Abstract
Obsessive–compulsive disorder (OCD) is a condition with substantial disability and subclinical obsessive–compulsive behaviors affect up to 1 billion people globally. Transcranial electrical stimulation (tES) is emerging as a potential treatment for OCD, yet its effectiveness is uncertain due to the limited number of randomized controlled trials conducted so far. Here we applied 3 meta-analytic approaches to evaluate the pooled therapeutic effect size of included randomized controlled trials (ntDCS = 15, ntACS = 1) (pair-wise meta-analysis), compare the efficacy of various tES interventions (network meta-analysis), and explore how treatment effect size correlates with induced electrical field in the target brain regions (meta-modeling). With these 3 distinct meta-analytic approaches, we first show a significant moderate effect of tES interventions (standardized mean difference 0.61; 95% confidence interval [0.37 to 0.85]; P < 0.001) as a whole on reducing OCD severity, with larger therapeutic effects of twice-daily intervention, and with other stimulation parameters also moderating efficacy. Next, we identify specific tES interventions with superior indicated and estimated effects, including entraining alpha frequency alternating stimulation of the medial prefrontal cortex, excitability-diminishing cathodal stimulation over the supplementary motor area and orbitofrontal cortex, and excitability-enhancing anodal stimulation of the lateral prefrontal cortex. Finally, we show based on computational approaches that longer stimulation targeting the medial prefrontal and frontopolar cortices has greater therapeutic effects and suggest an optimized intervention accordingly. We finally discuss other potentially effective tES interventions according to the latest neurobiological theory of OCD. The results contribute to establishing the clinical efficacy of tES for OCD treatment while suggesting future studies to further evaluate current and future approaches to enhance the efficacy of interventions.
Similar content being viewed by others
Main
Obsessive–compulsive disorder (OCD) imposes a substantial burden on mental health and is associated with substantial disability1,2. With a lifetime prevalence of 2–3% (ref. 3), OCD often starts during childhood and follows a chronic course, is accompanied by functional impairment and higher risk of early mortality4,5,6, and is often underdiagnosed and undertreated2. Individuals with OCD have unwanted and distressing thoughts (obsessions) and repetitive behaviors that the individual feels driven to perform (compulsions)2. While cognitive behavioral therapy and serotonin reuptake inhibitor medication are considered first-line treatments for OCD7, less than 40% of patients respond fully to these treatments8.
Non-invasive brain stimulation (NIBS) techniques, as applied across cognitive neuroscience and neurophysiology, offer a safe way to alter physiological parameters underlying human behavior and cognition (for example, cortical excitability, neuroplasticity, brain oscillations) in both healthy individuals and clinical populations9,10,11. NIBS, namely, deep repetitive transcranial magnetic stimulation (rTMS), was approved by the US Food and Drug Administration (FDA) for the treatment of OCD in 201812, suggesting that related interventions may also have therapeutic applications in OCD. Transcranial electrical stimulation (tES), another NIBS method, uses a weak electrical current to modulate brain activity13,14 and its effectiveness for OCD treatment remains unclear despite several advantages over rTMS, such as fewer side effects, affordability and home-based applicability. The most common tES technique uses direct electrical current (known as transcranial direct current stimulation-tDCS) to change brain excitability15, producing subthreshold neuronal membrane polarization16, resulting in excitability-enhancing effects by anodal stimulation and excitability-reducing effects by cathodal stimulation at the macroscale level with typical protocols17,18. Transcranial alternating current stimulation (tACS), the next most common tES technique, uses weak oscillating electric currents hypothesized to entrain ongoing brain rhythms19. Transcranial random noise stimulation (tRNS) changes brain excitability via alternating currents at a mix of low or high frequencies20,21.
The rationale behind the therapeutic use of NIBS includes restoring impaired plasticity or re-balancing abnormally altered brain functions including oscillatory activities (that is, up- or downregulation)9,10,22,23. In neuropsychiatric disorders including OCD, functional brain abnormalities (that is, hyper- or hypoactivity of specific brain regions and networks) are well documented. Functional imaging, lesion and deep brain stimulation studies have shown altered activations in several brain regions involved in OCD pathophysiology, including the pre-supplementary motor area (pre-SMA), insula, cingulate cortex, prefrontal cortex (PFC) and orbitofrontal cortex (OFC)24,25,26,27,28, and altered cortical–subcortical connectivity (that is, hyper–hypoconnectivity)29,30. Furthermore, abnormal changes in neurotransmitter systems across frontostriatal pathways (for example, glutamatergic and dopaminergic hyperactivity) and frontolimbic systems (for example, serotonergic and γ‑aminobutyric acid (GABA)‑ergic hypoactivity) are observed in OCD31,32,33. Some of the observed brain alterations (for example, thinner precentral area, higher medial PFC–ventral striatal connectivity) are associated with treatment response to tDCS and rTMS in some studies34,35. These are common rationales behind applying rTMS or tES for OCD treatment. While the efficacy of rTMS for OCD treatment has been confirmed in previous studies36,37, the efficacy of tDCS remains under investigation.
One reason is the insufficient number of randomized controlled trials (RCTs) that have applied tES for treatment of OCD. The recently published reviews and meta-analyses, and systematic reviews include 7 to 10 RCTs38,39 with quasi-experimental studies included40 or did not address the clinical efficacy of tES41,42,43. In the past 2 years, however, several RCTs have investigated the efficacy of tES, particularly tDCS, for OCD treatment44,45,46,47,48,49,50,51. These studies allow us to conduct multiple meta-analyses on the efficacy of tES intervention for OCD treatment with more included RCTs, as highlighted as a necessity in previous reports40. Consequently, we performed three meta-analyses and a systematic review to evaluate the effectiveness and safety of tES (including tDCS, tACS and tRNS) for patients with OCD.
To this end and to address the limitations of common meta-analytic approaches, we conducted three separate but related meta-analyses. (1) A classic (pairwise) meta-analysis to determine the pooled effect estimate, quantifying the overall impact of tES interventions on OCD treatment and (2) a network meta-analysis to compare the efficacy of various tES interventions against a sham condition52. The latter method provides a Bayesian summary of the rank distribution based on relative strength and estimated treatment effects53, enabling a comprehensive assessment of the relative efficacy of different tES interventions in a single analysis54. This approach is essential for our research question, given the diverse intervention and stimulation protocols used in tES interventions for OCD symptoms. (3) Lastly, a computational meta-analysis focused on the correlations between tDCS-induced electrical field density in brain regions and treatment effect size, a recently introduced meta-analytic method for tES studies55. This three-level meta-analysis has been conducted to evaluate the efficacy of a NIBS intervention and is also an updated meta-analysis that solely contains tES RCTs published up to July 2025 (PROSPERO ID CRD42023485717). Our analyses, while revealing a statistically significant moderate therapeutic effect of tES for OCD treatment, highlight important stimulation parameters in this respect, with specific intervention shown to be more effective and model-suggested tES interventions.
Results
Literature search and study characteristics
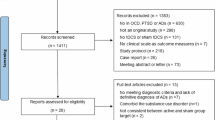
The study selection process is shown in Fig. 1. The final search yielded 829 records. After removing duplicates and screening abstracts against the inclusion criteria, 39 records were left for full-text assessment and data extraction. Twenty-two studies were excluded for the following reasons: open-label trials (n = 5)56,57,58,59,60, no control condition (n = 2)61,62, proceedings report (n = 2)63,64, lack of clinical measures or post-intervention Yale-Brown Obsessive Compulsive Scale (Y-BOCS) score (n = 4)65,66,67,68 and case studies (n = 9)69,70,71,72,73,74,75,76,77. At the end, 17 studies with 25 experimental conditions remained for pairwise and network meta-analyses and the meta-modeling analysis44,45,46,47,48,49,50,51,78,79,80,81,82,83,84,85,86. However, owing to evidence of publication bias, the study by Shafiezadeh et al.83 was identified as an outlier and excluded from the sensitivity analysis. In addition, five interventions from two studies51,80 were excluded from the network meta-analysis due to non-sham control conditions. This exclusion ensured that the network analysis primarily compared treatment arms with sham conditions. The Shafiezadeh et al. study83, however, was kept in the third meta-analysis as Spearman’s rank correlation effectively handles non-normally distributed data and is less affected by outliers than the parametric Pearson coefficient. As a result of these exclusions, 16 RCTs (15 tDCS, 1 tACS, concluding 23 effect sizes) remained in the final pairwise meta-analysis, 14 RCTs (13 tDCS, 1 tACS, concluding 18 intervention and 23 effect sizes) remained in the network meta-analysis, and 16 tDCS studies (concluding 24 effect sizes), remained for meta-modeling analysis (Fig. 1; results with the outlier study83 are in Supplementary Figs. 2 and 3). No tRNS RCT was among the included studies. In total, 551 patients with OCD were included (nactive = 318; nsham = 233) and sample size per study ranged from 23 to 80 participants. The summary of studies, including study design, blinding, age, gender, stimulation parameters, primary and secondary outcome measures, and major findings about intervention efficacy and tolerability, are outlined in Table 1 and Supplementary Table 1.
Of studies investigating the effects of tES on reducing OCD symptom severity, 17 RCTs with sham or active control conditions were included in the final analysis, resulting in a total of 25 experimental conditions (effect sizes). The pairwise meta-analysis comprised 16 studies (15 tDCS and 1 tACS) with 25 effect sizes following excluding an RCT with publication bias (n = 1), while the network meta-analysis included 14 studies (13 tDCS and 1 tACS) with 23 effect sizes following excluding RCTs with non-sham control (n = 2) and publication bias (n = 1). The meta-modeling analysis encompassed all tDCS 16 studies, with 24 effect sizes.
Dropout and side-effects analysis
First, we systematically analyzed reported side effects, dropout rates and serious adverse events, if reported, among the included RCTs. Supplementary Table 1 summarizes the dropout rate, side effects (with percentage) and serious adverse events. In 14 tES studies that reported dropout rates, there was no significant difference between active (14/231, 6.06%) and sham/control (14/192, 7.29%) groups (χ² = 0.26; P = 0.61; Fisher’s exact test, P = 0.69). The total sample sizes were 231 for the active group and 192 for the sham/control group, excluding studies that did not report side effects or dropouts (Supplementary Fig. 4a). None of the RCTs that documented side effects reported any serious adverse events (Supplementary Table 1). Regarding side effects, only seven studies numerically reported them. These studies varied in the domains assessed and the methods of reporting (for example, occurrence in patients as numbers/percentages versus average intensity). A detailed summary of the reported side effects is available in Supplementary Table 1 and Supplementary Fig. 4b. The average occurrence of commonly reported side effects among patients in the active versus sham conditions was as follows: tingling (active, 44.01%; sham, 34.40%), sleepiness (including drowsiness) (active, 38.9%; sham, 37.17%), skin redness (active, 22.05%; sham, 9.77%), itching (active, 19.7%; sham, 15.3%) and pain (including headache and neck pain; active, 11.91%; sham, 11.33%). The only tACS study reported minor adverse events in 8 patients of 25, including headache (8%), phosphenes (20%), tingling (16%) and itching (8%).
Pairwise meta-analysis of overall tES efficacy and stimulation-parameter effects
The pooled effect size for the included studies (16 RCTs) with 23 experimental conditions was significant with a medium effect size (standardized mean difference (SMD) = 0.61; 95% confidence interval (CI), [0.37 to 0.85]; P < 0.001). The following tES studies had significant effect size (in descending order) for reducing OCD (indicating larger effect size) compared with the sham intervention or baseline scores: Chu et al.51 with cathodal transcranial direct current stimulation (ctDCS) pre-SMA/left OFC (lOFC) + anodal transcranial direct current stimulation (atDCS) shoulder (SMD = 1.92; 95% CI, [0.57 to 3.26]); Perera et al.47 applying peak alpha tACS over the medial PFC (SMD = 1.67; 95% CI, [0.62 to 2.72]); Kumar et al.49 with ctDCS pre-SMA + atDCS left dorsolateral prefrontal cortex (lDLPFC; SMD = 1.30; 95% CI, [0.51 to 2.09]); Yekta et al.85 combining atDCS right dorsolateral prefrontal cortex (rDLPFC) + ctDCS lDLPFC (SMD = 1.17; 95% CI, [0.04 to 2.31]); Fineberg et al.44 with ctDCS-lOFC + atDCS shoulder (SMD = 1.12; 95% CI, [0.23 to 2.01]); and Alizadehgoradel et al.45 with atDCS lDLPFC + ctDCS pre-SMA at 2 mA (SMD = 0.99; 95% CI, [0.11 to 1.86]) and 1 mA (SMD = 0.92; 95% CI, [0.04 to 1.80]) (Fig. 2a).
a, Forest plot showing pooled effect sizes of tES interventions44,45,46,47,48,49,50,51,78,79,80,81,82,84,85,86 (n = 16, k = 23) with SMD, 95% CIs and measures of heterogeneity (SMD = 0.61; 95% CI, [0.37 to 0.85]; heterogeneity, I2 = 40%, τ2 = 0.131, P = 0.02). b, Forest plot of pooled and single effect size for anodal tDCS interventions over cortical regions (k = 14) including SMD and 95% CIs and measures of heterogeneity (SMD = 0.52; 95% CI, [0.21 to 0.83]; heterogeneity, I2 = 40%, τ2 = 0.129, P = 0.06). c, Forest plot of pooled and single effect size for cathodal tDCS interventions over cortical regions (k = 20), including SMD, 95% CIs and measures of heterogeneity (SMD = 0.56; 95% CI, [0.34 to 0.78]; heterogeneity: I2 = 25%, τ2 = 0.088, P = 0.15). d, Forest plot of pooled effect sizes for tES intervention at twice-daily (k = 10) versus once-daily (k = 11) frequency, including SMD, 95% CIs and measures of heterogeneity (SMD = 0.62; 95% CI, [0.36 to 0.88]; heterogeneity: I2 = 42%, τ2 = 0.141, P = 0.02). e, Forest plot of pooled and single effect size for treatment duration of 2 days (k = 2), 5 days or 1 week (k = 7), 2 weeks (k = 9), 4 weeks (k = 2) and 8 weeks (k = 1) of tES interventions (both tDCS and tACS), including SMD and 95% CIs and measures of heterogeneity (SMD = 0.61; 95% CI, [0.37 to 0.85]; heterogeneity: I2 = 40%, τ2 = 0.131, P = 0.02). f, Forest plot of pooled and single effect size for total sessions of 4 (k = 2), 10 (k = 13), 15 (k = 1), 20 (k = 5) and 24 (k = 2) of tES interventions (both tDCS and tACS), including SMD, and 95% CIs and measures of heterogeneity (SMD = 0.61; 95% CI, [0.37 to 0.85]; heterogeneity: I2 = 40%, τ2 = 0.131, P = 0.02). g, Forest plot of pooled and single effect size for stimulation intensity of 1 mA (k = 1), 1.5 mA (k = 2) and 2 mA (k = 20) in tES interventions, including SMD, 95% CIs and measures of heterogeneity (SMD = 0.61; 95% CI, [0.37 to 0.85]; heterogeneity, I2 = 40%, τ2 = 0.131, P = 0.02). SMD values were calculated using the Morris method. Larger SMD values indicate a larger reduction of OCD symptoms (measured by Y-BOCS). n represents the number of tES studies and k represents the number of effect sizes included in the main and secondary analyses. Squares represent individual study effect, diamonds represent pooled effect and lines represent CIs. SMD, standardized mean difference; CI, confidence interval; HK, Hartung-Knapp adjustment for random-effect meta-analysis.
In addition, we did secondary analyses of effect sizes with respect to important stimulation parameters (cortical region, polarity, treatment frequency per day, treatment duration, number of sessions and stimulation intensity). For cortical regions, regardless of stimulation polarity, significant reductions in OCD symptoms were observed in the lOFC as well as single effects reported for the lOFC + pre-SMA, medial PFC, and rDLPFC (Supplementary Fig. 5). However, as effect sizes derived from each target region are misleading due to the common practice of positioning anode and cathode electrodes on separate regions, we calculated effect sizes for each cortical region based on the stimulation polarity. The results show that only cathodal stimulation of the SMA (SMD = 0.52; k = 11; 95% CI, [0.16 to 0.87]; medium), regardless of anodal electrode placement and anodal stimulation of lDLPFC (SMD = 0.81; k = 4, 95% CI, [0.04 to 1.59]; large), regardless of cathodal electrode placement led to a significant reduction in OCD symptoms (Fig. 2b,c).
Next, analysis of stimulation frequency per day shows that both twice-daily (SMD = 0.83; k = 10; 95% CI, [0.44 to 1.23]; large) and once-daily (SMD = 0.42; k = 11; 95% CI, [0.06 to 0.78]; small) stimulation had a significant impact on reducing OCD symptom severity, albeit with large and small effects, respectively (Fig. 2d). Analysis of treatment duration (range 2 days, 5 days, 2 weeks, 3 weeks, 4 weeks, 6 weeks and 8 weeks), irrespective of target region or stimulation type/polarity, shows significant effects for 2-week tDCS (SMD = 0.60; k = 9; 95% CI, [0.06 to 1.13]; medium) and 5-day tDCS (SMD = 0.69; k = 7; 95% CI, [0.39 to 0.98]; medium). In 5-day tDCS studies, only 2 tDCS protocols had significant effects (SMD = 0.92; 95% CI, [0.04 to 1.80] for 1 mA; SMD = 0.99; 95% CI, [0.11 to 1.86] for 2 mA) and these protocols applied 10 tDCS sessions twice daily (Fig. 2e). In relation to this, we also analyzed the effect size obtained for total sessions (range 4, 10, 15, 20 and 24 sessions) regardless of treatment duration. Only the effect size of studies with 10 sessions (SMD = 0.52; k = 13; 95% CI, [0.20 to 0.84]; medium) and 20 sessions (SMD = 0.73; k = 5; 95% CI, [0.14 to 0.1.31]; medium to large) were significant (Fig. 2f). Finally, analysis of stimulation intensity (1 mA, 1.5 mA and 2 mA) showed a moderate effect size for 2-mA tDCS in reducing OCD symptoms (SMD = 0.57; k = 20; 95% CI [0.31 to 0.82]) (Fig. 2g).
In summary, meta-analysis of 23 effect sizes suggests a moderate overall impact of tES interventions on reducing OCD symptoms, and secondary analyses show significant effects of ctDCS of the SMA, atDCS of the lDLPFC, twice- and once-daily stimulation sessions, stimulation duration of 5 days and 2 weeks, total tDCS sessions of 10 and 20, and stimulation intensity of 2 mA for reducing OCD symptoms. To overcome the limitations of pairwise meta-analyses on specific tES protocols—such as pooled effect sizes that overlook stimulation parameters, sole reliance on behavioral measures, and focus of secondary analyses on a single parameter with low number of studies—we examined active treatment arms versus sham condition for reducing OCD symptoms (network meta-analysis) and assessed the correlation between behavioral changes in Y-BOCS and protocol-induced electrical fields (meta-modeling).
Network meta-analysis to identify superior tES interventions against sham
Twenty-five interventions from 17 tES RCTs (16 tDCS, 1 tACS) were initially included in the analysis. In addition to 1 study, due to publication bias83, 5 interventions from 2 studies51,80 had a non-sham control condition (for example, healthy volunteers or active control) and were excluded, which resulted in 14 RCTs with 18 interventions and 23 effect sizes. As the network analysis focuses on comparing treatment arms with sham conditions, we excluded these 3 interventions, lowering statistical heterogeneity from high (τ2 = 0.25, I2 = 60.1%; Q = 5.01, P = 0.08) to low (τ2 = 0.0, I2 = 0; Q = 0.88, P = 0.35). Figure 3a shows the network structure of the treatment arms for the primary outcomes (Y-BOCS). A total of 270 participants received the sham intervention and 264 were assigned to various tES interventions, including 1 alpha tACS and 15 tDCS interventions.
a, Network structure of the primary outcome. Each node represents a different tES intervention including sham (n = 18), with the size of the node proportional to the number of treatments. The edges between nodes represent direct comparisons between treatments (n = 23), with the thickness of the edges indicating the precision of the estimates (thicker lines represent smaller standard errors). The arrangement of nodes is optimized to enhance clarity. b, The network meta-analysis forest plot shows the SMD and 95% CIs for each treatment (n = 17) compared with the sham control (n = 1) in reducing OCD symptoms. Positive effect sizes indicate a greater reduction in OCD symptoms compared with sham. Squares represent individual study effect and lines represent CIs. c, Network ranking plots with each subplot representing the probability distribution of ranks for different treatments in the network meta-analysis. Higher probabilities at lower ranks indicate better treatment performance. The rankograms illustrate comparative effectiveness, with tACS_AFz showing the highest likelihood of being the most effective treatment, as reflected by its high SUCRA score, while the sham condition shows lower effectiveness as methodologically expected. We adopted the terminology from the papers for describing target regions which may not accurately explain the actual target region. Here is the clarification: Fz, FC1/FC2 and C3/C4 represent the supplementary and pre-SMAs; Fp1 denotes the lOFC and Fp2/F8 refers to the right OFC; F3 and F4 represent the lDLPFC and the rDLPFC, respectively. O1 and O2 represent the left and right cerebellum, respectively. AFz refers to the medial PFC. atDCS, anodal transcranial direct current stimulation; ctDCS, cathodal transcranial direct current stimulation; tACS, transcranial alternating current stimulation.
The following tES protocols significantly reduced OCD symptom severity compared with the sham control in descending order of effect size: peak alpha tACS over medial PFC (SMD = 1.67; 95% CI, [0.62 to 2.72]), ctDCS pre-SMA + atDCS lDLPFC (SMD = 1.30; 95% CI, [0.51 to 2.09]), ctDCS-lOFC + atDCS shoulder (SMD = 1.24; 95% CI, [0.39 to 2.10]), atDCS lDLPFC + ctDCS rDLPFC (SMD = 1.17; 95% CI, [0.04 to 2.31]), and atDCS lDLPFC + ctDCS pre-SMA at 2 mA (SMD = 0.99; 95% CI, [0.11 to 1.86]) and at 1 mA (SMD = 0.92; 95% CI, [0.04 to 1.80]) (Fig. 3b). We further analyzed the surface under the cumulative ranking (SUCRA) percentages representing the mean rank of interventions compared with an idealized hypothetical one based on 1,000 simulations. Among the top-7 interventions (>60% SUCRA) for reducing OCD severity, the above protocols ranked first (89.05%), second (78.56%), third (75.49%), fourth (72.72%), fifth (66.06%) and eighth (63.16%), respectively. In addition, interventions such as atDCS-O2 + ctDCS-O1 (SMD = 1; 64.55%), and ctDCS pre-SMA + atDCS shoulder (SMD = 1.02; 63.95%), ranked sixth and seventh for reducing OCD severity, showing promise for further study (Fig. 3c and Supplementary Tables 2 and 3).
Meta-modeling analysis of correlations between tDCS-induced electrical field and treatment response
The meta-modeling analysis was carried out on tDCS studies (n = 16) as this modeling approach is based on a consistent tDCS-induced polarity-dependent electrical field (while tACS electric fields have fluctuating polarities). The results show that tDCS is most effective in treating OCD symptoms if the electric fields target the medial prefrontal and frontopolar cortices (maximum performance–electric field correlation (PECE) = 0.351, P = 0.046, explained variance = 12.3%; Fig. 4a, first map). This was most strongly observed in the left hemisphere. The positive relationship between electric-field strength and effect size was even more pronounced when considering stimulation duration per treatment session (maximum PECE × min per day = 0.369, P = 0.038, explained variance = 13.2%, Fig. 4a, second map). Conversely, when the number of treatment days was taken into account, the relationship between electric fields and effect sizes did not reach significance (maximum PECE × number of days = 0.209, maximum PECE × min × day = 0.311, P > 0.05, Fig. 4a, third and fourth maps). Together, these results based on the included studies suggest that while longer stimulation durations at a given day benefit the therapeutic effect, longer total number of days of treatment may not.
a, PEC maps for correlations between effect size and electric-field strength (first map), cumulative charge in one session (E × min per day; second maps), cumulative charge across treatment days (E × number of days; third map), and total cumulative charge (E × min × day; fourth map). Color gradients indicate correlation strength: darker shades signify stronger positive correlations with symptom improvement, while lighter shades indicate negative correlations. b, The five functional brain regions (HCP-MMP atlas) with the largest average PECE × min per day values. Individual dots represent correlation values per brain model element within a region. Black dots represent individual correlation values per gray-matter element. The horizontal bar shows the median, the upper and lower boundaries show the 25th and 75th percentiles, respectively, and the whiskers show 5th–95th percentiles. c, Correlation between effect sizes and the perpendicular component of the electric field (PECperpE). The current direction follows an inward-to-outward (orange-to-blue) path. The most positive correlation between inward currents (perpE > 0) and effect sizes on the right flanks of gyri suggests a right-to-left current flow is associated with improvements in OCD symptoms. Gradient represents correlation with effect sizes; positive values (warmer shades per embedded color-bar key) indicate inward current flow associated with symptom improvement. d, An optimized montage showing the electric-field strength (normE) for improving OCD symptoms with tDCS. Anodes are shown in red and cathodes are shown in blue. Note that the largest normE values correspond to the largest PEC values as shown in a. e, The same as in d, but in this case, the perpendicular component of the electric field (perpE) is shown. Note that this distribution is similar to the one shown in c. 10pp, polar area 10; pOFC, posterior orbitofrontal cortex; OFC, orbitofrontal cortex; 47l, lateral area 47; 10d, dorsal area 10 according to the HCP-MMP atlas. PECE, performance–electric field correlation; normE, magnitude of the simulated electric field; perpE, perpendicular field component.
Subsequently, we mapped the functional regions associated with the largest effects associated with the PECE × min per day results using the Human Connectome Project multimodal parcellation (HCP-MMP) atlas87,88. Better treatment response was mostly associated with stimulation of the left dorsomedial PFC and OFC, corresponding to Brodmann areas 10, 11 and 47 (Fig. 4b). Finally, we used the PEC maps to suggest an optimized montage for tDCS based on the computational modeling results. Crucially, the current direction determines the placement of anodal and cathodal electrodes. Therefore, the PEC value was calculated for the perpendicular component of the electric field, which indicates the direction of current flow (from the anode (inward currents) to the cathode (outward currents); Methods). We found that a current flow from the right to the left PFC was positively correlated (PECperpE) with effect sizes, while a current flow from the left to the right PFC was negatively correlated with effect sizes (Fig. 4c). As such, a positive treatment outcome would follow from right-atDCS targeting the DLPFC and left-ctDCS targeting the frontopolar cortex (including the OFC and the ventromedial PFC (vmPFC)). The exact montage was modeled and resulted in anodes over F4 and F2 (1 mA each) and cathodes over AF3 and AF7 (−1 mA each), as shown in Fig. 4d,e.
Risk of bias and publication bias
Supplementary Fig. 6 presents the overall and individual risk of bias assessment results for the studies. Most included studies showed low (n = 8) to moderate (n = 8) risk of bias, while one (n = 1) showed high risk due to the randomization process. We marked six studies with ‘other bias’ for not using or reporting side-effect surveys. Only one study examined blinding efficacy47. However, we did not label the others with ‘other bias’, as the updated CONSORT guidelines do not recommend evaluating blinding efficacy89.
Publication bias was assessed using funnel plots (Supplementary Fig. 7a,b) and Egger’s test of the intercept, which indicated significant asymmetry (intercept = 3.386; 95% CI, [1.74–5.03]; t = 4.023; P = 0.0006). We additionally used the P-curve method to evaluate evidential value and identify potential P-hacking. Of the included studies, 7 (30.43%) reported P < 0.05 and 4 (17.39%) reported P < 0.025. The full P-curve showed no significant right-skewness (zFull = −1.317; pFull = 0.094), whereas the lower half was significantly right-skewed (zHalf = −1.876; pHalf = 0.03), indicating evidential value. The flatness test confirmed evidential value, as there was no flat distribution. The P-curve’s estimate of the true effect size (d = 0.508) was close to the meta-analytic estimate (SMD = 0.612), but the estimated statistical power was low (23%; 95% CI, 5–67.5%). These findings suggest that, despite small-study effects, the evidence base contains genuine effects, although the low power of individual studies warrants cautious interpretation.
Discussion
This study uses three distinct meta-analytic approaches to evaluate the efficacy of tES interventions for treating OCD symptoms. Different meta-analytic approaches were used to evaluate different aspects of treatment efficacy (for example, pooled effects, the effectiveness of interventions versus sham treatments, the ranking of promising interventions, the correlation between symptom reduction and induced electrical fields, and the prediction of optimized interventions) that cannot be obtained by one single analysis. Our analysis of 23 tES experimental conditions reveals a significant medium pooled effect size for reducing OCD symptoms (Fig. 5a). This effect is particularly strong for interventions that primarily downregulate the SMA and the OFC and upregulate the lDLPFC with cathodal and anodal stimulation, respectively, with a large effect size for twice-daily stimulation per day, and moderate effect for 5-day, 2-week, 10-session and 20-session interventions, and stimulation intensity of 2 mA. The top tES interventions, compared with sham conditions, were alpha tACS over the medial PFC, cathodal SMA-anodal lDLPFC tDCS, ctDCS of the OFC, and bilateral anodal right DLPFC tDCS (Fig. 5b). Finally, our computational analyses of behavior–electrical-field correlations, in line with results of network meta-analysis, show that tDCS is more effective when targeting the medial PFC and frontopolar cortex, particularly when session durations are longer. This analysis further suggests anodal stimulation of the right lateral and medial PFC cortices (F2–F4), combined with cathodal stimulation of the left frontopolar cortex (AF3–AF7), may more effectively reduce OCD severity due to larger PEC values, indicative of gray-matter modulation (Fig. 5c). We discuss key findings regarding OCD pathophysiology and stimulation parameters, evaluate the therapeutic efficacy of tES for treating OCD, and conclude with a discussion of promising future tES interventions.
OCD pathophysiology and tES intervention
Although the pathophysiology of OCD is complex and not fully understood, functional and structural abnormalities in the cortico–striato–thalamo–cortical (CSTS) circuits are well documented90,91,92. At the cortical level, DLPFC, medial PFC, OFC, anterior cingulate cortex (ACC), SMA and pre-SMA are critical regions within CSTS circuits characterized by altered functional and structural connectivity92,93,94. Specifically, overactivity in the OFC, medial PFC and SMA coupled with hyperconnectivity with subcortical regions is prominent in OCD92. At chemical levels, excitatory and inhibitory neurotransmitter systems (for example, glutamate, GABA) show imbalanced activity in patients with OCD compared with healthy controls33. The effectiveness of tES interventions in alleviating OCD symptoms can be attributed to at least two explanations related to the functional, structural and chemical abnormalities within CSTS circuits (Fig. 5d).
First, restoring and/or rebalancing functional abnormalities is the primary mechanism of effect for tES interventions. According to the multifaceted neural circuit theory of compulsivity92, three cortico–subcortical networks are involved in OCD-related pathophysiology (and behavior) that can be modulated by tES intervention. A hyperconnectivity and hyperactivity of medial OFC and vmPFC circuits that have projections to the ventral striatum and caudate is observed in patients with OCD, leading to compulsions and exaggerated habits against goal-directed behaviors. A similar hyperconnectivity is seen in the SMA–putamen network, which is also linked to compulsions. In contrast, hypoconnectivity in the lateral frontal–striatal network affects top-down executive control, and this loss contributes to the shift from goal-directed behavior to compulsive habits in OCD. Therapeutic effect of tES interventions that downregulate SMA and OFC activity (for example, ctDCS of the SMA and the OFC) can thus be due to modulating hyperactivity in these regions and their subcortical projections (for example, putamen, insula). A similar therapeutic effect is reported for low-frequency rTMS of the SMA95. Another source of effect for tES interventions is related to upregulation of both lDLPFC and the rDLPFC with anodal stimulation, which is again in line with therapeutic effects of high-frequency rTMS over the bilateral DLPFC95. Accordingly, the top-four tES interventions identified in our network analysis aimed either to downregulate the SMA, OFC and medial PFC, or to upregulate the DLPFC. Nonetheless, this explanation is speculative owing to the lack of pre- and post-tES neuroimaging data, and altered connectivity may vary with brain state (for example, resting versus task functional MRI (fMRI)). For instance, in atDCS-fMRI study, anodal stimulation of the frontopolar region reduced negative connectivity between the medial PFC and the right insula/putamen and increased its connectivity with the medial and superior frontal gyri in OCD96. Although these changes were linked to improved safety learning, no anxiolytic effects of anodal frontopolar tDCS were observed.
a,b, Major findings of pairwise (a) and network (b) meta-analyses (for detailed results see Figs. 2 and 3). c, Model-estimated and suggested tES intervention based on computational modeling with atDCS over F2–F4 coupled with cathodal AF3–AF7. d, Proposed mechanisms of tES effects on reducing OCD severity by regulating glutamate/GABA levels, upregulating the prefrontal–striatum pathway, and downregulating the SMA–putamen and medial PFC–insula pathways. vmPFC, ventromedial prefrontal cortex; vlPFC, ventrolateral prefrontal cortex; DLPFC, dorsolateral prefrontal cortex; SMA, supplementary motor area; OFC, orbitofrontal cortex; tDCS, transcranial direct current stimulation; GABA, gamma‑aminobutyric acid. Schematic diagrams and workflow illustrations were created with Microsoft Powerpoint and BioRender.com.
The second explanation relates to the imbalanced levels of glutamate and GABA in key OCD regions, which are the major neurometabolites underlying the tES mechanism of effects. A recent study with 7 T proton magnetic resonance spectroscopy found that glutamate levels in the SMA correlated with the Y‑BOCS compulsivity subscale and with measures of habitual responding in patients with OCD compared with healthy controls33. Excessive levels of glutamate and reduced levels of GABA in the ACC, and a correspondingly enhanced glutamate:GABA ratio (and hence altered excitatory:inhibitory balance), were also associated with enhanced habitual responding in these patients. Also, the excitatory/inhibitory effects of tES are linked to changes in glutamate and GABA concentrations. MRS studies showed a glutamate reduction after ctDCS and enhancement after atDCS while both atDCS and ctDCS reduce GABA97,98,99. Pharmacological studies using N‑methyl‑D‑aspartate (NMDA) receptor blockers and GABA agonists have also shown that after effects of atDCS/ctDCS and tACS are dependent on NMDA-receptor activity100,101. Thus, the efficacy of cathodal SMA tDCS may be partly due to balanced glutamate levels following the intervention. The therapeutic effects of tES interventions targeting the medial PFC, a key area in our predicted montage from the third analysis, can also be partially attributed to the modulation of glutamate and GABA in the hyperactive ACC92, which plays a role in conflict monitoring and inhibitory tasks in patients with OCD and receives projections from the medial PFC94,102.
Stimulation parameters and therapeutic efficacy
Optimizing tES interventions is crucial for effectively changing cortical activity, restoring brain excitability and balancing imbalanced functions in OCD. Systematically investigating different parameters such as treatment duration, stimulation repetition and stimulation intensity is important for evaluating the efficacy of tES interventions. Although this is not sufficiently addressed in current tES RCTs (only five studies examined effects of different intensities45, polarity80 and target regions44,51,78,83), our results are still informative in some aspects. For cortical targeting, downregulation of the SMA and the OFC, upregulation of the DLPFC, and entrainment of brain oscillations in the medial PFC yielded promising results, which align with rTMS meta-analyses37,103,104. The predicted optimized configuration based on symptom reduction effect size and induced electrical fields more specifically suggests upregulation of the rDLPFC and the medial PFC with anodal and downregulation of left frontopolar cortex (including OFC) with cathodal stimulation. Regarding stimulation intensity, 2 mA was the most commonly utilized dosage. Although the only reported RCT using 3 mA tDCS over the inferior frontal cortex showed limited efficacy for OCD treatment105, it remains unclear whether higher intensities (for example, ≥4 mA), delivered within established safety guidelines, improve outcomes, and this warrants further evaluation14,106,107.
Another important stimulation parameter was the relevance of stimulation frequency per day for reducing OCD symptoms. Our pairwise analysis showed a large therapeutic effect for twice-daily tES sessions versus the small yet significant effects for once-daily sessions. In relation to this, our meta-modeling showed that longer stimulation durations on a given day, rather than an increased number of sessions, yield greater therapeutic effects. These findings align with recent meta-analyses of rTMS RCTs, which found that longer rTMS sessions, rather than more overall sessions, are associated with larger clinical improvement37,103 and are further in line with the efficacy of accelerated rTMS (involving two or more sessions per day with short inter-session intervals) for OCD treatment108. Here it is noteworthy that twice-daily 3 mA tDCS over the inferior frontal cortex showed limited efficacy for OCD treatment105, suggesting that intensifying tDCS beyond 2 mA may not improve outcomes, as shown in the human motor cortex109. Both 1-week and 2-week interventions showed significant therapeutic effects, unlike the 4-week intervention, a finding consistent with a recent rTMS meta-analysis103 but requiring cautious interpretation due to the limited number of studies with 4-week intervention103.
In sum, systematic investigations of tES parameters are crucial for optimizing interventions and enhancing therapeutic effects, which is currently insufficient. In addition to stimulation parameters, different OCD subtypes (for example, compulsive or obsessive) should be considered for protocol development (due to different pathophysiological profiles and circuitry92), which has not been investigated by current tES interventions. Finally, it is essential for future tES studies to standardize their designs and report intervention protocols in accordance with the recently published Report Approval for Transcranial Electrical Stimulation (RATES) checklist110. This approach enhances the interpretability and reproducibility of findings while enabling more effective comparisons between tES studies.
Therapeutic efficacy of tES interventions for OCD treatment
rTMS, the most used NIBS modality in clinical settings, is FDA-approved for treating OCD12. tES interventions follow a similar rationale for modulating OCD-related brain abnormalities, yet are more easily portable, more affordable, have fewer side effects and are home usable, suggesting them as a potentially first-line NIBS treatment111 if proved effective. Our analysis showed that tES interventions show significant moderate therapeutic effects, comparable to the moderate efficacy reported for rTMS in recent meta-analyses (Hedges’ g = 0.50 to 0.65)37,95,103,112. While this pooled medium effect shows promise, it is somewhat limited due to the heterogeneity of tES protocols and the small number of similar protocols used for OCD treatment, which is the reason we did a three-level analysis. Our network meta-analysis and specifically meta-modeling analysis suggest that tES interventions with specific stimulation configurations (downregulating OFC, medial PFC and SMA) may hold promise, as discussed above. The few available tACS RCTs have also shown promising effects of alpha and beta-gamma tACS over the medial PFC and OFC47,113 and very promising results were also reported for gamma tACS for OCD treatment114,115. Future tES studies are required to assess the efficacy of promising interventions, and our model-suggested anodal F2–F4/cathodal AF3–AF7 protocol. With more tES studies emerging, analyzing clinical predictors of tES responses116,117 and meta-regression analyses of tES predictors of response will help maximize its efficacy.
Potential tES interventions and approaches for OCD treatment
Despite variations in design and stimulation parameters, the tES interventions included in this analysis can be broadly classified as downregulatory (for example, SMA/OFC ctDCS) or upregulatory (for example, DLPFC atDCS). They still need to be evaluated in larger trials, yet other potential tES interventions can be inferred from the recent OCD pathophysiology framework92. The ventrolateral PFC (VLPFC) is a key region showing reduced connectivity with the caudate in OCD and is linked to impaired cognitive flexibility118. A recent study also confirmed its hypoconnectivity with the dorsal putamen119. In another recent study120, reduced activity in the inferior frontal gyrus (including the VLPFC), alongside its hypoconnectivity with the putamen and the SMA, was linked to impaired prefrontal arbitration process and this was correlated with OCD severity. Accordingly, reduced activity and connectivity of the VLPFC with the dorsal striatum (caudate and putamen), associated with cognitive flexibility impairments, suggests that enhancing VLPFC activity via tES (for example, anodal tDCS) may help lower OCD severity by enhancing goal-directed behaviors over habitual ones (Fig. 6a). This was demonstrated in our recent study, where anodal tDCS over the dorsolateral PFC, combined with cathodal pre-SMA stimulation, improved inhibitory control and reduced attention bias toward OCD-related stimuli while reducing symptom severity in patients with OCD45.
OCD features a complex pathophysiology involving various cortical and subcortical regions that influence disease severity. Neuropsychological and cognitive deficits are integral to OCD psychopathology and closely linked to its pathophysiology. According to the latest framework about OCD psychopathology and pathophysiology92, several regions can be targeted by tES and other NIBS interventions. a, The VLPFC, an important region for the executive control system, is often hypoactive in OCD. This diminishes influence over the dorsal striatum and habit system, which can lead to compulsive behaviors. Enhancing VLPFC activity (for example, with atDCS) may benefit OCD by strengthening goal-directed behavior. b, The vmPFC is typically hyperactive in OCD. A downregulatory tES intervention targeting the medial PFC could alleviate negative feelings and urges to act by modulating the insula and caudate. c, The dorsal ACC, which is involved in action monitoring and interoceptive feelings through its connections to the insula, is also hyperactive in OCD. This hyperactivity can hinder action monitoring and amplify interoceptive urges, leading to compulsions. A downregulatory tES intervention on the dorsal ACC may help regulate these urges and improve action monitoring, potentially reducing compulsions. vmPFC, ventromedial prefrontal cortex; vlPFC, ventrolateral prefrontal cortex; ACC, anterior cingulate cortex; DLPFC, dorsolateral prefrontal cortex. Brain surface renderings are based on the MNI152 nonlinear 2009 (ICBM152) standard template.
Two additional potential interventions relate to the downregulation of the medial PFC, vmPFC and the modulation of dorsal ACC activity (Fig. 6b,c). Symptom provocation fMRI studies typically showed enhanced activation of the vmPFC121,122 and the ACC123,124 in OCD. This activation correlates with hyperconnectivity to the ventral striatum and particularly the insula, which plays a role in negative interoceptive experiences and subjective urges to action in OCD92,119,125. Downregulating vmPFC and dorsal ACC may reduce negative interoceptive feelings that drive compulsive behaviors, consistent with a recent network meta-analysis showing that inhibitory rTMS of medial PFC and anterior ACC are more effective than sham interventions104. However, evidence supports the effectiveness of deep high-frequency rTMS over the medial PFC and ACC36. This is partly due to difficulty in precisely defining the vmPFC, which can contain elements of several Brodmann areas—14, 25, 32 and 24—and some controversy on whether this region is generally hyperactive or hypoactive in OCD126,127. Nevertheless, these proposed interventions are grounded in neuroimaging findings and merit exploration in future studies (For a detailed review of the empirical background supporting these interventions, see ref. 92).
Looking ahead, home-based tES128, a unique advantage of tES versus rTMS, offers a promising avenue for ongoing treatment in natural settings, reducing anxiety-provoking clinic visits. Preliminary evidence from the only tACS study included in our analyses, which was delivered at home, suggests that tES can significantly alleviate OCD symptoms, with sustained effects observed at follow-up47 in line with recent works on depression129. Similarly, closed-loop tES130,131, adapting stimulation based on neural biomarkers, could further optimize efficacy by targeting dynamic symptom states such as obsessive ruminations. While primarily used in invasive neuromodulation132, these adaptive systems can inspire non-invasive tES innovations for precise, on-demand interventions aligned with real-world symptom fluctuations. Future research should focus on large-scale trials to validate these modalities, assess long-term safety and integrate them with existing therapies for optimized and personalized tES106,113,133 for OCD treatment.
Study strengths, limitations and conclusion
Our study comes with several strengths. It pioneers the use of a three-level meta-analysis in the NIBS field to assess the therapeutic efficacy of tES for OCD, offering a new perspective on evaluating NIBS therapeutic efficacy. In addition, it incorporates the latest literature, includes more RCTs and excludes studies with bias, thereby improving statistical power. Sub-analyses of stimulation parameters, some of which are being explored for the first time (for example, stimulation frequency per day), are other innovative aspects of our work. Yet, several limitations exist. First, pairwise and network analyses are based on heterogeneous studies, and the findings across analyses require careful consideration due to potential overlap (for example, the model-optimized protocol excluded the SMA region). Next, the meta-modeling results on stimulation duration in tDCS efficacy rely on varying durations and frequencies. Lastly, we did not perform meta-regression to examine the influence of study-level characteristics on effect size because it was beyond our study’s scope. Furthermore, the limited number of studies and inconsistent reporting of covariates made a robust meta-regression infeasible. This will remain an open research question for future systematic reviews and meta-analyses with larger, more consistent datasets. These limitations stem from the diversity of tES interventions, which could be addressed by more published RCTs; thus, current findings should be interpreted cautiously.
To conclude, this triple meta-analysis integrates three complementary meta-analytic approaches to provide a comprehensive analysis of eligible tES studies used to reduce OCD severity. Our results suggest that interventions that downregulate the OFC, medial PFC and SMA, or upregulate the lateral PFC show promise. Our analysis shows that tES is both safe and well tolerated in patients with OCD. However, many of the included studies implement strict inclusion criteria, often excluding individuals with multiple physical comorbidities, older adults or those at higher risk. Furthermore, in some studies conducted in low- and middle-income countries, patients unable to tolerate the intervention may be excluded. This underscores the importance of thoroughly examining the potential adverse effects of tES in patients with OCD. Future studies may build on findings from our pairwise, network and meta-modeling analyses, as well as the additional tES interventions with potential for OCD treatment (Fig. 6), to improve current tES protocols and investigate more effective modalities.
Methods
General study guidelines
This meta-analysis adhered to the latest update of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines134. The study protocol was registered in PROSPERO (CRD42023485717).
Search strategy and eligibility criteria
Two authors (M.A.S. and A.H.-H.) systematically searched multiple electronic databases, including PubMed, Embase, Web of Science, Cochrane CENTRAL and Scopus, from the inception of each database until 31 July 2025. The search terms included: [‘obsessive compulsive disorder’ OR ‘OCD’ OR ‘obsessive disorder’ OR ‘compulsive disorder’] AND [‘transcranial electrical stimulation’ OR ‘transcranial direct current stimulation’ OR ‘transcranial alternating current stimulation’ OR ‘transcranial random noise stimulation’ OR ‘tES’ OR ‘tDCS’ OR ‘tACS’ OR ‘tRNS’]. We also searched gray literature databases and examined the reference lists of relevant review articles and meta-analyses41,135,136,137. Peer-reviewed RCTs published in English were included in this metanalysis and systematic review. The inclusion criteria were: (1) studies with human participants diagnosed with OCD; (2) administration of tDCS, tACS or tRNS to patients on a stable medication regimen (if applicable) without concurrent psychotherapy; (3) provision of both pre- and post-intervention OCD scores; (4) RCTs with sham control or active control groups; and (5) assessing OCD symptoms with Y-BOCS. Full texts of potentially eligible studies were assessed, with disagreements resolved through discussion or a third reviewer. Duplicates were identified and removed using EndNote, followed by manual verification to ensure accuracy. Each study’s data were extracted independently by two reviewers using a standardized form, with discrepancies resolved via consensus.
Data extraction and outcome variables
Two authors (A.H.-H. and M.A.S.) independently extracted data from the included studies, including the first author’s name, publication year, study design and blinding, sample size, mean age, percentage of female participants, target and return electrode site/size, stimulation intensity, duration, number of sessions, polarity, frequency (if applicable), OCD primary measure, secondary OCD measure (if applicable) and other clinical measures, and major findings. The corresponding authors were contacted for missing data. Efficacy was measured by the change in OCD symptom scores following tES interventions (pre versus post, active versus sham), using the Y-BOCS as a validated rating scale. If studies used multiple scales for core symptoms, we relied on Y-BOCS clinician ratings. We also noted any secondary outcome measures related to efficacy, including anxiety and cognitive performance rating scales.
Risk of bias and publication bias assessment
Two authors (M.A.S. and A.H.-H.) independently assessed the risk of bias in each included study using the revised Cochrane Collaboration’s tool (v.2)138. It assesses biases from five domains arising due to: randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome, and selection of the reported result. To identify and adjust for the impact of studies with potential unpublished negative results and small-study effects, we performed publication bias analyses using funnel plots, Egger’s regression and P-curve methods.
Data synthesis and statistical analysis
Pairwise and network meta-analyses
Statistical analyses were conducted in R (v.4.1.2) using metafor, netmeta, meta and dmetar packages to compute the SMD139, perform the network meta-analysis, generate forest plots, pooling effect sizes, and carry out sensitivity analyses and publication bias assessments140,141. We used the Morris method142 for computing the SMDs of studies with a pre-test–post-test control group design (PPC). This method estimates a bias-corrected form of Hedge’s g for PPCs (gPPC). The primary outcome was the pre- to post-stimulation Y-BOCS total score change. SMD and variance for each study were computed with the escalc function of the metafor package and assuming an r = 0.5 pre-test–post-test correlation. Cross-over RCTs were included only if they reported pre- and post-intervention data separately for each treatment arm and implemented an adequate washout interval to minimize carry-over effects to qualify as an independent condition. When eligible, each treatment phase was treated as an independent comparison, with effect sizes calculated using the same methodology as parallel-group trials, while accounting for within-individual correlation (r = 0.5).
For the pairwise meta-analysis, we used Knapp–Hartung adjustments143 to calculate the 95% CI of the pooled effect. Furthermore, heterogeneity was assessed by Cochran’s Q (heterogeneity statistic), τ2 (estimated between-study variance) and I2 (proportion of total variability) in each condition. I2 values above 50% were considered as evidence for high between-study heterogeneity. In addition to the primary analyses, we conducted pre-specified subgroup analyses by cortical target, treatment duration, tDCS intensity, total number of sessions, frequency-per-day subgroup and stimulation site by polarity. For the network meta-analysis, a network was created comparing treatments to a reference group (‘sham’). Both fixed and random effect models were evaluated, with primary results derived from the random effect models. Network graphs and rankograms were used to visualize treatment connections, rankings and evidence consistency. SUCRA values were calculated to estimate the likelihood of each treatment being the most effective based on 1,000 simulated datasets of SUCRA values, ranging from 0% to 100% for different scenarios of available interventions and outcomes144. Finally, sensitivity analyses were conducted by omitting specific studies with high heterogeneity to assess robustness. Publication bias was visually assessed with funnel plots and checked using Egger’s test145. We also used the P-curve method146 to investigate the evidential value of studies effect, determining whether our meta-analysis indicates a true effect and estimating its size. This method also explicitly addresses questionable research practices, such as P-hacking.
Meta-modeling
To explore which brain regions are associated with positive effects of tDCS on OCD symptoms, we performed the meta-modeling approach developed by Wischnewski et al.55. This computational approach correlates tDCS-related electric fields with behavioral effect sizes. First, the electric-field strengths (normE) of each intervention were calculated for every surface of a standard adult European male head model (~30 years of age). The electric-field simulations were performed in SimNIBS 3.2.147,148 and standard realistic conductivities of head and brain tissues were used149. The gray matter was extracted for further analysis, which in this model consisted of 246,272 surface elements. As different montages were used in the included studies, this resulted in 19 electric-field distributions and a 246,272 × 19 matrix. Subsequently, each row of this matrix (19 values) was correlated with the 19 effect size values. This resulted in a column vector of 246,272 correlation values, with one correlation value for each gray-matter element (PECE). For the correlations, we used the non-parametric Spearman rank-order coefficient, as it can deal with non-normally distributed data and is less sensitive to outlying values compared with a parametric Pearson coefficient. Given the exploratory nature of this analysis, we report uncorrected one-sided P values.
We conducted additional analyses on factors affecting tDCS efficacy, specifically treatment duration (comprising daily stimulation duration (once or twice daily) and total session number). To account for this, we considered the ‘cumulative charge’ per day and over the entire treatment. For this, normE values were multiplied by (1) the total minutes of stimulation in one session (PECE × min per day), (2) the total number of days of the treatment (PECE × number of days), and (3) the total minutes of stimulation per treatment (minutes per session × number of days; PECE × min × day). After this, correlations were calculated in the same way as described above. To relate our findings to functional brain regions, the correlation values were averaged per brain region of the HCP-MMP atlas87,88 which consists of 360 parcels. Subsequently, we displaced the five regions with the largest average PEC values. To ensure that our results were not biased due to peculiarities of our head model, all analyses described above were repeated in another head model (F, ~30 years of age), which was obtained from the Human Connectome Project (https://www.humanconnectome.org). Inter-map correlations were calculated for each analysis (PECE, PECE × min per day, PECE × number of days and PECE × min × day), meaning that the values of each region of one head model were correlated to the values of the same region of the other head model.
Finally, we aimed to provide an optimized montage that targets the exact PEC map, which would result in the most beneficial results. To do so, we calculated the perpendicular direction of the electric field (perpE), which provides a measure of direction as it reflects locations of current influx and outflux147. As electrical current flows from the anodal to the cathodal electrode, an inward current (perpE > 0) occurs at a gyral bank facing the anode, while an outward current (perpE < 0) occurs at a gyral bank facing the cathode150 (perpE < 0; Supplementary Fig. 1). Accordingly, a correlation between effect sizes and perpE provides information about optimal placement of anodes and cathodes for tDCS, which is necessary for optimization. This PECperpE was calculated in the same way as the other PEC values, described above. PECperpE was calculated only for brain regions where PECE > 0, as the goal was to provide an optimized montage for OCD symptom improvement.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data that support the findings are available in the paper and are publicly available at https://osf.io/6taex/.
Code availability
The codes generated for data analyses are publicly available at https://osf.io/6taex/.
References
Vos, T. et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396, 1204–1222 (2020).
Stein, D. J. et al. Obsessive–compulsive disorder. Nat. Rev. Dis. Primers 5, 52 (2019).
Diagnostic and Statistical Manual of Mental Disorders (DSM-5®) (American Psychiatric Association, 2013).
Meier, S. M. et al. Mortality among persons with obsessive–compulsive disorder in denmark. JAMA Psychiatry 73, 268–274 (2016).
Wayne, K. et al. Harmonizing the neurobiology and treatment of obsessive–compulsive disorder. Am. J. Psychiatry 178, 17–29 (2021).
Fawcett, E. J., Power, H. & Fawcett, J. M. Women are at greater risk of OCD than men: a meta-analytic review of OCD prevalence worldwide. J. Clin. Psychiatry 81, 19r13085 (2020).
Romanelli, R. J., Wu, F. M., Gamba, R., Mojtabai, R. & Segal, J. B. Behavioral therapy and serotonin reuptake inhibitor pharmacotherapy in the treatment of obsessive-compulsive disorder: a systematic review and meta-analysis of head-to-head randomized controlled trials. Depress. Anxiety 31, 641–652 (2014).
Cuijpers, P. et al. Absolute and relative outcomes of psychotherapies for eight mental disorders: a systematic review and meta-analysis. World Psychiatry 23, 267–275 (2024).
Polanía, R., Nitsche, M. A. & Ruff, C. C. Studying and modifying brain function with non-invasive brain stimulation. Nat. Neurosci. 21, 174–187 (2018).
Fregni, F. & Pascual-Leone, A. Technology insight: noninvasive brain stimulation in neurology—perspectives on the therapeutic potential of rTMS and tDCS. Nat. Clin. Pract. Neurol. 3, 383–393 (2007).
Salehinejad, M. A. & Siniatchkin, M. Safety of noninvasive brain stimulation in children. Curr. Opin. Psychiatry https://doi.org/10.1097/YCO.0000000000000923 (2024).
Cohen, S. L., Bikson, M., Badran, B. W. & George, M. S. A visual and narrative timeline of US FDA milestones for transcranial magnetic stimulation (TMS) devices. Brain Stimul. 15, 73–75 (2022).
Bikson, M. et al. Transcranial electrical stimulation nomenclature. Brain Stimul. 12, 1349–1366 (2019).
Antal, A. et al. Low intensity transcranial electric stimulation: safety, ethical, legal regulatory and application guidelines (2017–2025: an update)—endorsed by the European Society for Brain Stimulation and by the International Federation for Clinical Neurophysiology. Clin. Neurophysiol. 2111436 (2025).
Nitsche, M. & Paulus, W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J. Physiol. 527, 633–639 (2000).
Jackson, M. P. et al. Animal models of transcranial direct current stimulation: methods and mechanisms. Clin. Neurophysiol. 127, 3425–3454 (2016).
Woods, A. J. et al. A technical guide to tDCS, and related non-invasive brain stimulation tools. Clin. Neurophysiol. 127, 1031–1048 (2016).
Stagg, C. J. & Nitsche, M. A. Physiological basis of transcranial direct current stimulation. The Neuroscientist 17, 37–53 (2011).
Wischnewski, M., Alekseichuk, I. & Opitz, A. Neurocognitive, physiological, and biophysical effects of transcranial alternating current stimulation. Trends Cogn. Sci. 27, 189–205 (2023).
Terney, D., Chaieb, L., Moliadze, V., Antal, A. & Paulus, W. Increasing human brain excitability by transcranial high-frequency random noise stimulation. Journal Neurosci. 28, 14147–14155 (2008).
van der Groen, O. et al. Using noise for the better: the effects of transcranial random noise stimulation on the brain and behavior. Neurosci. Biobehav. Rev. 138, 104702 (2022).
Fregni, F. et al. Evidence-based guidelines and secondary meta-analysis for the use of transcranial direct current stimulation in neurological and psychiatric disorders. Int. J. Neuropsychopharmacol. 24, 256–313 (2020).
Vicario, C. M., Salehinejad, M. A., Felmingham, K., Martino, G. & Nitsche, M. A. A systematic review on the therapeutic effectiveness of non-invasive brain stimulation for the treatment of anxiety disorders. Neurosci. Biobehav. Rev. 96, 219–231 (2019).
Li, H. et al. Neural primacy of the dorsolateral prefrontal cortex in patients with obsessive–compulsive disorder. NeuroImage Clin. 28, 102432 (2020).
de Wit, S. J. et al. Presupplementary motor area hyperactivity during response inhibition: a candidate endophenotype of obsessive–compulsive disorder. Am. J. Psychiatry 169, 1100–1108 (2012).
Norman, L. J. et al. Error processing and inhibitory control in obsessive–compulsive disorder: a meta-analysis using statistical parametric Maps. Biol. Psychiatry 85, 713–725 (2019).
Anticevic, A. et al. Global resting-state functional magnetic resonance imaging analysis identifies frontal cortex, striatal, and cerebellar dysconnectivity in obsessive-compulsive disorder. Biol. Psychiatry 75, 595–605 (2014).
Shephard, E. et al. Toward a neurocircuit-based taxonomy to guide treatment of obsessive–compulsive disorder. Mol. Psychiatry 26, 4583–4604 (2021).
Russman Block, S. et al. Resting-state connectivity and response to psychotherapy treatment in adolescents and adults with OCD: a randomized clinical trial. Am J Psychiatry 180, 89–99 (2023).
Beucke, J. C. et al. Abnormally high degree connectivity of the orbitofrontal cortex in obsessive–compulsive disorder. JAMA Psychiatry 70, 619–629 (2013).
Chakrabarty, K., Bhattacharyya, S., Christopher, R. & Khanna, S. Glutamatergic dysfunction in OCD. Neuropsychopharmacology 30, 1735–1740 (2005).
Maraone, A., Tarsitani, L., Pinucci, I. & Pasquini, M. Antiglutamatergic agents for obsessive-compulsive disorder: where are we now and what are possible future prospects? World J. Psychiatry 11, 568–580 (2021).
Biria, M. et al. Cortical glutamate and GABA are related to compulsive behaviour in individuals with obsessive compulsive disorder and healthy controls. Nat. Commun. 14, 3324 (2023).
Harika-Germaneau, G. et al. Investigating brain structure and tDCS response in obsessive–compulsive disorder. J. Psychiatric Res. 177, 39–45 (2024).
Dunlop, K. et al. Reductions in cortico-striatal hyperconnectivity accompany successful treatment of obsessive–compulsive disorder with dorsomedial prefrontal rTMS. Neuropsychopharmacology 41, 1395–1403 (2016).
Carmi, L. et al. Efficacy and safety of deep transcranial magnetic stimulation for obsessive–compulsive disorder: a prospective multicenter randomized double-blind placebo-controlled trial. Am. J. Psychiatry 176, 931–938 (2019).
Steuber, E. R. & McGuire, J. F. A meta-analysis of transcranial magnetic stimulation in obsessive–compulsive disorder. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 8, 1145–1155 (2023).
Moshfeghinia, R. et al. Efficacy and safety of transcranial direct current stimulation (tDCS) in patients with obsessive–compulsive disorder (OCD): a systematic review and meta-analysis of randomized controlled trials. Neurosci. Biobehav. Rev. 173, 106171 (2025).
Yan, L., Wang, Y. & Li, M. Transcranial direct current stimulation in patients with obsessive–compulsive disorder: a meta-analysis of randomised controlled trials. Int. J. Psychiatry Clin. Pract. 29, 1–8 (2025).
Green, P. E., Loftus, A. M. & Anderson, R. A. Transcranial direct current stimulation for obsessive compulsive disorder: a systematic review and CONSORT evaluation. J. Int. Neuropsychol. Soc. 30, 1003–1014 (2024).
Pinto, B. S. et al. The effects of transcranial direct current stimulation in obsessive–compulsive disorder symptoms: a meta-analysis and integrated electric fields modeling analysis. Biomedicines 11, 80 (2022).
Zhou, S. & Fang, Y. Efficacy of non-invasive brain stimulation for refractory obsessive–compulsive disorder: a meta-analysis of randomized controlled trials. Brain Sci. 12, 943 (2022).
Spiroiu, F. I., Minuzzi, L., Duarte, D., McCabe, R. E. & Soreni, N. Neurocognitive effects of transcranial direct current stimulation in obsessive–compulsive disorder: a systematic review. Int. J. Neurosci. 1–14 (2024).
Fineberg, N. A. et al. Feasibility, acceptability and practicality of transcranial stimulation in obsessive compulsive symptoms (FEATSOCS): a randomised controlled crossover trial. Compr. Psychiatry 122, 152371 (2023).
Alizadehgoradel, J. et al. Targeting the prefrontal-supplementary motor network in obsessive-compulsive disorder with intensified electrical stimulation in two dosages: a randomized, controlled trial. Transl. Psychiatry 14, 78 (2024).
Harika-Germaneau, G. et al. Treating refractory obsessive compulsive disorder with cathodal transcranial direct current stimulation over the supplementary motor area: a large multisite randomized sham-controlled double-blind study. Front. Psychiatry 15, 1338594 (2024).
Perera, M. P. N. et al. Home-based individualized alpha transcranial alternating current stimulation improves symptoms of obsessive–compulsive disorder: preliminary evidence from a randomized, sham-controlled clinical trial. Depress. Anxiety 2023, 9958884 (2023).
Agrawal, A., Agarwal, V., Kar, S. K. & Arya, A. Transcranial direct current stimulation as early augmentation in adolescent obsessive compulsive disorder: a pilot proof-of-concept randomized control trial. World J. Clin. Pediatr. 13, 93138 (2024).
Kumar, L., Gupta, B., Tripathi, A., Joshi, M. & Kar, S. K. A single-blind, randomized controlled trial of transcranial direct current stimulation in adults with obsessive–compulsive disorder as early augmentation. CNS Spectr. 30, e50 (2025).
Wang, Y. et al. Assessing the effectiveness of high-definition transcranial direct current stimulation for treating obsessive–compulsive disorder: results from a randomized, double-blind, controlled trial. Compr. Psychiatry 135, 152520 (2024).
Chu, C. S. et al. Comparing different montages of transcranial direct current stimulation in treating treatment-resistant obsessive compulsive disorder: a randomized, single-blind clinical trial. Medicina 61, 169 (2025).
Cipriani, A., Higgins, J. P. T., Geddes, J. R. & Salanti, G. Conceptual and technical challenges in network meta-analysis. Ann. Intern. Med. 159, 130–137 (2013).
Daly, C. H. et al. Empirical evaluation of SUCRA-based treatment ranks in network meta-analysis: quantifying robustness using Cohen’s kappa. BMJ Open 9, e024625 (2019).
Higgins, J. P. T. & Welton, N. J. Network meta-analysis: a norm for comparative effectiveness? Lancet 386, 628–630 (2015).
Wischnewski, M., Mantell, K. E. & Opitz, A. Identifying regions in prefrontal cortex related to working memory improvement: a novel meta-analytic method using electric field modeling. Neurosci. Biobehav. Rev. 130, 147–161 (2021).
Bation, R., Poulet, E., Haesebaert, F., Saoud, M. & Brunelin, J. Transcranial direct current stimulation in treatment-resistant obsessive–compulsive disorder: an open-label pilot study. Prog. Neuropsychopharmacol. Biol. Psychiatry 65, 153–157 (2016).
Najafi, K. et al. Efficacy of transcranial direct current stimulation in the treatment: resistant patients who suffer from severe obsessive–compulsive disorder. Indian J. Psychol. Med. 39, 573–578 (2017).
Harika-Germaneau, G. et al. Treating refractory obsessive–compulsive disorder with transcranial direct current stimulation: an open label study. Brain Behav. 10, e01648 (2020).
Dinn, W. M. et al. Treatment-resistant obsessive–compulsive disorder: insights from an open trial of transcranial direct current stimulation (tDCS) to design a RCT. Neurol. Psychiatry Brain Res. 22, 146–154 (2016).
Kumar, S., Kumar, N. & Verma, R. Safety and efficacy of adjunctive transcranial direct current stimulation in treatment-resistant obsessive–compulsive disorder: an open-label trial. Indian J. Psychiatry 61, 327–334 (2019).
Dadashi, M., Yousefi Asl, V. & Morsali, Y. Cognitive-behavioral therapy versus transcranial direct current stimulation for augmenting selective serotonin reuptake inhibitors in obsessive–compulsive disorder patients. Basic Clin. Neurosci. J. 11, 111–120 (2020).
Cheng, J. et al. Transcranial direct current stimulation improve symptoms and modulates cortical inhibition in obsessive–compulsive disorder: a TMS-EEG study. J. Affect. Disord. 298, 558–564 (2022).
Harika-Germaneau, G. et al. Transcranial direct current stimulation in refractory obsessive–compulsive disorder treatment: a randomized sham-controlled trial. Brain Stimul. 16, 279 (2023).
Mukherjee, A., Praharaj, S. & Rai, S. Effect of add-on cathodal transcranial direct current stimulation (c-TDCS) over pre-supplementary motor area (pre-SMA) in patients with obsessive compulsive disorder: a randomized sham controlled study. Eur. Psychiatry 64, S152–S153 (2021).
Cheng, J. et al. EEG microstates are associated with the improvement of obsessive–compulsive symptoms after transcranial direct current stimulation. J. Psychiatric Res. 176, 360–367 (2024).
Todder, D. et al. Immediate effects of transcranial direct current stimulation on obsession-induced anxiety in refractory obsessive–compulsive disorder: a pilot study. J. ECT 34, e51–e57 (2018).
Ruan, H. et al. Local effective connectivity changes after transcranial direct current stimulation in obsessive–compulsive disorder patients. J. Affect. Disord. 374, 116–127 (2025).
Rodriguez-Manrique, D. et al. Investigating the effects of brain stimulation on the neural substrates of inhibition in patients with OCD: a simultaneous tDCS-fMRI study. Transl. Psychiatry 15, 173 (2025).
Volpato, C. et al. Modulation of affective symptoms and resting state activity by brain stimulation in a treatment-resistant case of obsessive–compulsive disorder. Neurocase 19, 360–370 (2013).
Mondino, M., Haesebaert, F., Poulet, E., Saoud, M. & Brunelin, J. Efficacy of cathodal transcranial direct current stimulation over the left orbitofrontal cortex in a patient with treatment-resistant obsessive–compulsive disorder. J. ECT 31, 271–272 (2015).
Hazari, N. et al. Response to transcranial direct current stimulation in a case of episodic obsessive compulsive disorder. J. ECT 32, 144–146 (2016).
D’Urso, G. et al. Polarity-dependent effects of transcranial direct current stimulation in obsessive-compulsive disorder. Neurocase 22, 60–64 (2016).
Palm, U. et al. Prefrontal tDCS and sertraline in obsessive compulsive disorder: a case report and review of the literature. Neurocase 23, 173–177 (2017).
Narayanaswamy, J. C. et al. Successful application of add-on transcranial direct current stimulation (tDCS) for treatment of SSRI resistant OCD. Brain Stimul. 8, 655–657 (2015).
Silva, R. M., Brunoni, A. R., Miguel, E. C. & Shavitt, R. G. Transcranial direct current stimulation for treatment-resistant obsessive–compulsive disorder: report on two cases and proposal for a randomized, sham-controlled trial. Sao Paulo Med. J. 134, 446–450 (2016).
Alizadeh et al. The effects of transcranial direct current stimulation on obsession-compulsion, anxiety, and depression of a patient suffering from obsessive–compulsive disorder. J. Pract. Clin. Psychol. 4, 75–80 (2016).
Basu, I., Panda, S. P. & Yadav, P. Transcranial direct current stimulation as an augmentation therapy in patients with obsessive–compulsive disorder: a case series. Indian Psychiatry J. 33, S226–s8 (2024).
Akbari, S., Hassani-Abharian, P. & Tajeri, B. The effect of transcranial direct current stimulation (tDCS) on cerebellum in reduction of the symptoms of obsessive–compulsive disorder. Neurocase 28, 135–139 (2022).
Bation, R., Mondino, M., Le Camus, F., Saoud, M. & Brunelin, J. Transcranial direct current stimulation in patients with obsessive compulsive disorder: a randomized controlled trial. Eur. Psychiatry 62, 38–44 (2019).
D’Urso, G. et al. Transcranial direct current stimulation for obsessive–compulsive disorder: a randomized, controlled, partial crossover trial. Depress. Anxiety 33, 1132–1140 (2016).
Echevarria, M. A. N. et al. Increases in functional connectivity between the default mode network and sensorimotor network correlate with symptomatic improvement after transcranial direct current stimulation for obsessive–compulsive disorder. J. Affect. Disord. 355, 175–183 (2024).
Gowda, S. M. et al. Efficacy of pre-supplementary motor area transcranial direct current stimulation for treatment resistant obsessive compulsive disorder: a randomized, double blinded, sham controlled trial. Brain Stimul. 12, 922–929 (2019).
Shafiezadeh, S. et al. Effect of transcranial direct current stimulation on dorsolateral prefrontal cortex to reduce the symptoms of the obsessive–compulsive disorder. Basic Clin. Neurosci. 12, 675–680 (2021).
Silva, R.dM. F. d et al. Efficacy and safety of transcranial direct current stimulation as an add-on treatment for obsessive–compulsive disorder: a randomized, sham-controlled trial. Neuropsychopharmacology 46, 1028–1034 (2021).
Yekta, M., Rostami, R. & Fayyaz, E. Transcranial direct current stimulation of dorsolateral prefrontal cortex in patients with obsessive compulsive disorder to improve decision making and reduce obsession symptoms. Pract. Clin. Psychol. 3, 185–194 (2015).
Yoosefee, S. et al. The safety and efficacy of transcranial direct current stimulation as add-on therapy to fluoxetine in obsessive-compulsive disorder: a randomized, double-blind, sham-controlled, clinical trial. BMC Psychiatry 20, 570 (2020).
Glasser, M. F. et al. A multi-modal parcellation of human cerebral cortex. Nature 536, 171–178 (2016).
Wischnewski, M., Berger, T. A. & Opitz, A. Meta-modeling the effects of anodal left prefrontal transcranial direct current stimulation on working memory performance. Imaging Neurosci. 2, 1–14 (2024).
Boutron, I., Altman, D. G., Moher, D., Schulz, K. F. & Ravaud, P. CONSORT statement for randomized trials of nonpharmacologic treatments: a 2017 update and a CONSORT extension for nonpharmacologic trial abstracts. Ann. Intern. Med. 167, 40–47 (2017).
Park, H. et al. Aberrant cortico-striatal white matter connectivity and associated subregional microstructure of the striatum in obsessive–compulsive disorder. Mol. Psychiatry 27, 3460–3467 (2022).
Kim, B.-G. et al. White matter diffusion estimates in obsessive–compulsive disorder across 1653 individuals: machine learning findings from the ENIGMA OCD Working Group. Mol. Psychiatry 29, 1063–1074 (2024).
Robbins, T. W., Banca, P. & Belin, D. From compulsivity to compulsion: the neural basis of compulsive disorders. Nat. Rev. Neurosci. 25, 313–333 (2024).
Chamberlain, S. R. et al. Orbitofrontal dysfunction in patients with obsessive–compulsive disorder and their unaffected relatives. Science 321, 421–422 (2008).
Norman, L. J. et al. Structural and functional brain abnormalities in attention-deficit/hyperactivity disorder and obsessive–compulsive disorder: a comparative meta-analysis. JAMA Psychiatry 73, 815–825 (2016).
Fitzsimmons, S. M. D. D. et al. Repetitive transcranial magnetic stimulation for obsessive–compulsive disorder: a systematic review and pairwise/network meta-analysis. J. Affect. Disord. 302, 302–312 (2022).
Adams, T. G. et al. Transcranial direct current stimulation targeting the medial prefrontal cortex modulates functional connectivity and enhances safety learning in obsessive–compulsive disorder: results from two pilot studies. Depress. Anxiety 39, 37–48 (2022).
Stagg, C. J. et al. Polarity-sensitive modulation of cortical neurotransmitters by transcranial stimulation. J. Neurosci. 29, 5202–5206 (2009).
Stagg, C. J., Bachtiar, V. & Johansen-Berg, H. The role of GABA in human motor learning. Curr. Biol. 21, 480–484 (2011).
Stagg, C. J. et al. Relationship between physiological measures of excitability and levels of glutamate and GABA in the human motor cortex. J. Physiol. 589, 5845–5855 (2011).
Wischnewski, M. et al. NMDA receptor-mediated motor cortex plasticity after 20 Hz transcranial alternating current stimulation. Cereb. Cortex. 29, 2924–2931 (2019).
Nitsche, M. A. et al. Pharmacological modulation of cortical excitability shifts induced by transcranial direct current stimulation in humans. J. Physiol. 553, 293–301 (2003).
Salehinejad, M. A., Ghanavati, E., Rashid, M. H. A. & Nitsche, M. A. Hot and cold executive functions in the brain: a prefrontal-cingular network. Brain Neurosci. Adv. 5, 23982128211007769 (2021).
Dehghani-Arani, F. et al. Metaanalysis of repetitive transcranial magnetic stimulation (rTMS) efficacy for OCD treatment: the impact of stimulation parameters, symptom subtype and rTMS-induced electrical field. J. Clin. Med. 13, 5358 (2024).
Vinod, P. et al. Comparative efficacy of repetitive transcranial magnetic stimulation protocols for obsessive–compulsive disorder: a network meta-analysis. Asian J. Psychiatry 94, 103962 (2024).
Fatehi-Chenar, A. et al. Limited efficacy of 3 mA intensified tDCS of the right inferior frontal cortex for OCD treatment: a randomized, sham-controlled study. Preprint at medRxiv https://doi.org/10.1101/2025.04.29.25326694 (2025).
Salehinejad M. A. et al. Efficacy of optimized multichannel vs conventional tDCS for major depression treatment: a randomized controlled trial. Preprint at medRxiv https://doi.org/10.1101/2025.02.12.25322157 (2025).
Schlaug, G. et al. Safety and efficacy of transcranial direct current stimulation in addition to constraint-induced movement therapy for post-stroke motor recovery (TRANSPORT2): a phase 2, multicentre, randomised, sham-controlled triple-blind trial. Lancet Neurol. 24, 400–412 (2025).
Joseph, J. T., Vishwanath, R. & Praharaj, S. K. Efficacy and safety of accelerated transcranial magnetic stimulation for obsessive–compulsive disorder: a systematic review and meta-analysis. Asian J. Psychiatry 106, 104420 (2025).
Agboada, D., Mosayebi-Samani, M., Kuo, M.-F. & Nitsche, M. A. Induction of long-term potentiation-like plasticity in the primary motor cortex with repeated anodal transcranial direct current stimulation—better effects with intensified protocols? Brain Stimul. 13, 987–997 (2020).
Nejati, V. et al. Report Approval for Transcranial Electrical Stimulation (RATES): expert recommendation based on a Delphi consensus study. Nat. Protoc. (2025).
Brunoni, A. R. & Padberg, F. Spaced transcranial direct current stimulation for depression: the road less traveled. Am. J. Psychiatry 182, 231–233 (2025).
Perera, M. P. N. et al. Repetitive transcranial magnetic stimulation for obsessive–compulsive disorder: a meta-analysis of randomized, sham-controlled trials. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 6, 947–960 (2021).
Grover, S., Nguyen, J. A., Viswanathan, V. & Reinhart, R. M. G. High-frequency neuromodulation improves obsessive–compulsive behavior. Nat. Med. 27, 232–238 (2021).
Klimke, A., Nitsche, M. A., Maurer, K. & Voss, U. Case report: successful treatment of therapy-resistant OCD with application of transcranial alternating current stimulation (tACS). Brain Stimul. 9, 463–465 (2016).
Haller, N., Senner, F., Hasan, A., Padberg, F. & Palm, U. Gamma transcranial alternating current stimulation (γtACS) in obsessive–compulsive disorder: a case report. Fortschr. Neurol. Psychiatrie 88, 398–401 (2020).
Rostami, R. et al. Efficacy and clinical predictors of response to rTMS treatment in pharmacoresistant obsessive–compulsive disorder (OCD): a retrospective study. BMC Psychiatry 20, 372 (2020).
Rostami, R., Kazemi, R., Nitsche, M. A., Gholipour, F. & Salehinejad, M. A. Clinical and demographic predictors of response to rTMS treatment in unipolar and bipolar depressive disorders. Clinical Neurophysiology 128, 1961–1970 (2017).
Vaghi, M. M. et al. Specific frontostriatal circuits for impaired cognitive flexibility and goal-directed planning in obsessive–compulsive disorder: evidence from resting-state functional connectivity. Biol. Psychiatry 81, 708–717 (2017).
Naze, S. et al. Mechanisms of imbalanced frontostriatal functional connectivity in obsessive–compulsive disorder. Brain 146, 1322–1327 (2023).
Kim, T. et al. Neurocomputational model of compulsivity: deviating from an uncertain goal-directed system. Brain 147, 2230–2244 (2024).
Banca, P. et al. Imbalance in habitual versus goal directed neural systems during symptom provocation in obsessive–compulsive disorder. Brain 138, 798–811 (2015).
Apergis-Schoute, A. M. et al. Hyperconnectivity of the ventromedial prefrontal cortex in obsessive–compulsive disorder. Brain Neurosci. Adv. 2, 1–10 (2018).
van de Veerdonk, M. M. G. H. et al. The role of the dorsal anterior cingulate cortex in obsessive–compulsive disorder. Deep Brain Stimul. 3, 1–7 (2023).
McGovern, R. A. & Sheth, S. A. Role of the dorsal anterior cingulate cortex in obsessive–compulsive disorder: converging evidence from cognitive neuroscience and psychiatric neurosurgery. J. Neurosurg. 126, 132–147 (2017).
Jackson, S. R., Parkinson, A., Kim, S. Y., Schüermann, M. & Eickhoff, S. B. On the functional anatomy of the urge-for-action. Cogn. Neurosci. 2, 227–243 (2011).
Carmi, L. et al. Clinical and electrophysiological outcomes of deep TMS over the medial prefrontal and anterior cingulate cortices in OCD patients. Brain Stimul. 11, 158–165 (2018).
Cheng, Y. et al. Abnormal resting-state activities and functional connectivities of the anterior and the posterior cortexes in medication-naïve patients with obsessive–compulsive disorder. PLoS ONE 8, e67478 (2013).
Palm, U. et al. Home use, remotely supervised, and remotely controlled transcranial direct current stimulation: a systematic review of the available evidence. Neuromodulation 21, 323–333 (2018).
Woodham, R. D. et al. Home-based transcranial direct current stimulation treatment for major depressive disorder: a fully remote phase 2 randomized sham-controlled trial. Nat. Med. 31, 87–95 (2025).
Soleimani, G. et al. Closing the loop between brain and electrical stimulation: towards precision neuromodulation treatments. Transl. Psychiatry 13, 279 (2023).
Sellers, K. K. et al. Closed-loop neurostimulation for the treatment of psychiatric disorders. Neuropsychopharmacology 49, 163–178 (2024).
Langbein, J., Boddeti, U., Xie, W. & Ksendzovsky, A. Intracranial closed-loop neuromodulation as an intervention for neuropsychiatric disorders: an overview. Front. Psychiatry 16, 1479240 (2025).
Van Hoornweder, S., Stagg, C. J. & Wischnewski, M. Personalizing transcranial electrical stimulation. Trends Neurosci. 48, 663–678 (2025).
Page, M. J. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372, n71 (2021).
Brunelin, J. et al. Transcranial direct current stimulation for obsessive–compulsive disorder: a systematic review. Brain Sciences 8, 37 (2018).
D’Urso, G., Mantovani, A., Patti, S., Toscano, E. & de Bartolomeis, A. Transcranial direct current stimulation in obsessive–compulsive disorder, posttraumatic stress disorder, and anxiety disorders. J. ECT 34, 172–181 (2018).
Sá, K. N. et al. Evidence-based umbrella review of non-invasive brain stimulation in anxiety disorders. Eur. J. Psychiatry 37, 167–181 (2023).
Sterne, J. A. C. et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366, l4898 (2019).
Viechtbauer, W. & Cheung, M. W.-L. Outlier and influence diagnostics for meta-analysis. Res. Synth. Methods 1, 112–125 (2010).
Balduzzi, S., Rücker, G. & Schwarzer, G. How to perform a meta-analysis with R: a practical tutorial. Evid. Based Ment. Health 22, 153–160 (2019).
Harrer, M., Cuijpers, P., Furukawa, T. & Ebert, D. Doing Meta-analysis with R: A Hands-on Guide (Chapman and Hall/CRC, 2021).
Morris, S. B. Estimating effect sizes from pretest–posttest-control group designs. Organ. Res. Methods 11, 364–386 (2008).
Knapp, G. & Hartung, J. Improved tests for a random effects meta-regression with a single covariate. Stat. Med. 22, 2693–2710 (2003).
Bellos, I. Multicriteria decision-making methods for optimal treatment selection in network meta-analysis. Med. Decis. Making 43, 78–90 (2023).
Egger, M., Smith, G. D., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 315, 629–634 (1997).
Simonsohn, U., Nelson, L. D. & Simmons, J. P. P-curve and effect size: correcting for publication bias using only significant results. Perspect. Psychol. Sci. 9, 666–681 (2014).
Saturnino, G. B., Siebner, H. R., Thielscher, A. & Madsen, K. H. Accessibility of cortical regions to focal TES: dependence on spatial position, safety, and practical constraints. Neuroimage 203, 116183 (2019).
Thielscher, A., Antunes, A. & Saturnino, G. B. Field modeling for transcranial magnetic stimulation: a useful tool to understand the physiological effects of TMS? In Proc. 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society 222–225 (IEEE, 2015).
Windhoff, M., Opitz, A. & Thielscher, A. Electric field calculations in brain stimulation based on finite elements: an optimized processing pipeline for the generation and usage of accurate individual head models. Hum. Brain Mapp. 34, 923–935 (2013).
Jamil, A. & Nitsche, M. A. What effect does tDCS have on the brain? Basic physiology of tDCS. Curr. Behav. Neurosci. Rep. 4, 331–340 (2017).
Acknowledgements
We thank S. Karimi and A. Fathi Jouzdani for their technical assistance.
Funding
Open access funding provided by RWTH Aachen University.
Author information
Authors and Affiliations
Contributions
M.A.S.: conceptualization, methodology, investigation, supervision, visualization, formal analysis, writing—original draft, writing—review and editing. A.H.-.H.: investigation, methodology, visualization, formal analysis. M.W.: formal analysis, visualization, writing—original draft (meta-modeling section), writing—review and editing. A.A., M.B., D.M.B., J.B., A.R.B., Z.J.D., S.N., F.P., A.T.S., M.S., O.A.v.d.H. and C.M.V.: methodology, writing—review and editing. Z.V.: investigation, writing—review and editing. T.W.R. and M.A.N.: supervision, writing—review and editing. All authors reviewed and approved the paper.
Corresponding author
Ethics declarations
Competing interests
M.A.N. is a member of the Scientific Advisory Boards of Neuroelectrics and Precisis. D.M.B. receives research support from CIHR, NIH, Brain Canada and the Temerty Family through the CAMH Foundation and the Campbell Family Research Institute. He received research support and in-kind equipment support for an investigator-initiated study from Brainsway Ltd. He was the site principal investigator for three sponsor-initiated studies for Brainsway Ltd. He also receives in-kind equipment support from Magventure for investigator-initiated research. He received medication supplies for an investigator-initiated trial from Indivior. He is a scientific advisor for Sooma Oy Medical. F.P. is a member of the European Scientific Advisory Board of BrainsWay Inc. (Jerusalem, Israel), and the International Scientific Advisory Board of Sooma (Helsinki, Finland); he has received speaker honoraria from Mag&More GmbH, the neuroCare Group (Munich, Germany), and BrainsWay Inc.; his lab has received support with equipment from neuroConn GmbH (Ilmenau, Germany), Mag&More GmbH, and BrainsWay Inc. A.T.S. is scientific advisor for PlatoScience Medical and Alpha Brain Technologies, director of the Academy of Brain Stimulation (www.brainstimulation-academy.com) and the International Clinical TMS Certification Course (www.tmscourse.eu), receiving equipment support from MagVenture, Magstim, Deymed, Yingchi, and BrainsWay. T.W.R has consultancy for Cambridge Cognition, Supernus and Platea, and editorial honoraria for Springer Nature. These acknowledgments had no impact on the objectivity, integrity or value of the publication. The other authors declare no competing interests.
Peer review
Peer review information
Nature Mental Health thanks Reza Moshfeghinia and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–7 and Tables 1–3.
Source data
Source Data Fig. 2 (download XLSX )
Effect size and stimulation parameters of studies included in the pairwise meta-analysis.
Source Data Fig. 3 (download XLSX )
Effect sizes of the studies included in the network meta-analysis.
Source Data Fig. 4 (download XLSX )
Behavioral effect size and induced electrical field in meta-modeling studies.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Salehinejad, M.A., Hallajian, AH., Wischnewski, M. et al. Transcranial electrical stimulation for the treatment of obsessive–compulsive disorder: a triple meta-analysis. Nat. Mental Health 4, 634–652 (2026). https://doi.org/10.1038/s44220-026-00590-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44220-026-00590-z