Abstract
This scoping review examines the use of artificial intelligence (AI) in sexual and reproductive health (SRH) to guide future research and policymaking. We conducted searches across four electronic databases in October 2023. We included primary research studies applying AI on any SRH topic, regardless of language and timeframe. Our search retrieved 12,823 articles, of which abstracts and full texts were independently screened by two reviewers, yielding 2666 studies for final analysis. The predominant health topics were maternal health (44.9%; n = 1198); reproductive cancers (29.2%; n = 779), mainly cervical cancer; and infertility and fertility care (12.6%; n = 337). AI tools were primarily used to facilitate screening and diagnosis (74.2%, n = 1980), support treatment and care management (8.7%, n = 233), and understand health trends (8.6%, n = 230). While substantial progress has been made in certain domains, significant gaps remain in geographic representation, methodological rigor, and deployment studies with external validity.
Similar content being viewed by others
Introduction
Artificial intelligence (AI) refers to creating algorithms, processes, and computer programmes that can perform tasks and exhibit behaviours, such as learning, making decisions and predictions, without every step in the process being explicitly programmed by a human. The use of AI in health has traditionally focused on processing of large volumes of data, such as medical images and structured datasets1,2,3,4. Recent advancements in generative AI and natural language processinghave extended this computing ability to manage and interpret unstructured text data, such as those found in chatbots, clinical notes, and social media posts5,6 Such applications, combined with the growth in use of mobile devices, have stimulated interest in harnessing AI for broader health needs7,8,9,10,11,12,13.
The application of AI in sexual and reproductive health (SRH) represents an emerging field with potential to enhance healthcare delivery and access14,15,16,17,18,19,20,21,22. Health domains within SRH encompass topics such as contraception/family planning, infertility and fertility care, maternal health, sexually transmitted infections (STIs) including HIV, comprehensive abortion care, sexual health, and gender-based violence, as derived from the World Health Organization compendium of interventions for advancing universal health coverage23. These health topics are also ones often faced by populations in vulnerable situations or subjected to stigma24. As such, AI could be critical to expanding SRH access to populations underserved by traditional health service delivery mechanisms, but it could also put individuals in vulnerable situations at greater risk depending on how the AI is developed, regulated, and deployed.
Despite the growing body of literature on the use of AI in SRH, existing work either focuses on specific health domains, application types (e.g., chatbots, AI-assisted medical devices)17 or are exploratory assessments on the potential opportunities14,15,24. While this provides a valuable starting point, it also underscores the need for a systematic effort to organize the available evidence across this field. This scoping review examines the landscape of how AI is concretely being applied in SRH and encompasses a wide range of SRH topics, with no limitations on geographical coverage, allowing for an extensive and comprehensive overview of the field. By synthesizing data from a wide array of studies, this review seeks to provide a foundational understanding to inform research gaps and guide policymaking to ensure the effectiveness and equitable use of AI across SRH.
Results
Study characteristics
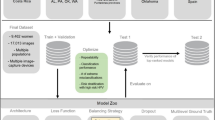
Our search retrieved a total of 12,823 citations. After the removal of 196 duplicates, both manually and through Covidence, 12,627 unique articles remained for title and abstract screening. Of these, 2666 studies were included for data extraction after fulfilling the inclusion criteria in the full-text screening (Fig. 1).
PRISMA flow diagram.
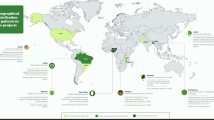
The highest proportion of studies was published in 2022 (n = 631, 23.7%), with the majority (n = 1628, 61.1%) published since 2021, while the oldest ones dating back to 198925,26. The majority of studies were conducted in high-income countries (n = 1344, 49.9%), followed by upper-middle-income countries (n = 964, 35.9%). The largest contribution of studies came from China (n = 662), followed by the United States (n = 523), India (n = 181), and the United Kingdom (n = 107) (Fig. 2).
Darker shades of blue represent greater number of studies.
The majority of included articles were validation studies (n = 1704, 63.9%), which described the process of developing AI models and testing their performance using subsets of the same dataset against a reference, such as standard diagnostic devices, physician decisions, or other AI models applied to the same dataset27,28,29,30. Observational studies accounted for 31.7% (n = 845) of the total and focused on applying AI models, mainly machine learning, from retrospective data for descriptive and inferential analyses, such as identifying linkages between environmental exposures and SRH outcomes or identifying patterns and risk factors31,32,33,34,35,36. A small portion of studies (n = 59, 2.2%) were sentiment analysis, using natural language processing (NLP) to extract text from social media platforms like Twitter (now X) to explore attitudes towards SRH topics, such as HPV vaccination, abortion37,38,39,40,41,42,43. Experimental studies (n = 43, 1.6%), in which there was an introduction of an AI intervention to different participant groups, were relatively limited and focused on evaluating the use of chatbots on topics, such as fertility support and preconception care to the intervention group, while the control group received either standard care or no intervention44,45. Qualitative studies (n = 13, 0.4%) used methods such as focus group discussions, interviews, user testing, and participatory design approaches to understand the feasibility, acceptability, and user experience of AI tools across different SRH domains, including contraception, HIV prevention, and SRH education46,47,48. Economic evaluations (n = 2, 0.08%) used AI to estimate cost inputs and evaluate the cost-effectiveness of SRH interventions, such in vitro fertilization procedures and HIV prevention programmes49,50.
Health domains and AI intended purpose
Maternal health emerged as the most represented SRH area, comprising 44.9% (n = 1198) of studies. Within this health domain, AI demonstrated diverse applications, with its predominant use being for screening purposes (n = 954, 80%), followed by understanding health trends (n = 96, 8%). This included predicting maternal complications (n = 392, 32.7%)51,52,53,54, such as postpartum haemorrhage, gestational diabetes, preeclampsia, as well as foetal and neonatal complications (n = 323, 26.9%)55,56,57, such as foetal growth restriction and spina bifida. AI was also employed in routine antenatal care (ANC) for monitoring maternal and foetal biometric and physiological measurements, such as gestational age and foetal weight58,59,60. During labour, AI applications extended to predicting the mode of delivery and supporting physicians through clinical decision support systems (CDSS)61,62,63. Machine learning was also commonly applied to understand health trends by analysing implications of maternal behavioural and environmental exposures, such as nutrition, smoking, and pollutant exposure, and their associations with pregnancy and birth outcomes64,65,66,67. For postnatal care, AI was used to evaluate mental health outcomes, particularly postpartum depression, and other mental health disorders68,69,70,71,72,73,74. These studies utilized various data sources, including electronic medical records (EMRs), research repositories with clinical parameters, and social media posts, to assess risk and provide early intervention68,69,70,71,72,73,74. AI modelling techniques were also used in clinical research and drug discovery in 5% (n = 62) of maternal health studies, to identify drug toxicity and biomarkers for pregnancy and foetal conditions75,76,77,78 (Fig. 3).
Darker shades of blue represent higher concentration of studies, such as screening and diagnosis for maternal health and reproductive organ cancers, whereas lighter shades of yellow indicate fewer numbers of studies.
Cervical cancer, as a specific gynaecological/reproductive cancer, represented 27.7% (n = 741) of all the studies, in which the primary intended purpose was screening and diagnosis (n = 612, 82.5%), followed by treatment and care management (n = 88, 11.8%)79,80,81,82. Other types of reproductive organ cancers, such as uterine, prostate, and ovarian cancers, comprised 1.4% (n = 38) of all studies. Many of the investigations utilized ultrasound and MRI data to develop diagnostic models capable of distinguishing benign from malignant masses and performing image segmentation for more precise diagnoses83,84. Across all reproductive organ cancers (n = 779, 29.2%), the primary outcomes focused on risk assessment85, cancer detection86, and disease prognosis87,88. Additionally, AI was used in radiation therapy and brachytherapy to enhance treatment precision by targeting affected areas, dynamically adjusting dosages, and projecting patient prognosis for clinical decision-making80,89 (Fig. 3).
In infertility and fertility care (n = 337, 12.6%), AI applications were prominent in supporting various steps of assisted reproductive technology, with AI being utilized in both laboratory set-ups and clinical care settings90,91,92,93,94,95,96,97,98. These ranged from the early stages of embryo selection, where machine learning techniques were employed to evaluate embryo quality and viability99,100, to predicting assisted reproductive technology outcomes by training models on selected clinical and laboratory features101,102. Assessment of fertility parameters, encompassing investigations of biological factors such as hormonal levels, reproductive organ health, formed the largest subset of outcomes (n = 182, 54%)98,103,104,105. Technologies such as computer-assisted sperm analysis, which traditionally automate sperm motility and morphology measurement, are now being augmented with AI models, such as You Only Look Once (YOLO). These AI-powered approaches enable advanced object detection and are applied to evaluate sperm parameters with greater speed and accuracy90,91. The primary data sources for training and applying the AI models were diagnostic images (n = 92, 27.2%) and laboratory specimens (n = 66, 19.5%).
The application of AI in HIV and other STIs encompassed 259 studies (9.7%), focusing primarily on risk assessment and early detection (n = 86, 33.2%) within key populations106,107, such as men who have sex with men as well as on predicting HIV-related complications (n = 105, 40.5%)108,109. In the context of HIV care and treatment using antiretroviral therapy (n = 25, 9.6%), AI models were developed for drug monitoring and toxicology predictions. Data sources for the HIV studies predominantly included EMRs with clinical patient-level information (n = 75, 28.9%), along with research repositories (n = 41, 15.8%), and survey data (n = 40, 15.4%).
Fewer studies were identified on other SRH domains, including intimate partner and sexual violence, sexual health, contraception, and abortion. For intimate partner and sexual violence (n = 23, 0.8%), AI was used primarily for online data mining (n = 12, 52.1%) to perform risk assessments and detection110,111, identify gender-based violence (GBV) narratives112, and evaluate legislation related to domestic violence113 as a way to better understand reporting trends and attitudes across online GBV discussions. In contraception and family planning (n = 22, 0.8%): AI was applied to gain insights into fertility and family planning usage on a broad scale. Studies utilized population-level surveys113,114 to capture overarching trends and patterns, while online data were employed to gauge public attitudes and perceptions of various contraceptive methods. The use of AI in sexual health (n = 12, 0.4%) was mainly through chatbots to provide health information115,116, as well as through machine learning models that mined social media data to analyse youth communication patterns and trends in sexual health discussions and risk behaviours117. For menopause (n = 9, 0.3%), AI was applied to forecast conditions associated with pre-menopausal and postmenopausal stages, including osteoporosis and endometrial alterations118,119,120. Additionally, AI was leveraged to assess healthcare behaviours and address the physiological requirements of menopausal women121. In abortion (n = 6, 0.2%), AI was employed to examine public discourse about abortion on social media platforms such as X (formerly Twitter)122,123,124, as well as using natural language processing to categorize abortion bills’ wording as restrictive or protective40.
Target end-users and populations of interest
Healthcare providers (n = 1792, 67.2%) emerged as the primary target end-users where AI was used to facilitate screening and diagnosis, primarily by identifying risk factors, or assist in care provision such as treatment options for HIV and in vitro fertilization125,126,127,128,129,130,131,132,133,134,135,136. Researchers (n = 628, 23.5%) were the second-largest end-users, where they used AI to analyse health trends using research repositories, social media data137,138, and to advance clinical and pharmacological research139,140,141. Health service users/clients (n = 146, 5.4%) were direct beneficiaries of AI-driven tools, such as conversational agents (i.e., chatbots) for accessing tailored health information115,142,143. Additionally, sensors and wearables were used for personal health monitoring, including tracking ovulation and monitoring foetal health at home144,145,146. The underlying populations within the health service users include women (women of reproductive age, postpartum, menopausal, and postmenopausal, overall n = 1106, 41.5%), pregnant women (n = 852, 32%)%), foetus (n = 251, 9.4%), people living with HIV (n = 172, 6.5%), and men (mainly involved in fertility care = 152, 5.7%), where semen samples often served as the unit of analysis147,148.
AI model development and lifecycle
The main data sources consisted of images derived from diagnostic devices, such as ultrasounds and MRI (n = 928, 34.8%); EMRs (n = 519, 19.5%); laboratory reports, including genomic and molecular data (n = 368, 13.8%); research repositories and registries for secondary data analysis (n = 335, 12.6%); signal data (n = 172, 6.5%) obtained through foetal electrocardiograms (ECG), cardiotocography (CTG), photoplethysmography (PPG), and sensors; and surveys (n = 154, 5.8%). Laboratory data included the examination of blood plasma for various biomarkers, semen analysis for fertility assessments, and amniotic fluid evaluation. The remaining data was derived from audio recordings, mobile applications collecting behavioural or user-reported data, such as fertility monitoring, self-reported questionnaire responses, regulatory authorities, and social media posts41,149,150,151. The data types used were varied, ranging from structured clinical data, such as patient records and diagnostic results, to unstructured data, including free-text online content and other narrative data, which were analysed to capture user experiences, emotions, and sentiment.
Actual model deployment demonstrating real-world applications was observed in 6.4% (n = 171) of all publications, such as in the introduction of conversational agents, understanding of population-level trends, or use in clinical environments for screening and clinical decision support systems152,153,154,155. The majority of studies were in the model evaluation stage (n = 1946, 72.9%). These were often conducted as validation studies to assess the performance of algorithms prior to deployment or observational studies to determine if machine learning models could accurately make predictions based on existing datasets92,156,157. Such studies employed training, validation, and test sets to develop and evaluate their AI models, utilizing performance metrics such as precision, recall, accuracy, and other relevant indicators to ensure reliability. However, external validation, which involves testing the models on entirely independent datasets from different sources, is not explicitly described in most of the studies. The next largest category comprised of studies in the earlier stage of model development (n = 549, 20.5%), with the primary objective of designing and training AI models or identifying key predictive features158,159,160,161,162,163.
Comparators often included traditional diagnostic methods (e.g., physician decisions, laboratory tests, or imaging modalities) and other AI models, providing benchmarks to evaluate improvements in efficiency and accuracy. The outcomes varied by study, with AI models frequently demonstrating superior performance in areas like predictive accuracy and diagnostic efficiency, though some studies highlighted challenges, such as overfitting or limited generalizability due to small sample sizes or lack of external validation. One study reported that AI performed slightly worse than the traditional predictive model against which it was compared156. We did not compare the performance of AI models across studies due to the heterogeneity in SRH domains, the nature of AI applications, and variations in data sources, model designs, and evaluation methods, which made cross-study comparisons unfeasible.
Discussion
The findings of this scoping review highlight the diversity of ways AI is used and studied in SRH, providing a comprehensive foundation to spur further analysis, research prioritization, and policy development. While AI was used across all country income categories, most studies were concentrated in high- and upper-middle-income countries, underscoring the uneven uptake in settings where SRH challenges are often most acute. A contributing factor of this imbalance is the greater availability of datasets from research registries and academic institutions in high-income settings, partly due to the more advanced digital infrastructure and greater availability of structured datasets for AI modelling13. Our analysis, which captured both the origin of datasets and their deployment, revealed a trend of ad hoc machine learning analyses on convenient datasets, drawn primarily from academic institutions in upper-income settings, often without a clear pathway toward real-world implementation. For example, several studies with authors in low- and middle-income countries leveraged datasets from research registries and academic institutions in high-income countries to develop and test AI models, highlighting the need for datasets that can enable the use of AI in local contexts. However, some studies also explicitly aimed to address this disparity by validating AI tools for use in rural or low-resource settings, such as exploring the use of image analysis on smartphones to offer affordable diagnostic devices, including ultrasounds and HIV viral load detection164,165,166,167,168,169,170,171,172,173.
Generally, in lower and lower-middle-income countries, AI applications were largely geared towards prediction tools addressing health system needs such as contraceptive service use, comprehensive HIV prevention and treatment, and maternal risk assessment174,175,176,177. In contrast, the use of AI in high-income settings extends to include advanced biomedical discovery and analyses that leverage imaging, molecular, and other complex datasets across multiple disciplines (e.g., environment and health)178,179,180. China, an upper-middle-income country with the highest number of studies, demonstrated a mixed pattern, applying AI both to conventional maternal health risk prediction and more advanced diagnostic applications using image analysis and unsupervised learning181,182.
Across all settings, the review identified a strong emphasis on AI for screening and diagnosis, particularly in maternal health and cervical cancer. This may reflect the suitability of traditional AI approaches, such as risk prediction and classification, as well as the availability of diverse data sources, including images from ultrasounds, signals from ECGs and sensors, and electronic medical records1,183,184. These findings align with other analyses highlighting the potential of AI for diagnostic tools and personalized treatment14,185. This review also identified less documented uses of AI in SRH, such as for drug discovery, understanding people’s views on highly debated topics, including the HPV vaccine, abortion, contraception, and conducting epidemiological analysis for correlations between exposures and SRH outcomes.
With the majority of studies in the model development and evaluation stages, experimental studies of deployed AI tools and economic evaluations were notably scarce within the reviewed literature, restricting insights into their real-world applications and scalability. These findings resonate with observations in the broader field of AI in health, where experimentation is rapidly growing, yet real-world AI implementation in clinical practices is not widespread5. This absence of external validation further limits the generalizability of AI models to broader populations and diverse settings, and potentially yield tools that are not vetted for use outside of research settings or in new contexts. In addition, the heterogeneity and lack of transparency in reporting data sources further undermined the robustness and scalability of some of the identified studies. With the development of frameworks such as SPIRIT-AI and CONSORT-AI, reporting should become more standardized and could be expanded to guide authors on key ethical considerations for reporting186,187. For example, although this review sought to extract data on adverse effects and ethical considerations, studies did not report these systematically or explicitly. Considering that SRH is often politicized and the use of AI in this field is fraught with concerns to uphold privacy, bodily autonomy, and rights14,24, a deeper analysis of the ethical implications of included studies can advance understanding of the overall use of AI in sexual and reproductive health and rights (SRHR).
This is one of the first scoping reviews to comprehensively map the literature of AI in SRH. One of the key strengths of this review is the extensive body of evidence and literature synthesized, spanning 2666 studies across all SRH domains and geographic locations. The review adhered to rigorous methodological standards, including registration of the protocol before starting the overview, we designed a sensitive search strategy that was run in several electronic databases without date or language restrictions. It also serves as a foundation for conducting sub-analyses across SRH domains and AI functionalities, while also providing a taxonomy for the classification of AI studies that can be broadened for use beyond SRH. Furthermore, the technical consultation of AI and SRH experts enriched the development of key themes and patterns in the formulation of the underlying taxonomy and data charting process22.
Despite its strengths, this review has several limitations. The heterogeneity in study designs and volume of studies provided valuable breadth, though it limited the ability to draw uniform conclusions on effect, which requires more extensive in-depth analyses across subgroups. The comprehensive inclusion criteria were consistent with scoping review methodology; however, future research efforts, particularly systematic reviews or meta-analyses, will need to differentiate across levels of study quality and maturity. In addition, the exclusion of HIV genomic and drug discovery may underestimate the level of AI investments and research in HIV. Lastly, the rapidly evolving pace of AI research and the extent of studies captured requires more efficient ways of continuously synthesizing the literature, including by leveraging AI to expedite updates to this review.
This structured synthesis distils the concrete uses of AI in SRH, highlights key patterns, and pinpoints critical areas for future research and impact. While substantial progress has been made in certain domains, significant gaps remain in geographic representation, methodological rigor, and deployment studies with external validity. Addressing these challenges requires a concerted effort to prioritize rigorous validation, equity, and sustainability for advancing the use of AI for SRH impact and fostering health equity and rights.
Methods
Identifying the research questions
The review was based on a published protocol and conducted using the established methodological framework by Arksey and O’Malley188,189. The PRISMA-ScR (Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews) format was used to report the findings (Supplemental Note 1).
The primary research question for this scoping review was to identify the characteristics of AI systems and tools being applied to SRH, in terms of health domains, intended purpose (e.g., screening, counselling, understanding health trends, health promotion and counselling), geographic scope, target users, implementation maturity, data sources, and outcomes of interest189. SRH domains were derived from the World Health Organization (WHO) Universal Health Coverage (UHC) Compendium and comprise maternal and perinatal health, contraception and family planning, infertility and fertility care, sexual health, female genital mutilation, intimate partner and sexual violence, reproductive cancers, sexually transmitted infections, including HIV23. Within HIV, genomic studies were excluded from this review due to their complexity and narrower focus on genomic research rather than the broader sexual and reproductive health dimensions of HIV; HIV studies focused on service delivery and access to care were included. There was no restriction on the language of the study, and where needed, articles were translated into English using Google Translate. The full version of the search strategy can be found in Supplemental Note 2.
Identifying relevant studies
We conducted searches across four electronic databases: MEDLINE (PubMed), Scopus, Web of Science, and CINAHL, covering all available studies up to October 2023. No start date was imposed for the search, allowing for the inclusion of all available studies regardless of their publication date. Table 1 outlines the inclusion and exclusion criteria for the studies. We included all primary research studies reporting on AI applications to aspects of SRH, provided they had a clearly described methodology, encompassing quantitative studies, qualitative studies, as well as programme evaluations and descriptions.
Study selection
A web-based tool for article screening and data extraction, Covidence (https://www.covidence.org/), was used to manage the screening and data extraction process (Table 1). Titles and abstracts were double screened across six independent reviewers (A.F., A.P.B., S.M., M.B., C.M. and T.T.), and any conflicts were resolved through discussion with screeners or by a third author. Articles that qualified for full-text review were independently double-screened across nine independent reviewers (S.M., T.T., M.A., G.M.P., G.M., S.T.N., R.W., J.D. and G.D.). No AI tools were employed at any stage of this review, including during the screening and data extraction processes.
Charting the data
A standardized chart for data extraction was developed consisting of the following data for each study: title, author, year, geographical location, study design, methodology, objectives, sample size, setting, SRH domain, AI intended purpose, target population, participant type, AI lifecycle, source of data, type of dataset, outcome, comparator, and any adverse effects or ethical considerations. Three authors (S.M., S.P. and T.T.) manually extracted data from all full-text articles and discussed the emergent responses. Twenty percent of the extracted data was rechecked (TT) for quality assurance and standardization (Table 2).
For the study design, we categorized studies into the following categories:
-
Validation studies, defined as studies that compare the accuracy of a measure with a gold standard or reference measure190.
-
Observational studies, which applied AI to analyse large datasets and draw inferences on the effects of an “exposure” or intervention191. These studies often leveraged surveys, case-control, and cohort designs and applied machine learning, a subset of AI, which focuses on the use of statistical and mathematical modelling techniques to define and analyse data192.
-
Experimental studies, which aim to assess the effects of an AI intervention that has been intentionally introduced on an outcome of interest (e.g., randomized controlled trials and quasi-experimental studies)193.
-
Qualitative studies, often conducted through interviews and focus group discussions, to explore the acceptability and feasibility of AI systems and tools.
-
Content and sentiment analysis, which includes the use of natural language processing to systematically identify, extract, quantify, and examine patterns of information194,195. Sentiment analysis is a subset of content analysis in which the focus is on mining data to explore attitudes, beliefs, and opinions194,195. Although these studies may also qualify as using qualitative methods, we included this as a separate category due to the specific way AI and natural language processing are applied to analyse unstructured text.
The intended AI purpose was based on a predefined classification developed by WHO through a consultative process used to develop a technical brief on the role of AI in sexual and reproductive health and rights (SRHR)22. Building off existing digital health classification frameworks196, the target population described the intended end-users of the AI tools across the following categories: clients/health service users, healthcare workers, health system managers responsible for oversight at facility levels, researchers, and policy makers. The participant type described the population of interest or research subjects, such as foetuses, pregnant women, adolescents, people living with HIV, women of reproductive age, men or women engaging with SRH services.
Collating, summarizing, and reporting the results
The extracted data were systematically organized across several key categories using tables to summarize observed patterns and accompanied by a narrative synthesis. In addition, we used the World Bank country classification for 2024197 to consolidate countries/geographic locations into country income groups.
We applied the AI lifecycle framework198 to classify and analyse the maturity of studies. The framework outlines five stages: data creation, data acquisition, model development, model evaluation, and model deployment, emphasizing a continuous process from data collection, pre-processing, to the training, validation, and application of AI models in real-world contexts198. The following definitions were used to delineate the stages across the AI lifecycle198:
-
Model development: Studies that primarily describe the process of formulating the algorithms and building an AI model. These studies may also include preliminary testing to validate the AI models, but the core focus of the results is the developmental process.
-
Model evaluation: Studies that focus on testing the performance and efficacy of AI models within a controlled research setting. These studies may include the AI model development process as part of the methodology, but the core focus of the results is the AI model’s performance.
-
Model deployment: Studies that introduce the use of AI models in real-world implementations and able to draw conclusions that extend beyond the performance of the AI model.
Data sources and types of datasets used in the studies were summarized to evaluate the nature and origin of the data underpinning the AI models. The study outcomes were categorized to highlight the range of specific areas covered under each SRH domain. Considering the lack of a taxonomy on AI data sources, we applied an iterative process to develop categorizations based on the first 100 extracted studies and refined over the course of the data extraction process.
Consulting stakeholders
We consulted stakeholders from the WHO technical expert group on AI for SRHR for feedback on classifying the functional purpose of the AI system and tools. This technical expert was convened at the inception of the scoping review to inform the development of a technical brief that would leverage findings from this scoping review. The early inputs from this consultative process were used to refine the research questions and data extraction needs.
Data availability
The authors declare that data generated and analysed during the current research are included in this article. Additional data available based on request to the corresponding author. The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of WHO concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. Dotted and dashed lines on maps represent approximate border lines for which there may not yet be full agreement.
References
Okeibunor, J. C. et al. The use of artificial intelligence for delivery of essential health services across WHO regions: a scoping review. Front. Public Health 11, 1102185 (2023).
World Health Organization. Regulatory Considerations on Artificial Intelligence for Health (World Health Organization, 2023).
Hernström, V. et al. Screening performance and characteristics of breast cancer detected in the Mammography Screening with Artificial Intelligence trial (MASAI): a randomised, controlled, parallel-group, non-inferiority, single-blinded, screening accuracy study. Lancet Digit. Health. https://doi.org/10.1016/S2589-7500(24)00267-X (2025).
Kim, Y. H. Artificial intelligence in medical ultrasonography: driving on an unpaved road. Ultrasonography 40, 313–317 (2021).
Esmaeilzadeh, P. Challenges and strategies for wide-scale artificial intelligence (AI) deployment in healthcare practices: a perspective for healthcare organizations. Artif. Intell. Med. 151, 102861 (2024).
Locke, S. et al. Natural language processing in medicine: a review. Trends Anaesth. Crit. Care 38, 4–9 (2021).
Pujari, S. et al. Artificial intelligence for global health: cautious optimism with safeguards. Bull. World Health Organ. 101, 364–364a (2023).
Rajpurkar, P., Chen, E., Banerjee, O. & Topol, E. J. AI in health and medicine. Nat. Med. 28, 31–38 (2022).
Bauer, G. R. & Lizotte, D. J. Artificial intelligence, intersectionality, and the future of public health. Am. J. Public Health 111, 98–100 (2021).
Benke, K. & Benke, G. Artificial intelligence and big data in public health. Int J. Environ. Res Public Health 15, 1–9 (2018).
Bhatt, P., Liu, J., Gong, Y., Wang, J. & Guo, Y. Emerging artificial intelligence-empowered mhealth: scoping review. JMIR Mhealth Uhealth 10, e35053 (2022).
Young, S. D., Crowley, J. S. & Vermund, S. H. Artificial intelligence and sexual health in the USA. Lancet Digit. Health 3, e467–e468 (2021).
Saxena, A. K., Ness, S. & Khinvasara, T. The influence of AI: the revolutionary effects of artificial intelligence in healthcare sector. J. Eng. Res. Rep. 26, 49–62 (2024).
Gbagbo, F. Y., Ameyaw, E. K. & Yaya, S. Artificial intelligence and sexual reproductive health and rights: a technological leap towards achieving sustainable development goal target 3.7. Reprod. Health 21, 196 (2024).
Abou Chawareb, E. et al. Sexual health in the era of artificial intelligence: a review of the literature. J. Sex. Med. 21, 267–279 (2024).
Wasson, E. J., Driver, K., Hughes, M. & Bailey, J. Sexual reproductive health chatbots: should we be so quick to throw artificial intelligence out with the bathwater?. BMJ Sex. Reprod. Health 47, 73 (2021).
Mills, R., Mangone, E. R., Lesh, N., Mohan, D. & Baraitser, P. Chatbots to improve sexual and reproductive health: realist synthesis. J. Med. Internet. Res. 25, e46761 (2023).
Delanerolle, G. et al. Artificial intelligence: a rapid case for advancement in the personalization of Gynaecology/Obstetric and Mental Health care. Women’s Health 17, 1–2 (2021).
Wang, R. et al. Artificial intelligence in reproductive medicine. Reproduction 158, R139–R154 (2019).
Rolfes, V. et al. Artificial intelligence in reproductive medicine—an ethical perspective. Geburtshilfe Frauenheilkd. 83, 106–115 (2023).
Buslón, N., Cortés, A., Catuara-Solarz, S., Cirillo, D. & Rementeria, M. J. Raising awareness of sex and gender bias in artificial intelligence and health. Front Glob. Women’s Health 4, 970312 (2023).
World Health Organization. The Role of Artificial Intelligence in Sexual and Reproductive Health and Rights: Technical Brief (World Health Organization, 2024).
World Health Organization. Sexual and Reproductive Health Interventions in the WHO UHC Compendium (World Health Organization, 2021).
Khosla, R., Mishra, V. & Singh, S. Sexual and reproductive health and rights and bodily autonomy in a digital world. Sex. Reprod. Health Matters 31, 2269003 (2023).
Chan, S. Y. et al. Computer-assisted image analysis of sperm concentration in human semen before and after swim-up separation: comparison with assessment by haemocytometer. Int. J. Androl. 12, 339–345 (1989).
Bassil, H. E. & Dripps, J. H. Real time processing and analysis of fetal phonocardiographic signals. Clin. Phys. Physiol. Meas. 10, 67–74 (1989).
Hu, H., Wang, H., Bai, Y. & Liu, M. Determination of endometrial carcinoma with gene expression based on optimized Elman neural network. Appl. Math. Comput. 341, 204–214 (2019).
Boudet, S., Houzé de l’Aulnoit, A., Peyrodie, L., Demailly, R. & Houzé de l’Aulnoit, D. Use of deep learning to detect the maternal heart rate and false signals on fetal heart rate recordings. Biosensors. https://doi.org/10.3390/bios12090691 (2022).
Bano, S. et al. FetNet: a recurrent convolutional network for occlusion identification in fetoscopic videos. Int. J. Comput. Assist. Radio. Surg. 15, 791–801 (2020).
Potluri, V. et al. An inexpensive smartphone-based device for point-of-care ovulation testing. Lab Chip 19, 59–67 (2018).
Orel, E. et al. Prediction of HIV status based on socio-behavioural characteristics in East and Southern Africa. PLoS ONE 17, e0264429 (2022).
Fox, H., Topp, S. M., Lindsay, D. & Callander, E. A cascade of interventions: a classification tree analysis of the determinants of primary cesareans in Australian public hospitals. Birth 48, 209–220 (2021).
Betts, K. S., Kisely, S. & Alati, R. Predicting common maternal postpartum complications: leveraging health administrative data and machine learning. Bjog 126, 702–709 (2019).
Khanam, R. et al. Performance of a validated spontaneous preterm delivery predictor in South Asian and Sub-Saharan African women: a nested case control study. J. Matern. Fetal Neonatal Med. 35, 8878–8886 (2022).
Jehan, F. et al. Multiomics characterization of preterm birth in low- and middle-income countries. JAMA Netw. Open 3, e2029655 (2020).
Hossain, M. I. et al. Performance evaluation of machine learning algorithm for classification of unintended pregnancy among married women in Bangladesh. J. Healthc. Eng. 2022, 1460908 (2022).
Boucher, J. C. et al. HPV vaccine narratives on Twitter during the COVID-19 pandemic: a social network, thematic, and sentiment analysis. BMC Public Health 23, 694 (2023).
Du, J., Xu, J., Song, H. Y. & Tao, C. Leveraging machine learning-based approaches to assess human papillomavirus vaccination sentiment trends with Twitter data. BMC Med. Inf. Decis. Mak. 17, 69 (2017).
Luo, X., Zimet, G. & Shah, S. A natural language processing framework to analyse the opinions on HPV vaccination reflected in Twitter over 10 years (2008–2017). Hum. Vaccin Immunother. 15, 1496–1504 (2019).
Valdez, D. & Goodson, P. Neutral or framed? A sentiment analysis of 2019 abortion laws. Sex. Res. Soc. Policy 19, 936–945 (2022).
Fatima, I. et al. Prediction of postpartum depression using machine learning techniques from social media text. Expert Syst. 36, e12409 (2019).
Chivers, B. R. et al. Perinatal distress during COVID-19: thematic analysis of an online parenting forum. J. Med. Internet. Res. 22, e22002 (2020).
Ravaldi, C., Mosconi, L., Bonaiuti, R. & Vannacci, A. The emotional landscape of pregnancy and postpartum during the COVID-19 pandemic in Italy: a mixed-method analysis using artificial intelligence. J. Clin. Med. 12, 6140 (2023).
Gardiner, P. et al. Using health information technology to engage African American women on nutrition and supplement use during the preconception period. Front Endocrinol. 11, 571705 (2020).
Jack, B. et al. Reducing Preconception Risks Among African American Women with Conversational Agent Technology. J. Am. Board Fam. Med. 28, 441–451 (2015).
Bonnevie, E. et al. Layla’s Got You: developing a tailored contraception chatbot for Black and Hispanic young women. Health Educ. J. 80, 413–424 (2021).
Peng, M. L. et al. Formative evaluation of the acceptance of HIV prevention artificial intelligence chatbots by men who have sex with men in Malaysia: focus group study. JMIR Form. Res. 6, e42055 (2022).
Wang, H. et al. An artificial intelligence chatbot for young people’s sexual and reproductive health in India (SnehAI): instrumental case study. J. Med. Internet. Res. 24, e29969 (2022).
Fragoulakis, V. et al. Follitropin Alpha for assisted reproduction: an analysis based on a non-interventional study in Greece. Curr. Med. Res. Opin. 38, 2227–2235 (2022).
Zang, X. et al. Prioritizing additional data collection to reduce decision uncertainty in the HIV/AIDS response in 6 US cities: a value of information analysis. Value Health 23, 1534–1542 (2020).
Chen, Y. et al. Machine-learning predictive model of pregnancy-induced hypertension in the first trimester. Hypertension Res. 46, 2135–2144 (2023).
Cubillos, G. et al. Development of machine learning models to predict gestational diabetes risk in the first half of pregnancy. BMC Pregnancy Childbirth 23, 469 (2023).
Liu, J. et al. Machine learning-based prediction of postpartum hemorrhage after vaginal delivery: combining bleeding high risk factors and uterine contraction curve. Arch. Gynecol. Obstet. 306, 1015–1025 (2022).
Zhu, H. et al. A computerized diagnostic model for automatically evaluating placenta accrete spectrum disorders based on the combined MR radiomics-clinical signatures. Sci. Rep. 12, 10130 (2022).
Chen, L., Tian, Y., Deng, Y. & Abdulhay, E. Neural network algorithm-based three-dimensional ultrasound evaluation in the diagnosis of fetal spina bifida. Sci. Program. 2021, 3605739 (2021).
Gong, Y. et al. Fetal congenital heart disease echocardiogram screening based on DGACNN: adversarial one-class classification combined with video transfer learning. IEEE Trans. Med. Imaging 39, 1206–1222 (2020).
Song, F. et al. Predicting the risk of fetal growth restriction by radiomics analysis of the placenta on T2WI: a retrospective case-control study. Placenta 134, 15–22 (2023).
Miyagi, Y. & Miyake, T. Potential of artificial intelligence for estimating Japanese fetal weights. Acta Med Okayama. https://doi.org/10.18926/AMO/61207 (2020).
Veerabhadrappa, S. T. & Vyas, A. L. Analysis and classification of three trimesters during normal pregnancy using bispectrum. IETE J. Res. 68, 2697–2706 (2022).
Xiao, Y., Lu, Y., Liu, M., Zeng, R. & Bai, J. A deep feature fusion network for fetal state assessment. Front Physiol. 13, 969052 (2022).
De Ramón Fernández, A., Ruiz Fernández, D. & Prieto Sánchez, M. T. Prediction of the mode of delivery using artificial intelligence algorithms. Comput. Methods Prog. Biomed. 219, 106740 (2022).
Guedalia, J. et al. Transporting an artificial intelligence model to predict emergency cesarean delivery: overcoming challenges posed by interfacility variation. J. Med. Internet. Res. 23, e28120 (2021).
Yu, S., Tan, K. K., Sng, B. L., Li, S. & Sia, A. T. H. Lumbar ultrasound image feature extraction and classification with support vector machine. Ultrasound Med. Biol. 41, 2677–2689 (2015).
Bodnar, L. M. et al. Machine learning as a strategy to account for dietary synergy: an illustration based on dietary intake and adverse pregnancy outcomes. Am. J. Clin. Nutr. 111, 1235–1243 (2020).
Fang, H., Johnson, C., Stopp, C. & Espy, K. A. A new look at quantifying tobacco exposure during pregnancy using fuzzy clustering. Neurotoxicol. Teratol. 33, 155–165 (2011).
Li, Q. et al. Effect of airborne particulate matter of 2.5μm or less on preterm birth: a national birth cohort study in China. Environ. Int. 121, 1128–1136 (2018).
Roh, M. E. et al. Association between indoor residual spraying and pregnancy outcomes: a quasi-experimental study from Uganda. Int. J. Epidemiol. 51, 1489–1501 (2022).
Amit, G. et al. Estimation of postpartum depression risk from electronic health records using machine learning. BMC Pregnancy Childbirth 21, 630 (2021).
Betts, K. S., Kisely, S. & Alati, R. Predicting postpartum psychiatric admission using a machine learning approach. J. Psychiatr. Res. 130, 35–40 (2020).
Fischbein, R., Cook, H. L., Baughman, K. & Díaz, S. R. Using machine learning to predict help-seeking among 2016–2018 Pregnancy Risk Assessment Monitoring System participants with postpartum depression symptoms. Women’s. Health 18, 17455057221139664 (2022).
Gopalakrishnan, A., Venkataraman, R., Gururajan, R., Zhou, X. & Zhu, G. Predicting women with postpartum depression symptoms using machine learning techniques. Mathematics 10, 4570 (2022).
Khapre, S., Stewart, R. & Taylor, C. An evaluation of symptom domains in the 2 years before pregnancy as predictors of relapse in the perinatal period in women with severe mental illness. Eur. Psychiatry 64, e26 (2021).
Shatte, A. B. R., Hutchinson, D. M., Fuller-Tyszkiewicz, M. & Teague, S. J. Social media markers to identify fathers at risk of postpartum depression: a machine learning approach. Cyberpsychol. Behav. Soc. Netw. 23, 611–618 (2020).
Zhang, Y., Wang, S., Hermann, A., Joly, R. & Pathak, J. Development and validation of a machine learning algorithm for predicting the risk of postpartum depression among pregnant women. J. Affect. Disord. 279, 1–8 (2021).
Lu, Q. et al. Longitudinal metabolomics integrated with machine learning identifies novel biomarkers of gestational diabetes mellitus. Free Radic. Biol. Med. 209, 9–17 (2023).
Eguchi, A., Sakurai, K., Watanabe, M. & Mori, C. Exploration of potential biomarkers and related biological pathways for PCB exposure in maternal and cord serum: a pilot birth cohort study in Chiba, Japan. Environ. Int. 102, 157–164 (2017).
Demailly, R., Escolano, S., Haramburu, F., Tubert-Bitter, P. & Ahmed, I. Identifying drugs inducing prematurity by mining claims data with high-dimensional confounder score strategies. Drug Saf. 43, 549–559 (2020).
Chen, S. et al. Association of the LEP gene with immune infiltration as a diagnostic biomarker in preeclampsia. Front. Mol. Biosci. https://doi.org/10.3389/fmolb.2023.1209144 (2023).
Jihong, C. et al. Automated intensity modulated radiation therapy treatment planning for cervical cancer based on convolution neural network. Technol. Cancer Res. Treat. 19, 1533033820957002 (2020).
Ecker, S. et al. Neural network-assisted automated image registration for MRI-guided adaptive brachytherapy in cervical cancer. Z. Med. Phys. 32, 488–499 (2022).
Chauhan, N. K., Singh, K., Kumar, A. & Kolambakar, S. B. HDFCN: a robust hybrid deep network based on feature concatenation for cervical cancer diagnosis on WSI pap smear slides. BioMed. Res. Int. 2023, 4214817 (2023).
Bhuvaneshwari, K. & Poornima, B. Cervical cancer cell identification & detection using fuzzy C mean and K nearest neighbor techniques. Int. J. Innov. Technol. Explor. Eng. 8, 1080–1084 (2019).
Lefebvre, T. L. et al. Development and validation of multiparametric MRI-based radiomics models for preoperative risk stratification of endometrial cancer. Radiology 305, 375–386 (2022).
Moro, F. et al. Developing and validating ultrasound-based radiomics models for predicting high-risk endometrial cancer. Ultrasound Obstet. Gynecol. 60, 256–268 (2022).
Ahmed, M. R., Rehana, H. & Asaduzzaman, S. Ovarian cancer substantial risk factor analysis by machine learning: a low incoming country perspective. Biointerface Res. Appl. Chem. 11, 8457–8466 (2020).
Amarsee, K. et al. Automatic detection and tracking of marker seeds implanted in prostate cancer patients using a deep learning algorithm. J. Med. Phys. 46, 80–87 (2021).
Grimley, P. M. et al. A prognostic system for epithelial ovarian carcinomas using machine learning. Acta Obstet. Gynecol. Scand. 100, 1511–1519 (2021).
Paik, E. S. et al. Prediction of survival outcomes in patients with epithelial ovarian cancer using machine learning methods. J. Gynecol. Oncol. 30, e65 (2019).
Ling, Y., Zhang, W., Li, Z., Pu, X. & Ren, Y. Application and comparison of several machine learning methods in the prognosis of cervical cancer. Eur. J. Gynaecol. Oncol. 43, 34–44 (2022).
Maurya, S. et al. Morphological analysis of metabolically dysregulated spermatozoa using Artificial Intelligence based approach. J. Integr. Sci. Technol. 11, 569–569 (2023).
Aristoteles, A., Syarif, A., Sutyarso, S. & Lumbanraja, F. R. Identification of human sperm based on morphology using the you only look once version 4 algorithm. Int. J. Adv. Comput. Sci. Appl. 13, 424–431 (2022).
Hassan, M. R., Al-Insaif, S., Hossain, M. I. & Kamruzzaman, J. A machine learning approach for prediction of pregnancy outcome following IVF treatment. Neural Comput. Appl. 32, 2283–2297 (2020).
Amini, P. et al. Factors associated with in vitro fertilization live birth outcome: a comparison of different classification methods. Int. J. Fertil. Steril. 15, 128 (2021).
Huang, B. et al. Using deep learning to predict the outcome of live birth from more than 10,000 embryo data. BMC Pregnancy Childbirth 22, 36 (2022).
Uyar, A., Bener, A. & Ciray, H. N. Predictive modeling of implantation outcome in an in vitro fertilization setting: an application of machine learning methods. Med. Decis. Mak. 35, 714–725 (2015).
Wang, R. et al. AI-Based Optimal Treatment Strategy Selection for Female Infertility for First and Subsequent IVF-ET Cycles. J. Med. Syst. 47, 87 (2023).
Mehrjerd, A., Rezaei, H., Eslami, S., Ratna, M. B. & Khadem Ghaebi, N. Internal validation and comparison of predictive models to determine success rate of infertility treatments: a retrospective study of 2485 cycles. Sci. Rep. 12, 7216 (2022).
Targosz, A., Przystałka, P., Wiaderkiewicz, R. & Mrugacz, G. Semantic segmentation of human oocyte images using deep neural networks. Biomed. Eng. OnLine 20, 40 (2021).
Bormann, C. L. et al. Consistency and objectivity of automated embryo assessments using deep neural networks. Fertil. Steril. 113, 781–787.e781 (2020).
Dirvanauskas, D., Maskeliunas, R., Raudonis, V. & Damasevicius, R. Embryo development stage prediction algorithm for automated time lapse incubators. Comput. Methods Prog. Biomed. 177, 161–174 (2019).
Wald, M. et al. Computational models for prediction of IVF/ICSI outcomes with surgically retrieved spermatozoa. Reprod. Biomed. Online 11, 325–331 (2005).
Tian, T. et al. A Bayesian network model for prediction of low or failed fertilization in assisted reproductive technology based on a large clinical real-world data. Reprod. Biol. Endocrinol. 21, 8 (2023).
Hicks, S. A. et al. Machine learning-based analysis of sperm videos and participant data for male fertility prediction. Sci. Rep. 9, 16770 (2019).
Li, M. et al. Evaluation of endometrial receptivity by ultrasound elastography to predict pregnancy outcome is a non-invasive and worthwhile method. Biotechnol. Genet Eng. Rev. 40, 284–298 (2024).
Koesoema Wijaya, R. A., Kusumaatmaja, A. & Rizal, D. M. Novel method to classify varicocele using electronic nose. Indones. J. Electr. Eng. Comput. Sci. 28, 165–173 (2022).
Mukherjee, G., Zhang, C., Kandaswamy, S., Gooding, H. & Orenstein, E. Current inpatient screening practices for sexual history and STIs: an opportunity to seize. Clin. Pediatr. 63, 350–356 (2024).
Wray, T. B. et al. Using smartphone survey data and machine learning to identify situational and contextual risk factors for HIV risk behavior among men who have sex with men who are not on PrEP. Prev. Sci. 20, 904–913 (2019).
Parra-Rodriguez, L. et al. Common antiretroviral combinations are associated with somatic depressive symptoms in women with HIV. AIDS 38, 167–176 (2024).
Yang, X. et al. Utilizing electronic health record data to understand comorbidity burden among people living with HIV: a machine learning approach. AIDS 35, S39–S51 (2021).
González-Prieto, Á, Brú, A., Nuño, J. C. & González-Álvarez, J. L. Hybrid machine learning methods for risk assessment in gender-based crime. Knowl. Based Syst. 260, 110130 (2023).
McDougal, L. et al. Opening closed doors: using machine learning to explore factors associated with marital sexual violence in a cross-sectional study from India. BMJ Open 11, e053603 (2021).
Silva, M. et al. Gender-based violence narratives in internet-based conversations in Nigeria: social listening study. J. Med. Internet. Res. 25, e46814 (2023).
Orts Llopis, M. Á. Las leyes sobre la vioLencia de género y doméstica en España y Reino Unido y la emoción: un estUdio léxico deL discUrso jUrídico desde eL anáLisis deL sentimiento. Rev. Llen. Dret 71, 171–192 (2019).
Udomboso, C. G. & Amoateng, A. Modelling trends in contraception usage in Nigeria and Ghana. J. Health Manag. 20, 277–290 (2018).
Liew, T. W., Tan, S.-M., Yoo, N. E., Gan, C. L. & Lee, Y. Y. Let’s talk about Sex!: AI and relational factors in the adoption of a chatbot conveying sexual and reproductive health information. Comput. Hum. Behav. Rep. 11, 100323 (2023).
Nadarzynski, T., Bayley, J., Llewellyn, C., Kidsley, S. & Graham, C. A. Acceptability of artificial intelligence (AI)-enabled chatbots, video consultations and live webchats as online platforms for sexual health advice. BMJ Sex. Reprod. Health 46, 210–217 (2020).
Stevens, R. et al. On sex, drugs, and alcohol: a mixed-method analysis of youth posts on social media in the United States. J. Child. Media 16, 514–531 (2022).
Selskyy, P., Sverstiuk, A., Slyva, A. & Selskyi, B. Prediction of the progression of endometrial hyperplasia in women of premenopausal and menopausal age based on an analysis of clinical and anamnestic indicators using multiparametric neural network clustering. Fam. Med. Prim. Care Rev. 25, 184–189 (2023).
Thawnashom, K., Pornsawad, P. & Makond, B. Machine learning’s performance in classifying postmenopausal osteoporosis Thai patients. Intell. Based Med. 7, 100099 (2023).
Zeitlin, J., Parides, M. K., Lane, J. M., Russell, L. A. & Kunze, K. N. A clinical prediction model for 10-year risk of self-reported osteoporosis diagnosis in pre-and perimenopausal women. Arch. Osteoporos. 18, 78 (2023).
Chao, Q. et al. Menopausal women’s health care method based on computer nursing diagnosis intelligent system. J. Healthc. Eng. 2021, 4963361 (2021).
Swanson, K. et al. Effect of recent abortion legislation on Twitter user engagement, sentiment, and expressions of trust in clinicians and privacy of health information: content analysis. J. Med. Internet. Res. 25, e46655 (2023).
Ujah, O. I., Olaore, P., Nnorom, O. C., Ogbu, C. E. & Kirby, R. S. Examining ethno-racial attitudes of the public in Twitter discourses related to the United States Supreme Court Dobbs vs. Jackson Women’s Health Organization ruling: a machine learning approach. Front. Global Women’s Health. https://doi.org/10.3389/fgwh.2023.1149441 (2023).
Valdez, D., Jozkowski, K. N., Montenegro, M. S., Crawford, B. L. & Jackson, F. Identifying accurate pro-choice and pro-life identity labels in Spanish: social media insights and implications for comparative survey research. Perspect. Sex. Reprod. Health 54, 166–176 (2022).
Alabi, O. et al. Robust fetoscopic mosaicking from deep learned flow fields. Int. J. Comput. Assist. Radiol. Surg. 17, 1125–1134 (2022).
Płotka, S. S. et al. Deep learning for estimation of fetal weight throughout the pregnancy from fetal abdominal ultrasound. Am. J. Obstetr. Gynecol. MFM. https://doi.org/10.1016/j.ajogmf.2023.101182 (2023).
Shahzad, S. et al. Sperm abnormality detection using sequential deep neural network. Mathematics 11, 515 (2023).
Xia, T.-H. et al. Establish a normal fetal lung gestational age grading model and explore the potential value of deep learning algorithms in fetal lung maturity evaluation. Chin. Med. J. 134, 1828–1837 (2021).
Yeo, L. & Romero, R. Fetal Intelligent Navigation Echocardiography (FINE): a novel method for rapid, simple, and automatic examination of the fetal heart. Ultrasound Obstet. Gynecol. 42, 268–284 (2013).
Hariton, E. et al. A machine learning algorithm can optimize the day of trigger to improve in vitro fertilization outcomes. Fertil. Steril. 116, 1227–1235 (2021).
Letterie, G. & Mac Donald, A. Artificial intelligence in in vitro fertilization: a computer decision support system for day-to-day management of ovarian stimulation during in vitro fertilization. Fertil. Steril. 114, 1026–1031 (2020).
Mirroshandel, S. A., Ghasemian, F. & Monji-Azad, S. Applying data mining techniques for increasing implantation rate by selecting best sperms for intra-cytoplasmic sperm injection treatment. Comput. Methods Prog. Biomed. 137, 215–229 (2016).
Wang, Y. et al. Task model-specific operator skill assessment in routine fetal ultrasound scanning. Int. J. Comput. Assist. Radiol. Surg. 17, 1437–1444 (2022).
Guh, R.-S., Wu, T.-C. J. & Weng, S.-P. Integrating genetic algorithm and decision tree learning for assistance in predicting in vitro fertilization outcomes. Expert Syst. Appl. 38, 4437–4449 (2011).
Lu, H., Hirst, J., Yang, J., Mackillop, L. & Clifton, D. Standardising the assessment of caesarean birth using an Oxford caesarean prediction score for mothers with gestational diabetes. Health. Technol. Lett. 9, 1–8 (2022).
Ekpenyong, M. E., Etebong, P. I. & Jackson, T. C. Fuzzy-multidimensional deep learning for efficient prediction of patient response to antiretroviral therapy. Heliyon 5, e02080 (2019).
Bulka, C. M. et al. Arsenic in private well water and birth outcomes in the United States. Environ. Int. 163, 107176 (2022).
Klein, A. Z., Sarker, A., Weissenbacher, D. & Gonzalez-Hernandez, G. Towards scaling Twitter for digital epidemiology of birth defects. NPJ Digit. Med. 2, 96 (2019).
Ailes, E. C. et al. Using supervised learning methods to develop a list of prescription medications of greatest concern during pregnancy. Matern. Child Health J. 24, 901–910 (2020).
Ghasemi, G. & Nemati-Rashtehroodi, A. QSAR modeling of benzimidazole derivatives as potent inhibitors of trichomoniasis/QSAR modellemesi ile Benzimidazole türevlerinin trikomoniasis için etkili inhibitörler olarak kullanılması. Turkish J. Biochem. 40, 492–499 (2015).
Kang, L. et al. Structure-activity relationship (SAR) model for predicting teratogenic risk of antiseizure medications in pregnancy by using support vector machine. Front Pharm. 13, 747935 (2022).
Bickmore, T., Zhang, Z., Reichert, M., Julce, C. & Jack, B. Promotion of preconception care among adolescents and young adults by conversational agent. J. Adolesc. Health 67, S45–S51 (2020).
Chervenak, J., Lieman, H., Blanco-Breindel, M. & Jindal, S. The promise and peril of using a large language model to obtain clinical information: ChatGPT performs strongly as a fertility counseling tool with limitations. Fertil. Steril. 120, 575–583 (2023).
Qin, M., Xu, Y., Liang, Y. & Sun, T. A wearable fetal movement detection system for pregnant women. Front. Med. 10, 1160373 (2023).
Jennings, V., Haile, L. T., Simmons, R. G., Spieler, J. & Shattuck, D. Perfect-and typical-use effectiveness of the Dot fertility app over 13 cycles: results from a prospective contraceptive effectiveness trial. Eur. J. Contracept. Reprod. Health Care 24, 148–153 (2019).
Sexual and reproductive health interventions in the WHO UHC Compendium https://www.who.int/publications/i/item/9789240022867.
Haugen, T. B. et al. Sperm motility assessed by deep convolutional neural networks into WHO categories. Sci. Rep. 13, 14777 (2023).
Gunderson, S. J. et al. Machine-learning algorithm incorporating capacitated sperm intracellular pH predicts conventional in vitro fertilization success in normospermic patients. Fertil. Steril. 115, 930–939 (2021).
Merz, A. A. et al. Population attitudes toward contraceptive methods over time on a social media platform. Am. J. Obstet. Gynecol. 224, 597.e591–597.e514 (2021).
Du, J. et al. Use of deep learning to analyze social media discussions about the human papillomavirus vaccine. JAMA Netw. Open 3, e2022025 (2020).
Movaghar, A., Mailick, M., Sterling, A., Greenberg, J. & Saha, K. Automated screening for Fragile X premutation carriers based on linguistic and cognitive computational phenotypes. Sci. Rep. 7, 2674 (2017).
Shara, N. et al. Early identification of maternal cardiovascular risk through sourcing and preparing electronic health record data: machine learning study. JMIR Med. Inform. 10, e34932 (2022).
Klein, A. Z. & Gonzalez-Hernandez, G. An annotated data set for identifying women reporting adverse pregnancy outcomes on Twitter. Data Brief. 32, 106249 (2020).
Rigla, M., Martínez-Sarriegui, I., García-Sáez, G., Pons, B. & Hernando, M. E. Gestational diabetes management using smart mobile telemedicine. J. Diab. Sci. Technol. 12, 260–264 (2018).
Kim, T. The impact of working hours on pregnancy intention in childbearing-age women in Korea, the country with the world’s lowest fertility rate. PLoS ONE 18, e0288697 (2023).
Day, T. G. et al. Prenatal diagnosis of hypoplastic left heart syndrome on ultrasound using artificial intelligence: how does performance compare to a current screening programme?. Prenat. Diagn. 44, 717–724 (2024).
Wang, F., Mao, R., Yan, L., Ling, S. & Cai, Z. A deep learning-based approach for rectus abdominis segmentation and distance measurement in ultrasonography. Front Physiol. 14, 1246994 (2023).
Marcus, J. L. et al. Use of electronic health record data and machine learning to identify candidates for HIV pre-exposure prophylaxis: a modelling study. Lancet HIV 6, e688–e695 (2019).
MacDowell, M. et al. Understanding birthing mode decision making using artificial neural networks. Med Decis. Mak. 21, 433–443 (2001).
Zhao, M. et al. Identification and immuno-infiltration analysis of cuproptosis regulators in human spermatogenic dysfunction. Front. Genet. 14, 1115669 (2023).
Candemir, C. Estimating the semen quality from life style using fuzzy radial basis functions. Int. J. Mach. Learn. Comput. 8, 44–48 (2018).
Fraiwan, L. et al. Time frequency analysis for automated sleep stage identification in fullterm and preterm neonates. J. Med. Syst. 35, 693–702 (2011).
Sadda, P. et al. Deep-learned placental vessel segmentation for intraoperative video enhancement in fetoscopic surgery. Int. J. Comput. Assist. Radiol. Surg. 14, 227–235 (2019).
Stockman, J., Friedman, J., Sundberg, J., Harris, E. & Bailey, L. Predictive analytics using machine learning to identify ART clients at health system level at greatest risk of treatment interruption in Mozambique and Nigeria. J. Acquir. Immune Defic. Syndr. 90, 154–160 (2022).
Das, R. et al. Performance analysis of machine learning algorithms and screening formulae for β-thalassemia trait screening of Indian antenatal women. Int. J. Med. Inf. 167, 104866 (2022).
Katebi, N., Sameni, R., Rohloff, P. & Clifford, G. D. Hierarchical attentive network for gestational age estimation in low-resource settings. IEEE J. Biomed. Health Inf. 27, 2501–2511 (2023).
Kulkarni, S. S., Katebi, N., Valderrama, C. E., Rohloff, P. & Clifford, G. D. CNN-based LCD transcription of blood pressure from a mobile phone camera. Front Artif. Intell. 4, 543176 (2021).
Schilpzand, M. et al. Automatic placenta localization from ultrasound imaging in a resource-limited setting using a predefined ultrasound acquisition protocol and deep learning. Ultrasound Med. Biol. 48, 663–674 (2022).
Simplified models to assess newborn gestational age in low-middle income countries: findings from a multicountry, prospective cohort study. BMJ Glob Health. https://doi.org/10.1136/bmjgh-2021-005688 (2021).
Revell, A. D. et al. An update to the HIV-TRePS system: the development of new computational models that do not require a genotype to predict HIV treatment outcomes. J. Antimicrob. Chemother. 69, 1104–1110 (2014).
Gomes, R. G. et al. A mobile-optimized artificial intelligence system for gestational age and fetal malpresentation assessment. Commun. Med. 2, 128 (2022).
Revell, A. D. et al. 2021 update to HIV-TRePS: a highly flexible and accurate system for the prediction of treatment response from incomplete baseline information in different healthcare settings. J. Antimicrob. Chemother. 76, 1898–1906 (2021).
Holmström, O. et al. Point-of-care digital cytology with artificial intelligence for cervical cancer screening in a resource-limited setting. JAMA Netw. Open 4, e211740 (2021).
Jaganath, D. et al. Evaluation of multi-antigen serological screening for active tuberculosis among people living with HIV. PLoS ONE 15, e0234130 (2020).
Kebede, S. D. et al. Prediction of contraceptive discontinuation among reproductive-age women in Ethiopia using Ethiopian Demographic and Health Survey 2016 Dataset: a Machine Learning Approach. BMC Med. Inform. Decis. Mak. 23, 9 (2023).
Togunwa, T. O., Babatunde, A. O. & Abdullah, K. -u-R. Deep hybrid model for maternal health risk classification in pregnancy: synergy of ANN and random forest. Front. Artif. Intell. 6, 1213436 (2023).
Rittenhouse, K. J. et al. Improving preterm newborn identification in low-resource settings with machine learning. PLoS ONE 14, e0198919 (2019).
Bahado-Singh, R. et al. Cell-free DNA in maternal blood and artificial intelligence: accurate prenatal detection of fetal congenital heart defects. Am. J. Obstet. Gynecol. 228, 76.e71–76.e10 (2023).
Dabi, Y. et al. Endometriosis-associated infertility diagnosis based on saliva microRNA signatures. Reprod. Biomed. online 46, 138–149 (2023).
Sivasankaran, S. & Jonnalagadda, S. Levonorgestrel loaded biodegradable microparticles for injectable contraception: preparation, characterization and modelling of drug release. Int. J. Pharmaceut. 624, 121994 (2022).
Fan, Z. et al. CAM-VT: A weakly supervised cervical cancer nest image identification approach using conjugated attention mechanism and visual transformer. Comput. Biol. Med. 162, 107070 (2023).
Lin, Q. & Fang, Z.-J. Establishment and evaluation of a risk prediction model for gestational diabetes mellitus. World J. Diab. 14, 1541 (2023).
Alowais, S. A. et al. Revolutionizing healthcare: the role of artificial intelligence in clinical practice. BMC Med. Educ. 23, 689 (2023).
Russell, S. J. Artificial Intelligence: A Modern Approach. Third edition edn (Prentice Hall, 2010).
Masilamani, V. et al. Leveraging artificial intelligence for advancements in reproductive health. Afr. J. Reprod. Health 28, 216–217 (2024).
Cruz Rivera, S., Liu, X., Chan, A. W., Denniston, A. K. & Calvert, M. J. Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension. Lancet Digit. Health 2, e549–e560 (2020).
Liu, X., Cruz Rivera, S., Moher, D., Calvert, M. J. & Denniston, A. K. Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension. Nat. Med. 26, 1364–1374 (2020).
Arksey, H. & O’Malley, L. Scoping studies: towards a methodological framework. Int. J. Soc. Res. Methodol. 8, 19–32 (2005).
Tamrat, T. et al. Exploring the use and implications of AI in sexual and reproductive health and rights: protocol for a scoping review. JMIR Res. Protoc. 13, e53888 (2024).
Fox, M. P., Lash, T. L. & Bodnar, L. M. Common misconceptions about validation studies. Int J. Epidemiol. 49, 1392–1396 (2020).
Carlson, M. D. & Morrison, R. S. Study design, precision, and validity in observational studies. J. Palliat. Med 12, 77–82 (2009).
Rubinger, L., Gazendam, A., Ekhtiari, S. & Bhandari, M. Machine learning and artificial intelligence in research and healthcare. Injury 54, S69–S73 (2023).
World Health Organization. Monitoring and Evaluating Digital Health Interventions: A Practical Guide to Conducting Research and Assessment (World Health Organization, 2016).
Zhang, C., Fan, C., Yao, W., Hu, X. & Mostafavi, A. Social media for intelligent public information and warning in disasters: an interdisciplinary review. Int. J. Inf. Manag. 49, 190–207 (2019).
Barbounaki, S. G., Gourounti, K. & Sarantaki, A. Advances of sentiment analysis applications in obstetrics/gynecology and midwifery. Mater. Sociomed. 33, 225–230 (2021).
World Health Organization. Classification of Digital Interventions, Services and Applications in Health: A Shared Language to Describe the Uses of Digital Technology for Health, 2nd edn (World Health Organization, 2023).
Metreau, E., Young, K. & Eapen, S. World Bank country classifications by income level for 2024–2025. World Bank Blogs. https://blogs.worldbank.org/en/opendata/world-bank-country-classifications-by-income-level-for-2024-2025 (2024).
Ng, M. Y., Kapur, S., Blizinsky, K. D. & Hernandez-Boussard, T. The AI life cycle: a holistic approach to creating ethical AI for health decisions. Nat. Med. 28, 2247–2249 (2022).
Acknowledgements
We thank Chellan Kumarasamy, Denise Schalet, and Daniel Belay for exchanging experiences and guidance during earlier stages of the review; Camila Volij for support on data extraction; Rebecca Carlson and Kavita Kothari for serving as the information specialists for refining and conducting the search strategy. We are grateful to participants from the WHO technical consultation on AI and SRHR for their insights in advancing this area of work. This work was funded by the UNDP-UNFPA-UNICEF-WHO-World Bank Special Programme of Research, Development and Research Training in Human Reproduction (HRP), a cosponsored programme executed by the World Health Organization.
Author information
Authors and Affiliations
Contributions
Conceptualization: T.T., L.S., Y.Z. and S.P.; methodology development: T.T., A.P.B., S.M., Y.Z. and L.S.; screening: S.M., T.T., A.P.B., A.F., G.M., M.B.A., R.W., M.B., C.M., G.M.P., S.T.N., J.D. and G.T.; data extraction: S.M., S.P. and T.T.; analysis: S.M., T.T., A.P.B. and S.P.; drafting: S.M. and T.T.; editing and review: S.M., T.T., A.P.B., S.P., A.F., G.M., M.B.A., R.W., M.B., C.M., G.M.P., S.T.N., S.A., J.D., Y.Z., G.T., R.M., M.A., S.D., K.K., R.S., S.A., G.P., S.P. and L.S.; funding acquisition: L.S. All authors read and agreed to the published version of the manuscript. The authors alone are responsible for the views expressed in this article and they do not necessarily represent the views, decisions or policies of the institutions with which they are affiliated.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mengistu, S., Tamrat, T., Betran, AP. et al. The use of artificial intelligence in sexual and reproductive health: a comprehensive scoping review. npj Womens Health 3, 70 (2025). https://doi.org/10.1038/s44294-025-00118-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44294-025-00118-3