Abstract
Background
Within the colorectal cancer (CRC) tumour microenvironment, tumour infiltrating lymphocytes (TILs) and tumour cell density (TCD) are recognised prognostic markers. Measurement of TILs and TCD using deep-learning (DL) on haematoxylin and eosin (HE) whole slide images (WSIs) could aid management.
Methods
HE WSIs from the primary tumours of 127 CRC patients were included. DL was used to quantify TILs across different regions of the tumour and TCD at the luminal surface. The relationship between TILs, TCD, and cancer-specific survival was analysed.
Results
Median TIL density was higher at the invasive margin than the luminal surface (963 vs 795 TILs/mm2, P = 0.010). TILs and TCD were independently prognostic in multivariate analyses (HR 4.28, 95% CI 1.87–11.71, P = 0.004; HR 2.72, 95% CI 1.19–6.17, P = 0.017, respectively). Patients with both low TCD and low TILs had the poorest survival (HR 10.0, 95% CI 2.51–39.78, P = 0.001), when compared to those with a high TCD and TILs score.
Conclusions
DL derived TIL and TCD score were independently prognostic in CRC. Patients with low TILs and TCD are at the highest risk of cancer-specific death. DL quantification of TILs and TCD could be used in combination alongside other validated prognostic biomarkers in routine clinical practice.
Similar content being viewed by others
Background
Colorectal cancer (CRC) is the third most commonly diagnosed malignancy globally and is the second most common cause of cancer death with over 44,000 new cases and over 16,800 deaths every year in the United Kingdom [1, 2]. In routine clinical practice, disease stage, as determined by the Union for International Cancer Control tumour node metastasis (TNM) system, is primarily used to predict CRC patient prognosis and guide treatment decisions along with other validated adverse pathological features [3,4,5]. However, patients diagnosed at the same disease stage may have differing outcomes [6, 7] and 20–50% of patients initially diagnosed with localised disease, will ultimately develop distant metastases [8]. There is therefore an urgent need for novel validated prognostic biomarkers that are reproducible, have a short turnaround time, and are easy to implement into routine pathology workflow, to further stratify patients who may benefit from adjuvant therapy or who may be cured by surgery alone.
Tumour infiltrating lymphocytes (TILs) are a major component of the tumour microenvironment and high numbers of TILs have been associated with improved survival in CRC and other tumour types [9,10,11]. There are several existing TIL classification systems that rely on the subjective quantification of the lymphocytic infiltration or the scoring of lymphoid aggregates or “Crohn’s-like lymphoid reaction” [10,11,12]. However, manually scoring TILs using the aforementioned systems is limited by high inter-observer variability, is time-consuming and therefore hasn’t been implemented into routine clinical practice [13, 14]. Ourselves and others, including Galon et al., have recognised that using immunohistochemistry (IHC) to quantitate the density of T-cell subsets is important, however, this remains expensive and time consuming to perform, hindering its use in routine clinical practice [15,16,17,18]. It is also important to note that quantification of TILs on haematoxylin and eosin (HE) slide includes the assessment of all lymphocytes (T and B cells), whereas IHC for T-cell subsets only assesses T cells, so values may differ between techniques. To date, quantification of TILs has focused on assessing the deep invasive margin (e.g. Jass score) and/or the central tumour region in resection specimens [9]. Little attention has been paid to the luminal surface of the tumour, despite this being the only area sampled in endoscopic biopsies. Although using IHC to explore T cell subsets has been performed in rectal biopsies, there has been minimal focus on quantification of TILs on HE in biopsies [19,20,21]. With increasing use of neoadjuvant treatment in CRC patients, biomarkers applied to routine endoscopic biopsy samples will play a key role in prognostication and prediction of response to treatment in the future.
An alternative method of quantifying TILs, without the need for IHC, is the use of deep-learning (DL) on a high resolution digitally scanned HE stained slide. The benefits of using DL in this capacity would be two-fold, in the clinical setting it would be cheap, rapid and eliminate pathologist inter-observer variability and in the research setting it would allow the analysis of large datasets quickly to generate robust data. DL detection of TILs on HE has been shown to be prognostic, with a high number of TILs associated with a favourable prognosis in a range of cancer types including breast cancer [22, 23], gastric cancer [24], and melanoma [25].
The number of lymphocytes also differs depending on the specific region of the tumour that has been assessed; several studies have found that the number of lymphocytes was greater at the invasive margin compared to the central tumour [11, 26, 27]. In these assessments, the luminal surface has always been incorporated into the “central tumour” region, and hasn’t been separately investigated.
In addition to TILs, the relative proportion of malignant epithelial cells and stroma, known as tumour cell density (TCD) or tumour-stroma ratio, has been identified as a prognostic factor in patients with potentially curative CRC with a high proportion of stroma associated with poorer survival [28,29,30,31,32,33]. As with TILs, TCD has been calculated in different ways between studies, with the majority being conducted manually either using a subjective estimation or an objective point counting technique at the luminal surface or invasive margin [28,29,30,31, 34]. More recently, TCD estimation has been carried out using DL [35, 36]. The benefit of combining TCD and TIL scores has recently been demonstrated through the Glasgow Microenvironment Score which is a prognostic marker in stage I-III CRC, however this system requires manual assessment of both TCD and TILs and neither marker was measured at the luminal surface [37]. DL measurement of TILs and TCD would have the benefit of being quicker, cheaper, allow analysis of large datasets, and potentially more objective and convenient compared to manual measurement.
We hypothesised that: (a) high DL-derived TIL density is associated with improved cancer-specific survival on both univariate and multivariate analysis, (b) combining DL-derived TIL and TCD values improves prognostication in CRC, (c) DL-derived TILs and TCD values assessed at the luminal surface could be used to inform neoadjuvant treatment decisions.
Methods
Patients and clinicopathological data
Patients who underwent potentially curative surgery for CRC at the Marienhospital, Düsseldorf, Germany between 1st January 1990 and 31st December 1995, were included in this retrospective study. None of the patients received pre-operative chemotherapy or radiotherapy. Patients undergoing palliative surgery were not included in this study. One representative HE stained tumour tissue section from the surgical resection specimen was scanned at x40 magnification (Aperio XT, Aperio Technologies, Vista, CA, USA). Only those with manually derived TCD values from a previous study were included (n = 144) [28]. Cases were excluded if the slides did not contain definite invasive adenocarcinoma or the full thickness of the primary tumour; slides with significant artefact were also excluded. Histopathological data were available for all patients including site of tumour, lymphovascular invasion status, maximum depth of invasion (pT category), lymph node involvement (pN category) and distant metastasis (pM category) according to TNM 5th edition [38]. In addition, mismatch repair status was available for all patients from a previous study [39].
This study conforms to the REMARK guidelines (refer to Supplementary Table S1) [40].
Annotation of tumour regions
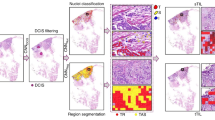
Digital slides were annotated using HeteroGenius-MIM image analysis software (HeteroGenius Ltd., Leeds, UK). In each case, three tumour regions were annotated manually by a pathologist (AW): (1) the whole invasive tumour area, including tumour and associated stroma; (2) a 2 mm deep strip at the luminal surface of the tumour, and (3) a 1 mm strip at the deep invasive margin of the tumour (Supplementary Fig. S1). Regions of ulcer slough or necrosis visible at low magnification were excluded from the annotation areas. Areas of dysplasia at the edges of the cancer were excluded from the annotation.
Image analysis pipeline to establish TIL density
For the detection of TILs, we used our published method [24] which classified cells into 9 different types: lymphocyte, plasma cell, granulocyte, fibroblast, endothelial cell, muscle, tumour, and normal epithelium [24]. This published model had a further ~55,000 annotated cells from a mixture of oesophagogastric cancer cases and colorectal cancer cases added and was trained for a further 9000 epochs. We developed a convolutional neural network model to differentiate between genuine tissue and background space. This model was trained using 147 tissue annotations and 159 background annotations for 2049 epochs. Manual quality control was performed as previously described in three independent regions [24]. TIL density was calculated by dividing the total number of lymphocytes detected within the region of interest by the genuine tissue area of the region of interest. No attempt was made to distinguish between TILs located in the stroma and TILs overlying tumour epithelium [24]. For an example of TILs segmentation in HE stained colorectal cancer, see Supplementary Fig. S2A, B.
Image analysis pipeline to establish TCD
The annotation previously used to determine TCD by manual point counting (9 mm2 region of interest placed at the luminal surface in the region of apparent highest TCD) [28] was uploaded to HeteroGenius-MIM image analysis software. None of the manual spot counting data was used to train the TCD model.
A convolutional neural network derived from a UNET framework [41] was trained using manual point-counting data from 300 +/− 15 points spread equidistant across the whole invasive tumour area from 101 chemo(radio)therapy naive rectal cancers from an independent series; this series was split 60% and 40% for training and testing.
The trained model was then applied to the 9 mm2 region of interest at the luminal surface in the current dataset. TCD was calculated by dividing tumour points by all informative points to give a percentage TCD. For an example of TCD segmentation in HE stained colorectal cancer, see Supplementary Fig. S2C, D.
Statistical analyses
The relationship between TIL density in different regions of the tumour and clinicopathological variables was analysed using the Kruskal–Wallis test. Differences in TIL density between tumour regions were firstly analysed using the Kruskal–Wallis test; if significant, Dunn’s Test with Bonferroni correction was performed to determine the statistical differences between each region. The Pearson correlation coefficient was calculated to analyse correlation between TIL density and TCD.
Cut-off values to dichotomise TCD and TIL density in each region into high or low were based on the best hazard ratio for survival. Dichotomised TCD at the luminal surface and TIL density at the invasive margin were combined to create four groups. The same categorisation was also performed when TIL density was measured at the luminal surface, rather than the invasive margin.
The primary study endpoint was cancer-specific survival, which was available for all patients. Patients who died within 30 days of surgery were excluded from the study. The median follow-up time was 5.3 years and 98 patients (77.2%) were alive at the end of the study period. The relationship of TIL density and TCD with cancer-specific survival was analysed using the Kaplan–Meier method, log-rank test and Cox proportional hazards models [42]. In multivariate analyses, factors found to be significant in univariate survival analysis were included. P-values < 0.05 were considered statistically significant.
Statistical analyses were performed using IBM SPSS Statistics (Version 29.0.0.0, IBM, Armonk, NY, USA), R, version 4.2.2 (R Foundation for Statistical Computing, Vienne, Austria), and python version 3.8. R packages used were: ggplot2 (version 3.4.4), ggpubr (version 0.6.0), FSA (version 0.9.5), survival (version 3.5-3), survminer (version 0.4.9), and dplyr (version 1.1.0); python packages used were lifelines (version 0.28.0).
Results
TIL density in different tumour regions
Of the 144 cases initially identified, 17 were excluded (Supplementary Fig. S3). 127 CRC cases were therefore included in the final analysis. The median (range) area of each annotation was 94.0 mm2 (22.1–439.3 mm2) in the whole invasive tumour, 18.5 mm2 (3.0–52.2 mm2) at the invasive margin, and 32.6 mm2 (7.2–109.8 mm2) at the luminal surface.
The median (range) TIL density varied between the different tumour regions; whole tumour 810 TILs/mm2 (221–3404 TILs/mm2), invasive margin 963 TILs/mm2 (296–3432 TILs/mm2), and luminal surface 795 TILs/mm2 (range: 259–3471 TILs/mm2). TIL density at the invasive margin was significantly greater than that at the luminal surface (P = 0.010) and across the whole tumour area (P = 0.015). TIL density in any of the regions measured was not significantly related to any clinicopathological variables (Table 1), including MMR status.
TIL density and TCD and survival
TIL density was dichotomised in the whole tumour area (low ≤ 940 TILs/mm2, n = 80, high n = 47) the invasive margin (low ≤ 1155 TILs/mm2, n = 78, high n = 49), and the luminal surface (low ≤ 978 TILs/mm2, n = 84, high n = 43). Patients with low TIL density at the invasive margin or across the whole tumour area had poorer cancer-specific survival than those with high TIL density (HR 3.83, 95% CI 1.47-9.98, P = 0.006; HR 3.16, 95% CI 1.21–8.22, P = 0.019, respectively (Table 2, and Fig. 1a, b)). TIL density at the luminal surface was not significantly associated with survival (HR 1.96, 95% CI 0.81–4.80, P = 0.138; Fig. 1c). When only pMMR cases were analysed (n = 106), low TIL density at the invasive margin and across the whole tumour remained significantly associated with poorer cancer-specific survival than those with high TIL density (HR 4.58 95% CI 1.37–15.33, P = 0.013; HR 3.11, 95% CI 1.07–9.07, P = 0.019, respectively).
P value derived by the log rank test. a TIL at the invasive margin, b TIL across the whole tumour area, c TIL at the luminal surface, d TCD in a 3 × 3 mm box at the luminal surface. TIL tumour infiltrating lymphocytes, TCD tumour cell density.
TCD at the luminal surface was dichotomised at 47% (low n = 30, high n = 97). There was no relationship between TCD and any clinicopathological variables, including MMR status (Supplementary Table S2). Patients with low TCD had significantly poorer cancer-specific survival than those with high TCD (HR 2.66, 95% CI 1.30–5.42, P = 0.007, Fig. 1d). When only pMMR cases were analysed (n = 106), low TCD remained significantly associated with poorer cancer-specific survival than those with high TCD (HR 2.23, 95% CI 1.00–4.97, P = 0.049).
When TIL density at the invasive margin and TCD at the luminal surface were included in a multivariate model along with pT, pN and lymphovascular invasion, both low TIL density and low TCD were significantly associated with poorer cancer-specific survival (HR 4.28, 95% CI 1.87–11.71, P = 0.004; HR 2.72, 95% CI 1.19–6.17, P = 0.017, respectively) when compared to patients with high TIL density and high TCD (Table 2).
When TILs in the whole tumour area was included in the model instead of TILs at the invasive margin, both low TIL density and low TCD remained significantly associated with cancer-specific survival (HR 3.74, 95% CI 1.34–10.48, P = 0.012; HR 2.80, 95% CI 1.22–6.39; P = 0.015, respectively) when compared to those with high TIL density and high TCD (Supplementary Table S4). Survival analysis for data censored at 5 years is provided in Supplement.
Combined TCD and TILs score and survival
Correlation between TILs at the invasive margin and TCD was explored and no correlation was found (Pearson correlation coefficient: -0.149, P = 0.094). When the dichotomised scores for TILs at the invasive margin and TCD were combined, patients in the high TCD and TILs had a significantly greater cancer-specific survival than those with low TCD and TILs group (HR 0.08, 95% CI 0.02–0.27, P = < 0.001; Table 2, Fig. 2a); this remained significant on multivariate analysis (HR 0.10, 95% CI 0.03–0.40, P = 0.001; Supplementary Table S5). Those in the other two groups also had a significantly greater cancer-specific survival compared to those in the low TCD and TILs group (Supplementary Table S5).
P value derived by the log rank test. a TIL at the invasive margin and TCD at the luminal surface, b Both TIL and TCD at the luminal surface. TIL tumour infiltrating lymphocytes, TCD tumour cell density, IM invasive margin, LS luminal surface.
When a further combined score was determined based on the dichotomised scores for both TCD and TILs at the luminal surface, patients in the high TCD and TILs group had a significantly greater cancer-specific survival compared to those in the low TCD and TILs group (HR 0.21 95% CI 0.08–0.56, P = 0.002; Fig. 2b). This remained significant on multivariate analysis (HR 0.29, 95% CI 0.10–0.81, P = 0.019; Supplementary Table S6).
Discussion
A significant proportion of patients diagnosed with CRC will ultimately develop incurable distant metastatic disease [8], however, the identification of patients with high-risk disease who are most likely to benefit from adjuvant/neoadjuvant treatment remains challenging. There is therefore a pressing clinical need to develop novel prognostic biomarkers to aid decision making and to better identify this subset of patients. We aimed to explore two recognised but currently underutilised prognostic markers, TILs and TCD, both derived by DL on HE slides. To date, most studies on TILs have focused on the immune infiltrate at the deep invasive margin, which is notably defined differently across various studies, with some focussing on the infiltrate in the central tumour region instead [11, 13, 16]. There has been little focus on the immune infiltrate at the luminal surface, which is surprising given that this area is sampled in diagnostic biopsies and therefore opens the possibility for being used in decision making for neoadjuvant treatment.
Our study compared TIL density in different tumour regions of interest and aimed to explore the combined measurement of TILs and TCD as a prognostic marker, particularly at the luminal surface, to understand whether measuring these markers in biopsies could be a valid approach. The prospective assessment of TCD in different regions of the tumour is not the focus of this study, but will be explored in the future.
This study has shown that TIL density at the invasive margin is significantly greater compared to that across the whole tumour area and at the luminal surface. This is not unexpected as the invasive margin is considered to be the interface between tumour and normal tissue and therefore the primary site of interaction between malignant and immune cells. Many existing classification systems assess TILs at the invasive margin; similarly the presence of a “Crohn’s-like reaction” is noted at the deep aspect of the tumour [10,11,12, 18].
As expected from the current literature, high TIL density at both the invasive margin and across the whole tumour area was independently associated with greater cancer-specific survival on multivariate analysis [9, 16]. Similarly a high TCD was associated with greater cancer-specific survival on multivariate analysis. When dichotomised scores for TCD at the luminal surface and TIL density at the invasive margin were combined, patients with a low score in both had the worst cancer-specific survival when compared to those with a high scores in both. This demonstrates that the combined TCD and TILs score was a stronger prognostic marker than that of TILs or TCD in isolation. When looking at the results for the other two groups, it appeared that TIL density at the invasive margin had a greater impact on survival than TCD at the luminal surface with the low TCD and high TIL group showing a similar hazard ratio to that of the high TCD and high TIL group.
As a single marker, TIL density at the luminal surface was not significantly associated with prognosis, but there was a trend between high TILs and improved cancer-specific survival as seen in other areas of the tumour. Given the size of the hazard ratio, this likely reflects a type 2 error and this analysis may well be significant in a larger series of patients. As expected from current literature, this study found that TCD at the luminal surface was an independent prognostic marker in CRC. In contrast to other studies that have largely focused on the invasive margin [29, 31,32,33], this measure was assessed at the luminal surface of the tumour in keeping with the previous work we have performed using manual assessment [28]. When TCD and TIL density scores from the luminal surface were combined, the combined score was a stronger prognostic marker than that of TCD in isolation; patients with low scores in both had the worst cancer-specific survival when compared to those with high scores in both.
The luminal surface of a resection specimen is essentially the same region sampled in a diagnostic endoscopic biopsy, with the depth of the luminal strip in our study chosen to replicate the depth of a typical biopsy sample. These prognostic scores are easy to obtain and could be combined with existing clinicopathological factors to augment clinical decision making; the luminal surface scoring in particular has the potential to be applied to biopsy specimens to identify the subset of patients with the worst outcomes and even influence neoadjuvant decision making. Biopsy samples offer unique challenges in comparison to resection samples in that they can be heterogeneous and are frequently small and fragmented. Despite this, they are increasingly being used for molecular biomarker testing and therefore decision making for neoadjuvant treatment. The composition of immune cells and TCD in biopsy samples has been found to be important by others [19, 43]. Studies utilising IHC to identify specific T-cell subsets in biopsies from patients with locally advanced rectal cancer prior to neoadjuvant chemo-radiotherapy have found high Immunoscore in the pre-treatment biopsy was associated with improved 5 year recurrence rates compared to low Immunoscore; however, these results are only from a subset of cases showing a complete response to neoadjuvant therapy [20, 21]. These studies rely on IHC and little work has been done on TIL detection on HE in CRC biopsies to date. The value of manually measuring TCD on biopsy samples has been demonstrated in oesophageal adenocarcinoma with low TCD associated with worse overall survival; however, there was significant inter-observer variation in TCD scoring between pathologists [43]. Low TCD in CRC biopsies has also been found to be significantly associated with the rate of lymph node metastasis in potentially curable CRC patients [44].
TCD and TILs have previously been combined to create a composite score called the Glasgow Microenvironment Score; this score has been found to be a prognostic biomarker in stage I-III CRC, however this system requires manual assessment of both TCD and TILs [37]. Another study in CRC has combined DL tumour stroma estimations and DL immune scores and found a beneficial effect of a combined score [35]; this study however looked at TCD and immune scores within the invasive margin component only; our study also considers the use of a combined TCD and TIL density score at the luminal surface.
To date, both TILs and TCD are not used in routine clinical practice largely due to the time-consuming nature of manual scoring by pathologists and also the subjective nature of such assessments. Measuring TILs and TCD by DL is objective, cheap and unlike Immunoscore can be carried out on HE without requiring additional tissue sections to be cut and stained from the block [16]. Digital scanning is increasingly used in routine clinical practice internationally and a move to DL assessment could save significant resources in a financially stretched health service. DL has been used to separately quantify TILs in the stromal and epithelial compartments on HE in triple negative breast cancer with high TILs in both regions associated with improved prognosis [22]. Quantifying TILs into epithelial and stromal compartments is therefore recommended in breast cancer management pathways [45], however there are no similar recommendations in CRC at the present time. Another study demonstrated that DL TIL quantification was both prognostic and predictive of response to adjuvant chemotherapy in stage II-III gastric cancer; similarly to our study, this work did not separate TILs into epithelial or stromal compartments [24]. DL has been used in CRC to detect “Crohn’s-like reaction” density at the invasive margin, where greater levels were also associated with improved prognosis, however, in contrast to our study, this methodology used an area based convolutional neural network instead of UNET based cell detection [46]. UNET based cell detection identifies and segments individual cells rather than being based on pixels, and may therefore be superior at calculating TIL density [47].
The current study has some limitations. This study used the 5th edition of the TNM classification [38] compared to the 8th edition used in current practice [48]. Serial changes to TNM staging could impact the multivariate analyses used in our study. We appreciate that the small size of this cohort and lack of a validation cohort is a significant limitation of this study in respect to validation of our cut-off values. However, this combined biomarker approach will be investigated in the recently published international FOxTROT trial of advanced but operable colon cancer, including a straight to surgery group (n = 354) and a neoadjuvant chemotherapy group (n = 699) [49], where diagnostic biopsy samples are also available alongside surgical resections.
In conclusion, we have explored the prognostic value of TIL density in different regions of the tumour along with TCD at the luminal surface. We have shown that luminal surface TCD and invasive margin TIL density measured on HE using DL based methods are both independently prognostic in CRC. Patients with combined low TCD and TIL score are at highest risk of death. The DL methodology for both biomarkers is cheap, objective, should be more reproducible and quicker to perform and could be used as an adjunct alongside routine biomarkers to stratify and identify those patients who may benefit from adjuvant therapy. TCD and TIL density when both measured at the luminal surface have the potential to be used on biopsy samples to aid prognostication in the neoadjuvant setting. Further studies are in progress to validate this work in an independent cohort and to investigate the prognostic value of TCD and TIL density in biopsy samples from patients with CRC. As TCD and TIL density have been found to be prognostic in other solid tumour types, this DL based method and combined biomarker approach should also be explored in other solid tumour types.
Data availability
The data that support the findings of this study are not openly available but are available from the corresponding author upon reasonable request. Data are located in controlled access data storage at the University of Leeds.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209–49.
Cancer Research UK. Bowel Cancer Statistics 2024. 2024. Available from: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/bowel-cancer.
Nagtegaal ID, Odze RD, Klimstra D, Paradis V, Rugge M, Schirmacher P, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76:182–8.
Gray R, Barnwell J, McConkey C, Hills RK, Williams NS, Kerr DJ. Adjuvant chemotherapy versus observation in patients with colorectal cancer: a randomised study. Lancet. 2007;370:2020–9.
Brierley JD, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours (Eighth edition). Oxford, UK: Wiley-Blackwell, 2017.
Morris EJA, Maughan NJ, Forman D, Quirke P. Who to treat with adjuvant therapy in Dukes B/stage II colorectal cancer? The need for high quality pathology. Gut. 2007;56:1419–25.
Kim HS, Kim KM, Lee SB, Kim GR, Han YD, Cho MS, et al. Clinicopathological and biomolecular characteristics of stage IIB/IIC and stage IIIA colon cancer: Insight into the survival paradox. J Surgical Oncol. 2019;120:423–30.
Cervantes A, Adam R, Roselló S, Arnold D, Normanno N, Taïeb J, et al. Metastatic colorectal cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34:10–32.
Alexander PG, McMillan DC, Park JH. The local inflammatory response in colorectal cancer - Type, location or density? A systematic review and meta-analysis. Cancer Treat Rev. 2020;83:101949.
Jass J. Lymphocytic infiltration and survival in rectal cancer. J Clin Pathol. 1986;39:585–9.
Klintrup K, Mäkinen JM, Kauppila S, Väre PO, Melkko J, Tuominen H, et al. Inflammation and prognosis in colorectal cancer. Eur J Cancer. 2005;41:2645–54.
Graham DM, Appelman HD. Crohn’s-like lymphoid reaction and colorectal carcinoma: a potential histologic prognosticator. Mod Pathol J U S Can Acad Pathol Inc. 1990;3:332–5.
Alexander PG, McMillan DC, Park JH. The local inflammatory response in colorectal cancer–type, location or density? A systematic review and meta-analysis. Cancer Treat Rev. 2020;83:101949.
Hynes SO, Coleman HG, Kelly PJ, Irwin S, O’Neill RF, Gray RT, et al. Back to the future: routine morphological assessment of the tumour microenvironment is prognostic in stage II/III colon cancer in a large population‐based study. Histopathology. 2017;71:12–26.
Anitei MG, Zeitoun G, Mlecnik B, Marliot F, Haicheur N, Todosi AM, et al. Prognostic and predictive values of the immunoscore in patients with rectal cancer. Clin Cancer Res J Am Assoc Cancer Res. 2014;20:1891–9.
Pagès F, Mlecnik B, Marliot F, Bindea G, Ou F-S, Bifulco C, et al. International validation of the consensus Immunoscore for the classification of colon cancer: a prognostic and accuracy study. Lancet. 2018;391:2128–39.
Galon J, Lanzi A. Immunoscore and its introduction in clinical practice. Q J Nucl Med Mol Imaging. 2020;64:152–61.
Williams C, Gray RG, Shires M, Zhang L, Zhao Z, Bai I, et al. Artificial intelligence-assisted evaluation of tumor infiltrating CD3+ and CD8+ T cells for prognostication and prediction of benefit from adjuvant chemotherapy in early stage colorectal cancer (CRC): A retrospective analysis of the QUASAR trial. 2023;41:204.
Yasuda K, Nirei T, Sunami E, Nagawa H, Kitayama J. Density of CD4(+) and CD8(+) T lymphocytes in biopsy samples can be a predictor of pathological response to chemoradiotherapy (CRT) for rectal cancer. Radiat Oncol. 2011;6:49.
El Sissy C, Kirilovsky A, Lagorce Pagès C, Marliot F, Custers PA, Dizdarevic E, et al. International Validation of the Immunoscore Biopsy in Patients With Rectal Cancer Managed by a Watch-and-Wait Strategy. J Clin Oncol. 2024;42:70–80.
El Sissy C, Kirilovsky A, Van den Eynde M, Muşină AM, Anitei MG, Romero A, et al. A Diagnostic Biopsy-Adapted Immunoscore Predicts Response to Neoadjuvant Treatment and Selects Patients with Rectal Cancer Eligible for a Watch-and-Wait Strategy. Clin Cancer Res. 2020;26:5198–207.
Balkenhol MCA, Ciompi F, Świderska-Chadaj Ż, van de Loo R, Intezar M, Otte-Höller I, et al. Optimized tumour infiltrating lymphocyte assessment for triple negative breast cancer prognostics. Breast. 2021;56:78–87.
Choi S, Cho SI, Jung W, Lee T, Choi SJ, Song S, et al. Deep learning model improves tumor-infiltrating lymphocyte evaluation and therapeutic response prediction in breast cancer. npj Breast Cancer. 2023;9:71.
Liu DHW, Kim Y-W, Sefcovicova N, Laye JP, Hewitt LC, Irvine AF, et al. Tumour infiltrating lymphocytes and survival after adjuvant chemotherapy in patients with gastric cancer: post-hoc analysis of the CLASSIC trial. Br J Cancer. 2023;128:2318–25.
Acs B, Ahmed FS, Gupta S, Wong PF, Gartrell RD, Sarin Pradhan J, et al. An open source automated tumor infiltrating lymphocyte algorithm for prognosis in melanoma. Nat Commun. 2019;10:5440.
Mlecnik B, Bindea G, Angell HK, Maby P, Angelova M, Tougeron D, et al. Integrative Analyses of Colorectal Cancer Show Immunoscore Is a Stronger Predictor of Patient Survival Than Microsatellite Instability. Immunity. 2016;44:698–711.
Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pagès C, et al. Type, Density, and Location of Immune Cells Within Human Colorectal Tumors Predict Clinical Outcome. Science. 2006;313:1960–4.
West NP, Dattani M, McShane P, Hutchins G, Grabsch J, Mueller W, et al. The proportion of tumour cells is an independent predictor for survival in colorectal cancer patients. Br J Cancer. 2010;102:1519–23.
Huijbers A, Tollenaar R, v Pelt G, Zeestraten E, Dutton S, McConkey C, et al. The proportion of tumor-stroma as a strong prognosticator for stage II and III colon cancer patients: validation in the VICTOR trial. Ann Oncol. 2013;24:179–85.
Mesker WE, Junggeburt JM, Szuhai K, de Heer P, Morreau H, Tanke HJ, et al. The carcinoma-stromal ratio of colon carcinoma is an independent factor for survival compared to lymph node status and tumor stage. Cell Oncol. 2007;29:387–98.
Park JH, Richards CH, McMillan DC, Horgan PG, Roxburgh CSD. The relationship between tumour stroma percentage, the tumour microenvironment and survival in patients with primary operable colorectal cancer. Ann Oncol. 2014;25:644–51.
Hansen TF, Kjær-Frifeldt S, Lindebjerg J, Rafaelsen SR, Jensen LH, Jakobsen A, et al. Tumor-stroma ratio predicts recurrence in patients with colon cancer treated with neoadjuvant chemotherapy. Acta Oncologica. 2018;57:528–33.
van Pelt GW, Sandberg TP, Morreau H, Gelderblom H, van Krieken J, Tollenaar R, et al. The tumour-stroma ratio in colon cancer: the biological role and its prognostic impact. Histopathology. 2018;73:197–206.
Hutchins GGA, Treanor D, Wright A, Handley K, Magill L, Tinkler-Hundal E, et al. Intratumoral stromal morphometry predicts disease recurrence but not response to 5-fluorouracil-results from the QUASAR trial of colorectal cancer. Histopathology. 2018;72:391–404.
Yang J, Ye H, Fan X, Li Y, Wu X, Zhao M, et al. Artificial intelligence for quantifying immune infiltrates interacting with stroma in colorectal cancer. J Transl Med. 2022;20:451.
Zhao K, Li Z, Yao S, Wang Y, Wu X, Xu Z, et al. Artificial intelligence quantified tumour-stroma ratio is an independent predictor for overall survival in resectable colorectal cancer. eBioMedicine. 2020;61:103054.
Knight K, Bigley C, Pennel K, Hay J, Maka N, McMillan D, et al. The Glasgow Microenvironment Score: an exemplar of contemporary biomarker evolution in colorectal cancer. J Pathol Clin Res. 2024;10:e12385.
Sobin LH, Fleming ID. TNM Classification of Malignant Tumors, fifth edition (1997). Union Internationale Contre le Cancer and the American Joint Committee on Cancer. Cancer. 1997;80:1803–4.
Grabsch H, Dattani M, Barker L, Maughan N, Maude K, Hansen O, et al. Expression of DNA double-strand break repair proteins ATM and BRCA1 predicts survival in colorectal cancer. Clin Cancer Res J Am Assoc Cancer Res. 2006;12:1494–500.
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM. REporting recommendations for tumour MARKer prognostic studies (REMARK). Br J Cancer. 2005;93:387–91.
Ronneberger O, Fischer P, Brox T, editors. U-net: Convolutional networks for biomedical image segmentation. In: Medical Image Computing and Computer-Assisted Intervention–MICCAI 2015: 18th International Conference, Proceedings, Part III 18. Cham, Switzerland. Springer: 2015.
Royston P, Sauerbrei W. Two Techniques for Investigating Interactions between Treatment and Continuous Covariates in Clinical Trials. Stata J. 2009;9:230–51.
Courrech Staal EF, Smit VT, van Velthuysen ML, Spitzer-Naaykens JM, Wouters MW, Mesker WE, et al. Reproducibility and validation of tumour stroma ratio scoring on oesophageal adenocarcinoma biopsies. Eur J Cancer. 2011;47:375–82.
Fu M, Chen D, Luo F, Li M, Wang Y, Chen J, et al. Association of the tumour stroma percentage in the preoperative biopsies with lymph node metastasis in colorectal cancer. Br J Cancer. 2020;122:388–96.
Salgado R, Denkert C, Demaria S, Sirtaine N, Klauschen F, Pruneri G, et al. The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: recommendations by an International TILs Working Group 2014. Ann Oncol. 2015;26:259–71.
Zhao M, Yao S, Li Z, Wu L, Xu Z, Pan X, et al. The Crohn’s-like lymphoid reaction density: a new artificial intelligence quantified prognostic immune index in colon cancer. Cancer Immunol Immunother. 2022;71:1221–31.
Falk T, Mai D, Bensch R, Çiçek Ö, Abdulkadir A, Marrakchi Y, et al. U-Net: deep learning for cell counting, detection, and morphometry. Nat Methods. 2019;16:67–70.
Weiser MR. AJCC 8th Edition: Colorectal Cancer. Ann Surgical Oncol. 2018;25:1454–5.
Morton D, Seymour M, Magill L, Handley K, Glasbey J, Glimelius B, et al. Preoperative chemotherapy for operable colon cancer: mature results of an international randomized controlled trial. J Clin Oncol. 2023;41:1541–52.
Funding
AW is supported by a Cancer Research UK Clinical Research Training Fellowship (award reference S4154). BIW is supported by the UKRI Centre for Doctoral Training in Artificial Intelligence for Medical Diagnosis and Care (EP/S024336/1). NW and PQ are supported by a Yorkshire Cancer Research programme grant L386. NW, PQ, HIG, JL and DM are supported in part by the National Institute for Health and Care Research (NIHR) Leeds Biomedical Research Centre (BRC) (NIHR203331). The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care. NW is supported by a Cancer Research UK Radiation Research Network Infrastructure Award (RadPath) and would like to acknowledge Cancer Research UK funding for the Leeds Radiotherapy Research Centre of Excellence (RRCOER-Jun24/100004).
Author information
Authors and Affiliations
Contributions
NW, HIG, DRM and AW conceived and designed the study. HIG, NW, PQ and DM supervised the study. WM and HIG collected the images and provided the database. BIW and DM provided the TCD data. AW and NW annotated the WSIs and performed the pathological review. BIW, JL, AW and DRM set up the image analysis pipeline. AW performed the quality control. AW, BIW and DRM did the computational analysis. AW and BIW carried out the statistical analysis. All authors had full access to the study data, discussed and reviewed the manuscript, and approved the manuscript for publication. AW, BIW, DRM, JL, HIG, PQ, WM, and NW wrote and revised the manuscript.
Corresponding author
Ethics declarations
Competing interest
HG received honoraria from Astra Zeneca and Bristol Myers Squibb not related to the study. NW has undertaken paid consultancy for Bristol Myers Squibb, GSK, Astellas, Pfizer and Amgen not related to this study. PQ declares research funding from Roche and honoraria for lectures by Roche, Bayer, Amgen, not related to the study. DM is director and shareholder of HeteroGenius Limited. AW received honoraria from Servier not related to this study.
Ethics approval and consent to participate
Ethical approval for the study was provided by North East – York Research Ethics Committee (ref. 08/H0903/62). The study was performed in accordance with local ethics regulation, the need for individual patient consent was waived by the ethics committee. The study was conducted in compliance with the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Westwood, A.C., Wilson, B.I., Laye, J. et al. Deep-learning enabled combined measurement of tumour cell density and tumour infiltrating lymphocyte density as a prognostic biomarker in colorectal cancer. BJC Rep 3, 12 (2025). https://doi.org/10.1038/s44276-025-00123-8
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44276-025-00123-8