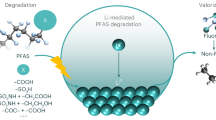
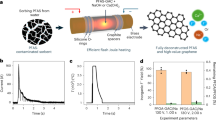
Abstract
Per- and polyfluoroalkyl substances (PFAS) are recalcitrant and bioaccumulative environmental pollutants. Whereas substantial efforts have been made to degrade PFAS, the potential for effectively using these fluoride resources has been overlooked. Here we develop an electrothermal fluorination method to selectively fluorinate brine salts using granular activated carbon (GAC)-sorbed aqueous film-forming foam (AFFF) as a fluorination agent. During this process, GAC and PFAS in AFFF are converted to graphene, whereas fluorine atoms are effectively mineralized into metal fluorides. Followed by washing and flash distillation, lithium can be recovered from other alkali and alkaline-earth metal cations in brine (Na+, Mg2+, K+, Ca2+) in the form of lithium fluoride, with an ~99% lithium purity and ~82% yield. The recovered lithium fluoride is demonstrated as an additive to stabilize electrolytes and improve the performance of lithium-ion batteries. Life-cycle assessment and techno-economic analysis indicate that this process greatly reduces greenhouse gas emissions and costs compared to the industrial lithium extraction method. This highlights the potential of the process to manage pollutants while providing a sustainable lithium supply, and this fluorination strategy shows promise to be extended to other metal extraction processes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data needed to support the conclusions in the paper are present in the paper and/or Supplementary Information. Additional data related to this paper are available upon reasonable request from the corresponding authors.
References
Semieniuk, G., Taylor, L., Rezai, A. & Foley, D. K. Plausible energy demand patterns in a growing global economy with climate policy. Nat. Clim. Change 11, 313–318 (2021).
Baars, J., Domenech, T., Bleischwitz, R., Melin, H. E. & Heidrich, O. Circular economy strategies for electric vehicle batteries reduce reliance on raw materials. Nat. Sustain. 4, 71–79 (2021).
Choubey, P. K., Kim, M.-S., Srivastava, R. R., Lee, J.-C. & Lee, J.-Y. Advance review on the exploitation of the prominent energy-storage element: lithium. Part I: from mineral and brine resources. Miner. Eng. 89, 119–137 (2016).
Mine production of lithium worldwide from 2010 to 2023. Statista www.statista.com/statistics/606684/world-production-of-lithium/ (2025).
Demand for lithium worldwide in 2020 and 2021 with a forecast from 2022 to 2035. Statista www.statista.com/statistics/452025/projected-total-demand-for-lithium-globally/ (2024).
Kesler, S. E. et al. Global lithium resources: relative importance of pegmatite, brine and other deposits. Ore Geol. Rev. 48, 55–69 (2012).
Yang, S., Zhang, F., Ding, H., He, P. & Zhou, H. Lithium metal extraction from seawater. Joule 2, 1648–1651 (2018).
Diallo, M. S., Kotte, M. R. & Cho, M. Mining critical metals and elements from seawater: opportunities and challenges. Environ. Sci. Technol. 49, 9390–9399 (2015).
Chen, X. et al. Spatially separated crystallization for selective lithium extraction from saline water. Nat. Water 1, 808–817 (2023).
Khalil, A., Mohammed, S., Hashaikeh, R. & Hilal, N. Lithium recovery from brine: recent developments and challenges. Desalination 528, 115611 (2022).
Amoatey, P. et al. A critical review of environmental and public health impacts from the activities of evaporation ponds. Sci. Total Environ. 796, 149065 (2021).
Flexer, V., Baspineiro, C. F. & Galli, C. I. Lithium recovery from brines: a vital raw material for green energies with a potential environmental impact in its mining and processing. Sci. Total Environ. 639, 1188–1204 (2018).
Swain, B. Recovery and recycling of lithium: a review. Sep. Purif. Technol. 172, 388–403 (2017).
Zhang, Q.-H., Li, S.-P., Sun, S.-Y., Yin, X.-S. & Yu, J.-G. Lithium selective adsorption on low-dimensional titania nanoribbons. Chem. Eng. Sci. 65, 165–168 (2010).
Baird, M. A. et al. Microporous polymer sorbents for direct lithium extraction. ACS Energy Lett. 9, 4361–4368 (2024).
Zhang, Y. et al. Congener-welded crystalline carbon nitride membrane for robust and highly selective Li/Mg separation. Sci. Adv. 10, eadm9620 (2024).
Zhang, S. et al. Solar-driven membrane separation for direct lithium extraction from artificial salt-lake brine. Nat. Commun. 15, 238 (2024).
Song, Y. et al. Solar transpiration–powered lithium extraction and storage. Science 385, 1444–1449 (2024).
Xu, R. et al. Continuous lithium extraction from brine by efficient redox-couple electrodialysis. Matter 7, 3876–3890 (2024).
Li, Z. et al. Lithium extraction from brine through a decoupled and membrane-free electrochemical cell design. Science 385, 1438–1444 (2024).
Shi, C. et al. Solvent extraction of lithium from aqueous solution using non-fluorinated functionalized ionic liquids as extraction agents. Sep. Purif. Technol. 172, 473–479 (2017).
Zhang, L. et al. Recovery of lithium from salt lake brine with high Na/Li ratio using solvent extraction. J. Mol. Liq. 362, 119667 (2022).
Sun, Y., Wang, Q., Wang, Y., Yun, R. & Xiang, X. Recent advances in magnesium/lithium separation and lithium extraction technologies from salt lake brine. Sep. Purif. Technol. 256, 117807 (2021).
Guo, H., Lv, M., Kuang, G., Cao, Y. & Wang, H. Stepwise heat treatment for fluorine removal on selective leachability of Li from lepidolite using HF/H2SO4 as lixiviant. Sep. Purif. Technol. 259, 118194 (2021).
Rosales, G. D., Pinna, E. G., Suarez, D. S. & Rodriguez, M. H. Recovery process of Li, Al and Si from lepidolite by leaching with HF. Minerals 7, 36 (2017).
Rosales, G. D., del Carmen Ruiz, M. & Rodriguez, M. H. Novel process for the extraction of lithium from β-spodumene by leaching with HF. Hydrometallurgy 147, 1–6 (2014).
Evich, M. G. et al. Per-and polyfluoroalkyl substances in the environment. Science 375, eabg9065 (2022).
Glüge, J. et al. An overview of the uses of per-and polyfluoroalkyl substances (PFAS). Environ. Sci. Process. Impacts 22, 2345–2373 (2020).
Ankley, G. T. et al. Assessing the ecological risks of per- and polyfluoroalkyl substances: current state-of-the science and a proposed path forward. Environ. Toxicol. Chem. 40, 564–605 (2021).
Economic Analysis for the Final Per- and Polyfluoroalkyl Substances National Primary Drinking Water Regulation (EPA, 2024); www.epa.gov/system/files/documents/2024-04/pfas-npdwr_final-rule_ea.pdf
Jafarinejad, S. A mini-review of full-scale drinking water treatment plants for per-and polyfluoroalkyl substances (PFAS) removal: possible solutions and future directions. Sustainability 17, 451 (2025).
Xiao, F. et al. Thermal decomposition of anionic, zwitterionic, and cationic polyfluoroalkyl substances in aqueous film-forming foams. Environ. Sci. Technol. 55, 9885–9894 (2021).
Alinezhad, A. et al. Mechanistic investigations of thermal decomposition of perfluoroalkyl ether carboxylic acids and short-chain perfluoroalkyl carboxylic acids. Environ. Sci. Technol. 57, 8796–8807 (2023).
Yang, N. et al. Solvent-free nonthermal destruction of PFAS chemicals and PFAS in sediment by piezoelectric ball milling. Environ. Sci. Technol. Lett. 10, 198–203 (2023).
Guan, Y. et al. Near-complete destruction of PFAS in aqueous film-forming foam by integrated photo-electrochemical processes. Nat. Water 2, 443–452 (2024).
Cheng, Y. et al. Flash upcycling of waste glass fibre-reinforced plastics to silicon carbide. Nat. Sustain. 7, 452–462 (2024).
Dong, Q. et al. Depolymerization of plastics by means of electrified spatiotemporal heating. Nature 616, 488–494 (2023).
Cui, B. et al. Waste to wealth: defect-rich Ni-incorporated spent LiFePO4 for efficient oxygen evolution reaction. Sci. China Mater. 64, 2710–2718 (2021).
Chen, W. et al. Nondestructive flash cathode recycling. Nat. Commun. 15, 6250 (2024).
Cheng, Y. et al. Rapid electrothermal rejuvenation of spent lithium cobalt oxide cathode. Energy Environ. Sci. 18, 6085–6093 (2025).
Cheng, Y. et al. Electrothermal mineralization of per- and polyfluoroalkyl substances for soil remediation. Nat. Commun. 15, 6117 (2024).
Yu, F. et al. Rapid self-heating synthesis of Fe-based nanomaterial catalyst for advanced oxidation. Nat. Commun. 14, 4975 (2023).
Sun, L. et al. Millisecond self-heating and quenching synthesis of Fe/carbon nanocomposite for superior reductive remediation. Appl. Catal. B 342, 123361 (2023).
Scotland, P. et al. Mineralization of captured perfluorooctanoic acid and perfluorooctane sulfonic acid at zero net cost using flash Joule heating. Nat. Water 3, 486–496 (2025).
Chen, Y. et al. Ultra-fast self-assembly and stabilization of reactive nanoparticles in reduced graphene oxide films. Nat. Commun. 7, 12332 (2016).
Dong, Q. et al. Programmable heating and quenching for efficient thermochemical synthesis. Nature 605, 470–476 (2022).
Jiang, R. et al. Ultrafast synthesis for functional nanomaterials. Cell Rep. Phys. Sci. 2, 100302 (2021).
Gagliano, E., Falciglia, P. P., Zaker, Y., Karanfil, T. & Roccaro, P. Microwave regeneration of granular activated carbon saturated with PFAS. Water Res. 198, 117121 (2021).
Abou-Khalil, C. et al. Enhancing the thermal mineralization of perfluorooctanesulfonate on granular activated carbon using alkali and alkaline-earth metal additives. Environ. Sci. Technol. 58, 11162–11174 (2024).
Wang, F., Lu, X., Li, X. Y. & Shih, K. Effectiveness and mechanisms of defluorination of perfluorinated alkyl substances by calcium compounds during waste thermal treatment. Environ. Sci. Technol. 49, 5672–5680 (2015).
Xiao, F. et al. Thermal stability and decomposition of perfluoroalkyl substances on spent granular activated carbon. Environ. Sci. Technol. Lett. 7, 343–350 (2020).
Camdzic, D., Dickman, R. A. & Aga, D. S. Total and class-specific analysis of per-and polyfluoroalkyl substances in environmental samples using nuclear magnetic resonance spectroscopy. J. Hazard. Mater. Lett. 2, 100023 (2021).
Deng, B. et al. Urban mining by flash Joule heating. Nat. Commun. 12, 5794 (2021).
Deng, B. et al. Flash separation of metals by electrothermal chlorination. Nat. Chem. Eng. 1, 627–637 (2024).
Universal Matter. Ltd https://www.universalmatter.com/ (2026).
Fan, X. Z. et al. Directing fluorinated solid electrolyte interphase by solubilizing crystal lithium fluoride in aprotic electrolyte for lithium metal batteries. Adv. Energy Mater. 14, 2303336 (2024).
Ramasubramanian, A. et al. Lithium diffusion mechanism through solid–electrolyte interphase in rechargeable lithium batteries. J. Phys. Chem. C 123, 10237–10245 (2019).
Chemanalyst www.chemanalyst.com/Pricing-data/ (2026).
An, J. W. et al. Recovery of lithium from Uyuni salar brine. Hydrometallurgy 117, 64–70 (2012).
Huang, T.-Y., Pérez-Cardona, J. R., Zhao, F., Sutherland, J. W. & Paranthaman, M. P. Life cycle assessment and techno-economic assessment of lithium recovery from geothermal brine. ACS Sustain. Chem. Eng. 9, 6551–6560 (2021).
OpenLCA software (Greendelta, 2026); https://www.openlca.org
GREET software (Argonne National Laboratory, 2025); https://greet.es.anl.gov/
ELCD database. Nexus https://nexus.openlca.org/database/ELCD (2018).
Stinn, C. & Allanore, A. Selective sulfidation of metal compounds. Nature 602, 78–83 (2022).
Acknowledgements
We thank W. Guo for the assistance of cryo-TEM characterizations, B. Chen for helpful discussion on XPS results, T. Terlier for developing ICP methods and X. Wang for the NMR test. The funding of the research is provided by the Air Force Office of Scientific Research (FA9550-22-1-0526, J.M.T.), the US Army Corps of Engineers, ERDC grant (W912HZ-21-2-0050, and W912HZ-24-2-0027, J.M.T.), Rice Academy Fellowship (Y. Cheng), the Strategic Environmental Research and Development Program (SERDP) award (ER22-3258, M.S.W.), and Rice WaTER Institute and Sustainability Institute Postdoctoral Fellowship (Y. Chung). The characterization equipment used in this project is partly from the Shared Equipment Authority (SEA) at Rice University.
Author information
Authors and Affiliations
Contributions
Y. Cheng and J.M.T. conceived the idea and designed the experiments. Y. Cheng conducted the thermodynamic analyses. Y. Cheng conducted the Li recovery experiments and most of the characterizations with the help of Q.L., P.S., T.X, J.S. and S.C. Y. Cheng and C.K. built the flash distillation system. C.G. provided the AFFF samples. L.E. built and maintained the electrical system. K.J.S. helped with the GC-MS detection. P.S., B.L. and T.S. helped with the NMR test. Y. Cheng conducted the electrochemical tests of the LIBs with the help of J.C. and O.E.O. Y. Chung and M.S.W. conducted the liquid characterizations. Y. Cheng and R.A.N. conducted the scale-up experiments with the help of H.Y. A.E.L. conducted the techno-economic analysis and life-cycle assessment and analysed the results with Y. Cheng. Y. Cheng and J.M.T. wrote and edited the paper. All aspects of the research were overseen by J.M.T. All authors have discussed the results and given approval to the final version of the paper.
Corresponding authors
Ethics declarations
Competing interests
Rice University owns intellectual property on the electrothermal fluorination strategy for lithium recovery from brine salt. A provisional patent was filed by Rice University, where Y. Cheng and J.M.T. are listed as the inventors, which has not yet been licensed. The other authors declare no competing interests.
Peer review
Peer review information
Nature Water thanks Onur Apul, Treavor Boyer, Yanan Chen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Physical properties of different chlorides components in brine salts.
a, Vapor pressure-temperature relationship. The horizontal dash line denotes atmospheric pressure of ~105 Pa. b, Water solubility at 25 °C.
Extended Data Fig. 2 Characterizations of field-collected AFFF before and after GAC adsorption.
a, Pictures of field-collected AFFF solution before (left) and after GAC adsorption (right). b, pH values of field-collected AFFF before and after GAC adsorption. c, Total dissolved organic carbon analysis of field-collected AFFF before and after GAC adsorption.
Extended Data Fig. 3 DSC results for AFFF decomposition.
a, AFFF/GAC. b, LiCl. c, AFFF/GAC mixed with LiCl with the F/Li ratio of 1.
Extended Data Fig. 4 XRD patterns of fluorination products from the reaction between AFFF/GAC with different metal chlorides at an input voltage of 100 V.
a, AFFF/GAC before ETF. b–f, Reaction products of AFFF/GAC with LiCl (b), NaCl (c), KCl (d), MgCl2 (e), and CaCl2 (f) after ETF.
Extended Data Fig. 5 GC-MS results of the fluorination evolved gas during the ETF process.
a, GC-MS chromatogram of the gases for the AFFF/GAC. b, Mass spectrum corresponds to the main peak with a retention time of 1.55 min in a. c, GC-MS chromatogram of the gases for the mixture of brine salt and AFFF/GAC (10% mole excessive of F). d, Mass spectrum corresponds to the main peak with a retention time of 1.55 min in c.
Extended Data Fig. 6 Scaling up sample mass to 60 g per batch.
a, Picture of the experiment setup for brine salt fluorinations. b, Picture of the sample during scale-up ETF. c, Real-time temperature curve recorded by an infrared thermometer with a detecting range of 1000–3000 °C. d, XRD patterns of fluorinated products. e, Purity and yield of the recovered Li after following washing and distillation processes.
Extended Data Fig. 7 31P NMR spectra of different electrolytes after storage for 1 month.
a, 31P NMR spectra. b, Zoom-in 31P NMR spectra. The peaks in the range from −15 to −25 ppm are ascribed to F2PO2−, degraded from LiPF6 in the electrolytes.
Extended Data Fig. 8 Morphology of the surface of Li chip in Li | |Li symmetric cells after cycling for 100 cycles.
a, Using the electrolyte without LiF. b, Using the electrolyte with LiF. The red circles labeled the formed lithium dendrites after cycling.
Extended Data Fig. 9 Material flow of different scenarios for lithium extraction from brine.
a, ETF process using AFFF/GAC as the fluorination agent. Gr denotes graphene. GAC and PFAS were converted to flash graphene during the process. b, Material flow of the industrial sorption process for lithium recovery from brine. c, Material flow of the industrial evaporation-precipitation process for lithium recovery from brine. C-brine-X means the concentrated brine, where X indicates different steps, and the chemicals in the brackets after are the components in the C-brine.
Extended Data Fig. 10 LCA and TEA results.
a, Average CED of four scenarios. b, Average GWP in CO2 equivalent of four scenarios. c, Average WRD of four scenarios. d, Average operating cost of four scenarios. e, Average revenue of four scenarios. f, Average profit of four scenarios.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Notes 1–3, Figs. 1–56, Tables 1–7 and References.
Supplementary Data 1 (download XLSX )
TEA and LCA.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 10 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cheng, Y., Lathem, A.E., Scotland, P. et al. Waste per- and polyfluoroalkyl substance-assisted flash fluorination for lithium recovery from brine. Nat Water 4, 369–380 (2026). https://doi.org/10.1038/s44221-026-00593-1
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44221-026-00593-1