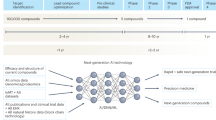
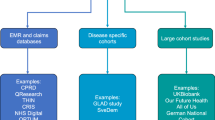
Abstract
Clinical trials are at the core of evidence-based medicine, but many are underpowered and fail to inform clinical practice. In mental health, the number of regulatory drug approvals has consistently lagged behind other areas of medicine, the effects of established therapies may vary, and comparative effectiveness data for available treatments are scarce. Thus, there is an urgent need for more efficient, faster and more collaborative ways of generating evidence. Traditional approaches of ‘one treatment, one trial’ are slow, inefficient, and limit comparability across trials. In contrast, platform trials use a shared infrastructure for many treatments, shared control group(s) and a master protocol that allows treatments to be added over time and ineffective ones to be dropped early. Here we present examples of platform trials in mental health (M-PACT, EU-PEARLDIVER, PUMA and RESiLIENT) and discuss their potential to increase speed, reduce operational costs and participant burden, and improve statistical power and comparability.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$79.00 per year
only $6.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Grimes, D. A. & Schulz, K. F. An overview of clinical research: the lay of the land. Lancet 359, 57–61 (2002).
Wouters, O. J., McKee, M. & Luyten, J. Estimated research and development investment needed to bring a new medicine to market, 2009-2018. JAMA 323, 844–853 (2020).
DiMasi, J. A., Grabowski, H. G. & Hansen, R. W. Innovation in the pharmaceutical industry: new estimates of R&D costs. J. Health Econ. 47, 20–33 (2016).
Chevance, A., Ravaud, P., Cornelius, V., Mayo-Wilson, E. & Furukawa, T. A. Designing clinically useful psychopharmacological trials: challenges and ways forward. Lancet Psychiatry 9, 584–594 (2022).
de Vries, Y. A., Schoevers, R. A., Higgins, J. P. T., Munafò, M. R. & Bastiaansen, J. A. Statistical power in clinical trials of interventions for mood, anxiety and psychotic disorders. Psychol. Med. 53, 4499–4506 (2023).
Leucht, S., Hierl, S., Kissling, W., Dold, M. & Davis, J. M. Putting the efficacy of psychiatric and general medicine medication into perspective: review of meta-analyses. Br. J. Psychiatry 200, 97–106 (2012).
Mullard, A. 2023 FDA approvals. Nat. Rev. Drug Discov. 23, 88–95 (2024).
Zhu, T. Challenges of psychiatry drug development and the role of human pharmacology models in early development—a drug developer’s perspective. Front. Psychiatry 11, 562660 (2020).
Tranberg, K. et al. Challenges in reaching patients with severe mental illness for trials in general practice—a convergent mixed methods study based on the SOFIA pilot trial. Pilot Feasibility Stud. 9, 182 (2023).
Gold, S. M., Landray, M. J., Medhurst, N. & Otte, C. Fast tracking informative clinical trials: lessons for mental health. Lancet Psychiatry 10, 376–378 (2023).
Adaptive Platform Trials Coalition. Adaptive platform trials: definition, design, conduct and reporting considerations. Nat. Rev. Drug Discov. 18, 797–807 (2019).
Wang, H. & Yee, D. I-SPY 2: a neoadjuvant adaptive clinical trial designed to improve outcomes in high-risk breast cancer. Curr. Breast Cancer Rep. 11, 303–310 (2019).
James, N. D. et al. STAMPEDE: Systemic Therapy for Advancing or Metastatic Prostate Cancer—a multi-arm multi-stage randomised controlled trial. Clin. Oncol. (R. Coll. Radiol.) 20, 577–581 (2008).
Peto, L., Horby, P. & Landray, M. Establishing COVID-19 trials at scale and pace: experience from the RECOVERY trial. Adv. Biol. Regul. 86, 100901 (2022).
Angus, D. C. et al. The REMAP-CAP (Randomized Embedded Multifactorial Adaptive Platform for Community-Acquired Pneumonia) Study. Rationale and design. Ann. Am. Thorac. Soc. 17, 879–891 (2020).
Griessbach, A. et al. Characteristics, progression and output of randomized platform trials: a systematic review. JAMA Netw. Open 7, e243109 (2024).
Blackwell, S. E. et al. Demonstration of a ‘leapfrog’ randomized controlled trial as a method to accelerate the development and optimization of psychological interventions. Psychol. Med. 53, 6113–6123 (2023).
Gold, S. M. et al. Control conditions for randomised trials of behavioural interventions in psychiatry: a decision framework. Lancet Psychiatry 4, 725–732 (2017).
Gold, S. M. et al. Platform trials and the future of evaluating therapeutic behavioural interventions. Nat. Rev. Psychol. 1, 7–8 (2022).
Blackwell, S. E., Woud, M. L., Margraf, J. & Schönbrodt, F. D. Introducing the leapfrog design: a simple Bayesian adaptive rolling trial design for accelerated treatment development and optimization. Clin. Psychol. Sci. 7, 1222–1243 (2019).
Koenig, F. et al. Current state-of-the-art and gaps in platform trials: 10 things you should know, insights from EU-PEARL. EClinicalMedicine 67, 102384 (2024).
Nguyen, Q. L. et al. Regulatory issues of platform trials: learnings from EU-PEARL. Clin. Pharmacol. Ther. 116, 52–63 (2024).
Gidh-Jain, M. et al. Developing generic templates to shape the future for conducting integrated research platform trials. Trials. 25, 204 (2024).
Bschor, T., Nagel, L., Unger, J., Schwarzer, G. & Baethge, C. Differential outcomes of placebo treatment across 9 psychiatric disorders: a systematic review and meta-analysis. JAMA Psychiatry 81, 757–768 (2024).
Huneke, N.T.M. Placebo effects in randomized trials of pharmacological and neurostimulation interventions for mental disorders: an umbrella review. Mol. Psychiatry 29, 3915–3925 (2024).
Papakostas, G. I. & Fava, M. Does the probability of receiving placebo influence clinical trial outcome? A meta-regression of double-blind, randomized clinical trials in MDD. Eur. Neuropsychopharmacol. 19, 34–40 (2009).
Jones, B. D. M. et al. Magnitude of the placebo response across treatment modalities used for treatment-resistant depression in adults: a systematic review and meta-analysis. JAMA Netw. Open 4, e2125531 (2021).
Viele, K. Allocation in platform trials to maintain comparability across time and eligibility. Stat. Med. 42, 2811–2818 (2023).
Bofill Roig, M., Glimm, E., Mielke, T. & Posch, M. Optimal allocation strategies in platform trials with continuous endpoints. Stat. Methods Med. Res. 33, 858–874 (2024).
Bofill Roig, M. et al. On model-based time trend adjustments in platform trials with non-concurrent controls. BMC Med. Res. Methodol. 22, 228 (2022).
Lee, K. M. & Wason, J. Including non-concurrent control patients in the analysis of platform trials: is it worth it? BMC Med. Res. Method. 20, 165 (2020).
Saville, B. R., Berry, D. A., Berry, N. S., Viele, K. & Berry, S. M. The Bayesian time machine: accounting for temporal drift in multi-arm platform trials. Clin. Trials 19, 490–501 (2022).
Bofill Roig, M., König, F., Meyer, E. & Posch, M. Commentary: Two approaches to analyze platform trials incorporating non-concurrent controls with a common assumption. Clin. Trials 19, 502–503 (2022).
Henssler, J., Alexander, D., Schwarzer, G., Bschor, T. & Baethge, C. Combining antidepressants vs antidepressant monotherapy for treatment of patients with acute depression: a systematic review and meta-analysis. JAMA Psychiatry 79, 300–312 (2022).
Nuñez, N. A. et al. Augmentation strategies for treatment resistant major depression: a systematic review and network meta-analysis. J. Affect. Disord. 302, 385–400 (2022).
Scott, F. et al. Systematic review and meta-analysis of augmentation and combination treatments for early-stage treatment-resistant depression. J. Psychopharmacol. 37, 268–278 (2023).
Köhler-Forsberg, O., Otte, C., Gold, S. M. & Østergaard, S. D. Statins in the treatment of depression: hype or hope? Pharmacol. Ther. 215, 107625 (2020).
Toba-Oluboka, T., Vochosková, K. & Hajek, T. Are the antidepressant effects of insulin-sensitizing medications related to improvements in metabolic markers? Transl. Psychiatry 12, 469 (2022).
Drevets, W. C., Wittenberg, G. M., Bullmore, E. T. & Manji, H. K. Immune targets for therapeutic development in depression: towards precision medicine. Nat. Rev. Drug Discov. 21, 224–244 (2022).
Köhler-Forsberg, O. et al. Efficacy of anti-inflammatory treatment on major depressive disorder or depressive symptoms: meta-analysis of clinical trials. Acta Psychiatr. Scand. 139, 404–419 (2019).
Deisenhofer, A. K. et al. Implementing precision methods in personalizing psychological therapies: barriers and possible ways forward. Behav. Res. Ther. 172, 104443 (2024).
Blackwell, S. E. Using the ‘leapfrog’ design as a simple form of adaptive platform trial to develop, test and implement treatment personalization methods in routine practice. Adm. Policy Ment. Health 51, 686–701 (2024).
Freitag, M.M. et al. Design considerations for a Phase II platform trial in major depressive disorder. Preprint at https://arxiv.org/abs/2310.02080 (2023).
Cunniffe, N. et al. Systematic approach to selecting licensed drugs for repurposing in the treatment of progressive multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 92, 295–302 (2021).
Furukawa, T. A. et al. Four 2 × 2 factorial trials of smartphone CBT to reduce subthreshold depression and to prevent new depressive episodes among adults in the community-RESiLIENT trial (Resilience Enhancement with Smartphone in LIving ENvironmenTs): a master protocol. BMJ Open 13, e067850 (2023).
Michopoulos, I. et al. Different control conditions can produce different effect estimates in psychotherapy trials for depression. J. Clin. Epidemiol. 132, 59–70 (2021).
Furukawa, T. A. et al. Dismantling, optimising and personalising internet cognitive behavioural therapy for depression: a systematic review and component network meta-analysis using individual participant data. Lancet Psychiatry 8, 500–511 (2021).
Cuijpers, P. et al. Psychotherapies for depression: a network meta-analysis covering efficacy, acceptability and long-term outcomes of all main treatment types. World Psychiatry 20, 283–293 (2021).
Karyotaki, E. et al. Association of task-shared psychological interventions with depression outcomes in low- and middle-income countries: a systematic review and individual patient data meta-analysis. JAMA Psychiatry 79, 430–443 (2022).
Author information
Authors and Affiliations
Contributions
S.M.G. and C.O. developed the overall concept. S.M.G. wrote the first draft of the manuscript and Table 1. S.M.G., J.B., M.M.F., F.K., M.P., E.R.K. and T.A.F. prepared the figures. E.R.K., S.M.G., B.L. and T.A.F. wrote Box 1. F.-L.M. and K.D. wrote Box 2. F.-L.M., K.D., J.B., M.M.F., F.K., M.P., J.A.R.-Q., F.B., O.K.-F., N.G., W.H., C.M.P., E.R.K., S.W., B.L., T.A.F. and C.O. edited, reviewed and refined all versions of the manuscript.
Corresponding author
Ethics declarations
Competing interests
S.M.G. reports honoraria from Hexal, Angelini and Tegus. O.K.-F. reports honoraria for lectures for Lundbeck Pharma A/S and consultant work for WCG Clinical. J.A.R.-Q. was on the speakers’ bureau and/or acted as consultant for Biogen, Idorsia, Casen-Recordati, Janssen-Cilag, Novartis, Takeda, Bial, Sincrolab, Neuraxpharm, Novartis, BMS, Medice, Rubió, Uriach, Technofarma and Raffo in the last three years. He also received travel awards (air tickets and hotel) for taking part in psychiatric meetings from Idorsia, Janssen-Cilag, Rubió, Takeda, Bial and Medice. The Department of Psychiatry chaired by him has received unrestricted educational and research support from the following companies in the last three years: Exeltis, Idorsia, Janssen-Cilag, Neuraxpharm, Oryzon, Roche, Probitas and Rubió. C.P. reports consultation and speaker’s fees from Boehringer-Ingelheim, Eli Lilly, Compass, Eleusis, GH Research, Lundbeck and Värde Partners. T.A.F. reports personal fees from Boehringer-Ingelheim, Daiichi Sankyo, DT Axis, Kyoto University Original, Shionogi, SONY and UpToDate, a grant from DT Axis and Shionogi, patent 7448125 concerning use of machine learning in internet cognitive–behavioural therapy (iCBT), and a pending patent 2022-082495 about prediction models for depression relapse, and intellectual properties for Kokoro-app (a smartphone CBT app) licensed to Mitsubishi-Tanabe. C.O. reports honoraria for lectures and/or scientific advice from Boehringer-Ingelheim, Janssen, Neuraxpharm, Oberberg Kliniken, Peak Profiling and Limes Klinikgruppe. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Mental Health thanks Urska Arnautovska, Simon Blackwell and Matt Muijen for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gold, S.M., Mäntylä, FL., Donoghue, K. et al. Transforming the evidence landscape in mental health with platform trials. Nat. Mental Health 3, 276–285 (2025). https://doi.org/10.1038/s44220-025-00391-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44220-025-00391-w