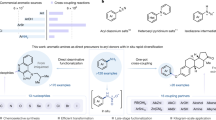
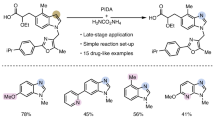
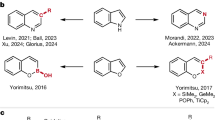
Abstract
The atom-by-atom manipulation of the core molecular framework has come into reality recently. In particular, the late-stage diversification of robust aza-arenes such as indole has been at the centre of research activity because of its importance in biological and pharmaceutical studies. The previous single-atom modifications of indoles were made possible through the insertion of a reactive nitrene species into the enamine-like C2–C3 bond, resulting in the incorporation of a new atom at the 3-position. Here we discovered an alternative reaction mode, in which the N-nitroso group underwent a sequential intramolecular translocation and deoxygenative rearrangement to afford regiochemically orthogonal 1,4-diazines that had been lacking in skeletal editing. This activation of the nearly inert aromatic C3–C9 bond was applicable to various indoles including bio-relevant molecules to furnish quinoxalines, a class of underexplored pharmacophores. The combined experimental and computational studies revealed the reaction mechanism for the unique selectivity pattern.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within this article and its Supplementary Information, which includes experimental details, characterization data, copies of NMR spectra for all new compounds and density functional theory calculation details. The computation output files and the variable time normalization analysis kinetic data are provided as source data. Crystallographic data for 3y have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition number CCDC 2418467, which can be accessed free of charge via https://www.ccdc.cam.ac.uk/structures/. All data are available from the corresponding author upon reasonable request. Source data are provided with the paper.
References
Jurczyk, J. et al. Single-atom logic for heterocycle editing. Nat. Synth. 1, 352–364 (2022).
Levin, M. D. Retrosynthetic simplicity. Synlett 35, 1471–1474 (2024).
Joynson, B. W. & Ball, L. T. Skeletal editing: interconversion of arenes and heteroarenes. Helv. Chim. Acta 106, e202200182 (2023).
Liu, Z., Sivaguru, P., Ning, Y., Wu, Y. & Bi, X. Skeletal editing of (hetero)arenes using carbenes. Chem. Eur. J. 29, e202301227 (2023).
Patel, C. K. et al. Skeletal editing through single-atom insertion and transmutation: an insight into a new era of synthetic organic chemistry. Synthesis 56, 3793–3814 (2024).
Xu, Y.-A., Xiang, S.-H., Che, J.-T., Wang, Y.-B. & Tan, B. Skeletal editing of cyclic molecules using nitrenes. Chin. J. Chem. 42, 2656–2667 (2024).
Sharma, R., Arisawa, M., Takizawa, S. & Salem, M. S. H. Remodelling molecular frameworks via atom-level surgery: recent advances in skeletal editing of (hetero)cycles. Org. Chem. Front. 12, 1633–1670 (2025).
Nogi, K. & Yorimitsu, H. Aromatic metamorphosis: conversion of an aromatic skeleton into a different ring system. Chem. Commun. 53, 4055–4065 (2017).
Yorimitsu, H. Aromatic metamorphosis: skeletal editing of aromatic rings. Acc. Chem. Res. 58, 1323–1334 (2025).
Yorimitsu, H., Vasu, D., Bhanuchandra, M., Murakami, K. & Osuka, A. Aromatic metamorphosis of dibenzothiophenes. Synlett 27, 1765–1774 (2016).
Saito, H. & Yorimitsu, H. Ring-expanding and ring-opening transformations of benzofurans and indoles with introducing heteroatoms. Chem. Lett. 48, 1019–1028 (2019).
Li, X., Xu, J. & Xu, Z.-G. Precision single-atom editing: new frontiers in nitrogen insertion andsubstitution for the generation of N-heterocycles.Org. Chem. Front. 11, 4041–4053 (2024).
Bi, X., Wei, H., Lu, H. & Song, S. Skeletal editing of cyclic scaffolds. CCS Chem 8, 607–692 (2026).
Lazzara, P. R. & Moore, T. W. Scaffold-hopping as a strategy to address metabolic liabilities of aromatic compounds. RSC Med. Chem. 11, 18–29 (2020).
Sicignano, M. & Costa, P. Drug design via single-carbon atom insertion. Nat. Catal. 7, 224–226 (2024).
Ma, C., Lindsley, C. W., Chang, J. & Yu, B. Rational molecular editing: a new paradigm in drug discovery. J. Med. Chem. 67, 11459–11466 (2024).
Reisenbauer, J. C., Green, O., Franchino, A., Finkelstein, P. & Morandi, B. Late-stage diversification of indole skeletons through nitrogen atom insertion. Science 377, 1104–1109 (2022).
Reisenbauer, J. C. et al. Direct access to quinazolines and pyrimidines from unprotected indoles and pyrroles through nitrogen atom insertion. Org. Lett. 25, 8419–8423 (2023).
Zhang, B.-S. et al. Electrochemical skeletal indole editing via nitrogen atom insertion by sustainable oxygen reduction reaction. Angew. Chem. Int. Ed. 63, e202407384 (2024).
Ghosh, B. et al. Sulfenylnitrene-mediated nitrogen-atom insertion for late-stage skeletal editing of N-heterocycles. Science 387, 102–107 (2025).
Kafle, P. et al. Photolysis-generated sulfenylnitrenes enable site-selective nitrogen-atom insertion into N-heterocycles. Chem 12, 102753 (2025).
Dherange, B. D., Kelly, P. Q., Liles, J. P., Sigman, M. S. & Levin, M. D. Carbon atom insertion into pyrroles and indoles promoted by chlorodiazirines. J. Am. Chem. Soc. 143, 11337–11344 (2021).
Joynson, B. W., Cumming, G. R. & Ball, L. T. Photochemically mediated ring expansion of indoles and pyrroles with chlorodiazirines: synthetic methodology and thermal hazard assessment. Angew. Chem. Int. Ed. 62, e202305081 (2023).
Wu, F.-P., Tyler, J. L., Daniliuc, C. G. & Glorius, F. Atomic carbon equivalent: design and application to diversity-generating skeletal editing from indoles to 3-functionalized quinolines. ACS Catal. 14, 13343–13351 (2024).
Guo, H., Qiu, S. & Xu, P. One-carbon ring expansion of indoles and pyrroles: a straightforward access to 3-fluorinated quinolines and pyridines. Angew. Chem. Int. Ed. 63, e202317104 (2024).
Saito, H., Otsuka, S., Nogi, K. & Yorimitsu, H. Nickel-catalyzed boron insertion into the C2–O bond of benzofurans. J. Am. Chem. Soc. 138, 15315–15318 (2016).
Tsuchiya, S., Saito, H., Nogi, K. & Yorimitsu, H. Manganese-catalyzed ring opening of benzofurans and its application to insertion of heteroatoms into the C2–O bond. Org. Lett. 19, 5557–5560 (2017).
Tsuchiya, S., Saito, H., Nogi, K. & Yorimitsu, H. Aromatic metamorphosis of indoles into 1,2-benzazaborins. Org. Lett. 21, 3855–3860 (2019).
Zou, X., Zou, J., Yang, L., Li, G. & Lu, H. Thermal rearrangement of sulfamoyl azides: reactivity and mechanistic study. J. Org. Chem. 82, 4677–4688 (2017).
Hyland, E. E., Kelly, P. Q., McKillop, A. M., Dherange, B. D. & Levin, M. D. Unified access to pyrimidines and quinazolines enabled by N–N cleaving carbon atom insertion. J. Am. Chem. Soc. 144, 19258–19264 (2022).
Woo, J. et al. Scaffold hopping by net photochemical carbon deletion of azaarenes. Science 376, 527–532 (2022).
Woo, J., Stein, C., Christian, A. H. & Levin, M. D. Carbon-to-nitrogen single-atom transmutation of azaarenes. Nature 623, 77–82 (2023).
Pearson, T. J. et al. Aromatic nitrogen scanning by ipso-selective nitrene internalization. Science 381, 1474–1479 (2023).
Bartholomew, G. L., Carpaneto, F. & Sarpong, R. Skeletal editing of pyrimidines to pyrazoles by formal carbon deletion. J. Am. Chem. Soc. 144, 22309–22315 (2022).
Bartholomew, G. L. et al. 14N to 15N isotopic exchange of nitrogen heteroaromatics through skeletal editing. J. Am. Chem. Soc. 146, 2950–2958 (2024).
Paschke, A.-S. K. et al. Carbon-to-nitrogen atom swap enables direct access to benzimidazoles from drug-like indoles. Nat. Chem. 17, 1750–1756 (2025).
Patel, S. C. & Burns, N. Z. Conversion of aryl azides to aminopyridines. J. Am. Chem. Soc. 144, 17797–17802 (2022).
Lu, H. et al. Carbon–nitrogen transmutation in polycyclic arenol skeletons to access N-heteroarenes. Nat. Commun. 15, 3772 (2024).
Cheng, Q. et al. Skeletal editing of pyridines through atom-pair swap from CN to CC. Nat. Chem. 16, 741–748 (2024).
Wang, Z., Xu, P., Guo, S.-M., Daniliuc, C. G. & Studer, A. C-to-N atom swapping and skeletal editing in indoles and benzofurans. Nature 642, 92–98 (2025).
Wu, F.-P. et al. Nitrogen-to-functionalized carbon atom transmutation of pyridine. Chem. Sci. 15, 15205–15211 (2024).
Kim, D. et al. Photocatalytic furan-to-pyrrole conversion. Science 386, 99–105 (2024).
Nguyen, H. M. H. et al. Synthesis of 15N-pyridines and higher mass isotopologs via Zincke imine intermediates. J. Am. Chem. Soc. 146, 2944–2949 (2024).
Conboy, A. & Greaney, M. F. Synthesis of benzenes from pyridines via N to C switch. Chem 10, 1940–1949 (2024).
Liu, S. et al. Halogencarbene-free Ciamician–Dennstedt single-atom skeletal editing. Nat. Commun. 15, 9998 (2024).
He, Y., Wang, J., Zhu, T., Zheng, Z. & Wei, H. Nitrogen atom insertion into arenols to access benzazepines. Chem. Sci. 15, 2612–2617 (2024).
Falcone, N. A., He, S., Hoskin, J. F., Mangat, S. & Sorensen, E. J. N-oxide-to-carbon transmutations of azaarene N-oxides. Org. Lett. 26, 4280–4285 (2024).
Spinnato, D., Leutzsch, M., Wang, F. & Cornella, J. Internal atom exchange in oxazole rings: a blueprint for azole scaffold evaluation. Synlett 35, 1015–1018 (2024).
Pasha, M. A., Jang, J., Bae, Y. & Shin, S. N-Insertion of diazonium salts into ketone derivatives. Angew. Chem. Int. Ed. 64, e202505341 (2025).
Bartholomew, G. L. et al. Phototransposition of indazoles to benzimidazoles: tautomer-dependent reactivity, wavelength dependence, and continuous flow studies. Angew. Chem. Int. Ed. 64, e202423803 (2025).
Paschke, A.-S. K. et al. Late-stage diversification of native tryptophan-containing peptides and peptide drugs through nitrogen atom insertion. J. Am. Chem. Soc. 148, 55–60 (2026).
Pennington, L. D. & Moustakas, D. T. The necessary nitrogen atom: a versatile high-impact design element for multiparameter optimization. J. Med. Chem. 60, 3552–3579 (2017).
Zeng, W. et al. FDA-approved pharmaceuticals: targets, pharmacological activities, and SAR studies. RSC Med. Chem. 15, 788–808 (2024).
Marshall, C. M., Federice, J. G., Bell, C. N., Cox, P. B. & Njardarson, J. T. An update on the nitrogen heterocycle compositions and properties of U.S. FDA-approved pharmaceuticals (2013–2023). J. Med. Chem. 67, 11622–11655 (2024).
Liu, F., Anand, L. & Szostak, M. Diversification of indoles and pyrroles by molecular editing: new frontiers in heterocycle-to-heterocycle transmutation. Chem. Eur. J. 29, e202300096 (2023).
Ikeda, M., Tabusa, F., Nishimura, Y., Kwon, S. & Tamura, Y. The reactions of some indoles with iodine azide: syntheses of 3-azidoindolenines, 2-azidomethylindoles, and 3a-azido-furo- and pyrrolo-[2,3-b]indoles. Tetrahedron Lett. 17, 2347–2350 (1976).
Borah, B. & Chowhan, L. R. Recent advances in the transition-metal-free synthesis of quinoxalines. RSC Adv. 11, 37325–37353 (2021).
Khatoon, H. & Faudzi, S. M. M. Exploring quinoxaline derivatives: an overview of a new approach to combat antimicrobial resistance. Eur. J. Med. Chem. 276, 116675 (2024).
Aakash, V. B., Ramalakshmi, N., Bhuvaneswari, S., Sankari, E. & Arunkumar, S. Comprehensive review on versatile pharmacology of quinoxaline derivative. Russ. J. Bioorg. Chem. 48, 657–677 (2022).
Suthar, S. K., Chundawat, N. S., Singh, G. P., Padrón, J. M. & Jhala, Y. K. Quinoxaline: a comprehension of current pharmacological advancement in medicinal chemistry. Eur. J. Med. Chem. Rep. 5, 100040 (2022).
Yashwantrao, G. & Saha, S. Recent advances in the synthesis and reactivity of quinoxaline. Org. Chem. Front. 8, 2820–2862 (2021).
Montana, M., Mathias, F., Terme, T. & Vanelle, P. Antitumoral activity of quinoxaline derivatives: a systematic review. Eur. J. Med. Chem. 163, 136–147 (2019).
Song, S.-M., Jin, J., Choi, J.-H., & Chung, W.-j. Synthesis of cis-thiiranes as diastereoselective access to epoxide congeners via 4π-electrocyclization of thiocarbonyl ylides. Nat. Commun. 13, 4818 (2022).
Im, J. K., Yang, B., Jeong, I., Choi, J.-H., & Chung, W.-j. N-Chlorination-induced, oxidative ring contraction of 1,4-dimethoxyphthalazines. Tetrahedron Lett. 61, 152048 (2020).
Im, J. K. et al. N-Chlorinative ring contraction of 1,4-dimethoxyphthalazines via a bicyclization/ring opening mechanism. Synthesis 53, 1760–1770 (2021).
Kim, H. E., Choi, J.-H., & Chung, W.-j. Synthesis of α‑ketoimidoyl fluorides via geminal fluorine-promoted azide rearrangement. Org. Lett. 23, 8810–8815 (2021).
Kim, H. E., Choi, J.-H. & Chung, W.-j. Fluorine-assisted rearrangement of geminal azidofluorides to imidoyl fluorides. J. Org. Chem. 88, 6878–6889 (2023).
Pace, C. J. & Gao, J. Exploring and exploiting polar−π interactions with fluorinated aromatic amino acids. Acc. Chem. Res. 46, 907–915 (2013).
Sekine, K., Mawatari, T. & Yamada, T. Synthesis of oxazolidin-2-ones by tandem cyclization of propargylic alcohols and phenyl isocyanate promoted by silver catalysts as π-Lewis acids. Synlett 26, 2447–2450 (2015).
Wang, Y., Kumar, R. K. & Bi, X. Silver-catalyzed organic reactions of isocyanides. Tetrahedron Lett. 57, 5730–5741 (2016).
Büchi, G., Lee, G. C. M., Yang, D. & Tannenbaum, S. R. Direct acting, highly mutagenic, α-hydroxy N-nitrosamines from 4-chloroindoles. J. Am. Chem. Soc. 108, 4115–4119 (1986).
Bravo, C., Hervés, P., Leis, J. R. & Peña, M. E. Kinetic study of the nitrosation of 3-substituted indoles. J. Chem. Soc., Perkin Trans. 2, 185–189 (1992).
Astolfi, P., Panagiotaki, M., Rizzolib, C. & Greci, L. Reactions of indoles with nitrogen dioxide and nitrous acid in an aprotic solvent. Org. Biomol. Chem. 4, 3282–3290 (2006).
Zheng, M. et al. Indole derivatives as potent inhibitors of 5-lipoxygenase: design, synthesis, biological evaluation, and molecular modeling. Bioorg. Med. Chem. Lett. 17, 2414–2420 (2007).
Curran, M. P. & Keating, G. M. Tadalafil. Drugs 63, 2203–2212 (2003).
Shamon, S. D. & Perez, M. I. Blood pressure-lowering efficacy of reserpine for primary hypertension. Cochrane Database Syst. Rev. https://doi.org/10.1002/14651858.CD007655.pub3 (2016).
Burés, J. Variable time normalization analysis: general graphical elucidation of reaction orders from concentration profiles. Angew. Chem. Int. Ed. 55, 16084–16087 (2016).
Li, G., Kanda, Y., Hong, S. Y. & Radosevich, A. T. Enabling reductive C–N cross-coupling of nitroalkanes and boronic acids by steric design of P(III)/P(V)═O catalysts. J. Am. Chem. Soc. 144, 8242–8248 (2022).
Hong, S. Y. & Radosevich, A. T. Chemoselective primary amination of aryl boronic acids by PIII/PV═O-catalysis: synthetic capture of the transient Nef Intermediate HNO. J. Am. Chem. Soc. 144, 8902–8907 (2022).
Bickelhaupt, F. M. & Houk, K. N. Analyzing reaction rates with the distortion/interaction-activation strain model. Angew. Chem. Int. Ed. 56, 10070–10086 (2017).
Knizia, G. Intrinsic atomic orbitals: an unbiased bridge between quantum theory and chemical concepts. J. Chem. Theory Comput. 9, 4834–4843 (2013).
Glendening, E. D., Reed, A. E., Carpenter, J. E. & Weinhold, F. NBO Version 3.1 (Theoretical Chemistry Institute, Univ. Wisconsin: Madison, 1990).
Acknowledgements
This research was supported by the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (grant nos. RS-2025-00517801, W.J.C. and RS-2024-00409659, W.J.C.). We thank the Surface Physical Property Laboratory at GIST Advanced Institute of Instrumental Analysis (GAIA) for the X-ray crystallographic analysis of 3y.
Author information
Authors and Affiliations
Contributions
W.J.C. conceived of the research concept. M.S. discovered the preliminary reactivity. W.J.C., M.S., and I.J. designed the synthetic strategy. M.S. and I.J. performed the leading synthetic work. M.S., I.J. and H.E.K. designed and performed the mechanistic experiments. H.E.K. and I.J. performed the main computational analysis. J. Jin performed the supporting computational analysis. H.M., J.K.I. and J. Jung performed the supporting synthetic work. J. Jo contributed to the initial idea formulation. W.J.C., M.S. and I.J. wrote the paper. All authors discussed the results, and contributed to editing the paper and preparing the Supplementary Information. M.S. and I.J. contributed equally to this work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Yifeng Chen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Joel Cejas-Sánchez, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information. (download PDF )
Experimental Details including Notes, Methods, Sections 1–4, Tables 1–3, Figs. 1–3 and NMR spectra.
Supplementary Data 1.
Crystallographic data for compound 3y, CCDC 2418467.
Source data
Source Data Fig. 5. (download XLSX )
Statistical source data file for the kinetic data for variable time normalization analysis in Fig. 5c.
Source Data Fig. 6. (download ZIP )
Computational data and Cartesian coordinates for Fig. 6.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Song, M., Jeong, I., Kim, H.E. et al. Regio-orthogonal single N-atom insertion into indoles via NO translocation. Nat. Synth (2026). https://doi.org/10.1038/s44160-026-01046-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-026-01046-z