Abstract
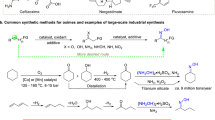
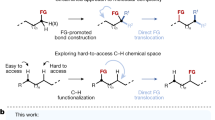
Direct co-conversion of earth-abundant N2 and CH4 to high-value C–N–O compounds is a promising synthetic method but remains challenging, as the activation of N2 and CH4 typically requires high-energy conditions that can cause product decomposition. Here we report a plasma-cascade process for synthesizing cyclohexanone cyanohydrin (Cy(OH)CN) via direct coupling of N2 and CH4 plasma with cyclohexanone. This catalyst-free process achieves a Cy(OH)CN formation rate of 0.60 mmol h−1 with a high selectivity of 95.8% towards the cyclohexanone-derived products under mild conditions, concurrently generating ammonia as a valuable coproduct. Comprehensive mechanistic studies reveal that plasma-generated ·CHx and hydrogen radicals enable direct α-carbonyl functionalization of cyclohexanone to α-CHx cyclohexanol intermediates, which then coupled mainly with excited-state N2 and are further activated by hydrogen radicals to generate Cy(OH)CN. This plasma-cascade strategy decouples the activation of inert molecules from the formation of relatively unstable C–N–O compounds, enabling selective cyanohydrin synthesis that avoids using high-cost NH3 and toxic HCN.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the article and its Supplementary information. Source data are provided with this paper.
References
Gregory, R. J. H. Cyanohydrins in nature and the laboratory: biology, preparations, and synthetic applications. Chem. Rev. 99, 3649–3682 (1999).
Brunel, J.-M. & Holmes, I. P. Chemically catalyzed asymmetric cyanohydrin syntheses. Angew. Chem. Int. Ed. 43, 2752–2778 (2004).
North, M., Usanov, D. L. & Young, C. Lewis acid catalyzed asymmetric cyanohydrin synthesis. Chem. Rev. 108, 5146–5226 (2008).
Bracco, P., Busch, H., Langermann, J. & Hanefeld, U. Enantioselective synthesis of cyanohydrins catalysed by hydroxynitrile lyases—a review. Org. Biomol. Chem. 14, 6375–6389 (2016).
Delagrange, S. & Schuurman, Y. HCN synthesis from methane and ammonia over platinum. Catal. Today 121, 204–209 (2007).
Goodwin, C. M. et al. Operando probing of the surface chemistry during the Haber–Bosch process. Nature 625, 282–286 (2024).
Soloveichik, G. Electrochemical synthesis of ammonia as a potential alternative to the Haber–Bosch process. Nat. Catal. 2, 377–380 (2019).
Chen, J. J. et al. Beyond fossil fuel-driven nitrogen transformations. Science 360, eaar6611 (2018).
Hou, Y. & Deng, D. Membrane electrode assembly: another technical roadmap for lithium-mediated nitrogen reduction?. Joule 7, 1679–1683 (2023).
MacFarlane, D. R. et al. A roadmap to the ammonia economy. Joule 4, 1186–1205 (2020).
Zhou, H. et al. Organocatalytic stereoselective cyanosilylation of small ketones. Nature 605, 84–89 (2022).
Mo, K., Yang, Y. & Cui, Y. A homochiral metal-organic framework as an effective asymmetric catalyst for cyanohydrin synthesis. J. Am. Chem. Soc. 136, 1746–1749 (2014).
Akhtar, R., Gaurav, K. & Khan, S. Applications of low-valent compounds with heavy group-14 elements. Chem. Soc. Rev. 53, 6150–6243 (2024).
Xu, P., Shen, C., Xu, A., Low, K.-H. & Huang, Z. Desymmetric cyanosilylation of acyclic 1,3-diketones. Angew. Chem. Int. Ed. 61, e202208443 (2022).
Jin, Z. et al. Hydrophobic zeolite modification for in situ peroxide formation in methane oxidation to methanol. Science 367, 193–197 (2020).
Kang, W., Lee, C. C., Jasniewski, A. J., Ribbe, M. W. & Hu, Y. Structural evidence for a dynamic metallocofactor during N2 reduction by Mo-nitrogenase. Science 368, 1381–1385 (2020).
Anderson, J. S., Rittle, J. & Peters, J. C. Catalytic conversion of nitrogen to ammonia by an iron model complex. Nature 501, 84–87 (2013).
Légaré, M.-A. et al. Nitrogen fixation and reduction at boron. Science 359, 896–900 (2018).
Fu, X. et al. Continuous-flow electrosynthesis of ammonia by nitrogen reduction and hydrogen oxidation. Science 379, 707–712 (2023).
Gibson, E. K. et al. Probing the role of a non-thermal plasma (NTP) in the hybrid NTP catalytic oxidation of methane. Angew. Chem. Int. Ed. 56, 9351–9355 (2017).
Stere, C. E. et al. Non-thermal plasma activation of gold-based catalysts for low temperature water-gas shift catalysis. Angew. Chem. Int. Ed. 56, 5579–5583 (2017).
Mehta, P. et al. Overcoming ammonia synthesis scaling relations with plasma-enabled catalysis. Nat. Catal. 1, 269–275 (2018).
Guo, Z., Yi, Y., Wang, L., Yan, J. & Guo, H. Pt/TS‑1 catalyst promoted C–N coupling reaction in CH4-NH3 plasma for HCN synthesis at low temperature. ACS Catal. 8, 10219–10224 (2018).
Kamarinopoulou, N. S., Wittreich, G. R. & Vlachos, D. G. Direct HCN synthesis via plasma-assisted conversion of methane and nitrogen. Sci. Adv. 10, eadl4246 (2024).
Xu, S. et al. Sustaining metal-organic frameworks for water-gas shift catalysis by non-thermal plasma. Nat. Catal. 2, 142–148 (2019).
Zhao, H. et al. In situ identification of NNH and N2H2 by using molecular-beam mass spectrometry in plasma-assisted catalysis for NH3 synthesis. ACS Energy Lett. 7, 53–58 (2022).
Xu, X., Zhao, X., Tang, J., Duan, Y. & Tian, Y.-H. Direct amination of benzene with molecular nitrogen enabled by plasma-liquid interactions. Angew. Chem. Int. Ed. 61, e202203680 (2022).
Liu, W. & Tian, Y. Observing C–N bond formation in plasma: a case study of benzene and dinitrogen coupling via an arylnitrenium ion intermediate. Phys. Chem. Chem. Phys. 26, 18016–18020 (2024).
Hosseini, H., Saleem, M., Marotta, E. & Paradisi, C. Nitrogen-containing organic products from the treatment of liquid toluene with plasma-activated N2 gas. Plasma Process Polym. 18, e2100012 (2021).
Begley, A. I. et al. Excited-state N atoms transform aromatic hydrocarbons into N‑heterocycles in low-temperature plasmas. J. Phys. Chem. A 126, 1743–1754 (2022).
Kin, J. et al. Adsorption and decomposition of cyclohexanone (C6H10O) on Pt (111) and the (2 × 2) and (√3 × √3)R30o-Sn/Pt(111) surface alloys. Langmuir 26, 16401–16411 (2010).
Machmud, A. & Chang, M. B. Conversion of C4F8 via plasma catalysis over Al2O3/Zr/SO4−2 catalyst: effects of H2O(g). Chem. Eng. J. 500, 157467 (2024).
Adhikari, E. R., Samara, V. & Ptasinska, S. Influence of O2 or H2O in a plasma jet and its environment on plasma electrical and biochemical performances. J. Phys. D 51, 185202 (2018).
Zhang, H. et al. Plasma activation of methane for hydrogen production in a N2 rotating gliding arc warm plasma: a chemical kinetics study. Chem. Eng. J. 345, 67–78 (2018).
Snoeckx, R. et al. Influence of N2 concentration in a CH4/N2 dielectric barrier discharge used for CH4 conversion into H2. J. Hydrog. Energy 38, 16098–16120 (2013).
Hagelaar, G. J. M. & Pitchford, L. C. Solving the Boltzmann equation to obtain electron transport coefficients and rate coefficients for fluid models. Plasma Sources Sci. Technol. 14, 722–733 (2005).
Alves, L. L. The IST-LISBON database on LXCat. J. Phys. Conf. Ser. 565, 012007 (2014).
Pancheshnyi, S., Eismann, B., Hagelaar, G. J. M. & Pitchford, L. C. Computer code zdplaskin, version 2.0a (University of Toulouse, 2008); http://www.zdplaskin.laplace.univ-tlse.fr
Manion, J. A. et al. NIST Chemical Kinetics Database, NIST Standard Reference Database 17, Version 7.0 (Web Version), Release 1.6.8, Data version 2015.09 (National Institute of Standards and Technology, 2026).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Kresse, G. & Hafner, J. Ab initio molecular-dynamics simulation of the liquid-metal-amorphous-semiconductor transition in germanium. Phys. Rev. B 49, 14251–14269 (1994).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Zhao, Y. & Truhlar, D. G. A new local density functional for main-group thermochemistry, transition metal bonding, thermochemical kinetics, and noncovalent interactions. J. Chem. Phys. 125, 194101 (2006).
Hjorth Larsen, A. et al. The atomic simulation environment—a Python library for working with atoms. J. Phys.: Condens. Matter 29, 273002 (2017).
Acknowledgements
We acknowledge financial support from the National Natural Science Foundation of China (grant nos. 22588201 and 22225204 to D.D., 22372019 and U24A20487 to R.H. and 22272170 to L.Y.), the National Key R&D Program of China (grant nos. 2022YFA1504500 to D.D. and 2024YFA1510103 to R.H.), the State Key Laboratory of Catalysis in the Dalian Institute of Chemical Physics (DICP) (grant nos. 2024SKL-A-002 and N-22-02) and the DICP (grant no. DICP I202506). We thank A. Goldbach from DICP for providing valuable suggestions.
Author information
Authors and Affiliations
Contributions
D.D., R.H. and L.Y. conceived and supervised the project. H.Z. carried out the experiments. Z.L. and L.Y. performed DFT calculations. D.L. and H.Z. conducted in situ MBMS characterization. C.W., L.H. and H.L. provided technical support for in situ MBMS device. Y.Y. conducted optical emission spectroscopy experiments. D.L. performed plasma kinetics simulation. H.Z., Z.L., D.L., R.H., L.Y. and D.D. discussed the results, wrote and revised the paper. All authors contributed to scientific discussion.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Xiaolei Fan, Ali Mesbah, Richard van de Sanden and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alexandra Groves, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Figs. 1–21, Tables 1–6 and Refs. 1–8.
Supplementary Video 1 (download MP4 )
Detailed configuration of DBD plasma reactor.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, H., Liu, Z., Li, D. et al. Direct plasma synthesis of a high-value C–N–O compound with inert N2 and CH4. Nat. Synth (2026). https://doi.org/10.1038/s44160-026-01055-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-026-01055-y