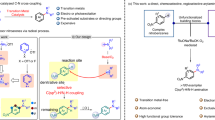
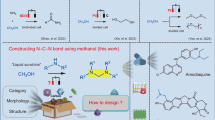
Abstract
Nitrogen-containing aromatic compounds, such as aromatic imines and aromatic oximes, are vital in the pharmaceutical and agrochemical industries. However, their synthesis via conventional C–N coupling strategies remains dependent on fossil-derived feedstocks and energy-intensive processes. Here we report a paired electrolysis integrating lignin cleavage with subsequent C–N coupling to synthesize these compounds. The system uses a Pd1/TivO2-NS cathode (palladium single atoms confined in titanium vacancies on monolayer TivO2 nanosheets) and a PbO2 plate anode in phosphate electrolyte. During electrolysis, lignin-derived polyols generated by cathodic cleavage are oxidized to carbonyl intermediates at the anode while nitroarenes are reduced to amines at the cathode, enabling C–N bond formation. Using poplar lignin and nitrobenzene, an aryl imine yield of 74.6% (based on β-O-4 units) is achieved. Compatibility with various nitrogen sources, including substituted nitroaromatics and nitrogen oxides (NOx), is also investigated. This work demonstrates the possibility of using lignin as a sustainable alternative to petroleum-based synthesis of nitrogen-containing aromatic compounds.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Supporting data for the study are available in this article and its Supplementary Information. Source data are provided with this paper.
References
Hili, R. & Yudin, A. K. Making carbon–nitrogen bonds in biological and chemical synthesis. Nat. Chem. Biol. 2, 284–287 (2006).
Zhou, T., Gao, P., Lalancette, R., Szostak, R. & Szostak, M. Gold-catalysed amine synthesis by reductive hydroamination of alkynes with nitroarenes. Nat. Chem. 16, 1–11 (2024).
Hartwig, J. F. Evolution of a fourth generation catalyst for the amination and thioetherification of aryl halides. Acc. Chem. Res. 41, 1534–1544 (2008).
Irrgang, T. & Kempe, R. Transition-metal-catalyzed reductive amination employing hydrogen. Chem. Rev. 120, 9583–9674 (2020).
Patel, M., Saunthwal, R. K. & Verma, A. K. Base-mediated hydroamination of alkynes. Acc. Chem. Res. 50, 240–254 (2017).
De Risi, C., Pollini, G. P. & Zanirato, V. Recent developments in general methodologies for the synthesis of α-ketoamides. Chem. Rev. 116, 3241–3305 (2016).
Ragauskas, A. J. et al. Lignin valorization: improving lignin processing in the biorefinery. Science 344, 1246843 (2014).
Zhang, C. & Wang, F. Catalytic lignin depolymerization to aromatic chemicals. Acc. Chem. Res. 53, 470–484 (2020).
Dong, L. et al. Sustainable production of dopamine hydrochloride from softwood lignin. Nat. Commun. 14, 4996 (2023).
Afanasenko, A. M. et al. Clean synthetic strategies to biologically active molecules from lignin: a green path to drug discovery. Angew. Chem. Int. Ed. 63, e202308131 (2024).
Ma, Z. et al. Synthesis of aromatic amides from lignin and its derivatives. Nat. Commun. 16, 3476 (2025).
Cui, T. et al. Atomically dispersed Pt–N3C1 sites enabling efficient and selective electrocatalytic C–C bond cleavage in lignin models under ambient conditions. J. Am. Chem. Soc. 143, 9429–9439 (2021).
Wang, C. et al. Enhancing β-O-4 linkage cleavage with ultrahigh faradaic efficiency via electrochemical–chemical tandem catalysis for lignin valorization. Adv. Mater. 37, e08234 (2025).
Zhou, Y., Klinger, G. E., Hegg, E. L., Saffron, C. M. & Jackson, J. E. Multiple mechanisms mapped in aryl alkyl ether cleavage via aqueous electrocatalytic hydrogenation over skeletal nickel. J. Am. Chem. Soc. 142, 4037–4050 (2020).
He, Y. et al. Aqueous electrocatalytic hydrogenation depolymerization of lignin β-O-4 linkage via selective Caryl–O(C) bond cleavage: the regulation of adsorption. J. Am. Chem. Soc. 146, 32022–32031 (2024).
Zhang, C. et al. Catalytic strategies and mechanism analysis orbiting the center of critical intermediates in lignin depolymerization. Chem. Rev. 123, 4510–4601 (2023).
Majumder, P. S. & Gupta, S. Hybrid reactor for priority pollutant nitrobenzene removal. Water Res. 37, 4331–4336 (2003).
Gupta, S. & Ronen, Z. Biological treatment of nitroaromatics in wastewater. Water 16, 901 (2024).
Gloag, L., Somerville, S. V., Gooding, J. J. & Tilley, R. D. Co-catalytic metal–support interactions in single-atom electrocatalysts. Nat. Rev. Mater. 9, 173–189 (2024).
Medford, A. J. et al. From the Sabatier principle to a predictive theory of transition-metal heterogeneous catalysis. J. Catal. 328, 36–42 (2015).
Shi, Z. et al. Phase-dependent growth of Pt on MoS2 for highly efficient H2 evolution. Nature 621, 300–305 (2023).
Liu, P. et al. Photochemical route for synthesizing atomically dispersed palladium catalysts. Science 352, 797–800 (2016).
Cui, W.-G. et al. Insights into the pH effect on hydrogen electrocatalysis. Chem. Soc. Rev. 53, 10253–10311 (2024).
Cerón-Carrasco, J. P. et al. Solvent polarity scales: determination of new ET(30) values for 84 organic solvents. J. Phys. Org. Chem. 27, 512–518 (2014).
Lewis, N. B., Bisbey, R. P., Westendorff, K. S., Soudackov, A. V. & Surendranath, Y. A molecular-level mechanistic framework for interfacial proton-coupled electron transfer kinetics. Nat. Chem. 16, 343–352 (2024).
Manbeck, G. F., DiMarco, B. N., Rotundo, L., Polyansky, D. E. & Ertem, M. Z. Correlated solvent coordinates accelerate multi-donor proton-coupled electron transfer. Chem. Sci. https://doi.org/10.1039/D5SC06787A (2026).
Sebastián-Pascual, P., Herzog, A., Zhang, Y., Shao-Horn, Y. & Escudero-Escribano, M. Electrolyte effects in proton–electron transfer reactions and implications for renewable fuels and chemicals synthesis. Nat. Catal. 8, 986–999 (2025).
Warburton, R. E., Soudackov, A. V. & Hammes-Schiffer, S. Theoretical modeling of electrochemical proton-coupled electron transfer. Chem. Rev. 122, 10599–10650 (2022).
Syed, K. A., Pang, S.-F., Zhang, Y., Zeng, G. & Zhang, Y.-H. Micro-Raman observation on the HPO42− association structures in an individual dipotassium hydrogen phosphate (K2HPO4) droplet. J. Phys. Chem. A 116, 1558–1564 (2012).
Quinet, O., Champagne, B. & Rodriguez, V. Experimental and theoretical investigation of the Raman and hyper-Raman spectra of acetonitrile and its derivatives. J. Chem. Phys. 124, 244312 (2006).
Schwan, J., Ulrich, S., Batori, V., Ehrhardt, H. & Silva, S. Raman spectroscopy on amorphous carbon films. J. Appl. Phys. 80, 440–447 (1996).
Fleger, Y., Mastai, Y., Rosenbluh, M. & Dressler, D. Surface enhanced Raman spectroscopy of aromatic compounds on silver nanoclusters. Surf. Sci. 603, 788–793 (2009).
Larkin, P., Makowski, M., Colthup, N. & Flood, L. Vibrational analysis of some important group frequencies of melamine derivatives containing methoxymethyl, and carbamate substituents: mechanical coupling of substituent vibrations with triazine ring modes. Vib. Spectrosc. 17, 53–72 (1998).
Ellis, G., Naffakh, M., Marco, C. & Hendra, P. Fourier transform Raman spectroscopy in the study of technological polymers part 1: poly(aryl ether ketones), their composites and blends. Spectrochim. Acta A 53, 2279–2294 (1997).
Do, V. H. & Lee, J. M. Transforming adsorbate surface dynamics in aqueous electrocatalysis: pathways to unconstrained performance. Adv. Mater. 37, 2417516 (2025).
Kiani, D. & Wachs, I. E. Practical considerations for understanding surface reaction mechanisms involved in heterogeneous catalysis. ACS Catal. 14, 16770–16784 (2024).
Meijers, S. & Ponec, V. Infrared spectroscopic study of the adsorption of nitro compounds and amines on cobalt oxide. J. Catal. 149, 307–316 (1994).
Dinodia, M. N-heterocycles: recent advances in biological applications. Mini Rev. Org. Chem. 20, 735–747 (2023).
Ley, J. P. & Bertram, H.-J. Hydroxy-or methoxy-substituted benzaldoximes and benzaldehyde-O-alkyloximes as tyrosinase inhibitors. Bioorg. Med. Chem. 9, 1879–1885 (2001).
Elsherbiny, D. A. et al. Bioactive tri-component nanofibers from cellulose acetate/lignin//N-vanillidene-phenylthiazole copper-(II) complex for potential diaper dermatitis control. Int. J. Biol. Macromol. 205, 703–718 (2022).
Kaya, I., Solak, E. & Kamaci, M. Synthesis and multicolor, photophysical, thermal, and conductivity properties of poly(imine)s. J. Taiwan Inst. Chem. Eng. 123, 328–337 (2021).
Wang, L. et al. Balancing mechanical performance and repair ability of epoxy and its composites via dual dynamic mechanism. Polymer 336, 128881 (2025).
Bae, S. J. et al. Design, synthesis, and evaluation of (E)-N-substituted benzylidene–aniline derivatives as tyrosinase inhibitors. Eur. J. Med. Chem. 57, 383–390 (2012).
Sahni, T., Sharma, S., Verma, D., Kaur, H. & Kaur, A. Synthesis and in vitro fungitoxic evaluation of syringaldehyde Schiff bases and β-lactams. Org. Prep. Proced. Int. 54, 370–379 (2022).
Cheng, W. et al. Lattice-strained metal–organic-framework arrays for bifunctional oxygen electrocatalysis. Nat. Energy 4, 115–122 (2019).
Abraham, M. J. et al. GROMACS: high performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 1, 19–25 (2015).
Wang, J., Wolf, R. M., Caldwell, J. W., Kollman, P. A. & Case, D. A. Development and testing of a general amber force field. J. Comput. Chem. 25, 1157–1174 (2004).
Maier, J. A. et al. ff14SB: improving the accuracy of protein side chain and backbone parameters from ff99SB. J. Chem. Theory Comput. 11, 3696–3713 (2015).
Darden, T., York, D. & Pedersen, L. Particle mesh Ewald: an N⋅log(N) method for Ewald sums in large systems. J. Chem. Phys. 98, 10089–10092 (1993).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169 (1996).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for open-shell transition metals. Phys. Rev. B 48, 13115 (1993).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953 (1994).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758 (1999).
Henkelman, G. & Jónsson, H. A dimer method for finding saddle points on high dimensional potential surfaces using only first derivatives. J. Chem. Phys. 111, 7010–7022 (1999).
Henkelman, G., Uberuaga, B. P. & Jónsson, H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 113, 9901–9904 (2000).
Acknowledgements
This work was supported by the National Key R&D Program of China (grant no. 2023YFA1507400) and the National Natural Science Foundation of China (grant nos. U24A20498 and 22425021).
Author information
Authors and Affiliations
Contributions
Y. Zou and S.W. conceived the idea and directed the research. Y.H. designed the experiments. Y.H., S.M. and X.Z. performed research; Y.H. and Yajing Zhang contributed to the theoretical calculations. Z.L., Y.Y. and Z.X. analysed data; Yuhao Zhang and Q.L. performed SR–FTIR measurements; T.T.T.N. and C.-L.D. performed XANES measurements. Y. Zou and Y.H. wrote the paper. All authors have approved the final version of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Haohong Duan, Jason Chun-Ho Lam and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Eric Piechota, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Figs. 1–64 and Tables 1–4.
Source data
Source Data Fig. 1 (download XLSX )
Source data for Fig. 1a–f,h.
Source Data Fig. 2 (download XLSX )
Source data for Fig. 2a,b,d,e.
Source Data Fig. 3 (download XLSX )
Source data for Fig. 3a–d.
Source Data Fig. 4 (download XLSX )
Source data for Fig. 4d–f.
Source Data Fig. 5 (download XLSX )
Source data for Fig. 5a–h and EQCM.
Source Data Fig. 6 (download XLSX )
Source data for Fig. 6a–d.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, Y., Mo, S., Zeng, X. et al. Paired electrolysis enables selective C–N coupling for the synthesis of nitrogenous aromatics. Nat. Synth (2026). https://doi.org/10.1038/s44160-026-01057-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44160-026-01057-w