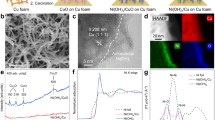
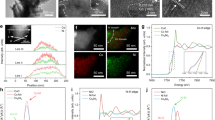
Abstract
Plasma-electrolysis tandem systems can facilitate ammonia (NH3) production from air and water under ambient conditions via an ‘air → NOx → NOx− → NH3’ pathway. However, NH3 yields are limited due to a lack of process intensification between plasma generation and electrocatalytic conversion. Here we report a modified cobalt cathode enriched with twin boundaries (TBs) and stacking faults (SFs) (TB/SF-Co) for selective NO2− reduction. Mechanistic analyses reveal that TB/SF defects upshift the Co d-band centre which strengthens NO2− adsorption and promotes hydrogenation of intermediates. The material achieves a Faradaic efficiency of 96.7% for NO2−-to-NH3 conversion at a partial current density of −1.73 A cm−2. Coupling a cobalt cathode enriched with twin boundaries and stacking faults with a microwave plasma module enables efficient air-to-NH3 synthesis, achieving an air-to-NO2− rate of 0.745 mol h−1 and NO2−-to-NH3 rate of 0.619 mol h−1 with >95% Faradaic efficiency at a total current of more than 101 A in 100 cm2 parallel electrolyzers, which rivals or exceeds contemporary benchmarks.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in the published article and its Supplementary Information. Source data are provided with this paper.
References
MacFarlane, D. R. et al. A roadmap to the ammonia economy. Joule 4, 1186–1205 (2020).
Guo, J. & Chen, P. Catalyst: NH3 as an energy carrier. Chem 3, 709–712 (2017).
Jiang, M. et al. Effective N2 activation strategies for electrochemical ammonia synthesis. Chem 11, 102441 (2025).
Ye, D. & Tsang, S. C. E. Prospects and challenges of green ammonia synthesis. Nat. Synth. 2, 612–623 (2023).
Guo, W., Zhang, K., Liang, Z., Zou, R. & Xu, Q. Electrochemical nitrogen fixation and utilization: theories, advanced catalyst materials and system design. Chem. Soc. Rev. 48, 5658–5716 (2019).
Qing, G. et al. Recent advances and challenges of electrocatalytic N2 reduction to ammonia. Chem. Rev. 120, 5437–5516 (2020).
Suryanto, B. H. R. et al. Challenges and prospects in the catalysis of electroreduction of nitrogen to ammonia. Nat. Catal. 2, 290–296 (2019).
Li, S., Fu, X., Nørskov, J. K. & Chorkendorff, I. Towards sustainable metal-mediated ammonia electrosynthesis. Nat. Energy 9, 1344–1349 (2024).
John, J., MacFarlane, D. R. & Simonov, A. N. The why and how of NOx electroreduction to ammonia. Nat. Catal. 6, 1125–1130 (2023).
Simonov, A. N., Koper, M. T. M. & MacFarlane, D. R. Practical prospects of electroreduction of dilute NOx streams to ammonia. Nat. Chem. Eng. 2, 398–401 (2025).
Liu, W. et al. Efficient ammonia synthesis from the air using tandem non-thermal plasma and electrocatalysis at ambient conditions. Nat. Commun. 15, 3524 (2024).
Guo, X. et al. Highly stable perovskite oxides for electrocatalytic acidic NOx− reduction streamlining ammonia synthesis from air. Angew. Chem. Int. Ed. 64, e202410517 (2024).
Ge, X. Controlling the reaction pathways of mixed NOxHy reactants in plasma-electrochemical ammonia synthesis. J. Am. Chem. Soc. 146, 35305–35312 (2024).
Ren, Y. et al. Microscopic-level insights into the mechanism of enhanced NH3 synthesis in plasma-enabled cascade N2 oxidation-electroreduction system. J. Am. Chem. Soc. 144, 10193–10200 (2022).
Liu, W. et al. Full runner electrolyzer stack for industrial-current-density NOx−-mediated ammonia synthesis from air and water. Nat. Commun. 16, 5716 (2025).
Liang, W. P. et al. Cascade N2 reduction process with DBD plasma oxidation and electrocatalytic reduction for continuous ammonia synthesis. Energy Environ. Sci. 57, 14558–14568 (2023).
Ding, J. et al. A cascade jet plasma oxidation-electroreduction system using Pd-Ni dual-site catalyst for sustainable ammonia production from air. Adv. Funct. Mater. 34, 2410768 (2024).
Wu, A. et al. Direct ammonia synthesis from the air via gliding arc plasma integrated with single atom electrocatalysis. Appl. Catal. B 299, 120667 (2021).
Meng, S.-L. et al. Cobaloxime: selective nitrite reduction catalysts for tandem ammonia synthesis. Energy Environ. Sci. 16, 1590–1596 (2023).
Sun, J. et al. A hybrid plasma electrocatalytic process for sustainable ammonia production. Energy Environ. Sci. 14, 865–872 (2021).
Liu, Y. et al. Efficient tandem electroreduction of nitrate into ammonia through coupling Cu single atoms with adjacent Co3O4. Nat. Commun. 15, 3619 (2024).
Chen, F.-Y. et al. Efficient conversion of low-concentration nitrate sources into ammonia on a Ru-dispersed Cu nanowire electrocatalyst. Nat. Nanotechnol. 17, 759–767 (2022).
Hu, Q. et al. Reaction intermediate-mediated electrocatalyst synthesis favors specified facet and defect exposure for efficient nitrate–ammonia conversion. Energy Environ. Sci. 14, 4989–4997 (2021).
Bozal-Ginesta, C., Pablo-García, S., Choi, C., Tarancón, A. & Aspuru-Guzik, A. Developing machine learning for heterogeneous catalysis with experimental and computational data. Nat. Rev. Chem. 9, 601–616 (2025).
Kim, M. et al. Artificial intelligence to accelerate the discovery of N2 electroreduction catalysts. Chem. Mater. 32, 709–720 (2019).
Wang, Z. et al. Machine learning-accelerated discovery of A2BC2 ternary electrides with diverse anionic electron densities. J. Am. Chem. Soc. 145, 26412–26424 (2023).
Liu, C. et al. Upcycling surplus acetone into long-chain chemicals using a tandem electro-biosystem. Nat. Sustain. 8, 806–817 (2025).
Li, Z. et al. Electroreduction-driven distorted nanotwins activate pure Cu for efficient hydrogen evolution. Nat. Mater. 24, 424–432 (2025).
Johnson, C. L. et al. Effects of elastic anisotropy on strain distributions in decahedral gold nanoparticles. Nat. Mater. 7, 120–124 (2007).
Song, M. et al. Oriented attachment induces fivefold twins by forming and decomposing high-energy grain boundaries. Science 367, 40–45 (2020).
Yang, K. et al. Unveiling the reaction mechanism of nitrate reduction to ammonia over cobalt-based electrocatalysts. J. Am. Chem. Soc. 146, 12976–12983 (2024).
Han, S. et al. Ultralow overpotential nitrate reduction to ammonia via a three-step relay mechanism. Nat. Catal. 6, 402–414 (2023).
Wang, S., Zhou, Z., Zhou, R., Fang, Z. & Cullen, P. J. Effect of solution pH on the characteristics of pulsed gas-liquid discharges and aqueous reactive species in atmospheric air. J. Appl. Phys. 130, 103302 (2021).
Chen, F.-Y., Elgazzar, A., Pecaut, S. & Qiu, C. Electrochemical nitrate reduction to ammonia with cation shuttling in a solid electrolyte reactor. Nat. Catal. 7, 1032–1043 (2024).
Lu, X. & Zhao, C. Electrodeposition of hierarchically structured three-dimensional nickel-iron electrodes for efficient oxygen evolution at high current densities. Nat. Commun. 6, 6616 (2015).
Shin, H., Hansen, K. U. & Jiao, F. Techno-economic assessment of low-temperature carbon dioxide electrolysis. Nat. Sustain. 4, 911–919 (2021).
Bond, K., Butler-Sloss, S., Lovins, A., Speelman, L., & Topping, N. X-change: Electricity—On Track for Net Zero (RMI, 2023); https://rmi.org/wp-content/uploads/dlm_uploads/2023/07/rmi_x_change_electricity_2023.pdf
Guo, S. et al. Electrocatalytic hydrogenation of quinolines with water over a fluorine-modified cobalt catalyst. Nat. Commun. 13, 5297 (2022).
Xie, W., Gong, Y. & Yu, K. Simple and accurate method for determining dissolved inorganic carbon in environmental water by reaction headspace gas chromatography. J. Sep. Sci. 41, 1091–1095 (2017).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Kresse, G. & Furthmüller, J. Efficiency of ab initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comp. Mater. Sci. 6, 15–50 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximationmade simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Nørskov, J. K. et al. Origin of the overpotential for oxygen reduction at a fuel-cell cathode. J. Phys. Chem. B 108, 17886–17892 (2004).
Acknowledgements
This work was supported by the National Key R&D Program of China (grant no. 2020YFA0710000, G.Y.), the Joint Funds of the National Natural Science Foundation of China (grant no. U22A20391, G.Y.), the National Natural Science Foundation of China (grant no. 22302154, H.O.), the Innovation Capability Support Program of Shaanxi (grant no. 2023-CX-TD-26, G.Y.), the Key Projects in Shaanxi Province (grant no. 2024CY2-GJHX-75, H.L.), the Program of Introducing Talents of Discipline to Universities (grant no. B23025, G.Y.) and Fundamental Research Funds for the Central Universities (grant no. xtr022021007, G.Y.). We also acknowledge technical support from Xi’an Taikang Biotechnology Co., Ltd. in relation to the microwave plasma system and thank R. Zhou for his valuable assistance with plasma diagnostics.
Author information
Authors and Affiliations
Contributions
W.L. designed and synthesized the TB/SF-Co electrocatalyst, performed DFT calculations, constructed the microwave plasma system and the scaled MEA electrolyzer, conducted electrochemical experiments, analysed the data and wrote the paper. H.O. contributed to data analysis and interpretation, assisted with experimental design, validated key results and critically revised the paper. Y. Lv performed the MPAA–ENitR system evaluation. B.X. analysed and interpreted the DFT results. Y.R. and W.T. synthesized large-area electrodes and carried out electrode characterization. H.L. and Y. Li revised the paper. G.Y. contributed substantially to research ideas, experimental design, data analysis, supervision of the project, paper preparation and funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Ali Jalili, Guoxiong Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Eric Piechota, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–57, Tables 1–5 and Notes 1–3.
Supplementary Data 1 (download XLSX )
Statistical source data of figures in Supplementary Information.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, W., Ou, H., Lv, Y. et al. Ampere-level ammonia synthesis through grain boundary engineering. Nat. Synth (2026). https://doi.org/10.1038/s44160-026-01026-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-026-01026-3