alkaline hydrogen evolution reaction under low overpotential. J. Am. Chem. Soc. 144, 6028–6039 (2022).
Zhang, S. et al. Integrating electrophilic and nucleophilic dual sites on heterogeneous bimetallic phosphide via enhancing interfacial electronic field to boost hydrazine oxidation and hydrogen evolution. Appl. Catal. B 324, 122207 (2023).
Hu, Y. et al. Cooperative Ni(Co)-Ru-P sites activate dehydrogenation for hydrazine oxidation assisting self-powered H2 production. Angew. Chem. Int. Ed. 62, 202308800 (2023).
Zhang, S., Wei, X., Dai, S., Wang, H. & Huang, M. Efficient hydrazine electro-oxidation achieved by tailored electron injection into Fe (III) sites activating dehydrogenation. Adv. Funct. Mater. 34, 2311370 (2023).
Guan, X. et al. Identifying the roles of Ru single atoms and nanoclusters for energy-efficient hydrogen production assisted by electrocatalytic hydrazine oxidation. Appl. Catal. B 323, 122145 (2023).
Zhong, X. et al. Rechargeable zinc–air batteries with an ultra-large discharge capacity per cycle and an ultra-long cycle life. Adv. Mater. 35, e2301952 (2023).
Liu, Z. et al. Breaking the reversibility barrier in Zn–air systems with dual-electrolyte engineering. Adv. Mater. 37, 2507851 (2025).
Zhong, X. et al. Flexible zinc–air batteries with ampere-hour capacities and wide-temperature adaptabilities. Adv. Mater. 35, e2209980 (2023).
Li, H. et al. Dual-plating aqueous Zn–iodine batteries enabled by halogen-complexation chemistry for large-scale energy storage. Energy Environ. Sci. 18, 3160–3168 (2025).
Zhang, Q. et al. Constructing bipolar dual-active sites through high-entropy-induced electric dipole transition for decoupling oxygen redox. Adv. Mater. 36, 2401018 (2024).
Li, T. et al. Ultrafast synthesis of an efficient urea oxidation electrocatalyst for urea-assisted fast-charging Zn–air batteries and water splitting. Energy Environ. Sci. 18, 4996–5008 (2025).
Li, Y. et al. Main-group element-boosted oxygen electrocatalysis of Cu–N–C sites for zinc–air battery with cycling over 5000 h. Nat. Commun. 15, 8365 (2024).
Jiang, P. et al. Entropy-driven competitive adsorption sites tailoring unlocks efficient hybrid conversion Zn–air batteries. Angew. Chem. Int. Ed. 64, e202504188 (2025).
Qiao, J. et al. Precisely constructing orbital-coupled Fe─Co dual-atom sites for high-energy-efficiency Zn–air/iodide hybrid batteries. Adv. Mater. 36, 7 (2024).
Zhang, G. et al. Copper collector generated Cu+/Cu2+ redox pair for enhanced efficiency and lifetime of Zn–Ni/air hybrid battery. ACS Nano 16, 17139–17148 (2022).
Li, Z. et al. Zinc–alcohol–air batteries with ultra-narrow cyclic voltage windows. Energy Environ. Sci. 18, 1002–1010 (2025).
Wei, Z. et al. Starch-mediated colloidal chemistry for highly reversible zinc-based polyiodide redox flow batteries. Nat. Commun. 15, 3841 (2024).
Wang, C. et al. High-voltage and dendrite-free zinc-iodine flow battery. Nat. Commun. 15, 6234 (2024).
Wang, Q. et al. Rescue of dead MnO2 for stable electrolytic Zn–Mn redox-flow battery: a metric of mediated and catalytic kinetics. Natl Sci. Rev. 11, nwae230 (2024).
Huang, S. et al. A universal coulombic efficiency compensation strategy for zinc-based flow batteries. Adv. Mater. 36, e2406366 (2024).
De Luna, P. et al. What would it take for renewably powered electrosynthesis to displace petrochemical processes?. Science 364, eaav3506 (2019).
Wang, B. et al. Rational design of NiFe LDH@Ni3N nano/microsheet arrays as a bifunctional electrocatalyst for overall water splitting. J. Mater. Chem. A 8, 17202–17211 (2020).
Lin, C. et al. In-situ reconstructed Ru atom array on α-MnO2 with enhanced performance for acidic water oxidation. Nat. Catal. 4, 1012–1023 (2021).
Hohenberg, P. & Kohn, W. Inhomogeneous electron gas. Phys. Rev. 136, B864–B871 (1964).
Kohn, W. & Sham, L. J. Self-consistent equations including exchange and correlation effects. Phys. Rev. 140, A1133–A1138 (1965).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Wang, V., Xu, N., Liu, J.-C., Tang, G. & Geng, W.-T. VASPKIT: a user-friendly interface facilitating high-throughput computing and analysis using VASP code. Comput. Phys. Commun. 267, 108033 (2021).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
Kühne, T. D. et al. CP2K: an electronic structure and molecular dynamics software package-Quickstep: efficient and accurate electronic structure calculations. J. Chem. Phys. 152, 194103 (2020).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant no. 92572103, G.Z.), Guangdong Basic and Applied Basic Research Foundation (2023B1515120099, G.Z.), Guangdong Innovative and Entrepreneurial Research Team Program (2021ZT09L197, G.Z.), and Shenzhen Science and Technology Program (KQTD20210811090112002, G.Z.). This work made use of the TEM facilities at the Institute of Materials Research, Tsinghua Shenzhen International Graduate School (Tsinghua SIGS).
Author information
Authors and Affiliations
Contributions
G.Z. and Z.Z. conceived the project. Z.Z., T.L., B.W. and G.J. synthesized the materials. Z.Z. and F.-Y.Z. carried out the materials characterization and analysed the data. Z.Z. conducted the TEAs. Y.Z. and B.H. conducted theoretical simulations. X.Z., Z.L. and Q.F. provided experimental insights. All authors participated in paper preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Hanfeng Liang, Duy Thanh Tran and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Peter Seavill, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–14, Notes 1–3 and Tables 1–8.
Supplementary Data 1 (download XLSX )
Source data for Supplementary Figs. 1–14.
Source data
Source Data Fig. 1 (download XLSX )
Source data for Fig. 1.
Source Data Fig. 2 (download XLSX )
Source data for Fig. 2.
Source Data Fig. 3 (download XLSX )
Source data for Fig. 3.
Source Data Fig. 4 (download XLSX )
Source data for Fig. 4.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, Z., Zheng, FY., Zhu, Y. et al. A precursor-valence-gating strategy for controllable synthesis of metal nanocrystal catalysts with varied periodicities. Nat. Synth (2026). https://doi.org/10.1038/s44160-026-01022-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-026-01022-7