Abstract
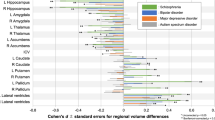
Psychotic and mood disorders, including schizophrenia and bipolar disorder, are increasingly viewed as part of a psychosis spectrum disorder (PSD) continuum, sharing genetic and neurobiological features. This systematic review and meta-analysis examines fractional anisotropy and mean diffusivity using diffusion tensor imaging in PSD. Across 96 studies (N = 4,424 PSD, N = 5,004 controls for fractional anisotropy; N = 1,607 PSD, N = 1,709 controls for mean diffusivity), fractional anisotropy reductions were consistently observed in the corpus callosum, whereas mean diffusivity increases were found in cortico-spinal projections. Controlling for age and gender strengthened these findings, suggesting that they contribute to PSD pathophysiology rather than reflecting disease progression. Subgroup analyses revealed overlapping but distinct patterns in schizophrenia and bipolar disorder. These findings support a transdiagnostic model of psychosis, with corpus callosum abnormalities as a potential biomarker. Future longitudinal studies are needed to clarify causality and clinical implications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$79.00 per year
only $6.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
All neuroimaging outputs (.nii.gz files and HTML reports) used in the analyses are available in Supplementary Information submitted with the paper. A summary table detailing study characteristics, methods and main findings is also included. Source data are provided with this paper.
Code availability
The SDM code is publicly accessible at https://www.sdmproject.com/.
References
Fusar-Poli, P. et al. Transdiagnostic psychiatry: a systematic review. World Psychiatry 18, 192–207 (2019).
Heckers, S. et al. Structure of the psychotic disorders classification in DSM-5. Schizophr. Res. 150, 11–14 (2013).
Keshavan, M. S. et al. A dimensional approach to the psychosis spectrum between bipolar disorder and schizophrenia: the Schizo-Bipolar Scale. Schizophr. Res. 133, 250–254 (2011).
Lee, P. H., Feng, Y.-C. A. & Smoller, J. W. Pleiotropy and cross-disorder genetics among psychiatric disorders. Biol. Psychiatry 89, 20–31 (2021).
Merola, G. P. et al. Transdiagnostic markers across the psychosis continuum: a systematic review and meta-analysis of resting state fMRI studies. Front. Psychiatry 15, 1378439 (2024).
Meda, S. A. et al. Multivariate analysis reveals genetic associations of the resting default mode network in psychotic bipolar disorder and schizophrenia. Proc. Natl Acad. Sci. USA 111, E2066–E2075 (2014).
O’Donnell, L. J. & Westin, C.-F. An introduction to diffusion tensor image analysis. Neurosurg. Clin. N. Am. 22, 185–196 (2011).
Vos, S. B., Jones, D. K., Jeurissen, B., Viergever, M. A. & Leemans, A. The influence of complex white matter architecture on the mean diffusivity in diffusion tensor MRI of the human brain. Neuroimage 59, 2208–2216 (2012).
Alexander, A. L. et al. Characterization of cerebral white matter properties using quantitative magnetic resonance imaging stains. Brain Connect. 1, 423–446 (2011).
Yang, X., Cao, D., Liang, X. & Zhao, J. Schizophrenia symptomatic associations with diffusion tensor imaging measured fractional anisotropy of brain: a meta-analysis. Neuroradiology 59, 699–708 (2017).
Vederine, F.-E., Wessa, M., Leboyer, M. & Houenou, J. A meta-analysis of whole-brain diffusion tensor imaging studies in bipolar disorder. Prog. Neuropsychopharmacol. Biol. Psychiatry 35, 1820–1826 (2011).
Xu, M. et al. Structural connectivity associated with familial risk for mental illness: a meta-analysis of diffusion tensor imaging studies in relatives of patients with severe mental disorders. Hum. Brain Mapp. 43, 2936–2950 (2022).
Clark, K. A. et al. Mean diffusivity and fractional anisotropy as indicators of disease and genetic liability to schizophrenia. J. Psychiatr. Res. 45, 980–988 (2011).
Tønnesen, S. et al. White matter aberrations and age-related trajectories in patients with schizophrenia and bipolar disorder revealed by diffusion tensor imaging. Sci. Rep. 8, 14129 (2018).
Kelly, S. et al. Widespread white matter microstructural differences in schizophrenia across 4322 individuals: results from the ENIGMA Schizophrenia DTI Working Group. Mol. Psychiatry 23, 1261–1269 (2018).
Favre, P. et al. Widespread white matter microstructural abnormalities in bipolar disorder: evidence from mega- and meta-analyses across 3033 individuals. Neuropsychopharmacology 44, 2285–2293 (2019).
Birur, B., Kraguljac, N. V., Shelton, R. C. & Lahti, A. C. Brain structure, function, and neurochemistry in schizophrenia and bipolar disorder-a systematic review of the magnetic resonance neuroimaging literature. npj Schizophr. 3, 15 (2017).
Reddaway, J. T. et al. Genomic and imaging biomarkers in schizophrenia. Curr. Top. Behav. Neurosci. 40, 325–352 (2018).
Canales-Rodríguez, E. J. et al. Widespread intra-axonal signal fraction abnormalities in bipolar disorder from multicompartment diffusion MRI: sensitivity to diagnosis, association with clinical features and pharmacologic treatment. Hum. Brain Mapp. 44, 4605–4622 (2023).
Friedman, J. I. et al. Diffusion tensor imaging findings in first-episode and chronic schizophrenia patients. Am. J. Psychiatry 165, 1024–1032 (2008).
Collinson, S. L. et al. Corpus callosum morphology in first-episode and chronic schizophrenia: combined magnetic resonance and diffusion tensor imaging study of Chinese Singaporean patients. Br. J. Psychiatry 204, 55–60 (2014).
Klaassen, A.-L. et al. Reduced anterior callosal white matter in risk for psychosis associated with processing speed as a fundamental cognitive impairment. Schizophr. Res. 264, 211–219 (2024).
Francis, A. N. et al. Abnormalities of the corpus callosum in non-psychotic high-risk offspring of schizophrenia patients. Psychiatry Res. 191, 9–15 (2011).
Popoola, O. et al. Neuropsychiatric manifestations of partial agenesis of the corpus callosum: A case report and literature review. Case Rep. Psychiatry 2019, 5925191 (2019).
Ghavipisheh, M., Jahromi, L. R., Ahrari, I. & Jahromi, M. G. Complete agenesis of corpus callosum with a rare neuropsychiatric presentation: A case report. Radiol. Case Rep. 18, 1442–1445 (2023).
Walterfang, M. & Velakoulis, D. Callosal morphology in schizophrenia: what can shape tell us about function and illness? Br. J. Psychiatry 204, 9–11 (2014).
Vallée, A. Neuroinflammation in schizophrenia: the key role of the WNT/β-catenin pathway. Int. J. Mol. Sci. 23, 2810 (2022).
Rund, B. R. Does active psychosis cause neurobiological pathology? A critical review of the neurotoxicity hypothesis. Psychol. Med. 44, 1577–1590 (2014).
Kristensen, T. D. et al. Fibre density and fibre-bundle cross-section of the corticospinal tract are distinctly linked to psychosis-specific symptoms in antipsychotic-naïve patients with first-episode schizophrenia. Eur. Arch. Psychiatry Clin. Neurosci. 273, 1797–1812 (2023).
Cetin-Karayumak, S. et al. White matter abnormalities across the lifespan of schizophrenia: a harmonized multi-site diffusion MRI study. Mol. Psychiatry 25, 3208–3219 (2020).
Stone, W. S. et al. Neurodegenerative model of schizophrenia: growing evidence to support a revisit. Schizophr. Res. 243, 154–162 (2022).
Kochunov, P. & Hong, L. E. Neurodevelopmental and neurodegenerative models of schizophrenia: white matter at the center stage. Schizophr. Bull. 40, 721–728 (2014).
Whitford, T. J. et al. Corpus callosum abnormalities and their association with psychotic symptoms in patients with schizophrenia. Biol. Psychiatry 68, 70–77 (2010).
Innocenti, G. M., Ansermet, F. & Parnas, J. Schizophrenia, neurodevelopment and corpus callosum. Mol. Psychiatry 8, 261–274 (2003).
Martin, B., Giersch, A., Huron, C. & van Wassenhove, V. Temporal event structure and timing in schizophrenia: preserved binding in a longer ‘now’. Neuropsychologia 51, 358–371 (2013).
Walther, S. et al. Aberrant hyperconnectivity in the motor system at rest is linked to motor abnormalities in schizophrenia spectrum disorders. Schizophr. Bull. 43, 982–992 (2017).
Kraguljac, N. V. et al. Neuroimaging biomarkers in schizophrenia. Am. J. Psychiatry 178, 509–521 (2021).
Kempton, M. J. & McGuire, P. How can neuroimaging facilitate the diagnosis and stratification of patients with psychosis?. Eur. Neuropsychopharmacol. 25, 725–732 (2015).
Tarcijonas, G. & Sarpal, D. K. Neuroimaging markers of antipsychotic treatment response in schizophrenia: An overview of magnetic resonance imaging studies. Neurobiol. Dis. 131, 104209 (2019).
Szeszko, P. R. et al. White matter changes associated with antipsychotic treatment in first-episode psychosis. Neuropsychopharmacology 39, 1324–1331 (2014).
Penadés, R. et al. Brain effects of cognitive remediation therapy in schizophrenia: a structural and functional neuroimaging study. Biol. Psychiatry 73, 1015–1023 (2013).
Mittal, V. A. et al. Exercise treatments for psychosis: a review. Curr. Treat. Options Psychiatry 4, 152–166 (2017).
Hsu, M.-C., Huang, Y.-S. & Ouyang, W.-C. Beneficial effects of omega-3 fatty acid supplementation in schizophrenia: possible mechanisms. Lipids Health Dis. 19, 159 (2020).
Voineskos, A. N. et al. Effects of repetitive transcranial magnetic stimulation on working memory performance and brain structure in people with schizophrenia spectrum disorders: A double-blind, randomized, sham-controlled trial. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 6, 449–458 (2021).
Kirschner, M. et al. Cortical and subcortical neuroanatomical signatures of schizotypy in 3004 individuals assessed in a worldwide ENIGMA study. Mol. Psychiatry 27, 1167–1176 (2022).
Morgan, K. D. et al. Differing patterns of brain structural abnormalities between black and white patients with their first episode of psychosis. Psychol. Med. 40, 1137–1147 (2010).
Gong, Q. et al. A neuroanatomical signature for schizophrenia across different ethnic groups. Schizophr. Bull. 41, 1266–1275 (2015).
Cardenas-Iniguez, C. & Gonzalez, M. R. Recommendations for the responsible use and communication of race and ethnicity in neuroimaging research. Nat. Neurosci. 27, 615–628 (2024).
Rauch, J. M. & Eliot, L. Breaking the binary: gender versus sex analysis in human brain imaging. Neuroimage 264, 119732 (2022).
Menzler, K. et al. Men and women are different: diffusion tensor imaging reveals sexual dimorphism in the microstructure of the thalamus, corpus callosum and cingulum. Neuroimage 54, 2557–2562 (2011).
Page, M. J. et al. PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. Br. Med. J. 372, n160 (2021).
Rethlefsen, M. L. et al. PRISMA-S: an extension to the PRISMA statement for reporting literature searches in systematic reviews. Syst. Rev. 10, 39 (2021).
Spijker, R., Dinnes, J., Glanville, J. & Eisinga, A. in Cochrane Handbook for Systematic Reviews of Diagnostic Test Accuracy (eds Deeks, J. J. et al.) https://doi.org/10.1002/9781119756194.ch6 (Wiley, 2023).
Ouzzani, M., Hammady, H., Fedorowicz, Z. & Elmagarmid, A. Rayyan—a web and mobile app for systematic reviews. Syst. Rev. 5, 210 (2016).
Müller, V. I. et al. Ten simple rules for neuroimaging meta-analysis. Neurosci. Biobehav. Rev. 84, 151–161 (2018).
Parlatini, V. et al. White matter alterations in attention-deficit/hyperactivity disorder (ADHD): a systematic review of 129 diffusion imaging studies with meta-analysis. Mol. Psychiatry 28, 4098–4123 (2023).
Aoki, Y., Cortese, S. & Castellanos, F. X. Research review: diffusion tensor imaging studies of attention-deficit/hyperactivity disorder: meta-analyses and reflections on head motion. J. Child Psychol. Psychiatry 59, 193–202 (2018).
Radua, J. et al. A new meta-analytic method for neuroimaging studies that combines reported peak coordinates and statistical parametric maps. Eur. Psychiatry 27, 605–611 (2012).
Radua, J. et al. Anisotropic kernels for coordinate-based meta-analyses of neuroimaging studies. Front. Psychiatry 5, 13 (2014).
Chen, L. et al. A systematic review and meta-analysis of tract-based spatial statistics studies regarding attention-deficit/hyperactivity disorder. Neurosci. Biobehav. Rev. 68, 838–847 (2016).
Rorden, C. From MRIcro to MRIcron: the evolution of neuroimaging visualization tools. Neuropsychologia 207, 109067 (2025).
Holmes, C. et al. Enhancement of MR images using registration for signal averaging. J. Comput. Assist. Tomogr. 22, 324–333 (1998).
Jiang, B., He, D., Guo, Z. & Gao, Z. Effect-size seed-based d mapping of resting-state fMRI for persistent insomnia disorder. Sleep Breath. 24, 653–659 (2020).
Albajes-Eizagirre, A., Solanes, A., Vieta, E. & Radua, J. Voxel-based meta-analysis via permutation of subject images (PSI): theory and implementation for SDM. Neuroimage 186, 174–184 (2019).
Higgins, J. P. T., Thompson, S. G., Deeks, J. J. & Altman, D. G. Measuring inconsistency in meta-analyses. Br. Med. J. 327, 557–560 (2003).
Acknowledgements
The preregistered protocol can be retrieved at https://osf.io/urk4y.
Author information
Authors and Affiliations
Contributions
G.P.M.: conceptualization, methodology, formal analysis, investigation, writing—original draft, writing—review and editing. L.T.: conceptualization, methodology, formal analysis, validation, writing—original draft, writing—review and editing. L.F.S.: conceptualization, methodology, formal analysis, investigation, writing—original draft, writing—review and editing. F.D.: writing—review and editing. C.P.: supervision, writing—review and editing. D.V.D.V.: supervision, writing—review and editing. G.C.: supervision, writing—review and editing. V.R.: supervision, writing—review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Mental Health thanks Marta Garrido and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Tables 1–6 (download XLSX )
This file contains all supplementary tables: (1) Demographic and clinical characteristics (FA studies); (2) Demographic and clinical characteristics (MD studies); (3) CoCoPop (Condition, Context and Population) criteria; (4) Quality assessment; (5) Criteria quality assessment; (6) CRediT author statement.
Source data
Source Data Table 1 (download ZIP )
Blob reports for uncorrected results.
Source Data Table 2 (download ZIP )
Blob reports for FA, age- and gender-corrected results.
Source Data Table 2 (download ZIP )
Blob reports for MD, age- and gender-corrected results.
Source Data Table 3 (download ZIP )
Blob reports for results by diagnostic group, age- and gender-corrected.
Supplementary Dataset 1 (download XLSX )
Extracted data points of included studies for Fig. 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Merola, G.P., Tarchi, L., Saccaro, L.F. et al. A systematic review and meta-analysis of transdiagnostic impairments in white matter integrity across the psychosis continuum. Nat. Mental Health 4, 298–306 (2026). https://doi.org/10.1038/s44220-025-00573-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44220-025-00573-6