Abstract
Cement production accounts for 8% of global CO2 emissions, necessitating its deep decarbonization. This paper reviews: (i) electrolysis-based methods to produce cement precursors, and (ii) electrified process heat technologies, along with heat storage approaches. We highlight scaled-up calciner and kiln designs and the associated technoeconomic factors. Addressing renewable energy intermittency, and the need for grid upgrades and strategic infrastructure investments are critical to enabling the transition to low-carbon cement manufacturing.
Similar content being viewed by others
Introduction
Rising global temperatures, intensifying hurricanes, severe floods, prolonged droughts, and rampant wildfires—all are devastating manifestations of climate change. In the 20th century, the global average number of natural disasters per year remained below 100; however, since the dawn of the 21st century, this frequency has escalated dramatically—now reaching ~400 events annually1. This significant increase in natural disasters is closely linked to climate change2,3, driven predominantly by greenhouse gas (GHG) emissions—90% of which are CO2 emissions4,5,6. These emissions result, primarily, from the burning of fossil fuels such as coal, oil, and natural gas for energy, transportation, and industrial processes7,8,9. In response to this crisis, decarbonization programs and policies have garnered substantial attention as essential mechanisms to promote—or, where necessary, mandate—the adoption of low-carbon technologies. These initiatives aim to foster sustainable growth while mitigating the long-term impacts of climate change.
Concrete is the dominant construction material in the world by a distance and, arguably, the most important man-made material. The most critical manufactured ingredient that endows concrete with its properties is Portland cement (PC), 4.5 billion tons of which is produced annually10 to cater for buildings and infrastructure construction. The carbon footprint of cement production (1.57 billion tons of CO2 emissions in 202311) is staggering; to put this in perspective, if cement production were a country, it would rank as the fourth-largest emitter of CO2 globally surpassed only by China, the U.S, and India11. The world’s population is projected to grow to 10 billion by 205012; with 60% of the people expected to live in cities by 203013, coupled with an increasing number of climate refugees displaced by extreme events14, dramatic urban expansion is anticipated. This will necessitate the construction of new infrastructure as well as the repair, rehabilitation, and revitalization of aging structures. Consequently, global production of PC is projected to increase by over 30% by 205015,16.
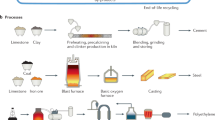
The fundamentals of PC manufacturing have not changed significantly over the many decades of its existence. Limestone excavated from the earth is ground and mixed with other source materials including quartz and clay; then the mixture is calcined using fossil fuels in a long rotating kiln at high temperatures. The resulting hard clinker is cooled, crushed, mixed with gypsum (and, sometimes, with a small amount of limestone), and ground into PC. Under current manufacturing practices, PC alone contributes to ~8% of all anthropogenic CO2 emissions, representing more than 25% of emissions from the industrial sector17,18. The major sources of CO2 emissions from PC manufacturing18,19,20 are (Fig. 1a, b): (1) ~40–50% due to calcination, where CaCO3—the primary precursor for PC production—is heated until it decomposes into CaO and CO2; (2) ~ 40% from the combustion of fuels (out of which ~82% is still fossil fuels, mostly coal and pet coke21) essential for generating the high temperatures (~1500 °C) needed for PC production; and (3) ~10% from product cooling, milling, and transportation. PC manufacturing is also highly energy intensive, requiring ~3.6 GJ of energy per ton21, supplied almost exclusively by fossil fuels. In terms of total energy use, cement manufacturing accounts for two-thirds of the total energy use in the production of non-metallic materials. The growing demand for PC, coupled with the challenge of meeting this demand without increasing CO2 emissions, underscores the urgent need for transformative steps toward more sustainable PC production.
a The major sources of CO2 emissions from PC manufacturing. Exhibit from “Laying the foundation for zero-carbon cement”, May 2020, McKinsey & Company, www.mckinsey.com. Copyright (c) 2024 McKinsey & Company. Reprinted by permission; and b percentage emissions breakdown for each step of cement manufacture based on refs. 105,251 and Fig. 1a.
As shown in Fig. 1b, ~85% of CO2 emissions from cement production is due to inherent process emissions from calcination and the heat required to attain calcination and clinkering temperatures, which cannot be decarbonized by external effects such as green grid, and will need more challenging measures. This does not consider decarbonization of electricity currently needed by the sector, which is considered an easier-to-attain goal. Thus, getting to a net-zero emissions scenario will require novel decarbonization strategies for the major share of the emissions, many of which do not exist yet at scale. Hence, cement industry is categorized as a difficult-to-decarbonize sector22,23. The barriers to deep decarbonization of cement stem from the immense volume of material produced within a capital-intensive framework—one characterized by slim profit margins; absence of short-term economic incentives for adopting changes; limited availability of cost-effective alternatives to PC; the century-long familiarity with PC among users and decision-makers; and a pervasive inertia within the construction sector, where there is hesitancy to experiment with materials not yet incorporated into established standards, specifications, and codes. However, as per the Paris Agreement24, GHG emissions need to fall by around half by 2050 to keep global warming to well below 2 °C above preindustrial levels.
There are four plausible pathways to enable deep decarbonization of cement. They include energy efficiency enhancements in cement manufacturing25, substitution of fossil fuels with carbon-efficient energy sources26, enhanced clinker substitution27 (i.e., reducing the share of cement content in concrete by blending with pozzolans, fine limestone, calcined clays, etc.), and a suite of innovative, high-risk/high-reward technologies. Figure 2 shows that achieving the Paris Agreement target of <2 °C by 2050 relies primarily on innovative technologies that current research efforts are converging onto. Energy efficiency enhancements and current fuel switching approaches (e.g., use of biomass, municipal solid waste, natural gas, etc.) make only minor dents in direct CO2 emissions28. Clinker substitution is practiced extensively in the industry, primarily through the use of supplementary cementitious materials (SCMs) that include both inert (e.g., quartz and limestone)29,30,31,32 and reactive variants (e.g., slag, silica fume, fly ash, and calcined clays such as metakaolin)33,34,35,36 that enhance the strength and durability of concrete. The use of municipal solid waste ash and other solid wastes (e.g., mine tailings, red mud, recycled concrete aggregates and fines) as SCMs are also being explored to reduce clinker content37,38,39. The common, specification-compliant clinker substitution materials like fly ash and slag are byproducts of other CO2-intensive industries and are becoming scarcer. As an example, there is a 30% demand-supply gap for fly ash in the U.S., owing to the reduced reliance on coal for electric power40. The adoption of non-conventional SCMs in concrete is also limited by environmental regulations, particularly due to the presence of toxic compounds and heavy metals. Recent studies41,42,43,44 have also investigated low-carbon, Ca-containing materials (e.g., wollastonite and recycled concrete aggregates and fines) that could replace limestone, either partially or entirely, in the PC manufacturing process. Although the use of alternative raw materials may fall short of achieving full carbon neutrality compared to alternative fuels or electrification, it provides a notable advantage by requiring fewer infrastructure upgrades. This entails less capital investment and allows for a quicker transition, rendering it a more practical option for reducing carbon emissions in the short to medium term.
The innovative high-risk approaches include novel synthesis routes for PC production including electrochemical approaches45, use of carbon-efficient raw materials41, and carbon capture, utilization, and storage (CCUS) methods. Across these approaches, electrification strategies rooted in renewable energy are pivotal, providing the requisite energy to drive these processes. When paired with carbon-efficient raw materials and methods designed to avoid CO₂ emissions, these strategies stand out as a crucial lever in the decarbonization of PC production46.
Overview
The recent surge in interest surrounding the decarbonization of cement manufacturing is evident in the substantial investments being made by governmental agencies both within the U.S. and internationally. These investments span the spectrum from foundational research to the commercial deployment of promising technologies, underscoring the urgency of the challenge. Moreover, this trend presents a strategic opportunity for companies engaged in pioneering advanced manufacturing and scaling up innovative solutions to secure a significant competitive advantage in an increasingly sustainability-driven market. Traditional cement manufacturers, companies in allied areas (e.g., chemicals, admixtures, and energy), and climate technology startups are active in the area of cement decarbonization. Prominent cement manufacturers (e.g., Holcim, Ash Grove, Cemex, Heidelberg, UltraTech, etc.) focus mostly on improving the energy and process efficiency in existing manufacturing facilities and adding new energy-efficient sub-process systems including CCUS to enhance the production of PC, portland limestone cements47,48, and limestone-calcined clay cements (LC3)49,50. Several startups in this field are exploring the use of low-carbon or carbon-free raw materials, electrochemical processes (e.g., Sublime Systems51, Brimstone52, Chement53, ZeroCAL54), electric arc furnaces (e.g., Cambridge Electric Cement55), rotodynamic heating (e.g., Coolbrook56), vertical calciners (e.g., Leilac57), and carbonated cements (e.g., Fortera58). This review does not delve into specific technologies deployed by various companies—traditional firms and startups alike—aimed at producing novel SCMs, incorporating CO₂ in concrete curing, or developing and integrating CCUS into cement production; however, their contributions are no less significant. Instead, the focus of this paper is twofold: (i) exploring novel, low-temperature electrochemical technologies for PC production that can be powered by renewable energy sources—technologies which, when coupled with effective CO₂ separation methods, yield a CO₂-free production process; and (ii) examining electrified heating technologies capable of sustainably achieving and maintaining the high temperatures essential for pyroprocessing.
Advanced manufacturing methods powered by renewable energy, like electrolysis, are emerging as promising avenues for producing green cement45,59,60,61. One of the principal benefits of electrochemical calcination of limestone, other than the energy-efficient production of lime (CaO) or hydrated lime (Ca(OH)2), is the straightforward separation and sequestration of CO2—which results in substantial reduction of the carbon footprint62. Solar thermal electrochemical process (STEP), a variant of this method, enables the formation of CaO and solid carbonaceous products at temperatures lower than conventional decarbonization (800 °C), significantly reducing emissions and energy consumption. By combining the primary carbon-free product of electrolysis, namely Ca(OH)2 or CaO, with SiO2 (though not trivial), it is possible to produce tri- and di-calcium silicates (C3S and C2S), which are the primary cement phases. This process can be carried out with a significantly lower carbon footprint compared to conventional manufacturing methods45.
Several electrified heating technologies—such as resistance heating, plasma heating, induction heating, and microwave heating—are increasingly viable for industrial scaling, with costs now approaching those of traditional fossil fuel-based heat sources for cement production63,64. Among these, resistance heating and solar-based heating stand out as leading candidates for large-scale deployment, owing to their comparatively lower capital requirements and proven effectiveness in sectors like chemicals and glass manufacturing65,66. Conversely, microwave, induction, and plasma heating, while highly innovative and potentially more impactful in the longer run, necessitate substantial modifications to existing infrastructure67. Microwave and induction heating methods, being bulk heating methods, are more energy-efficient, but not easily integrated with the current manufacturing processes, and thus are challenging for scale-up, while plasma heating, though capable of being modularly integrated in a kiln, presents energy efficiency and temperature control challenges68. Nonetheless, these technologies present compelling alternatives for niche applications, particularly in the production of specialized, high-value cements. As these methods advance toward industrial scalability, their implementation in targeted markets or for unique cement types could foster focused emissions reductions—bridging gaps left by conventional techniques.
The abovementioned methods may alter the chemistry and performance of PC, potentially leading to non-compliance with current construction standards69,70. More research is therefore needed before these manufacturing schemas can attain industrial translation. To address carbon emissions in the interim, a favorable approach involves CCUS20,71,72 from industrial operations. CCUS would serve as a transitional strategy to mitigate carbon emissions while industries work towards adopting sustainable low-carbon alternatives. However, the integration of CCUS within the cement industry is not expected to reach commercial-scale deployment before 203073. The delay is primarily due to the substantial technical, financial, and logistical challenges involved. Key obstacles include the high costs associated with CCUS technology, the need for extensive infrastructure to capture and transport CO₂, and the limited availability of suitable storage sites such as salt caverns. Additionally, regulatory hurdles, energy demands of the capture process, and the complexity of retrofitting existing cement plants further delay widespread adoption. In parallel, advancements in artificial intelligence and digital twin technology are transforming the cement industry74,75,76,77. Engineering and application software now span the entire plant lifecycle, with platforms designed to collect, analyze, and display data from multiple sources, whether within a single plant or across multiple facilities. By leveraging these technologies, manufacturers can optimize every phase of the manufacturing process—from raw material procurement to final product delivery—enhancing both energy efficiency and cost-effectiveness. The combination of CCUS and advanced digital tools represents a significant step forward in reducing carbon emissions and advancing towards a sustainable future in cement production.
In the following sections of the paper, we comprehensively explore multiple processing pathways for sustainable cement and precursor production through novel, promising techniques—both current and emerging—that employ electrification through integration of a wide array of technologies, including electrochemical synthesis of cement precursors and various electrically driven methods for high-temperature processing. Succinct techno-economic analyses of such technologies are also provided to glean insights into their viability, scalability, and integration strategies applicable to both existing and future cement production infrastructures, which ultimately influence decision-making. The review also provides perspectives on the limitations of these approaches and discusses potential solutions to address these challenges.
Electrolysis for production of cement and its precursors
Electrolysis—an innovative technique employing direct electric current to drive non-spontaneous reactions—presents a promising pathway to decarbonize the cement industry45,78,79. This process enables the decomposition of limestone—one of the key reactions in cement production—into lime (CaO) or hydrated lime (Ca(OH)2, also known as portlandite) using electricity as the energy source, eliminating the need for fossil fuels80,81,82,83. The process is typically divided into two half-reactions: (1) reduction at the cathode and (2) oxidation at the anode. The exact dynamics of half-cell reactions during electrolysis are influenced by a myriad of factors, including the composition of the precursor, the design of the electrochemical cell, the chemistry of the electrolyte, and the operating temperature, among others. In many processes—though not all of them—the cathode produces H2, a clean energy source that can be harnessed in subsequent stages of cement production. Concurrently, the anode yields CO2—often mixed with O2 or other gases—which can be efficiently captured to facilitate net-zero carbon emissions. In addition to using electrolysis for limestone decomposition, some studies54,84,85,86,87,88 have investigated its application with feedstocks for alternative cement production, such as calcium- and magnesium-rich solid wastes and seawater. For instance, electrolysis has been used for mineral deposition of dominant ions, such as magnesium and calcium, from seawater to produce alkali carbonates (e.g., MgCO3 and CaCO3) and alkali hydroxides (e.g., Mg(OH)2 and Ca(OH)2). Figure 3a shows the reductions in carbon emissions achieved through electrolysis compared to conventional cement production, demonstrating a reduction of at least 60% in CO2 emissions.
a Comparison of CO2 emissions resulting from conventional and electrochemical cement production methods, while accounting for auxiliary equipment such as crusher, transportation, grinder, cooler, and more. Data used in this figure was drawn from Zhang et al.105; b A concise schematic of multiple electrolytic approaches for cement production, details of which are provided in sections “Aqueous electrolysis”, “Solar thermal electrolysis” and “Seawater electrolysis”.
This section will explore three key approaches that harness electrolysis in cement production as shown by the schematic in Fig. 3b: (1) aqueous electrolysis, conducted at low to ambient temperatures; (2) high-temperature electrolysis, specifically molten carbonate systems; and (3) seawater electrolysis, which focuses on recent advancements in extracting calcium and magnesium from seawater while simultaneously enhancing the ocean’s capacity to absorb atmospheric CO2. Note that the electrolysis methods essentially create the cement precursors, lime or portlandite.
Aqueous electrolysis
Ellis et al.45 pioneered an aqueous electrolysis method to convert CaCO3 into Ca(OH)2, as shown in Fig. 4. This process also produces valuable byproducts, including H2 (at the cathode) and a CO2-O2 gas mixture (at the anode). The electrochemical cell, also known as an electrolyzer, comprises two chambers, each housing a half-cell (anodic and cathodic) connected via a cross-tube and separated by cation exchange membranes (CEMs) and anion exchange membranes (AEMs), respectively. The electrochemical reactions in the half-cells produce protons (H+) via oxygen evolution reaction (OER) at the anode and hydroxide ions (OH−) via hydrogen evolution reaction (HER) at the cathode, establishing a pH gradient that accelerates the dissolution of CaCO3 in the acidic environment of the anode chamber. The resulting Ca2+ ions migrate through the CEM towards the cathodic chamber, combining with OH− ions to form and precipitate Ca(OH)2 within the cross-tube.
Electrochemical production of Ca(OH)2 from CaCO3. Here, the decarbonation cell leverages the pH gradient created by neutral-water electrolysis to facilitate the dissolution of CaCO3 at the acidic anode (using HCl or other acids), while simultaneously promoting the precipitation of Ca(OH)2 in regions where the pH is ≥ 12.5 (alkalinity produced using NaOH or NaNO3 or NaClO4). Concurrently, H2 gas is generated at the cathode, while O2 and CO2 gases are released at the anode. Adapted from ref. 45.
In the original study45, platinum (Pt) electrodes were employed; however, proposals for more economical electrode materials are emerging89,90,91, tailored to the electrolyte’s pH—for instance, nickel, copper, or stainless steel for the cathode, and aluminum, tin, or lead for the anode. Despite these alternatives, Pt electrodes are frequently favored for their exceptional catalytic activity and chemical durability—attributes that are particularly advantageous during prolonged exposure to concentrated electrolytes such as strong acids (e.g., HCl) and bases [e.g., sodium hydroxide (NaOH), sodium perchlorate (NaClO4), or sodium nitrate (NaNO3)92,93]. As an example, Mowbray et al.59 utilized platinum on carbon (Pt/C) electrodes, demonstrating enhanced activity and selectivity for the hydrogen evolution reaction.
In the electrolysis of limestone or other calcium-rich materials, the capture and sequestration of emitted gases are critical for thoroughly mitigating carbon emissions. The process illustrated in Fig. 4 offers a significant advantage: the elevated CO2 concentration (66%vol, as compared to the conventional 20%vol) simplifies the gas separation process. This increased concentration reduces the energy demands associated with separation and enhances the feasibility of CO2 utilization. CCUS is anticipated to play a central role in managing this CO2 stream, contributing up to 36% and 48% of the total emissions reductions by 2050, according to projections by the Global Cement and Concrete Association and the International Energy Agency (IEA), respectively28,94. By streamlining this component of the process—as shown in recent studies by Zhou et al.95 and Xie et al.96.—the overall efficiency and environmental impact of the cement production can be markedly improved97. In another study, Lu et al.84 proposed a scalable electrochemical decarbonization approach that produces carbon-free calcium silicates—rather than lime or portlandite—from limestone and recycled concrete sourced from demolition waste. Like other electrification methods, Lu’s electrochemical scheme facilitates the collection of CO₂, O₂, and H₂, which can be utilized for mineralization, fuel synthesis, and power generation. Additionally, this innovative method yields high-value SCMs to further decarbonize the cement industry, along with a calcium (bi)carbonate-rich solution capable of mitigating ocean acidification. A standout advantage of this approach is its compatibility with the existing infrastructure of the cement industry, enabling rapid integration and scalability in the near term. This makes it a practical and forward-thinking solution for advancing sustainable cement manufacturing practices.
Electrolyte chemistry is a crucial variable that profoundly affects the energy demand, carbon footprint, cost, and efficiency of lime’s electrolytic production. CaCO3 is not soluble in water and requires a strong acidic environment to dissolve; therefore, to achieve electrolysis at low temperatures, the chosen electrolyte must meet several criteria, such as high ionic conductivity, stability under electrochemical conditions, and the ability to promote dissociation or ionization of CaCO3. Furthermore, for the precipitation of Ca(OH)2, an abundance of hydroxyl ions (OH‒) is required; this is typically accomplished using a strong base such as NaOH. This requirement of a pH gradient—with high local acidity in one half-cell and high local alkalinity in the other—limits the range of viable electrolytes [N.B.: The use of chelating agents offers a notable alternative; this was achieved, recently, using a novel ZeroCAL process described below]. Martinez et al.91 highlighted the importance of optimizing electrolyte chemistry and concentration—alongside applied potential—for efficient CaCO3 electrolysis. Based on this and other related studies98,99,100,101,102, it is clear that sustainably sourced electrolytes, such as recycled/reused acids (e.g., dilute HCl and HNO3) and bases (e.g., Ca2+-enriched NaNO3 or NaClO4 solutions), will be needed to replace carbon-intensive ones such as NaOH, which is produced via the chlor-alkali process. Simply put, ascertaining and comprehensive testing of alternative, carbon-efficient acidic and basic electrolytes—such as mixed electrolytes, organic solvents, and protic compounds—are crucial towards reducing operational costs as well as lessening the environmental impact of producing electrolytes102,103,104. An integrated environmental impact assessment—encompassing reductions in carbon emissions, energy consumption, and waste generation—combined with economic feasibility studies is important, factoring in production costs, supply chain logistics, and industrial scalability. This area requires significant further research, to ensure that large-scale cement production can be accomplished in a cost- and carbon-efficient manner.
The design of the electrochemical cell—and the associated electrochemical parameters (e.g., electrolyzer voltage)—collectively represent another pivotal factor in determining the efficiency of lime production via electrolysis. Despite limited research in this area, existing studies highlight the substantial advantages of optimized cell configurations. For instance, Zhang et al.105, demonstrated the effectiveness of bipolar membrane reactors, achieving nearly 100% current efficiency while producing clean gas streams that can be converted into valuable carbonaceous products. Similarly, Rouxhet et al.106 investigated limestone decarbonization using a two-compartment electrolyzer, separated by a conductive membrane, showing its effectiveness in enhancing reaction performance.
Among the electrochemical parameters, electrolyzer voltage is a critical one that directly affects both reaction specificity and energy consumption. Mowbray et al.59 demonstrated that replacing the conventional OER with the anodic hydrogen oxidation reaction (HOR) for proton (for reaction with CaCO3) generation significantly reduced energy consumption, as HOR operates at a lower voltage (1.8 V) compared to OER (4.2 V). Moreover, the HOR electrolyzer exhibited a combined membrane and anode overpotential of 0.11 V, which is in stark contrast to the 1.14 V observed for the OER electrolyzer. In another study, Zhang et al.107 proposed the use of potassium ions (K+)—sourced from KCl(aq)—as a charge carrier across the membrane. Findings from this study also underscore the importance of optimizing electrochemical cell design, particularly with respect to current density and operating voltage. The proposed design changes allow the electrolyzer to operate with near 100% current efficiency at a cell voltage of 2.9 V. Optimization of such parameters directly influence energy consumption and the concentration and mobility of ions participating in electrochemical processes within the cell. Ion concentrations, in turn, impacts the kinetics of Ca(OH)2 precipitation on both the membrane and the cathode. Studies106 have also demonstrated that modifying the electrochemical cell design to enable the gradual and sequential addition of CaCO3—rather than processing the entire material in a single batch—enhances the regulation of Ca(OH)2 precipitation by maintaining stable pH levels within the anodic chamber. This approach ensures a more balanced migration of Ca²⁺ and H⁺ ions, effectively minimizing pH gradients at the cathode and reducing the likelihood of undesirable precipitation. By preventing electrode passivation and preserving electrolyzer performance, these advancements in cell design offer a promising route toward more efficient and sustainable lime production.
Leão et al.54 recently introduced the ZeroCAL process (Fig. 5), an innovative acid base electrochemical process—combining aqueous flow-electrolysis with an electrolytic pH-swing process85,87,88—that utilizes limestone as a Ca source and seawater as an electrolyte to synthesize Ca(OH)2 under ambient conditions, while completely eliminating CO2 emissions. The process consists of three steps including dissolution, separation/recovery, and electrolysis: (Step 1) Ethylenediaminetetraacetic acid (EDTA)-promoted dissolution108,109,110 of CaCO3, wherein the chelation of Ca ions by EDTA effectively removes Ca from the solution thus maintaining an undersaturated solution concerning Ca which in turn stimulates further dissolution of CaCO3; (Step 2a) nanofiltration to separate the Ca-EDTA complex from bicarbonate ions87,88; (Step 2b) acid-promoted decomplexation of Ca from EDTA ensuring complete chelator recovery and the production of a Ca-rich stream; and (Step 3) ultrafast precipitation of Ca(OH)2 using electrolytically generated alkalinity86,111. This sequence produces HCl, sodium bicarbonate (NaHCO3), and H2 and O2 gases as co-products. The HCl is recovered and reused for EDTA regeneration and pH regulation, thereby minimizing chemical waste. The co-produced NaHCO3, while potentially dischargeable to marine environments, must be managed with consideration of local ecological constraints rather than assuming the ocean as an unrestricted sink. Therefore, any discharge should comply with environmental regulations to prevent localized impacts. Additionally, the process generates H2 and O2 gases which may be captured and utilized as zero-carbon energy carriers, supporting on-site power generation and reducing overall emissions112,113,114,115,116.
Process flow diagram of the ZeroCAL process54, which uses seawater-mediated electrolysis to produce Ca(OH)2. The so-produced Ca(OH)2 can be used as a carbon-free precursor and integrated with conventional or electrically operated cement manufacturing system.
In addition to the aforementioned studies demonstrating the potential of electrolysis for sustainable cement manufacturing, new startups (e.g., Sublime Systems51), have initiated efforts to scale up the commercial production of lime—and, by extension, PC—through an electrolysis-based approach utilizing limestone. Despite these advancements, several technical challenges remain for large-scale implementation: (1) the need to replace cost- and carbon-intensive electrodes, electrolytes (both acidic and basic), and membranes separating the two half-cells; (2) lack of engineering optimization in the design of electrolyzers capable of enabling continuous operation, where CaCO3 can be fed continuously while simultaneously allowing the extraction of pure lime; (3) unclear influences of impurities from industrial-grade limestone; and (4) lack of a comprehensive gas capture and separation process. Nevertheless, ongoing research and innovation in this field show promising potential for overcoming these challenges. By advancing these technologies, the cement industry could move closer to sustainable practices, aligning with global climate goals and fostering a more environmentally responsible approach to building and infrastructure development.
Solar thermal electrolysis
Solar thermal electrochemical process (STEP)78,79,117,118,119,120, an innovative single-chamber electrolysis method proposed by Licht120 that harnesses solar energy (Qsolar) to drive the electrolysis of molten carbonates at a lower electrolysis potential (Eelectrolysis) than conventional ambient conditions. This process enables the decomposition of CaCO3 into CaO and solid carbon (C) at temperatures well below 800 °C through the reaction: CaCO3 + Qsolar + Eelectrolysis → CaO + C + O2. Notably, at elevated temperatures, the carbon may react with the other byproduct, oxygen, yielding carbon monoxide (CO) instead: CaCO3 + Qsolar + Eelectrolysis → CaO + CO + ½O2, as shown in Fig. 6. When operated at lower temperatures, STEP virtually eliminates CO2 emissions typically produced during the calcination of CaCO3 [N.B.: thermal decomposition of CaCO3 at > 800 °C generates ~ 1 ton of CO2 and consumes 1.4 MWh of energy per ton of the product121,122,123,124]. This approach not only enhances energy efficiency and cost-effectiveness but also captures carbon as a valuable solid byproduct rather than releasing it as a gas, making it a more sustainable method for lime production61,79.
Representation of the STEP for direct conversion of CaCO3 to CaO, where CO2 is eliminated, and the formation of C or CO byproducts is temperature-dependent. Here M represents alkali metals (e.g., Li, Na, K, etc.).
STEP benefits from reduced operational temperatures, which contribute to significant energy savings. Recent research has demonstrated the potential for even lower operational temperatures (around 400 °C) for limestone-to-lime conversion by employing a mixture of alkali carbonate electrolytes79 (e.g., Na2CO3, K2CO3, and Li2CO3) with lower eutectic melting points, further reducing the energy demand and projected cost. Several studies119,120,125,126,127,128,129,130,131,132 have extended the application of STEP to incorporate CO2 capture and conversion—for example, by splitting CO2 into valuable fuels and carbon-based products. Furthermore, in addition to lime production from limestone, STEP has been applied to a wide range of chemical processes, including separation of iron from hematite ore, wastewater treatment, synthesis of organic compounds, and industrial applications including CCUS119,128,129,130. Kaplan et al.131 demonstrated the feasibility of using STEP as a CCUS technique that converts CO2 in dilute flue gas streams to CO (a potent reducing agent for industrial production of various metals and alloys) even in the presence of contaminants like SO2132. More recent research has explored molten alkali borate-carbonate mixtures for CCUS, which significantly increased CO2 uptake from 3% to 60%, thereby improving process efficiency133,134.
Despite these advancements in STEP, further improvements are required to enhance its industrial applicability—particularly in terms of lowering the operating temperature, enhancing the chemical stability and durability of the electrodes, and optimizing the recovery and utilization of products. Scaling up STEP also remains a challenge; current implementations demonstrate a CO2 capture capacity of only ~2 tons per day135,136. To achieve impactful global CO2 mitigation, additional enhancements are needed in electrochemical system design, including the recycling of molten carbonates—whose thermal stability under high-temperature and continuous operation requires further research. Moreover, integrating alternative renewable energy sources, such as wind or hydropower, is essential to maintain uninterrupted operation when solar energy is insuficcient137.
Seawater electrolysis
Seawater, an abundant and alkaline resource (pH 8.0–8.2), contains a mixture of cations such as Ca2+, Mg2+, and Na+ along with anions like HCO3−, Cl−, and SO42−. These ionic species exist at large abundances, typically ranging from 0.01 to 0.6 moles per liter; this contributes to the high ionic strength of seawater138. The pioneering method of extracting and precipitating mineral compounds from seawater was introduced by Hilbertz139. This work demonstrated the application of electrolysis to seawater while leveraging its intrinsic alkalinity and high ionic strength to catalyze redox reactions. These electrochemical reactions, when controlled and optimized, not only produce useful gaseous byproducts such O2 and H2 but also promote the precipitation of minerals such as CaCO3, Ca(OH)2, magnesium carbonate (MgCO3), and magnesium hydroxide (Mg(OH)2). Crucially, the increase in local pH around the cathode, brought about by the release of hydroxide ions (OH−), drives the chemical reactions with dissolved cations in the seawater140. This process thereby facilitates the formation of these mineral compounds, which could be used in various environmental and industrial applications including the production of low-carbon PC and alternative cements.
Recent studies141,142 have explored electrochemical methods to efficiently recover Ca- and Mg-rich precipitates from seawater, thereby providing a sustainable avenue for generating primary as well as supplementary cementitious materials. The electrochemical approach detailed in these investigations, once optimized, holds the potential to enable large-scale extraction and recovery of minerals from seawater. This advancement could facilitate the production of magnesium-based cements, which are not only eco-friendly but also offer properties conducive to modern construction needs143. These Mg-based cements produce compressive strengths comparable to PC and are particularly suited for unreinforced applications. Notably, such applications constitute approximately 15% of all concrete structures144.
A series of recent publications have introduced a seawater mineralization process87,88—titled Equatic—that uses electrolysis to emulate the natural CO2 removal processes of the oceans to extract and store atmospheric CO2 in the form of solid CaCO3 and aqueous magnesium (bi)carbonate (MgCO3 and Mg(HCO3)2). In the cathodic chamber of an electrochemical reactor (Fig. 7), the electrolysis of seawater yields OH−, elevating the pH from 8.2 to 10.5. At this elevated pH, all dissolved carbon (C) along with Ca and Mg species are precipitated as CaCO3 and Mg(OH)2 respectively. The introduction of additional CO2—e.g., from industrial flue gas—into this high-pH solution triggers two simultaneous reactions: (i) the additional precipitation of CaCO3, and (ii) dissolution of solid Mg(OH)2 resulting in the ultimate storage of CO2 as aqueous bicarbonate and mineral carbonate species. The authors proposed that, following the removal of the solid CaCO3 via filtration, the carbonate ion-rich catholyte solution could be returned to the ocean—thus enhancing and activating its ability to capture yet more CO2. The authors also claim that this process—implemented on a global scale—possesses the capacity to extract >10 billion tons of CO2 from the atmosphere (dissolved in seawater) and produce more than 15 billion tons of calcite each year88. Uniquely, the so-produced calcite could function as a precursor for electrolytic production of Ca(OH)2 as discussed above in, without having to resort to mining of this mineral—this would not only enhance circularity but also offer end-to-end certainty around CO2 abatement. Beyond its direct CO2 capture benefits, this electrochemical pathway offers additional industrial synergies. The excess calcite could satisfy the demands of the global carbonate market (15 billion tons/year145) including fillers and aggregates146,147. Additionally, H2 and O2—the byproducts of electrolysis—could be used as industrial fuels.
A schematic of the Equatic process illustrates the primary steps involved in CO2 removal, highlighting the major inlet and outlet feeds. This process leads to the formation of carbonate solids and dissolved CO2 in aqueous form (Cases 1 and 2a) and, alternatively, the formation of carbonate solids exclusively (Case 2b). Adapted from ref. 87.
Chen et al.141 proposed a novel approach that utilizes ultrafiltration membranes for Ca(OH)2 production, achieving over 90% recovery of Ca in the seawater. These membranes provide a cost-effective alternative to conventional AEM and CEM, offering not just better chemical durability but also superior resistance to Cl⁻ oxidation (thus mitigating the release of Cl2 gas from the anode). This durability, combined with the membranes’ lower cost, enhances their reusability. The method allows for the direct precipitation of Ca(OH)2, which can be calcined at <500 °C (using renewably sourced electricity) to produce CaO without emitting CO2. The same method can be adapted to produce Mg(OH)2 from seawater, which can subsequently be carbonated (e.g., using CO2 from industrial flue gas streams) at low temperatures to generate MgCO3, a precursor for Mg-based cement or an SCM for concrete. Although the energy requirements per ton of the product are comparable to those of conventional processes, utilizing renewable energy enables this method to achieve a carbon-negative footprint148.
Badjatya et al.148 introduced a membrane-less electrolyzer technique to extract alkaline earth metals—primarily Mg—from seawater, promoting the production of Mg-based cements. This method circumvents the traditional barriers associated with electrolytic processes, such as membrane fouling and degradation, thereby enhancing efficiency and scalability. Additionally, other studies149,150 are exploring chlorine-mediated and calcium-looping based electrochemical processes for the Gigaton-scale removal of CO2 from seawater. These processes not only aim to utilize the extracted CO2 for the regeneration of carbonate materials but also replenish the seawater’s capacity to absorb more CO2 accumulated in the atmosphere once it is returned to the ocean.
While the full potential of seawater electrolysis and large-scale CO2 removal (from the atmosphere) and utilization in cement production is still under investigation, the integration of these processes with renewable energy sources highlights a dual benefit. They can be entirely powered by sustainable energy, reducing the reliance on fossil fuels, and byproducts, such as H2, can be repurposed for energy generation. This holistic approach represents a significant stride towards mitigating climate change while advancing technologies for the production of sustainable construction materials. Though electrolysis of seawater has been demonstrated in various laboratory-scale experiments, scaling up these processes industrially requires solving various fundamental and engineering challenges. Critical challenges include the optimization of electrode compositions and designs in relation to the chemistry of seawater, electrolytes (e.g., salts or additives that are added to optimize ionic conductivity of the electrolytic medium), and products; system hydrodynamics and mass-and-charge transfer to reduce their energy intensity, ensuring effective species separation at the electrodes; a deeper understanding of how process parameters—temperature, feed cross-flow velocity, applied voltage, and flux—influence product quality and yield; and the integration with renewable energy. The relatively large abundances of Cl− and SO42⁻ in seawater introduce added complexity to the system (e.g., the risk of release of gaseous Cl2 and SO2/SO3), which necessitate careful environmental evaluations142. Table 1 summarizes the laboratory feasibility, scale-up potential, and key considerations for various electrochemical methods aimed at decarbonizing cement production. Overcoming these challenges would conceivably require the convergence of experiments with multiphysics models including: electric models to analyze field distributions and currents, and the impact of electrode geometry and composition on reaction efficiency; chemical models to describe reaction kinetics and product formation, optimizing electrolyte composition, electrode properties, and operational parameters to maximize selectivity and yield; and fluid dynamics models for mass-transfer and concentration-gradient optimizations. Furthermore, integrating these electrochemical processes with low-carbon energy sources is essential for achieving carbon-neutrality in cement production.
Electrified heating technologies
In the production of PC through conventional means, it is imperative to achieve and maintain a clinkering temperature of ~1500 °C since C3S (the cement chemistry notation for tricalcium silicate or alite), the major strength-imparting phase of PC forms at this temperature. Formation of cement phases, including C3S, C2S (dicalcium silicate), C3A (tricalcium aluminate), and C4AF (tetracalcium aluminoferrite) follows a sequential reaction scheme as shown in Eqs. (1)–(5) below. Equation (1) is called the limestone calcination reaction, and the subsequent reactions are the cement mineralogical phase formation reactions, generally termed clinkering reactions.
Historically—and even today—the vast majority of cement plants worldwide have relied on the combustion of fossil fuels to achieve this temperature. Although highly effective, fossil fuel-based heating poses significant environmental challenges. In addition to the release of large amounts of CO2 as a result of fossil fuel combustion (e.g., in cement manufacturing, ~40% of total CO2 emitted is attributed to process heating), other harmful air pollutants such as sulfur dioxide, nitrogen oxides, and particulate matter are released, which have the potential to impact human health and the environment. Thus, in the quest for sustainability and reduced carbon emissions, recent advancements have fostered the development and pilot- and plant-scale testing of various electrified heating technologies. Governments, cement companies, and academic institutions have come together in this regard, forming consortia to take advantage of this critical decarbonization lever. For instance, European Union’s Horizon Europe research and innovation program has put together the ELECTRA consortium with partners spanning eight countries to develop knowledge on how to provide heat to processes that require elevated temperatures. Their efforts cover plasma heating, resistive heating, rotary kiln, and fluidized bed technologies151. Recently, the U.S Department of Energy, through its 7th Clean Energy Manufacturing Innovation Institute, Electrified Processes for Industrial Excellence (EPIXC)152, has funded academia-industry collaborative research aimed at advancing translational electrification technologies for heavy industries. Within the cement industry sector of EPIXC, the focus lies on electrified calcination, plasma and induction-based heating, and related cutting-edge technologies.
Theoretical analysis determines that an electric kiln results in a specific energy density of 2.68 GJ/t of clinker, a value that is lower than the most efficient dry kiln currently in use153. Electrified technologies for many low- and medium-temperature heat processes (up to 600 °C) have currently matured, but electrified process heating for high temperature processes such as cement production (requiring up to 1500 °C) is extremely challenging. However, the elimination of fossil fuels from these processes also likely results in maximum environmental and human health benefits, and energy efficiency. To realize the decarbonization benefits associated with electrification of process heating, a significant expansion of renewable electricity generation capacity is needed, coupled with updates to grid infrastructure and mitigation pathways for intermittency of renewable electricity (e.g., through heat storage). Cement plants operate continuously and could provide options to consume unused renewable energy (in a future abundant renewables scenario), thereby facilitating grid-balancing. Addressing grid intermittency requires a combination of strategies, including the deployment of large-scale energy storage solutions such as molten salt, batteries, and thermal energy storage integrated within cement plants (thermal energy storage solutions are discussed in section “TES and other emerging low-carbon cement manufacturing technologies”). Additionally, demand-side flexibility measures, such as dynamic load management and smart grid integration, could enable cement plants to adjust power consumption in response to fluctuations in renewable energy availability, ensuring stable operation even under variable supply conditions. Another alternative proposed is the use of small modular nuclear reactors for energy-intensive industrial operations, which would afford electrification of the plant, with excess power being sent to the grid. Promising efforts on these fronts are emerging, which are expected to make innovative electrified heating methods such as resistive, inductive, plasma, microwave, and even solar based heating technologies attractive. Each of these technologies present unique advantages and challenges, which are critically reviewed in subsequent subsections.
Resistive heating
Resistive element heating, also known as Joule heating or ohmic heating, is a method of generating heat by passing electric current through a conductive material. Resistive heating has been widely implemented in various industries due to its simplicity, efficiency, and better process temperature control. The application of resistive element heating in cement manufacturing has been the subject of several feasibility studies over the past two decades.
Resistive element heating can be of two types—direct heating or indirect heating. Direct heating involves heating the material through a heat source placed inside the heating chamber. The heat source, in this case the heating elements, can directly transmit heat to the material through radiation, or through conduction if the heating elements are in contact with the material. Indirect heating involves heating the material through a heat source placed outside the heating chamber. This heat source either heats the chamber and heat is transferred to the material through conduction, or the source heats a gas (air) by convection, and the gas in turn heats and calcines the material through convection.
One of the earliest comprehensive studies on the electrification of cement production was presented in a technical report by the European Cement Research Academy in 2009154. The report examined various electric heating technologies, including resistive heating, and concluded that while technically feasible, significant challenges remained in terms of energy efficiency and cost-effectiveness. In 2016, Gabaldón-Estevan et al.155 published a theoretical analysis of the potential for electrifying the cement industry. Their study highlighted resistive heating as a promising technology for certain stages of cement production, particularly in preheating and calcination processes, where the temperature requirements are <1000 °C. Hornberger et al.156 demonstrated limestone decomposition at temperatures around 950 °C (comparable to those in conventional calcination processes), achieving an energy consumption of 3.2 GJ per ton of CaO in a pilot-scale resistive heating-based calcination setup. To address the intermittency of renewable electricity sources, a study by Bai et al.157 proposed a system combining resistive heating with high-temperature thermal storage using molten salts, potentially enabling more flexible operation of electrified cement kilns.
One significant barrier to the upscaling of resistance heating technologies is the substantial capital investment required. The transition involves not only the high costs of implementing new electrified technologies but also the expense of replacing existing cement plants or unit operations with entirely new facilities or subprocesses. Replacing existing cement plants or building electrified cement manufacturing plants fall under long-term goals towards industrial decarbonization. While technologies advance towards these long-term goals, as a short-term advancement towards decarbonization, relevance should be given to flexible technologies that can replace select subprocesses—especially those with large carbon footprint—and can be directly incorporated into the already existing cement plants as retrofit measures. The EU-funded CEMCAP project158 investigated resistive heating for calcination, concluding that resistive heating could be a viable option for new cement plants or major retrofits, particularly in regions with access to low-cost, low-carbon electricity.
In recent times, several projects have been initiated to upscale the existing state-of-the-art in electrification of cement plants incorporating resistive element heating. To realize the said short-term goals of incorporating electrified technologies into already existing cement plants, researchers at VDZ (German Cement Works Association) have developed modular heating elements159. These elements, made from advanced ceramics, are used as inserts inside or outside the heating chamber. When electric current is passed through these elements, heat is generated via the Joule effect. These elements can withstand temperatures up to 1600 °C and have demonstrated energy efficiencies of up to 85% in their laboratory tests. Launched in 2020, the ELSE 2 (Electrification of Limestone Calcination)160 project aims to develop and demonstrate a novel electric calciner design that could reduce CO2 emissions by up to 90% compared to conventional methods. Their project includes plans for a pilot plant capable of processing 100 tons of limestone per day. This electrifies only the calcination step in cement production, with the clinkering still powered by conventional (fossil-fuel) energy sources. Another notable example is the Decarbonate project in Finland161, which examined the calcination of limestone in an electrically heated rotary kiln. As part of the initiative, a mobile pilot plant featuring an electrified rotary calciner with a capacity of over 100 kg/h was constructed. This plant produced high-quality calcined material and achieved a CO2 concentration of 98% (vol. dry). In 2019, Heidelberg Cement announced a project to test electric heating in cement production at their plant in Slite, Sweden162. The project aims to achieve full-scale electric cement production by 2030, potentially reducing the plant’s CO2 emissions by up to 1.8 million tons annually.
In the short term, decoupling calcination operations from clinkering in a cement plant allows for a reduction in fuel-related CO2 emissions and a potentially shorter kiln length, in addition to enabling the capture of flue gas with CO2 concentrations approaching 100% (as opposed to <25% in conventional coal-fired processing163,164) which can be used in the production of chemicals and fuels. This makes resistance heating a viable retrofit for existing cement plants to reduce their CO2 footprint through a judicious combination of emission reduction and CCUS. Moreover, with the advent of blended cements such as limestone-calcined clay cements (LC3), electrification presents further opportunities to the cement producer in terms of emission reduction, and energy- and cost-efficiency. Calcined clays, which can be produced at temperatures below 800 °C, enable calcination of limestone and clays in electric rotary kilns or flash calciners that could be fueled by renewable energy. A schematic design of how the resistive element heating technology can be incorporated indirectly into a rotary calciner for co-calcination of limestone and clay is provided in Fig. 8165. These calciners could also be modified for steam injection, which has been reported to further depress the calcination temperature of limestone by up to 100 °C166, resulting in further emission reductions and enhancing energy efficiency. As the cement industry moves towards increased production of blended cements, and with paucity of traditional SCMs such as fly ash and slag looming in the horizon, such novel calcination systems are likely to become more prevalent.
Schematic of resistive element heating-based rotary calciner for limestone and clay calcination165.
Despite promising advancements, several challenges inhibit the widespread adoption of resistive heating in cement production. Energy efficiency—mainly due to uncontrolled heat losses—remains a concern, as electric heating systems may require 10–20% more primary energy than conventional methods, depending on insulation and electricity sources68. Renewable energy production must substantially increase over the next decade to satisfy decarbonization goals, along with widespread adoption of large-scale energy storage technologies. Retrofitting costs, estimated at $200 million or more per plant, operational expenses tied to regional electricity prices, uncertainties with respect to grid efficiency, energy delivery and distribution, and reduced material throughput (because of inefficiencies in converting electricity to heat), further complicate cost competitiveness167,168. While resistive heating can achieve high thermal efficiencies in laboratory-scale setups, large-scale implementation faces significant challenges. Contrary to results from modeling studies, uniform heat distribution, especially in rotary kilns, poses engineering challenges in designing industrial-scale electrical kilns, or retrofitting existing kilns with electrical resistance heating elements. More research and translational studies are needed in this domain.
Inductive heating
Inductive heating is a process that utilizes electromagnetic induction to heat electrically conductive materials. This technology operates by generating an alternating magnetic field through a coil, which induces an electric current (eddy current) in the conductive material placed within the field. The resistance of the material to this induced current generates heat, allowing for rapid, efficient, and uniform heating without direct contact with a heating element169. Electromagnetic heating directly converts electrical energy into thermal energy rapidly, thereby minimizing heat loss and enhancing energy efficiency, often shortening processing times by >50% as compared to conventional rotary kilns. It is important to note that for materials with high electric conductivity, induction heating is highly efficient as the electric current is directly induced in the material to be heated. In the context of cement manufacturing, where the cement raw meal has low electric conductivity, thermal energy must be transferred indirectly through a carrier fluid or through heat exchange from a conductive or a magnetic material170.
Results from a few studies have identified induction heating as a viable option for replacing conventional methodologies in the calcination and clinkering processes. The ELSE project (ELSE-Phase 1)171 assessed the feasibility of several electricity-to-heat methodologies, and identified induction heating as one among the promising technologies for cement production (though they favored resistance heating in their final analysis). A few studies have also highlighted the potential of the use of electromagnetic induction-based heating for limestone and clay calcination as well as cement production71,172. Another notable implementation of this technology is the “Blue Cement” concept173, which takes a holistic approach, combining an electrified calciner incorporating induction-based heating with carbon capture techniques. This integrated strategy, proposed for limestone calcination, aims to achieve near-zero emissions by addressing both process heat and inherent CO2 emissions from limestone decomposition. Figure 9 shows the proposed reactor incorporating induction-based heating for limestone calcination.
Quevedo Parra et al.174, in their technoeconomic assessment of theoretical electrified scenarios, studied a fully electrified cement plant consisting of a pre-calciner heated via magnetic induction. The assessment was conducted in Aspen plus, a process simulating software, wherein the pre-calciner was operated at 920 °C to account for higher CO2 pressure. The induction heating based pre-calciner was followed by combustion of alternative fuels in the rotary kiln. Their results indicate a total energy demand (fuel + electricity) of ~4.75 GJ per ton of clinker in their electrified plants as opposed to 3.7 GJ/ton of clinker for the reference plant. However, 2 GJ/ton of the total energy demand of the reference plant was sourced from the combustion of fossil fuels, whereas all the energy demand for the electrified plants were either sourced from electrified sources or from the combustion of alternate renewable fuels. Furthermore, the simulated electrified cement plants saw about 92.8% reduction in the total CO2 emissions.
Despite its promising potential, several challenges remain to be addressed before the technology can be upscaled. As an example, the CemZero project68 investigated the possibility of using induction heating technology for raw meal clinkering. The researchers calcined ~25 kg raw meal in a 9 kW high frequency induction furnace with an energy supply of 19.8 kWh. The test lasted for 3 h, which resulted in a temperature rise of 550 °C, far from that required for calcination or clinkering. Thus, the feasibility of direct and indirect induction heating in increasing the kiln temperatures to the required levels should be thoroughly understood. As mentioned earlier, the low electrical conductivity of the cement raw meal necessitates the requirement of additional conductive materials to aid heat transfer to the raw meal. Typically, nanomaterials such as graphene are used as conductive materials required for heat transfer, but this significantly compromises the cost-efficiency and scalability of the technique. Another challenge lies in the higher energy demand for induction-based systems. As discussed, process modeling suggests that an electrified cement plant using an induction-based pre-calciner may require a total energy input of 4.75 GJ per ton of clinker, which is higher than the 3.7 GJ/ton required by conventional fossil-fuel-fired plants174. Although this additional energy demand is offset by the elimination of fossil fuel combustion, it raises cost and efficiency concerns, particularly in regions where access to low-cost, low-carbon electricity is limited. Moreover, the capital costs for developing and scaling up induction-based reactors tailored for cement production remain significantly high. From a heat transfer perspective, since the primary coil must be cooled continuously with water or coolants to remove the internal losses and the heat transfer from the load to the coil, maintaining thermal efficiency remains a challenge. In conclusion, the available body of literature indicates that, while inductive heating is likely a viable alternative for decarbonizing the cement manufacturing process, current technologies must address significant knowledge- and scale-up gaps for successful implementation.
Plasma heating
Plasma heating involves generating a hot, ionized plasma beam by applying electrical energy to a gas. It is typically initiated by an arc discharge between an anode and a cathode within a plasma torch, at the same time, ensuring a continuous flow of gas. The process of heating cement raw materials via thermal plasma requires carrier gases like oxygen, carbon dioxide, air, steam, or other inert gases such as nitrogen and argon (choice of the gas based on enthalpy, reactivity, and cost), with heat transfer primarily occurring through convection71. For cement manufacturing using the thermal plasma-based process, CO2 can be used as the working gas since it is emitted during the calcination process. The gas flow stabilizes the arc and defines its path by cooling the outer layers175. Note that only a small portion of the gas attains the plasma state, which heats the remainder of the gas. The type and flow rate of the gas are generally adjusted to influence the heat transfer to the raw material to be heated. Plasma torches have substantially higher maximum temperatures than the fossil fuel-powered flame. A typical plasma arc can reach temperatures between 1500 °C and 10,000 °C, requiring cooling of the electrodes to extend their service life. Tungsten and copper are the most commonly used cathode and anode materials, respectively. Direct current thermal plasma torches are commonly used as they can sustain more stable and longer arcs175. Another fundamental difference between conventional coal-based heating and plasma-based heating pertains to heat transfer mechanisms within the kiln since no ash and fuel particles which radiate heat are present in the latter case176.
Burman et al.177 investigated whether the use of plasma torches in cement manufacturing is feasible. This work developed and evaluated a heat transfer model for cement production in a kiln by using plasma torches. Their results indicated that kiln operation with a plasma torch requires much higher gas temperatures, around ~400 °C more, to achieve the desired bed temperatures. It is to be noted that, due to the complexity, the process kinetics was not implemented in the model developed. Similar heat transfer models are also reported in176,178. In order to reduce the heat loss due to high flue gas temperatures, these models suggest increasing the production rate of the clinker (more material to consume heat) and tilting the plasma (increasing heat transfer to the kiln bed). Future work involving measurements or detailed computational fluid dynamics modeling of the gas and solid phase temperatures and their distribution in a kiln is required to optimize the process.
In the CemZero project68, a pre-feasibility study assessed the potential for electrifying the cement production process using plasma technology for heat transfer in a cement kiln. Various scenarios for full electrification were simulated and assessed in a laboratory setting. It was determined that, from a process perspective, using CO2 as a plasma gas offered several advantages. It is chemically compatible with the cement production process and helps prevent the formation of nitrogen oxides (NOx). Moreover, their results indicated better clinkering with CO2 as the plasma gas. In conclusion, it was recommended to explore plasma-based electrification of cement manufacturing on a larger scale for further investigation. The CemZero project also considered a combination of resistive or inductive heating to meet the heat demand in the precalciner, and plasma-based heating for clinkering in the kiln. Here, air is the plasma gas, which is pre-heated to 3470 °C before entering the kiln. Plasma gas is mixed with secondary air from the clinker cooler to achieve an outlet temperature of the clinker of 1450 °C174. A schematic of this process is shown in Fig. 10. Another potential benefit of this approach where calcination and clinkering are decoupled is that the kiln length can be considerably shortened, because of the relatively rapid clinkering reactions in the presence of hot plasma. It is also important to note that while plasma heating reduces solid byproduct generation such as ash, the introduction of high volumes of carrier gases like CO2, air, or argon into the system may alter the flue gas composition and flow, potentially increasing the downstream requirements for gas cleaning, heat recovery, or CO2 separation and reuse.
In another project by the British Mineral Products Association179, a simulation study and a 30-min physical clinkering test was conducted at an industrial cement kiln to evaluate the use of a plasma burner in the calciner. The plasma burner successfully operated in the harsh calciner environment and provided the required heat to the process, producing about 70 kW of thermal energy. However, due to the short test duration and the limited thermal output, no definitive conclusions could be made regarding the effectiveness of the electrified thermal input. At an industrial scale, SaltX Technology and SMA Mineral’s Electric Arc Calciner (EAC)180, uses the principle of plasma-based heating for zero emission quicklime (ZEQL) production. This technology has been further elaborated in section “Scaled-up electrified calciner and kiln designs”. Figure 11 presents a schematic of the EAC technology used for clean quicklime production.
A significant challenge in utilizing plasma technology lies in maintaining efficient heat transfer. When fuel and ash particles are removed, a substantial portion of the heat—carried primarily by photons (radiation)—is lost. To compensate, gas radiation from either air or CO₂ must be employed; however, these gases exhibit varying dissociation behaviors depending on the operation of the plasma torch. This variability inevitably alters the radiative heat transfer to the clinker bed, necessitating precise quantification to ensure consistent quality of the final product. It is also worth mentioning that, if the bed temperatures are not effectively maintained, heating the cement raw meal to very high temperatures may produce undesired phase transitions (such as melting or evaporation of some species), which could affect cement chemistry, and consequently the quality and properties of the cement. The extremely high operating temperatures of plasma torches, which can reach up to 10,000 °C, necessitates robust cooling systems to prevent excessive wear on the electrodes and maintain process stability, significantly increasing energy consumption and maintenance costs. Moreover, plasma heating requires careful control of the gas composition and flow rate to ensure efficient heat transfer, as only a small portion of the gas actually attains the plasma state. Therefore, to realize efficient application of plasma-based heating in cement manufacture, it is recommended that focused research should include: (1) optimization of the rate of heat transfer to the clinker bed, particularly in the burning zone; (2) design and operational parameters for managing the net heat flux from the plasma gas to the clinker bed; (3) quality and properties of the clinker formed as a function of the process parameters; (4) NOx formation; (5) controllability of the process owing to high temperatures reached by the arc; and (6) productivity changes resulting from changing the energy source177. Once the technological challenges are mitigated, it is also important to evaluate which of the unit operations can be converted efficiently to electrified technologies so as to operate efficiently in coordination with a plasma-powered kiln.
Microwave heating
The fundamental difference between microwave (µw) heating and other conventional heating methods is the mode of interaction with the materials to be heated. Microwave radiation—typically used within the frequency range of 300 MHz to 300 GHz and wavelengths of 1 mm to 1000 mm—stimulates polar molecules within the material to oscillate; this rapid motion results in friction and, subsequently, heat. Two dominant µw frequencies, 0.915 ± 0.013 GHz and 2.45 ± 0.05 GHz, are widely used in industrial µw applications181. The efficacy of the heating mechanism hinges on the dielectric properties of the material; notably, the dielectric constant and the loss factor, which dictate how well the material absorbs and converts microwave energy into thermal energy. A major advantage microwave heating poses over other methods is that the majority of the electromagnetic energy (carried by the µw photons) is absorbed, and heat is generated volumetrically, leading to a rapid rise in the material’s temperature. This process represents a significant difference from conventional heating methods, wherein the energy carrier—whether hot gas or a heating element—primarily heats the surface of the material, with the bulk of the material subsequently being heated through conduction. As µw heating leverages electromagnetic waves that penetrate the material, causing the molecules throughout the volume of the material to vibrate and generate heat internally, it allows for uniform heating throughout the volume of the material, not just at the surface. Owing to these characteristics, the generated energy is solely absorbed by the raw material, making it the hottest part of the system. Therefore, when employing µw heating, it is not necessary to elevate the kiln temperatures beyond what the raw materials themselves require according to the thermochemical demands of the process. Consequently, µw heating can lead to substantial energy savings and enhanced control over process conditions.
Some of the earliest works on microwave based cement manufacture date back to series of experiments conducted by Quéméneur et al.182,183. The clinkering of various raw meals was examined in these studies, where the raw meals were subjected to microwave heating at 2.45 GHz monomode exposure. In their initial study182, it was reported that plasma formed near the sample (the presence of alumina that has a high dielectric loss at high temperatures likely played a role in initiating the plasma phase) and temperatures of around 1450 °C were achieved within the sample. Qualitative product characterization revealed that the produced clinker had comparable properties to industrial-grade clinker. Li et al.184,185 were able to successfully produce alite (the major phase in PC, which only forms at temperatures between 1350 and 1500 °C) and alternative cements through microwave sintering. Their findings—based on X-ray diffraction, isothermal calorimetry, and surface area measurements—indicate a better quality of alite being produced from microwave sintering as compared to conventional methods. It was also found that the sintering occurred at temperatures that were lower by 100 °C to 150 °C as compared to those attained by resistive heating-based methods. In another series of studies, Fang et al.186,187 also demonstrated the feasibility of producing commercial-grade PC and colored cements using microwave processing. Their experiments resulted in a 100 °C reduction in clinkering temperature and preferential alite growth due to Fe2O3 acting as a microwave susceptor (since metallic additives adsorb microwave radiation efficiently and rapidly convert them to heat through resistive heating).
Buttress et al.188 reviewed µw processing of cement and concrete materials and acknowledged the technical feasibility of cement production through µw-based clinkering. However, their estimates for the energy required to heat a clinker raw meal to 1400 °C ranged from 9.3 to 17.5 GJ/ton at 500 W to 2000 W, significantly higher than the 3.7 GJ/ton required by conventional pyroprocessing methods154. This led them to conclude that while industrial-scale µw clinkering would be more carbon efficient, it would likely be highly energy-intensive. Vermeiren et al.67 highlighted the substantial reflection losses and the need to account for the temperature-dependency of the dielectric properties of the raw materials—which influence how materials absorb microwave energy—to accurately predict the efficiency and effectiveness of microwave heating. Development of an Efficient Microwave System for Material Transformation in energy INtensive processes for an improved Yield (DESTINY), one of European Union’s Horizon 2020 project, utilized 915 MHz microwave technology for continuous material processing in the cement industry189. The overall system was conceptualized as mobile cellular kilns (shipping container-sized) in a modular plant190, enabling the unique possibility of production on-site-of-demand.
The efficiency of µw clinkering methods depend largely on raw material properties, such as the dielectric properties, electrical permittivity, magnetic permeability, the mode of microwave heating (monomode or multimode), and the heating chamber characteristics such as cavity shape and dimensions. Clinker raw meal components including limestones, clays, and quartz exhibit low microwave absorption at room temperature, making initial µw heating challenging without specialized equipment. However, as temperatures rise to around 500 °C for clays and 800 °C for limestones, these materials become efficient µw absorbers, enabling effective heating. Another important parameter governing the efficiency of µw systems is the mode of heating. Monomode systems focus microwave energy into a few hot spots, achieving high local field intensities, making them ideal for fast heating of low-loss materials like cement raw materials. However, they are sensitive to changes in load characteristics and are limited in scalability. Multimode systems, on the other hand, distribute energy more evenly with many hot and cold spots, providing more flexibility for large-scale applications but with slower and less efficient heating.
To advance µw heating technology in the cement industry, feasibility must be validated at scale with respect to the starting materials used in cement manufacturing, the energy demands of the process, cost and throughput, and the resulting chemistry and uniformity of the product. With regard to industrial upscaling, Vermeiren et al.67 discuss three possible approaches: (1) direct volumetric heating of the raw materials with microwave radiation; (2) susceptor-assisted heating, wherein a high µw absorbing material called the “susceptor” is added to the system, which, at low temperatures heats the sample through resistive heating, and once the temperature is high enough, the sample is heated via both indirect radiation from the susceptor and direct volumetric µw heating; and (3) a combination of electric heating and microwave heating, which involves an initial heating of the sample through electric heating followed by microwave heating at higher temperatures when the hot material is able to adsorb microwave radiation more efficiently. Studies highlight both lab-scale and pilot-scale implementations for these three approaches but conclude that the most feasible approach is the combination of electric and microwave heating67,188,191. Frequencies around 2.45 GHz, with power ranging from 700 W to 30 kW have been generally adopted. For clay calcination, µw kilns have been used to achieve high feed rates (up to 40 kg/h), while maintaining temperatures between 450 °C and 650 °C192,193,194. In limestone calcination, susceptor-assisted heating techniques—such as silicon carbide (SiC) susceptor tubes and copper oxide coatings—show notable benefits in heating rates and calcination efficiency195,196,197,198. Experiments on clinkering have used µw enhancement to reduce processing temperatures184,186,199,200,201. Applications for processing cement raw meal and sulfoaluminate cement clinkerization suggest potential for upscaling, as demonstrated in the DAPhNE project202 and in the large-scale setups with batch processing sizes of ~25 kg by Fall et al.203, which highlighted potential energy savings of ~36% and a reduction of around 270 million tons of CO2 per year in the U.S. when this technology is used.
The major challenge inhibiting the widespread utilization of µW technology lies in the dielectric properties of cement raw materials, which influence how effectively they absorb microwave radiation. While susceptor-assisted heating is a potential solution, it introduces additional material costs and process complexities that must be optimized for industrial viability. Scalability also presents a critical hurdle. While monomode microwave systems achieve high local field intensities, they are sensitive to changes in input load characteristics and are challenging to scale up. On the other hand, multimode systems distribute energy more evenly but suffer from hot and cold spots, leading to non-uniform heating and reduced efficiency. Industrial-scale implementation requires optimized cavity designs and heating chamber geometries to ensure consistent temperature distribution throughout the material. Moreover, reflection losses and process control pose additional technical barriers. A substantial portion of microwave energy may be reflected rather than absorbed, reducing heating efficiency and necessitating precise tuning of the microwave frequency and power based on the material’s evolving dielectric properties at high temperatures. Furthermore, when susceptors are mixed with the raw materials to enhance microwave absorption, they may remain as residues in the clinker or require separation post-processing, potentially affecting product purity or introducing additional steps for material recovery and reuse. Economic feasibility also remains a significant concern. The high capital costs associated with microwave generators, susceptors, and specialized kiln modifications pose barriers to widespread adoption. Further research on µW chamber designs to enhance efficiency and process control, accounting for the raw materials’ varying µW susceptibility, is critical for advancing this technology in PC manufacture.
Solar heating
With the advent of cheap solar energy, solar-thermal power is a sustainable and potentially economical alternative to fossil fuels for a number of industrial applications including generating thermal energy required for solid-state clinkering reactions that are part of cement manufacturing. Concentrated solar-thermal power (CSP) technology harnesses solar energy by using mirrors or lenses to concentrate sunlight onto a small area, creating high temperatures that can be used to provide heat to industrial processes. Here, the concentrated sunlight heats a heat transfer fluid (working fluid), which in turn delivers the heat that is used for material production, or for storage. Combined solar heat and power (CSHP) systems can generate both thermal energy and electrical power from solar energy. The process typically utilizes solar collectors to capture sunlight, which heats working fluids such as water or oil, and can then be passed through the kilns to transfer heat to the raw feed or can produce steam or vapors which are capable of driving turbines and ultimately generate electricity. This dual output enhances overall efficiency compared to systems that only generate electricity or heat. The key component of both the technologies is a solar reactor, which is responsible for the conversion of energy types. Typically, the solar reactor is integrated with existing cement plants by the means of two designs, as discussed by Pitz-Paal et al.204. In the first design, known as the top of-tower (TT) design, the solar reactor is placed on top of the existing pre-heater tower. The advantage that this design poses over other designs is that the optical losses are minimal205,206. The second model, also known as the beam-down (BD) design, has the solar reactor fixed on the ground. An additional reflector is installed on top of the pre-heater tower to focus the solar radiation on the reactor. The advantage here is that the heat transfer efficiency is maintained in heating the raw materials effectively207,208.
In the available literature, CSP technology has primarily been proposed for the calcination step in cement production, which requires temperatures around 900 °C—sufficient for the near-complete decomposition of CaCO3 into CaO and CO2. This temperature requirement is comparatively lower than that needed for clinkering reactions. Since the calcination step is responsible for the majority of CO2 emissions in the cement manufacturing process, integrating CSP with CCUS technologies offers a promising path to eliminate these emissions. Studies investigating the use of solar energy for limestone calcination date back to the 1980s; the key among them are Badie et al.209, Flamant et al.210, and Salman and Khraishi211, where small solar furnaces of 1–2 kW power were used to perform thermal decomposition of limestone. Calcination rates of 65-70% were obtained with maximum furnace temperatures ranging from 600 to 1300 °C. Furthermore, Imhof212 examined the potential for integrating solar power into a cement plant producing 3000 tons of clinker daily. The study proposed a hybrid system using both traditional fossil fuels and solar energy for the calcination step. The findings suggested that incorporating solar energy could lead to a moderate ~9% reduction in total CO2 emissions.
More recently, Gonzalez et al.213 conducted a case study of the technical and economic aspects of using solar thermal technology in the cement production process. Three options related to the application of CSP technology into cement production were evaluated in their study, out of which they concluded that the best solution is a central tower with a solar reactor at the top of the tower, similar to the TT design described in Pitz-Paal et al.204. Furthermore, their evaluation reveals that ~40% CO2 emission reduction is possible by using CSP technology in the calcination step. Moumin et al.65 proposed a solar cement plant design based on the German Aerospace Centre’s solar calciner model, evaluating the heliostat field, and analyzing the solar calciner’s energy balance while exploring various scenarios. The findings revealed a CO2 emission reduction potential of 14–17% through the implementation of this technology. Meier et al.214 conducted a study on the economic viability of using concentrated solar energy for lime production. They examined both TT and BD solar lime plants, evaluating three solar input levels (1, 5, and 25 MWth). Their findings indicated that only the 25 MWth plant could achieve payback within eight years of installation, with the potential to eliminate 95% of fossil fuel emissions in lime production. The researchers concluded that using concentrated solar energy for small lime plants under 5 MWth is not economically viable unless costs can be reduced, or higher product prices can be achieved.
CHSP technology is more suited for low-temperature domestic applications215,216; however, Cemex and Synhelion set up a pilot batch production unit in 2022 to produce clinker from concentrated solar radiation by connecting traditional clinker production process with Synhelion’s solar receiver217. Another case of the industrial application uses computer vision to optimize the solar receivers’ position, thus allowing for maximum energy collection throughout the day. Their technology focuses on powering the industrial furnaces directly rather than relying on the grid, making the manufacturing process much more conservative in terms of emission control218. A schematic of the possible pathways of utilizing concentrated solar power (either to provide heat required for industrial processes, to generate solar electricity, or to synthesize solar fuels) is presented in Fig. 12.
Pathways towards utilization of concentrated solar power for industrial applications (not specifically for cement production). Image courtesy of U.S. Department of Energy254.
While CSP and CSHP technologies present potential for decarbonizing cement manufacturing, they are not devoid of shortcomings. Abanades et al.219 have highlighted key challenges that need to be overcome in order to achieve confidence in the CST and CSHP technologies to be implemented on a large scale. These issues include material constraints at extreme temperatures, short particle residence times ultimately leading to incomplete reactions, and sensitivity to raw material particle sizes. Moreover, some reactor designs suffer from batch processing limitations or semi-continuous operation, causing inefficiencies. Heat and mass transfer limitations can also result in non-uniform conversion and thermal gradients. Directly irradiated reactors face risks of particle deposition on optical windows, potentially causing damage. Additionally, certain reactor types are prone to dust formation from particle attrition and may experience clogging issues. These factors collectively impact the efficiency, practicality, and continuous operation of solar reactors in industrial applications.
TES and other emerging low-carbon cement manufacturing technologies
A notable innovation in the field of decarbonization of the cement manufacturing process is thermal heat storage (TES) and waste heat recovery and utilization. Beshore et al.220 analyzed the dry process cement plants in the USA to identify the possible waste heat recovery sources and develop TES based systems that can be used to generate electricity. Engin et al.221 evaluated the possible waste heat recovery pathways in a cement plant and identified: (1) kiln exhaust gases, (2) hot air from the cooler stack, and (3) radiation from the kiln surfaces, as heat loss sources that can be considered for heat recovery. Waste heat from these sources can either directly be used to pre-heat the raw feed or can be recovered by means of waste heat recovery steam generators and further be used to run turbines and generate electrical energy.
Thermal batteries capable of storing thermal energy can be utilized to deliver the required thermal energy for the intermediate steps of cement production. Electrified Thermal Solutions developed thermal batteries termed Joule Hives222 that can store thermal energy and deliver heat to industrial processes, grid storage purposes, and can even be used as standalone batteries. Rondo Energy’s heat battery223 works on similar principles and can be used to provide heat for industrial applications. This battery converts low-cost, intermittent renewable electricity into high-temperature heat using electric heating elements. This heat is stored in bricks, heated up to 1500 °C through thermal radiation, with minimal energy loss over time. When needed, the stored heat is released as superheated air or steam, capable of reaching over 1000 °C. The modular, scalable design enables it to meet the continuous or on-demand heat requirements of various industrial processes, providing a zero-emission, cost-effective alternative to conventional boilers. The process outline for the Rondo heat battery is provided in Fig. 13. Furthermore, wide utilization of TES in developing calcined clays (which is fast emerging as a sustainable SCM, or a component of blended cements) is becoming an area of interest for many studies. Though partial replacement of PC with calcined clays results in significantly lower CO2 emissions, it should be noted that the clay calcination and clinkerization processes still rely heavily on the combustion of fuels to generate heat. The ECoClay project224 explores the possible utilization of renewable energy such as wind power, and solar power, with additional integration of technologies such as TES with battery electric storage systems for the calcination of clays.
The process outline for a thermal energy storage battery. Specific case of the Rondo heat battery. Image courtesy of: Rondo Energy223. In the figure, (1): turning intermittent electricity to heat; (2) thermal radiation warming bricks up to 1500 °C; (3) on-demand or continuous delivery of heat; (4) superheated air- or steam-based heat delivery; and (5) heat utilization in an industrial process.
Other innovative heating methodologies are also being used for providing thermal energy input to the kiln/calciner. For instance, RotoDynamic Heater (RDH), developed by Coolbrook56, generates heat through friction and compression within a rotating tubular structure, reaching temperatures up to 1700 °C without combustion. Its compact design and high-temperature capability make it a promising option for clinker production. Several large industrial players such as CEMEX, UltraTech cement, ABB, and Shell have partnered with Coolbrook to further the implementation of the RDH at commercial scale. Furthermore, Self-Propagating High-Temperature (SHS) synthesis, or combustion synthesis, is another potential technology that can be used to develop various classes of cements. SHS, usually carried out on pelletized samples containing a mixture of raw materials and a bio-based fuel, involves initiating a high-temperature reaction at a specific point or volumetrically, which releases significant energy that propagates through the material autonomously, eliminating the need for external heat sources225,226. Through SHS, the duration and energy of the synthesis process can be reduced. Another key advantage this process has over conventional process and other electrified processes is that the synthesis can be carried out at much lower operational temperatures (~450 °C) as the synthesis reactions proceed owing to the intrinsic energy release from the combustion of biofuels that ignite at ~450 °C. Agrawal et al.227 applied this technology for the calcination step in cement manufacturing, achieving a reduction in the energy use by over 50% as opposed to the conventional calcination process. Heat transfer modeling of SHS-based limestone calcination demonstrated temperatures of ~1350 °C internally in the sample, which strengthens the feasibility of the utilization of this technique for cement production228,229. While SHS is a potentially carbon- and energy-efficient method for synthesizing cement, the process flow and unit operations are significantly different from conventional cement manufacturing. However, if the limestone calcination stage can be electrified using any of the technologies described in this paper (with or without attendant CCUS), the energy-consuming and large-footprint kiln in a cement plant could be replaced by a smaller SHS reactor, providing a viable option for retrofitting of existing infrastructure for low-carbon cement manufacturing.
Scaled-up electrified calciner and kiln designs
In conventional cement production, calcination of limestone occurs primarily in a pre-calciner, typically heated by exhaust gases, while the clinkering reactions take place in a rotary kiln fueled by combustion of natural gas or coal. Some plants also employ vertical shaft kilns for calcination of both limestone and raw meal. As electrified heating technologies continue to evolve, a critical consideration for their feasibility is the efficiency of heat transfer to the raw materials and their net carbon reduction potential. These aspects depend heavily on factors such as the heat transfer coefficient of the heating chamber and the effective surface area available for heat exchange. Therefore, the development of efficient, scalable, and reliable electrified calciner designs will be a key driver for the cement industry’s transition toward more sustainable production methods.
To keep the discussions succinct, the terms “kilns” and “calciners” are generically termed “heating chambers” in this section which examines various commercial and pre-commercial electrified heating chamber designs. The discussion predominantly focuses on heating chambers that use resistance heating elements, as their designs are generally similar to other electrification methods such as inductive, microwave, or solar heating. Note that the scaled-up systems actively under consideration are for the calcination sub-process in cement manufacturing, since electrified technologies still need to overcome major challenges to get to temperatures high enough for clinkering. The designs based on plasma technology are treated separately due to the need for carrier gas to transfer heat to the raw materials, as well as their potential to facilitate clinkering reactions.
Based on their orientation, heating chamber designs can be broadly categorized into vertical and horizontal configurations230. Vertical designs include drop tube calciners, entrainment calciners, shaft kilns, and fluidized bed calciners, while rotary kilns and tunnel calciners represent horizontal designs. The drop tube calciner, similar to the entrainment calciner, features a vertical tube where raw meal is dropped from the top and calcined as it descends231. This design has been employed in the LEILAC57 project at a pilot scale, handling feed rates of up to 40 tons per hour. However, scaling this technology to meet the production requirements of a typical cement plant would necessitate a number of drop tubes and heating elements, which would result in substantial heat loss due to the increased surface area, increased complexity, and the need to maintain uniformity in all the drop tubes. The limited contact time between particles and heating surfaces also reduces heat transfer efficiency, making the industrial-scale feasibility of an electrified drop tube calciner challenging. In contrast, tunnel calciners comprise a horizontal chamber through which raw meal is conveyed—typically on belts or within troughs—while radiant heat is supplied by heating elements positioned above. This configuration offers improved control over residence time; however, the static nature of the raw meal within the tunnel calciner significantly hampers heat transfer efficiency. With conduction serving as the primary mechanism for heat penetration into the deeper layers of the material, the process struggles to achieve optimal thermal performance.
Rotary calciners have undergone extensive modifications to allow for electrification, with designs featuring both external232 and internal heating233, corresponding to indirect and direct heating as discussed in an earlier section. In externally heated rotary calciners, electrical heating elements surround the rotating cylinder, transferring heat through the cylinder’s shell to the raw meal inside. While the rotary calciner is relatively simple and easily scalable, externally heated versions suffer from significant heat loss through the cylinder’s surface, necessitating expensive, high-temperature-resistant materials. Internally heated rotary calciners, on the other hand, place the heating elements directly inside the rotating cylinder, improving heat transfer efficiency. However, this design presents mechanical challenges, as the heating elements must endure the abrasive and dusty conditions inside the calciner. Nonetheless, experimental studies have achieved calcination rates of up to 95% with this approach.
Among the various designs, the fluidized bed calciner offers the highest heat transfer efficiency due to the excellent mixing of particles in a fluidized state234. In this configuration (an example shown in Fig. 14235), raw meal particles are suspended in an upward-flowing gas stream, promoting efficient heat and mass transfer. One challenge with fluidized bed calciners is the poor fluidization behavior of fine raw meal particles. However, research has demonstrated that mixing raw meals with coarser inert particles can significantly improve fluidization quality. Computational particle fluid dynamics simulations236 have also supported the viability of this concept under hot flow conditions, making the fluidized bed design one of the most efficient options for effective heat transfer.
Design of an externally heated batch fluidized bed reactor for calcination. Adapted from Schwing Technologies235.
Several commercial projects are actively pursuing electrification in cement production. The LEILAC (Low Emissions Intensity Lime And Cement) project57 has contributed valuable insights into the potential of electric heating in cement production. While primarily focused on carbon capture, the project’s pilot plant in Belgium has explored electric heating options for calcination, demonstrating the feasibility of direct separation of CO2 from calcination using electrically (indirectly) heated steel vessel. They employed a direct separation vertical reactor, similar to a drop-tube, which is heated via electricity or alternate fuels. The heat from a furnace is radiated into the calciner tube, releasing CO2 trapped in the raw material. A combination of resistive element heating along with other heating technologies (e.g., biomass, hydrogen) has also been deemed as a more flexible option if the logistical and economic demands can be met. The calciner can be scaled for increased production using a modular array of steel tubes. The LEILAC technology, along with conceptual integration of the LEILAC system in a cement plant is shown in Fig. 15. LEILAC claims this technology can be used as a retrofit strategy and integrated into existing cement plants. The technology is also capable of switching energy sources to enable grid load balancing. LEILAC is implementing their technology at a commercial scale in selected cement plants in Germany, Poland, and the U.S237.
Another notable development is SaltX’s Electric Arc Calciner (EAC), adaptable for several industries, which uses electric arc technology to generate the high temperatures needed for limestone calcination. The electric arc creates a plasma environment for rapid heating of raw materials, potentially reducing the residence time compared to other methods. ZEQL is SaltX’s fully electrified quicklime plant, which includes a calcination unit and a carbon capture unit238. However, challenges such as electrode wear and process control are critical issues that remain to be addressed in scaling up of the process. As described earlier, uncontrolled plasma heating could result in unwanted phase changes and a poor-quality product.
Insights from technoeconomic analysis towards advancing the adoption of electrified technologies
While extensive experimental and limited scaled-up feasibility studies have demonstrated the potential of both electrochemical methods and alternative heating technologies in decarbonizing the cement industry, widespread adoption of these technologies ultimately hinges on their scalability and economic viability in a competitive market. A thorough understanding of the technoeconomic landscape is crucial as it not only informs policy initiatives and investment decisions, but also identifies key cost drivers that require targeted innovation to enable market penetration. Technoeconomic analysis (TEA) generally follows the schematic process outlined in Fig. 16. The first step in TEA of any process involves designing the process flow, which provides a detailed representation of the sequence of operations, equipment needs, and material flows within the system. This design is typically created using process simulation tools, such as Aspen Plus or ECLIPSE, based on experimental data and/or theoretical/numerical models. The process flow diagram includes all major components such as reactors, separators, heat exchangers, and utilities, as well as input-output streams of raw materials, intermediates, and products. Assumptions such as steady-state operating conditions, negligible impact of secondary reactions, and ideal mixing conditions, are often made to ensure practical and computational feasibility of the analysis. The input data to the process simulation models include the reaction kinetics, mass and energy conversion rates, equipment specifications, and operational constraints, alongside economic variables including current market prices of raw materials, energy costs, labor expenses, and potential revenue streams. Next, mass and energy balance are conducted, ensuring that all inputs and outputs are accounted for within the process boundaries. This step helps estimate resource consumption, product production, and emissions, which gives insights into the efficiency of the process at industry-scale. Cost analysis in TEA involves estimating both capital expenditures (CAPEX) and operating expenditures (OPEX), along with requisite additional metrics. In the context of cement production, CAPEX includes initial investments in infrastructure, equipment procurement, installation costs, and infrastructure development. OPEX encompasses ongoing costs such as raw material procurement, energy consumption, maintenance, labor, and administrative expenses. For cement production specifically, the cost of clinker or cement per ton and the CO2 emission per ton are crucial metrics relating to economic and environmental efficiency of the process.
A template demonstrating typical technoeconomic analysis outline for a manufacturing process.
The following subsections provide a succinct economic analysis for some of the electrolysis-based methods and electrified heating technologies for cement manufacture which have been presented in the previous sections. The section focuses on an overview of economic analysis of the technologies that have shown promise, rather than a detailed evaluation and critique of the TEA process.
Electrolysis-based cement production
The electrochemical cement production paradigm faces substantial technological and economic challenges, primarily stemming from significant capital investments and scalability concerns. Key cost drivers include the complex designs of electrolyzer and the ability to provide high throughput, advanced membrane functionality, optimization of catalytic systems, and the need for durable, wear-resistant components. Furthermore, the process exhibits a notably high energy demand for limestone electrolysis, ranging from 5.9 to 11.2 GJ per ton of cement61,105,239. This demand corresponds to the production of one ton of high-purity portlandite as follows: 6.5 GJ‒Zhang et al.19, 6.9 GJ‒Miao et al.60, 7.2 GJ‒ZeroCAL process54, and 10.7 GJ‒Ellis et al.45. These numbers are two to four times higher than the U.S. average energy demand for conventional cement production240.
To address these energy demands, recent studies59,61,174,241 have reported TEA of strategies that utilize gaseous byproducts of electrolysis as energy sources. One approach involves using the O2 stream for oxy-fuel combustion of alternative fuels, in conjunction with H2 combustion for indirect kiln electrification. A recent analysis241 estimated that producing O2 via electrolysis could cost as low as $13 per ton when transported via pipelines, compared to $49 per ton from traditional air separation units. The combined use of O2 and H2 has the potential to lower electricity consumption by ~55%, and reducing the energy demand to 4.8 GJ per ton of clinker in a fully electrified cement plant174. Additionally, a promising scenario presented by Lu et al.84 demonstrated that wollastonite-based cement production (where there is no emission of CO2) could achieve a lower energy demand of 3.96 GJ per ton of cement. However, consistent cement production using this method faces challenges due to the reliance on a stable supply of O2 and H2, especially when depending on variable renewable energy sources. Addressing these supply issues is crucial for achieving the stability required for large-scale industrial implementation of electrochemical cement production.
The most important advantage of electrolysis-based cement production lies in its potential to drastically reduce emissions. Electrolysis, coupled with electrified kiln heating methods or gaseous by-product energy sources offer transformative pathways to decarbonize cement production, likely achieving >90% reduction in process-related CO2 emissions without the need for CCUS105,242,243,244. This approach results in an overall simplification of the decarbonization process, which is crucial in ensuring translation of technologies at-scale. Importantly, the economic feasibility of this transition will be greatly enhanced if low-cost renewable energy is abundantly available, as demonstrated by Liu et al.245. A complementary strategy involves capturing and utilizing pure CO2 from limestone calcination, which further enhances the economic and environmental benefits of electrolysis-based cement manufacturing. This method is particularly advantageous for existing cement plants, as it offers a cost-effective route for decarbonization without requiring capital-intensive infrastructure needed for new plants.
Economic projections61,96,246 suggest that electrochemical cement production could achieve cost parity with conventional methods when supported by carbon pricing mechanisms like carbon taxes, which are taking shape in several economies across the globe. Transitioning to electrified production with partial use of green H2 for kiln heating offers a viable path to decarbonization, which is reported to reduce emissions by up to 80% compared to conventional methods61. Total reduction of emissions is a distinct possibility when all the manufacturing and auxiliary sub-processes are entirely powered by renewable energy54,61,245. Furthermore, this shift has the potential to align the net present value of decarbonizing cement, creating a globally sustainable construction industry. Despite the technological, scale-up, and economic challenges that researchers and industry are working hard to overcome, electrochemical means of cement production paradigm represents a promising solution to deeply decarbonize the cement industry247.
Alternate heating technologies for cement production
Recent economic analysis indicates that, while having the potential to reduce the CO2 emissions by 95–100%, electrification of the process-heat source results in a 95–113% increase in the cost of clinker (COC)248, when CAPEX, OPEX, and electricity, fuel, and CCUS costs are considered. The cost of electricity and OPEX are reported to be the major contributing factors to the increased COC, which in the absence of structured incentives, will drive the industry away from the adoption of decarbonization technologies. Furthermore, the CO2 abatement costs, or the cost of avoided CO2 (CAC), typically lies between $69 and 94 per ton of CO2248. While this number is higher than that for other decarbonization strategies such as material efficiency and circularity, and CCUS alone ($45 per ton of CO2 on average), electrification of process heat generation still holds the potential for net-zero cement manufacturing provided the knowledge and implementation barriers elucidated in the previous sections are overcome.
Though TEA results are highly dependent upon the use-case that is modeled, geographic conditions, and the initial assumptions used, a few of them are presented here to provide the economic contexts under which the technological advancements become implementable at-scale. CAPEX estimates for implementing resistive heating in a cement plant are largely dependent on the retrofitting costs of the required sub-processes, and range between $50 million and $215 million depending on the size of the cement plant66,154. The OPEX structure for resistive heating-based cement plants is dominated by electricity costs, which typically account for ~70% of total operational expenses. At current average industrial electricity prices in the U.S., the operational costs are reported to be in the neighborhood of $100 per ton of cement produced66,154,249 (note that this is roughly half the market price of cement). This is at least 30–40% higher than that of conventional fossil fuel-based production. It is therefore imperative that industrial electricity costs should drop substantially for the electrical resistance-based methods to achieve cost parity. The COC produced using resistive heating-based method is currently estimated to be approximately twice as that made using conventional fossil-fuel driven production, primarily due to the high energy requirements associated with electrified heating250. Despite these challenges, resistive heating technologies combined with carbon capture methods that redirect CO2 to other businesses and lead to significant CAC values could enable the industry to achieve desired levels of economic certainty by offering competitive COC values.
Inductive heating systems present a different economic challenge, characterized by higher initial investment but potentially lower maintenance costs. The CAPEX overhead for inductive heating systems is estimated at $190–300 million for a 1 million ton annual capacity cement plant, which is approximately 40% higher than resistive heating systems for the same plant size, in most cases57,174. With electricity consumption of ~2.8–3.2 GJ per ton of clinker, the OPEX structure for inductive heating-based cement production could be 15–20% lower than resistive heating-based methods57,174. The COC and CAC for inductive heating-based methods will be at-par with those for the resistive heating-based methods, with prices being on the higher end if substantial indirect heating infrastructure is required68.
Based on the CemZero report68 which conducted an economic analysis for a cement plant with a production capacity of 1.3 million tons per year incorporating plasma-based heating and CCS, the CAPEX overhead for plasma heating is estimated at approximately $320 million. This figure is considerably higher than the CAPEX associated with resistive and inductive heating technologies for a plant of the same scale. Plasma-based manufacturing is also a very electricity-intensive operation (which accounts for ~75% OPEX costs), resulting in total OPEX ranging from $100–130 per ton of cement produced68. The COC for plasma heating based-cement production is ~70–93% higher than the conventional methods; however, the CAC for plasma heating is reported to be highly competitive at $82 per ton of CO2174. This value is applicable for the cases where CO2 is used as the carrier gas. However, the CAC estimates become significantly higher if other gases are used for heat transfer. However, plasma-based production has the capability to produce cement in an ultra-fast manner, thereby making the productivity enhancement pay for a part of the high CAPEX and OPEX. The high OPEX in this case, which accounts for the high COC, necessitates the availability of cheap, abundant, and unobstructed renewables-based electricity for the plasma-based production to be economically viable.
Microwave heating-based cement manufacturing is reported to present moderate capital costs but significant operational costs and challenges68. The CAPEX for microwave heating systems is estimated at $195–250 million overhead for a 3 million ton annual capacity cement plant, positioning it between resistive and inductive heating in terms of initial investment154. The primary capital costs are associated with microwave generators, waveguides, and specialized reactor designs. Owing to the low raw materials heating efficiency, microwave heating-based cement production poses higher COC compared to other heating technologies and the CAC remains competitive only in regions with affordable, renewables-based electricity.
Solar heating-based cement production is also reported to be capital investment intensive; however, it presents a low operational cost structure in the places where solar-power is abundant. CAPEX overhead for solar heating installations ranges from $215–320 million65 for typical 1–2 million ton per year plants, largely due to the high cost of solar concentrators and necessary thermal storage solutions. OPEX values are estimated to range from $60–80 per ton of clinker65, which is substantially lower than the other electrified heating options. In high solar power-intensive geographic locations, solar based heating presents highly competitive COC and CAC values.
A concise summary of the electrified heating technologies is presented in tabular form in Table 2. The foregoing discussion indicates that resistive heating could be an attractive option for near-term implementation, given its lowest CAPEX and OPEX among the technologies examined, as well as technology maturation. However, there are still unresolved challenges related to the serviceability of heating elements in the harsh kiln environment. Plasma heating, despite having higher CAPEX and OPEX, offers the advantage of consistently providing high temperatures and the potential recyclability of CO2 from limestone calcination as the plasma gas. Its economic viability could improve significantly with technological advancements, particularly in electrode longevity and the advent of cheap electricity from renewables. Microwave heating presents an interesting case with mid-range costs, but its practical implementation faces challenges due to the poor microwave absorption of cement raw materials. Solar heating, while having the highest CAPEX amongst the options considered, offers the potential for near-zero direct CO2 emissions and long-term operational cost stability in the regions with solar power abundance. It is important to note that these technologies are at various levels of readiness, and their economic profiles are likely to change as technologies mature. One common thread in all of these is that the economic and environmental viability of these technologies are highly dependent on renewable electricity availability and cost. Furthermore, carbon pricing, incentive structures, and regional policy frameworks will play a key role in scaled-up applications and translation of electrified technologies. Future cost reductions through technological advancement and economies of scale, combined with the adoption of increasingly stringent carbon regulations by governments and agencies, could significantly improve the chances of large-scale adoption of electrified cement production technologies.
Perspective and summary on electrification for deep decarbonization of cement manufacturing
Cement production remains one of the largest contributors to global CO₂ emissions, accounting for nearly 8% of all anthropogenic emissions—a staggering figure that underscores the urgency of developing transformative technologies for deep decarbonization. While traditional approaches have relied heavily on incremental improvements, it is increasingly clear that radical innovation is necessary to address the dual challenges of carbon emissions and energy intensity in cement manufacturing. High-risk, high-reward pathways—such as electrochemical synthesis of cement precursors, novel carbon-efficient raw materials, and CCUS—are emerging as promising alternatives, yet their success hinges critically on electrification technologies powered by renewable energy sources.
Electrification strategies offer a unique opportunity to decouple cement production from fossil fuel dependence, particularly when paired with carbon-free or carbon-efficient precursor production methods. Advanced electrolysis approaches—ranging from aqueous electrolysis to solar-thermal and seawater-based variants—show significant potential to reduce emissions by as much as 60%, enabling the synthesis of low-carbon precursors like lime or portlandite. Similarly, alternative heating technologies, including resistive, inductive, solar, plasma, and microwave heating, offer pathways for achieving the extreme temperatures required for clinker production. However, each of these solutions faces distinct challenges: the immense energy demand, supply-chain bottlenecks that could prohibit or limit the use of certain materials or technologies in certain areas of the world, technical limitations in achieving thermal uniformity and peak temperatures at scale, and prohibitive capital costs remain barriers to widespread industrial implementation. Moreover, risks presented by these novel technologies in cement production are exacerbated by the extremely high capital costs (~$1B for a 1 million tons per year plant) and opportunity costs resulting from downtime needed to maintain and service these new technologies; a representative 1–1.5 million tons per year plant could represent ~$100–200 million in opportunity costs if they experience a downtime of one year.
Addressing these challenges will require the convergence of material science, energy systems engineering, and advanced modeling approaches. Innovative refractory materials that enhance thermal efficiency under non-uniform heating, coupled with improved electrode and electrolyte designs for electrolysis systems, are critical for overcoming current technical bottlenecks. Hybrid heating systems, integrating renewable electricity with supplemental methods like plasma or microwaves, could provide a pragmatic bridge between conventional and fully electrified processes. Furthermore, advanced control systems leveraging real-time monitoring and multiphysics modeling—spanning electrical, chemical, and fluid dynamics—will play a pivotal role in optimizing energy efficiency and process reliability, and are likely to be critical components of a low-carbon cement manufacturing ecosystem.
The electrification of materials manufacturing holds immense promise for reducing carbon emissions; however, this transition can only achieve its full environmental potential if electricity production itself becomes sustainable. Materials manufacturing processes—especially cement production—are highly energy-intensive, demanding reliable and continuous power supplies. To meet this demand sustainably, renewable energy sources such as solar, wind, hydroelectric, and geothermal must be integrated at scale into the energy grid, complemented by advancements in energy storage technologies like batteries or green hydrogen. These solutions not only decarbonize power generation but also align with long-term goals of net-zero emissions. However, the intermittency of renewable energy sources remains a major challenge. Without robust energy storage solutions, fluctuations in power availability could introduce instability into cement manufacturing operations. Additional challenges such as the need for substantial grid infrastructure upgrades, and the geographic mismatch between renewable energy availability and industrial hubs exist. Moreover, the cost of transitioning to renewable power—alongside the development of technologies to improve energy efficiency within manufacturing—is often prohibitive without supportive policies and incentives. Addressing these challenges requires collaborative efforts among governments, industry leaders, and researchers to create a resilient, sustainable, and equitable energy system capable of powering the material foundations of modern society while minimizing environmental harm.
Looking forward, the pathway to decarbonizing cement manufacturing lies at the intersection of technological innovation, renewable energy integration, and systems-level optimization. Pilot-scale demonstrations will be instrumental in addressing economic and operational uncertainties, while fostering collaboration between academia, industry stakeholders, and policymakers will ensure the translation of these breakthroughs into scalable solutions. By investing in electrification technologies and their enabling systems, the cement industry can pave the way for a cleaner, more sustainable future—one where deep decarbonization is not merely aspirational but achievable.
Data availability
No datasets were generated or analyzed during the current study.
References
Number of recorded natural disaster events. Our World in Data. https://ourworldindata.org/grapher/number-of-natural-disaster-events (accessed 19 Aug 2024).
Benevolenza, M. A. & DeRigne, L. The impact of climate change and natural disasters on vulnerable populations: a systematic review of literature. J. Hum. Behav. Soc. Environ. 29, 266–281 (2019).
Islam, S., Chu, C., Smart James, C. R. & Liew, L. Integrating disaster risk reduction and climate change adaptation: a systematic literature review. Clim. Dev. 12, 255–267 (2020).
United Nations. Causes and Effects of Climate Change. https://www.un.org/en/climatechange/science/causes-effects-climate-change (accessed 19 Aug 2024) (United Nations, 2024).
Manabe, S. Role of greenhouse gas in climate change**. Tellus Dyn. Meteorol. Oceanogr. 71, 1620078 (2019).
Filonchyk, M., Peterson, M. P., Zhang, L., Hurynovich, V. & He, Y. Greenhouse gases emissions and global climate change: examining the influence of CO2, CH4, and N2O. Sci. Total Environ. 935, 173359 (2024).
Rissman, J. et al. Technologies and policies to decarbonize global industry: review and assessment of mitigation drivers through 2070. Appl. Energy 266, 114848 (2020).
Boa Morte, I. B., Araújo, O., de, Q. F., Morgado, C. R. V. & de Medeiros, J. L. Electrification and decarbonization: a critical review of interconnected sectors, policies, and sustainable development goals. Energy Storage Sav 2, 615–630 (2023).
Davis, S. J. et al. Net-zero emissions energy systems. Science 360, eaas9793 (2018).
Worldsteel Association. Buildings and infrastructure. worldsteel.org. https://worldsteel.org/steel-topics/buildings-and-infrastructure/ (accessed 27 Oct 2024).
Annual CO₂ emissions from cement. Our World in Data. https://ourworldindata.org/grapher/annual-co2-cement (accessed 2 Mar 2025).
United Nations. Growing at a slower pace, world population is expected to reach 9.7 billion in 2050 and could peak at nearly 11 billion around 2100. https://www.un.org/development/desa/en/news/population/world-population-prospects-2019.html (accessed 27 Sep 2024).
United Nations. The World’s Cities in 2018-Data Booklet (United Nations, 2018).
University of Southern California. Sea level rise could reshape the United States, trigger migration inland. ScienceDaily. https://www.sciencedaily.com/releases/2020/01/200122150021.htm (accessed 27 Oct 2024).
Cheng, D. et al. Projecting future carbon emissions from cement production in developing countries. Nat. Commun. 14, 8213 (2023).
Schneider, M. The cement industry on the way to a low-carbon future. Cem. Concr. Res. 124, 105792 (2019).
Watts, J. Concrete: the most destructive material on earth. The Guardian. February 25, 2019. https://www.theguardian.com/cities/2019/feb/25/concrete-the-most-destructive-material-on-earth (accessed 24 Sep 2020).
Mishra, U. C., Sarsaiya, S. & Gupta, A. A systematic review on the impact of cement industries on the natural environment. Environ. Sci. Pollut. Res. 29, 18440–18451 (2022).
Supriya et al. Low-CO2 emission strategies to achieve net zero target in cement sector. J. Clean. Prod. 417, 137466 (2023).
Benhelal, E., Shamsaei, E. & Rashid, M. I. Challenges against CO2 abatement strategies in cement industry: a review. J. Environ. Sci. 104, 84–101 (2021).
Cement. IEA. https://www.iea.org/energy-system/industry/cement (accessed 5 Nov 2024).
Watari, T., Cao, Z., Hata, S. & Nansai, K. Efficient use of cement and concrete to reduce reliance on supply-side technologies for net-zero emissions. Nat. Commun. 13, 4158 (2022).
Nehdi, M. L., Marani, A. & Zhang, L. Is net-zero feasible: systematic review of cement and concrete decarbonization technologies. Renew. Sustain. Energy Rev. 191, 114169 (2024).
The Paris Agreement—Publication|UNFCCC. https://unfccc.int/documents/184656 (accessed 5 Nov 2024).
Mokhtar, A. & Nasooti, M. A decision support tool for cement industry to select energy efficiency measures. Energy Strat. Rev. 28, 100458 (2020).
Mikulčić, H., Klemeš, J. J., Vujanović, M., Urbaniec, K. & Duić, N. Reducing greenhouse gasses emissions by fostering the deployment of alternative raw materials and energy sources in the cleaner cement manufacturing process. J. Clean. Prod. 136, 119–132 (2016).
Shah, I. H., Miller, S. A., Jiang, D. & Myers, R. J. Cement substitution with secondary materials can reduce annual global CO2 emissions by up to 1.3 gigatons. Nat. Commun. 13, 5758 (2022).
IEA. Technology Roadmap—Low-Carbon Transition in the Cement Industry. https://www.iea.org/reports/technology-roadmap-low-carbon-transition-in-the-cement-industry (accessed 25 Oct 2024).
Cook, R., Ma, H. & Kumar, A. Influence of size-classified and slightly soluble mineral additives on hydration of tricalcium silicate. J. Am. Ceram. Soc. 103, 2764–2779 (2020).
Schöler, A. et al. Early hydration of SCM-blended portland cements: a pore solution and isothermal calorimetry study. Cem. Concr. Res. 93, 71–82 (2017).
Dhandapani, Y., Santhanam, M., Kaladharan, G. & Ramanathan, S. Towards ternary binders involving limestone additions—a review. Cem. Concr. Res. 143, 106396 (2021).
Tavares, L. R. C. et al. Influence of quartz powder and silica fume on the performance of portland cement. Sci. Rep. 10, 21461 (2020).
Bhat, R., Han, T., Sant, G., Neithalath, N. & Kumar, A. A comprehensive analysis of hydration kinetics and compressive strength development of fly ash-portland cement binders. J. Build. Eng. 88, 109191 (2024).
Mondal, S. K., Clinton, C., Ma, H., Kumar, A. & Okoronkwo, M. U. Effect of Class C and Class F fly ash on early-age and mature-age properties of calcium sulfoaluminate cement paste. Sustainability 15, 2501 (2023).
Bullerjahn, F. & Bolte, G. Composition of the reactivity of engineered slags from bauxite residue and steel slag smelting and use as SCM for portland cement. Constr. Build. Mater. 321, 126331 (2022).
Qureshi, L. A., Ali, B. & Ali, A. Combined effects of supplementary cementitious materials (silica fume, GGBS, fly ash and rice husk ash) and steel fiber on the hardened properties of recycled aggregate concrete. Constr. Build. Mater. 263, 120636 (2020).
Siddique, R. Use of municipal solid waste ash in concrete. Resour. Conserv. Recycl. 55, 83–91 (2010).
Siddique, R. Utilization of Municipal Solid Waste (MSW) ash in cement and mortar. Resour. Conserv. Recycl. 54, 1037–1047 (2010).
Shih, P.-H., Chang, J.-E. & Chiang, L.-C. Replacement of raw mix in cement production by municipal solid waste incineration ash. Cem. Concr. Res. 33, 1831–1836 (2003).
ASH01-ARTBA-Forecast-Update, 2020. https://acaa-usa.org/wp-content/uploads/2024/09/ASH01-ARTBA-Forecast-Update.pdf.
Abdul-Wahab, S. A., Al-Dhamri, H., Ram, G. & Chatterjee, V. P. An overview of alternative raw materials used in cement and clinker manufacturing. Int. J. Sustain. Eng. 14, 743–760 (2021).
Costa, F. N. & Ribeiro, D. V. Reduction in CO2 emissions during production of cement, with partial replacement of traditional raw materials by civil construction waste (CCW). J. Clean. Prod. 276, 123302 (2020).
Her, S., Park, J., Li, P. & Bae, S. Feasibility study on utilization of pulverized eggshell waste as an alternative to limestone in raw materials for portland cement clinker production. Constr. Build. Mater. 324, 126589 (2022).
Schneider, M., Romer, M., Tschudin, M. & Bolio, H. Sustainable cement production—present and future. In: Proc. 13th International Congress on Chemistry of Cement. 41, 642–650 (2011).
Ellis, L. D., Badel, A. F., Chiang, M. L., Park, R. J.-Y. & Chiang, Y.-M. Toward electrochemical synthesis of cement—an electrolyzer-based process for decarbonating CaCO3 while producing useful gas streams. Proc. Natl. Acad. Sci. 117, 12584–12591 (2020).
Plaza, M. G., Martínez, S. & Rubiera, F. CO2 capture, use, and storage in the cement industry: state of the art and expectations. Energies 13, 5692 (2020).
Tsivilis, S., Chaniotakis, E., Badogiannis, E., Pahoulas, G. & Ilias, A. A study on the parameters affecting the properties of portland limestone cements. Cem. Concr. Compos. 21, 107–116 (1999).
Bushi, L., Meil, J. An environmental life cycle assessment of portlandlimestone and ordinary portland cements in concrete. Technical brief (2013).
Scrivener, K., Martirena, F., Bishnoi, S. & Maity, S. Calcined clay limestone cements (LC3). Cem. Concr. Res. 114, 49–56 (2018).
Scrivener, K. L. Options for the Future of Cement (2014).
Technology. Sublime Systems. https://sublime-systems.com/technology/ (accessed 5 Nov 2024).
Brimstone | Technology. Brimstone. https://www.brimstone.com/technology (accessed 5 Nov 2024).
Wexler, P. H. Turning Gray into Green. Medium. https://patriciahalfenwexler.medium.com/turning-gray-into-green-60a21c446760 (accessed 5 Nov 2024).
Leão, A., et al. ZeroCAL: Eliminating carbon dioxide emissions from limestone’s decomposition to decarbonize cement production. ACS Sustain. Chem. Eng. acssuschemeng.4c03193. https://doi.org/10.1021/acssuschemeng.4c03193 (2024).
Product and technology—Cambridge Electric Cement. https://cambridgeelectriccement.com/the-product/ (accessed 14 Oct 2024).
Industrial Process Heating with RotoDynamic Heater. Coolbrook. https://coolbrook.com/electrification-solutions/rdh-industrial-process-heating/ (accessed 5 Nov 2024).
Hills, T. P., Sceats, M., Rennie, D. & Fennell, P. LEILAC: low cost CO2 capture for the cement and lime industries. Energy Procedia 114, 6166–6170 (2017).
How We Do It—Fortera Corporation. https://forteraglobal.com/how-we-do-it/ (accessed 5 Nov 2024).
Mowbray, B. A. W., Zhang, Z. B., Parkyn, C. T. E. & Berlinguette, C. P. Electrochemical cement clinker precursor production at low voltages. ACS Energy Lett. 8, 1772–1778 (2023).
Zhang, Z., Mowbray, B., Parkyn, C., Kim, Y., Berlinguette, C. Electrolytic cement clinker production sustained through orthogonalization of ion vectors. ChemRxiv, https://doi.org/10.26434/chemrxiv-2023-0zcw9 (2023).
Miao, R. K. et al. Electrified cement production via anion-mediated electrochemical calcium extraction. ACS Energy Lett. 4694–4701 (2023).
Bahnasawy, N., Al Anany, S. & Allam, N. K. Electrochemical catalysis for the production of green cement: towards decarbonizing the cement industry. Catal. Sci. Technol. 14, 4087–4105 (2024).
How electrification can help industrial companies cut costs | McKinsey. https://www.mckinsey.com/industries/electric-power-and-natural-gas/our-insights/plugging-in-what-electrification-can-do-for-industry (accessed 17 Sep 2024).
Dunant, C. F., Joseph, S., Prajapati, R. & Allwood, J. M. Electric recycling of portland cement at scale. Nature 629, 1055–1061 (2024).
Moumin, G. et al. CO2 emission reduction in the cement industry by using a solar calciner. Renew. Energy 145, 1578–1596 (2020).
Laurini, B., Zong, Y., Træholt, C., Parra, S., Shanmugasundaram, M. Techno-economic analysis on the integration of an electrified clay calcination process into a cement plant. https://doi.org/10.46855/energy-proceedings-10989 (2024).
Vermeiren, J., Dilissen, N., Goovaerts, V. & Vleugels, J. Electrification of clinker and calcination treatments in the cement sector by microwave technology—a review. Constr. Build. Mater. 428, 136271 (2024).
Wilhelmsson B., Kollberg C., Larsson J., Eriksson M. CemZero—a feasibility study evaluating ways to reach sustainable cement production via the use of electricity. https://www.cement.heidelbergmaterials.se/sites/default/files/assets/document/65/de/final_cemzero_2018_public_version_2.0.pdf.pdf (accessed 7 Aug 2024).
ACI Committee 318, Building Code Requirements for Structural Concrete (ACI 318-08) and Commentary (American Concrete Institute, 2008).
Mindess, S. Developments in the Formulation and Reinforcement of Concrete (Woodhead Publishing, 2019).
Schneider, M., Hoenig, V., Ruppert, J. & Rickert, J. The cement plant of tomorrow. Cem. Concr. Res. 173, 107290 (2023).
Meijssen, M., Becattini, V. & Mazzotti, M. Integrated carbon capture and utilization in the cement industry: a comparative study. ACS Sustain. Chem. Eng. 12, 2709–2718 (2024).
Goldman, S., et al. Pathways to Commercial Liftoff: Low-Carbon Cement. https://liftoff.energy.gov/wp-content/uploads/2023/12/20230921-Pathways-to-Commercial-Liftoff-Cement.pdf (Department of Energy, 2023).
Gonçalves, J. P. et al. Toward smart and sustainable cement manufacturing process: analysis and optimization of cement clinker quality using thermodynamic and data-informed approaches. Cem. Concr. Compos. 147, 105436 (2024).
Ateş, K. T., Şahin, C., Kuvvetli, Y., Küren, B. A. & Uysal, A. Sustainable production in cement via artificial intelligence based decision support system: case study. Case Stud. Constr. Mater. 15, e00628 (2021).
Kim, B., Kim, M., Jeong, J. Estimation of greenhouse gas emissions in cement manufacturing process through blockchain and SSL based IoT data analysis. In: Gervasi, O., Murgante, B., Misra, S., Garau, C., Blečić, I., Taniar, D., Apduhan, B. O., Rocha, A. M. A. C., Tarantino, E., Torre, C. M., Karaca, Y., Eds. Computational Science and Its Applications—ICCSA 2020; 634–645 (Springer International Publishing, 2020).
Usman, M., Ahmad, I., Ahsan, M., Caliskan, H. Prediction and optimization of emissions in cement manufacturing plant under uncertainty by using artificial intelligence-based surrogate modeling. Environ. Dev. Sustain. https://doi.org/10.1007/s10668-024-05068-5 (2024).
Licht, S. Efficient solar-driven synthesis, carbon capture, and desalinization, STEP: solar thermal electrochemical production of fuels, metals, bleach. Adv. Mater. 23, 5592–5612 (2011).
Licht, S. et al. STEP cement: solar thermal electrochemical production of CaO without CO2 emission. Chem. Commun. 48, 6019 (2012).
Simoni, M. et al. Decarbonising the lime industry: state-of-the-art. Renew. Sustain. Energy Rev. 168, 112765 (2022).
Dowling, A., O’Dwyer, J. & Adley, C. C. Lime in the limelight. J. Clean. Prod. 92, 13–22 (2015).
Ochoa George, P. A., Gutiérrez, A. S., Cogollos Martínez, J. B. & Vandecasteele, C. Cleaner production in a small lime factory by means of process control. J. Clean. Prod. 18, 1171–1176 (2010).
Sagastume Gutiérrez, A., Van Caneghem, J., Cogollos Martínez, J. B. & Vandecasteele, C. Evaluation of the Environmental Performance of Lime Production in Cuba. J. Clean. Prod. 31, 126–136 (2012).
Lu, X. K., Zhang, W., Ruggiero, B. N., Seitz, L. C., Li, J. Scalable electrified cementitious materials production and recycling. Energy Environ. Sci. https://doi.org/10.1039/D4EE03529A, 10.1039.D4EE03529A (2024).
Castaño, S. V., La Plante, E. C., Collin, M., Sant, G. & Pilon, L. A pilot-process for calcium hydroxide production from iron slag by low-temperature precipitation. J. Environ. Chem. Eng. 10, 107792 (2022).
Kirchofer, A., Becker, A., Brandt, A. & Wilcox, J. CO2 mitigation potential of mineral carbonation with industrial alkalinity sources in the United States. Environ. Sci. Technol. 47, 7548–7554 (2013).
La Plante, E. C. et al. Electrolytic seawater mineralization and the mass balances that demonstrate carbon dioxide removal. ACS EST Eng. 3, 955–968 (2023).
La Plante, E. C. et al. Saline water-based mineralization pathway for gigatonne-scale CO2 management. ACS Sustain. Chem. Eng. 9, 1073–1089 (2021).
Park, H. S. et al. Comparison of two processes forming CaCO3 precipitates by electrolysis. Energies 9, 1052 (2016).
Ramirez-Amaya, D. et al. M. Comparison of the electrochemical decarbonation of different-grade limestones used in cement manufacturing. Cem. Concr. Res. 174, 107307 (2023).
Martínez, N. P. et al. High-performance and low-cost electrochemical reactor for limestone decarbonation applied to clinker production—a validation at laboratory scale. J. Clean. Prod. 468, 143112 (2024).
Daubinger, P., Kieninger, J., Unmüssig, T. & Urban, G. A. Electrochemical characteristics of nanostructured platinum electrodes—a cyclic voltammetry study. Phys Chem Chem Phys 16, 8392–8399 (2014).
Jacobse, L., Raaijman, S. J. & Koper, M. T. M. The reactivity of platinum microelectrodes. Phys. Chem. Chem. Phys. 18, 28451–28457 (2016).
GCCS | Concrete Future - The GCCA 2050 Cement and Concrete Industry Roadmap for Net Zero Concrete. GCCS | Staging. https://gccassociation.org/concretefuture/ (accessed 1 May 2025).
Zhou, C., Ni, J., Chen, H. & Guan, X. Harnessing electrochemical pH gradient for direct air capture with hydrogen and oxygen by-products in a calcium-based loop. Sustain. Energy Fuels 5, 4355–4367 (2021).
Xie, Q., Wan, L., Zhang, Z., Luo, J. Electrochemical transformation of limestone into calcium hydroxide and valuable carbonaceous products for decarbonizing cement production. iScience, 26 https://doi.org/10.1016/j.isci.2023.106015 (2023).
Goto, K. et al. Effect of CO2 purity on energy requirement of CO2 capture processes. Energy Procedia 37, 806–812 (2013).
Ma, X. et al. Sustainable electrolytes: design principles and recent advances. Chem. Eur. J. 30, e202400332 (2024).
Singh, B., Ghosh, S., Aich, S. & Roy, B. Low temperature solid oxide electrolytes (LT-SOE): a review. J. Power Sources 339, 103–135 (2017).
Pozio, A., Bozza, F., Nigliaccio, G., Platter, M. & Monteleone, G. Development perspectives on low-temperature electrolysis. Energ Ambiente Innov. 1, 66–72 (2021).
Li, W. et al. Low-temperature water electrolysis: fundamentals, progress, and new strategies. Mater. Adv. 3, 5598–5644 (2022).
Ayers, K. et al. Perspectives on low-temperature electrolysis and potential for renewable hydrogen at scale. Annu. Rev. Chem. Biomol. Eng. 10, 219–239 (2019).
Grigoriev, S. A., Fateev, V. N., Bessarabov, D. G. & Millet, P. Current status, research trends, and challenges in water electrolysis science and technology. Int. J. Hydrog. Energy 45, 26036–26058 (2020).
Asghari, E., Abdullah, M. I., Foroughi, F., Lamb, J. J. & Pollet, B. G. Advances, opportunities, and challenges of hydrogen and oxygen production from seawater electrolysis: an electrocatalysis perspective. Curr. Opin. Electrochem. 31, 100879 (2022).
Zhang, Z. et al. Cement clinker precursor production in an electrolyser. Energy Environ. Sci. 15, 5129–5136 (2022).
Rouxhet, R., Loudeche, M., Santoro, R. & Proost, J. Low-temperature water electrolysis under a sustained ph-gradient for electrochemically-induced decarbonation of limestone into hydrated lime. J. Electrochem. Soc. 171, 094504 (2024).
Zhang, Z., et al. Electrolytic cement clinker precursor production sustained through orthogonalization of ion vectors. Energy Environ. Sci. 10.1039.D4EE04881D. https://doi.org/10.1039/D4EE04881D (2025).
Hart, J. R. Ethylenediaminetetraacetic acid and related chelating agents. In Ullmann’s Encyclopedia of Industrial Chemistry. p a10_095.pub2. https://doi.org/10.1002/14356007.a10_095.pub2 (Wiley-VCH Verlag GmbH & Co. KGaA, 2011).
Zhu, Y., Fan, W., Zhou, T. & Li, X. Removal of chelated heavy metals from aqueous solution: a review of current methods and mechanisms. Sci. Total Environ. 678, 253–266 (2019).
Dow. Chelation Chemistry: General Concepts of the Chemistry of Chelation. https://www.dow.com/content/dam/dcc/documents/en-us/app-tech-guide/113/113-01388-01-chelation-chemistry-general-concepts-of-the-chemistry-of-chelation.pdf (2021).
U.S. Energy Information Administration. Quarterly Coal Distribution Report—Energy Information Administration. https://www.eia.gov/coal/distribution/quarterly/ (accessed 18 Apr 2018).
IPCC—Intergovernmental Panel on Climate Change. Annex III: technology-specific cost and performance parameters. https://www.ipcc.ch/site/assets/uploads/2018/02/ipcc_wg3_ar5_annex-iii.pdf (2014).
BBC. Climate change: the massive CO2 emitter you may not know about. https://www.bbc.com/news/science-environment-46455844 (2018).
McKinsey & Company. Laying the foundation for zero-carbon cement. https://www.mckinsey.com/industries/chemicals/our-insights/laying-the-foundation-for-zero-carbon-cement (2020).
Stathopoulos, P., Sleem, T. & Paschereit, C. O. Steam generation with stoichiometric combustion of H2/O2 as a way to simultaneously provide primary control reserve and energy storage. Appl. Energy 205, 692–702 (2017).
Wang, Y. et al. Life Cycle Assessment of combustion-based electricity generation technologies integrated with carbon capture and storage: a review. Environ. Res. 207, 112219 (2022).
Meier, A., Bonaldi, E., Cella, G. M., Lipinski, W. & Wuillemin, D. Solar chemical reactor technology for industrial production of lime. Sol. Energy 80, 1355–1362 (2006).
Ren, J., Lau, J., Lefler, M. & Licht, S. The minimum electrolytic energy needed to convert carbon dioxide to carbon by electrolysis in carbonate melts. J. Phys. Chem. C 119, 23342–23349 (2015).
Wang, B., Gu, D., Dong, J., Yuan, D. & Zhu, L. STEP chemistry: a fundamental insight into solar thermal electrochemical process. Energy Convers. Manag. 152, 99–109 (2017).
Licht, S. STEP (solar thermal electrochemical photo) generation of energetic molecules: a solar chemical process to end anthropogenic global warming. J. Phys. Chem. C 113, 16283–16292 (2009).
Bustillo Revuelta, M. Lime. In Construction Materials; Springer Textbooks in Earth Sciences, Geography and Environment 167–193 (Springer International Publishing, 2021).
FitzGerald D., et al. Documentation of changes implemented in the ecoinvent database v3.10. https://19913970.fs1.hubspotusercontent-na1.net/hubfs/19913970/Knowledge%20Base/Database/Releases/Change%20Report%20v3.10%20-%2020231214.pdf (2023).
Wernet, G. et al. The Ecoinvent Database Version 3 (Part I): overview and methodology. Int. J. Life Cycle Assess. 21, 1218–1230 (2016).
Kellenberger, D., et al. Life Cycle Inventories of Building Products. https://db.ecoinvent.org/reports/07_BuildingProducts.pdf?area=463ee7e58cbf8.
Licht, S. et al. A new solar carbon capture process: Solar Thermal Electrochemical Photo (STEP) carbon capture. J. Phys. Chem. Lett. 1, 2363–2368 (2010).
Deng, Y. et al. Molten salt electrolysis: promising technology to capture and transform CO2 into valuable carbon materials. Sustain. Energy Fuels https://doi.org/10.1039/D4SE00819G (2024).
Weng, W., Tang, L. & Xiao, W. Capture and electro-splitting of CO2 in molten salts. J. Energy Chem. 28, 128–143 (2019).
Ren, J. et al. Recent Advances in solar thermal electrochemical process (STEP) for carbon neutral products and high value nanocarbons. Acc. Chem. Res. 52, 3177–3187 (2019).
Thapaliya, B. P. et al. Low-temperature molten salt electrochemical CO2 upcycling for advanced energy materials. ACS Appl. Mater. Interfaces 16, 2251–2262 (2024).
Yang, Z., Yin, H., Deng, B. & Wang, D. Electrolyte engineering for efficient molten-carbonate electrolysis of CO2. Chem. Eng. J. 473, 145146 (2023).
Kaplan, V., Wachtel, E. & Lubomirsky, I. CO2 to CO electrochemical conversion in molten Li2CO3 is stable with respect to sulfur contamination. J. Electrochem. Soc. 161, F54–F57 (2014).
Kaplan, V., Wachtel, E. & Lubomirsky, I. Conditions of stability for (Li2CO3+ Li2O) melts in air. J. Chem. Thermodyn. 43, 1623–1627 (2011).
Bromberg, L., Nitzsche, M. P. & Hatton, T. A. Capture and electrochemical conversion of CO2 in molten alkali metal borate–carbonate blends. Nanoscale 14, 13141–13154 (2022).
Nitzsche, M. P., Bromberg, L. & Hatton, T. A. Capture and electrochemical reduction of CO2 using molten alkali metal borates. ACS Sustain. Chem. Eng. 11, 11012–11018 (2023).
Carbon Corp. Carbon XPRIZE. https://www.xprize.org/prizes/carbon/teams/carboncorp (accessed 20 Oct 2024).
Licht, S. Carbon dioxide to carbon nanotube scale-up. arXiv October 19, 2017. http://arxiv.org/abs/1710.07246 (accessed 21 Oct 2024).
Pornrungroj, C. et al. Bifunctional perovskite-BiVO4 tandem devices for uninterrupted solar and electrocatalytic water splitting cycles. Adv. Funct. Mater. 31, 2008182 (2021).
Ho, H.-J. & Iizuka, A. Mineral carbonation using seawater for CO2 sequestration and utilization: a review. Sep. Purif. Technol. 307, 122855 (2023).
Hilbertz, W. Electrodeposition of minerals in sea water: experiments and applications. IEEE J. Ocean. Eng. 4, 94–113 (1979).
Li, Y., Xie, L. & Su, T. Carbon dioxide mineralization by electrode separation for quick carbon reduction and sequestration in acidified seawater. Carbon Manag 14, 2202167 (2023).
Chen, Q., Choo, Y., Akther, N., Shon, H. K. & Naidu, G. Generating sustainable cement material from seawater with low-cost ultrafiltration (UF) membrane electrolysis. Chem. Eng. J. 494, 153007 (2024).
Devi, N. et al. Mechanistic insights into electrodeposition in seawater at variable electrochemical potentials. Adv. Sustain. Syst. 8, 2300446 (2024).
Bernard, E. et al. MgO-based cements—current status and opportunities. RILEM Tech. Lett. 8, 65–78 (2023).
National Academies of Sciences, Engineering, and Medicine; Division on Earth and Life Studies; Board on Chemical Sciences and Technology; Committee on Developing a Research Agenda for Utilization of Gaseous Carbon Waste Streams. Gaseous Carbon Waste Streams Utilization: Status and Research Needs (National Academies Press, 2019).
United States Geological Survey. Limestone; A Crucial and Versatile Industrial Mineral Commodity. https://pubs.usgs.gov/fs/2008/3089/ (accessed 4 Apr 2018).
United States Geological Survey. Crushed Stone Statistics and Information. https://pubs.usgs.gov/periodicals/mcs2020/mcs2020-stone-crushed.pdf (2020).
Driver, J. G. et al. Global decarbonization potential of CO2 mineralization in concrete materials. Proc. Natl. Acad. Sci. 121, e2313475121 (2024).
Badjatya, P. et al. Carbon-negative cement manufacturing from seawater-derived magnesium feedstocks. Proc. Natl. Acad. Sci. 119, e2114680119 (2022).
Hills, T., Leeson, D., Florin, N. & Fennell, P. Carbon capture in the cement industry: technologies, progress, and retrofitting. Environ. Sci. Technol. 50, 368–377 (2016).
Kim, S. et al. Asymmetric chloride-mediated electrochemical process for CO2 removal from oceanwater. Energy Environ. Sci. 16, 2030–2044 (2023).
The project. ELECTRA. https://www.electra-horizon.eu/the-project/ (accessed 19 Nov 2024).
Home—Electrified processes for industry without carbon - EPIXC. https://epixc.org/ (accessed 19 Nov 2024).
Dhas, S., Blok, K. & Kleijn, R. Decarbonisation Pathways for the Cement Industry. Master Thesis (TU Delft, 2021).
Hoenig, V. ECRA CCS Project-Report on Phase III.TR-ECRA-119/2012. Technical Report (ECRA, 2012).
Gabaldón-Estevan, D., Mezquita, A., Ferrer, S. & Monfort, E. Unwanted effects of european union environmental policy to promote a post-carbon industry. the case of energy in the European Ceramic Tile Sector. J. Clean. Prod. 117, 41–49 (2016).
Hornberger, M., Spörl, R. & Scheffknecht, G. Calcium looping for CO2 capture in cement plants—Pilot Scale Test. Energy Procedia 114, 6171–6174 (2017).
Bai, Z., Liu, Q., Lei, J., Li, H. & Jin, H. A polygeneration system for the methanol production and the power generation with the solar–biomass thermal gasification. Energy Convers. Manag. 102, 190–201 (2015).
Gazzani, M., et al. CEMCAP Framework for Comparative Techno Economic Analysis of CO2 Capture from Cement Plants
VDZ Activity Report 2015–2018 published. VDZ. https://www.vdz-online.de/en/news-1/vdz-activity-report-2015-2018-published (accessed 17 Sep 2024) (2015).
Combined calcination and CO2 capture in cement clinker production by use of CO2-neutral electrical energy – Phase 2 (ELSE 2). Climit. https://climit.no/en/project/combined-calcination-and-co2-capture-in-cement-clinker-production-by-use-of-co2-neutral-electrical-energy-phase-2-else-2/ (accessed 17 Sep 2024).
Eemeli T., et al. Reducing the climate impacts of industry through carbon-neutral energy and the circular economy—decarbonate. 2022.
Heidelberg Cement to install the world’s first full-scale CCS facility in a cement plant. https://www.heidelbergmaterials.com/en/pr-15-12-2020 (accessed 17 Sep 2024).
Bosoaga, A., Masek, O. & Oakey, J. E. CO2 Capture technologies for cement industry. Energy Procedia 1, 133–140 (2009).
Baker, R. W., Freeman, B., Kniep, J., Huang, Y. I. & Merkel, T. C. CO2 capture from cement plants and steel mills using membranes. Ind. Eng. Chem. Res. 57, 15963–15970 (2018).
Electrification of Lime and Cement Production – Neithalath Lab. https://faculty.engineering.asu.edu/neithalath/electrification-of-lime-and-cement-production/ (accessed 19 Nov 2024).
Deng, Y. et al. The steam-assisted calcination of limestone and dolomite for energy savings and to foster solar calcination processes. J. Clean. Prod. 363, 132640 (2022).
Roelofsen, O., Somers, K., Speelman, E., Witteveen, M. Plugging in: what electrification can do for industry. McKinsey & Co. 2020, 28.
Pisciotta, M., Pilorgé, H., Davids, J. & Psarras, P. Opportunities for cement decarbonization. Clean. Eng. Technol. 15, 100667 (2023).
Lucía, O., Maussion, P., Dede, E. J. & Burdío, J. M. Induction heating technology and its applications: past developments, current technology, and future challenges. IEEE Trans. Ind. Electron. 61, 2509–2520 (2014).
Effects of changing permeability and resistivity. Conduct. Induction Heat. 123–129.
Tokheim, L.-A., et al. Combined calcination and CO2 capture in cement clinker production by use of electrical energy. In Proc. Trondheim CCS Conference CO2 Capture, Transport and Storage. 4, 101–109 (2019).
ECRA Action Report 2023.
“Blue” cement production with electrified cement kilns | Strategic Energy Alliance. https://strategic-energy-alliance.stanford.edu/blue-cement-production-electrified-cement-kilns (accessed 13 Nov 2024).
Quevedo Parra, S. & Romano, M. C. Decarbonization of cement production by electrification. J. Clean. Prod. 425, 138913 (2023).
Venkatramani, N. Industrial plasma torches and applications. Curr. Sci. 83, 254–262 (2002).
Fakt, A., Nilsson, M. Demonstration scale of a rotary kiln for cement production using a thermal plasma: a scaling and heat transfer study (2023).
Burman, T., Engvall, J. Evaluation of usage of plasma torches in cement production (2019).
Petersson, J. Thermal plasma in a rotary kiln for cement production: an investigation of the heat transfer mechanisms (2022).
Casey, D. Options for switching UK cement production sites to near zero CO2 emission fuel: technical and financial feasibility.
Technology. ZEQL. https://www.zeql.com/technology (accessed 13 Nov 2024).
Xiao, Y. & Xue, Y. A review on application of microwave in cement life cycle. Renew. Sustain. Energy Rev. 199, 114498 (2024).
Quemeneur, L., Choisnet, J. & Raveau, B. Les micro-ondes peuvent-elles etre utilisees pour la clinkerisation des crus de cimenterie?. Mater. Chem. Phys. 8, 293–303 (1983).
Quéméneur, L., Choisnet, J., Raveau, B., Thiebaut, J. M. & Roussy, G. Microwave clinkering with a grooved resonant applicator. J. Am. Ceram. Soc. 66, 855–859 (1983).
Li, H., Agrawal, D. K., Cheng, J. & Silsbee, M. R. Formation and hydration of C3S prepared by microwave and conventional sintering. Cem. Concr. Res. 29, 1611–1617 (1999).
Li, H., Agrawal, D. K., Cheng, J. & Silsbee, M. R. Microwave sintering of sulphoaluminate cement with utility wastes. Cem. Concr. Res. 31, 1257–1261 (2001).
Fang, Y., Roy, D. M. & Roy, R. Microwave clinkering of ordinary and colored portland cements. Cem. Concr. Res. 26, 41–47 (1996).
Fang, Y., Chen, Y., Silsbee, M. R. & Roy, D. M. Microwave sintering of flyash. Mater. Lett. 27, 155–159 (1996).
Buttress, A., Jones, A. & Kingman, S. Microwave processing of cement and concrete materials—towards an industrial reality?. Cem. Concr. Res. 68, 112–123 (2015).
Van Reusel, K., Di Barba, P., Dughiero, F., Forzan, M. & Mognaschi, M. E. Two complementary examples of electrification in the cement industry. Int. J. Appl. Electromagn. Mech. 75, 193–197 (2024).
The Project » Destiny. Destiny. https://www.destinyh2020andbeyond.eu/the-project/ (accessed 20 Nov 2024).
Makul, N., Rattanadecho, P. & Agrawal, D. K. Applications of microwave energy in cement and concrete – a review. Renew. Sustain. Energy Rev. 37, 715–733 (2014).
Giulietti, N., et al. Measuring material temperature in high-temperature microwave-based heating process: a virtual sensor approach. In: Proc. IEEE International Workshop on Metrology for Industry 4.0 & IoT (MetroInd4.0&IoT); 451–455 (IEEE, 2023).
Dorn, C. et al. KPI and LCA evaluation of integrated microwave technology for high temperature processes. Procedia CIRP 29, 492–497 (2015).
Zhang, L. et al. Comparison of microwave and conventional heating routes for kaolin thermal activation. J. Cent. South Univ. 27, 2494–2506 (2020).
Hwang, D. J. et al. Characteristics of precipitated calcium carbonate by hydrothermal and carbonation processes with mega-crystalline calcite using rotary microwave kiln. J. Ind. Eng. Chem. 20, 2727–2734 (2014).
Hwang, D. J. et al. Preparation of CaCO3 using mega-crystalline calcite in electrical furnace and batch type microwave kiln. J. Ind. Eng. Chem. 19, 1507–1516 (2013).
Hwang, D. J. et al. Calcination of mega-crystalline calcite using microwave and electric furnaces. J. Ind. Eng. Chem. 18, 1956–1963 (2012).
Corrêa, B. H. B. C., Neto, J. C. B., França, A. B. & Cordeiro, A. C. C. New process to obtain unslaked lime through microwave hybrid heating and its fluid dynamics computational modeling. J. Eng. Exact Sci. 7, 12300–12e (2021).
Ke, K., Ma, B. G., Wang, X. L., Li, X. G. Formation of tricalcium silicate prepared by electric and microwave sintering. Adv. Mater. Res. 148–149, 1119–1123 (2011).
Long, S., Yan, C. & Dong, J. Microwave-promoted burning of portland cement clinker. Cem. Concr. Res. 32, 17–21 (2002).
Long, S., Dong, J. & Yan, C. Microwave promoted clinkering of sulfoaluminate cement. Cem. Concr. Res. 32, 1653–1656 (2002).
DAPhNE—Adaptive Production Systems and Measurement and Control Equipment for Optimal Energy Consumption and near-to-Zero Emissions in Manufacturing Processes. https://www.cordis.europa.eu/docs/results/314/314636/final1-publishable-summary-daphne.pdf.
Fall, M. L., et al. Energy Efficient Microwave Hybrid Processing of Lime for Cement, Steel, and Glass Industries; DOE/EE0003472; Ceralink Inc. https://doi.org/10.2172/1034621 (2012).
Pitz-Paal, R., Buck, R., Heller, P., Hirsch, T., Steinmann, W.-D. Solar thermal power production. In Transition to Renewable Energy Systems 307–338 (John Wiley & Sons, Ltd, 2013).
Falcone, P. K. A Handbook for Solar Central Receiver Design; SAND-86-8009, https://doi.org/10.2172/6545992 (Sandia National Lab. (SNL-CA), 1986).
Romero, M., Marcos, M. J., Osuna, R., Fernández, V. Design and implementation plan of a 10 MW solar tower power plant based on volumetric-air technology in Seville (Spain). Sol. Eng. 89–98. 2000
Yogev, A., Kribus, A., Epstein, M. & Kogan, A. Solar “tower reflector” systems: a new approach for high-temperature solar plants. Int. J. Hydrog. Energy 23, 239–245 (1998).
Kribus, A. High-Performance, Low-cost solar central receiver power plants (1997).
Badie, J. M. et al. 52 Decarbonation of calcite and phosphate rock in solar chemical reactors. Chem. Eng. Sci. 35, 413–420 (1980).
Flamant, G., Hernandez, D., Bonet, C. & Traverse, J.-P. Experimental aspects of the thermochemical conversion of solar energy; decarbonation of CaCO3. Sol. Energy 24, 385–395 (1980).
Salman, O. A. & Khraishi, N. Thermal decomposition of limestone and gypsum by solar energy. Sol. Energy 41, 305–308 (1988).
Imhof, A. Solar cement plants - an interesting challenge for business and science. ZKG Int. 53, 448–456 (2000).
Gonzalez, R. S., Flamant, G. Technical and economic feasibility analysis of using concentrated solar thermal technology in the cement production process: hybrid approach—a case study. https://doi.org/10.1115/ES2013-18143.
Meier, A., Gremaud, N. & Steinfeld, A. Economic evaluation of the industrial solar production of lime. Energy Convers. Manag. 46, 905–926 (2005).
Freeman, J., Hellgardt, K. & Markides, C. N. An assessment of solar-powered organic rankine cycle systems for combined heating and power in UK domestic applications. Appl. Energy 138, 605–620 (2015).
Stein, W. H. & Buck, R. Advanced power cycles for concentrated solar power. Sol. Energy 152, 91–105 (2017).
CEMEX and Synhelion achieve breakthrough in cement production with solar energy. Corporate Website. https://www.cemex.com/w/cemex-and-synhelion-achieve-breakthrough-in-cement-production-with-solar-energy (accessed 14 Oct 2024).
Solar Thermal Technology. https://www.heliogen.com/technology/ Heliogen.(accessed 14 Oct 2024).
Abanades, S. & André, L. Design and demonstration of a high temperature solar-heated rotary tube reactor for continuous particles calcination. Appl. Energy 212, 1310–1320 (2018).
Beshore, D., Jaeger, F., Gartner, E. Thermal energy storage/waste heat recovery applications in the cement industry (1979).
Engin, T. & Ari, V. Energy auditing and recovery for dry type cement rotary kiln systems––a case study. Energy Convers. Manag. 46, 551–562 (2005).
Technology – Electrified Thermal Solutions. https://electrifiedthermal.com/technology/ (accessed 14 Oct 2024).
How It Works. Rondo Energy. https://rondo.com/how-it-works (accessed 14 Oct 2024).
ECOCLAYTM. FLSmidth Cement | Research and Development | ECOCLAYTM. https://www.flsmidth-cement.com/solutions/research-and-development/ecoclay-a-partnership-to-decarbonize-the-cement-industry (accessed 14 Oct 2024).
Lis, J. Self-propagating high-temperature synthesis. In: Encyclopedia of Materials: Technical Ceramics and Glasses (ed. Pomeroy, M.) 40–58 (Elsevier, 2021).
Merzhanov, A., Sytschev, A. Self-propagating high-temperature synthesis of materials; 301–322 (2002).
S. Agrawal, S. S. Volaity, S. Kilambi, A. Kumar, N. Neithalath. A low-carbon approach for lime production using self-propagating high temperature synthesis-driven limestone calcination. Renew. Sustain. Energy Rev. 210 (2025).
S. S. Volaity, et al. Numerical simulation of heat transfer in self-propagating high-temperature synthesis (SHS)-based calcination of limestone. Fuel, 386 https://doi.org/10.1016/j.fuel.2024.134226 (2025).
Volaity, S. S. et al. Reaction kinetics in a novel low-temperature limestone calcination approach examined via numerical simulations and isoconversional methods. Thermochim. Acta 750, 180039 (2025).
Jacob, R. M. CO2 Capture through Electrified Calcination in Cement Clinker Production. Doctoral thesis. https://openarchive.usn.no/usn-xmlui/handle/11250/3084303 (accessed 14 Nov 2024) (University of South-Eastern Norway, 2023).
Usterud, M. H., Jacob, R. M., Tokheim, L.-A. Modeling and simulation of an electrified drop-tube calciner. Scand. Simul. Soc. 279–286 (2022).
Jacob, R. M. & Tokheim, L.-A. Electrified calciner concept for CO2 capture in pyro-processing of a dry process cement plant. Energy 268, 126673 (2023).
Jacob, R. M. & Tokheim, L.-A. Novel design of a rotary calciner internally heated with electrical axial heaters: experiments and modelling. Results Eng 17, 100992 (2023).
Ahmed, I., Duchesne, M., Tan, Y. & Lu, D. Y. Electrically heated fluidized beds─a review. Ind. Eng. Chem. Res. 63, 4205–4235 (2024).
Schwing Technologies: Fluidized Bed Process Technique. https://www.schwing-technologies.com/fluidized-bed-process-technique (accessed 14 Nov 2024).
Jacob, R. M. & Tokheim, L.-A. CPFD simulation of an electrically heated fluidized bed calciner with binary particles. Energy Convers. Manag. X 20, 100444 (2023).
Leilac Projects - Adbri, Boral, Bacchus Marsh. Leilac. https://www.leilac.com/projects/ (accessed 19 Nov 2024).
SaltX Technology. SaltX Technology. https://www.saltxtechnology.com/ (accessed 13 Nov 2024).
Ji, T., et al. Limestone conversion to cement clinker precursor in a zero-gap electrolyzer. Chemistry. https://doi.org/10.26434/chemrxiv-2024-fvxrb (2024).
Galitsky, C., Worrell, E., Galitsky, C. Energy efficiency improvement and cost saving opportunities for cement making. An ENERGY STAR guide for energy and plant managers; LBNL-54036-Revision, 927882; p LBNL-54036-Revision, 927882. https://doi.org/10.2172/927882 (2008).
Nhuchhen, D. R., Sit, S. P. & Layzell, D. B. Decarbonization of cement production in a hydrogen economy. Appl. Energy 317, 119180 (2022).
Mccoy, S. & Rubin, E. An engineering-economic model of pipeline transport of CO2 with application to carbon capture and storage. Int. J. Greenh. Gas Control 2, 219–229 (2008).
Hepburn, C. et al. The technological and economic prospects for CO2 utilization and removal. Nature 575, 87–97 (2019).
Gardarsdottir, S. O. et al. Comparison of technologies for CO2 capture from cement production—Part 2: cost analysis. Energies 12, 542 (2019).
Liu, X., Jin, K., Li, X. & Yang, R. Low-carbon cement manufacturing enabled by electrified calcium looping and thermal energy storage. Int. J. Greenh. Gas Control 129, 103986 (2023).
Loudeche, M., Rouxhet, R. & Proost, J. Development of a new type of electrochemical reactor for low temperature lime and cement production. ECS Meet. Abstr. MA2023-01, 1603–1603 (2023).
Zhang, Z. B. Low-temperature electrochemical cement clinker production. ACS Energy Lett. 8, 3927–3934 (2023).
Material Economics (2019). Industrial Transformation 2050—pathways to net-zero emissions from EU heavy industry.
KG, Q. G. & C. Technology Papers. Kickstarter Q3i Typo3 11 headless. https://www.ecra-online.org/research/technology-papers (accessed 15 Dec 2024).
Karlsson, I., Toktarova, A., Rootzén, J., Odenberger, M. Technical Roadmap: Cement Industry.
Nie, S. et al. Analysis of theoretical carbon dioxide emissions from cement production: methodology and application. J. Clean. Prod. 334, 130270 (2022).
Energy Technology Perspectives 2017—Analysis. IEA. https://www.iea.org/reports/energy-technology-perspectives-2017 (accessed 10 Nov 2024).
SMA Mineral. https://smamineral.se/en/ (accessed 13 Nov 2024).
Department of Energy, Washington, DC: US Concentrating Solar-Thermal Power Basics. Energy.gov. https://www.energy.gov/eere/solar/concentrating-solar-thermal-power-basics (accessed 14 Nov 2024).
Industrial Decarbonisation, CO₂ Capture & Environmental Solutions. Calix. https://calix.global/ (accessed 13 Nov 2024).
Acknowledgements
The authors acknowledge U.S. Department of Energy’s 7th Clean Energy Manufacturing Innovation Institute—Electrified Processes for Industrial Excellence (EPIXC; DEEE0010725); the Future Manufacturing Program of the U.S National Science Foundation (DMR 2228782); and the center for carbon-efficient and advanced manufacturing of materials and structures (CAMMS) at Arizona State University for supporting this work.
Author information
Authors and Affiliations
Contributions
S.S.V. and B.K.A. prepared the drafts of the manuscript along with all the figures; T.H. and J.H. added to the overview and sections on electrolysis and edited those sections; S.S. and G.S. prepared and edited the introduction, overview and perspective sections; A.K. and N.N. reorganized the manuscript, added context and content to all the sections, and carried out comprehensive editing of the document. All authors critically reviewed the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Volaity, S.S., Aylas-Paredes, B.K., Han, T. et al. Towards decarbonization of cement industry: a critical review of electrification technologies for sustainable cement production. npj Mater. Sustain. 3, 23 (2025). https://doi.org/10.1038/s44296-025-00068-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44296-025-00068-6
This article is cited by
-
Impact of partial cement dust replacement in unsaturated polyester: assessing material performance and waste valorization for sustainable management
Scientific Reports (2026)
-
A critical review of 3D printed fiber-based geopolymer concrete (3DP-FGC): fresh properties, mechanical performance, and current limitations
Journal of Building Pathology and Rehabilitation (2026)
-
Stabilization of Sandy Soil Using Polymer-Based Soil Treatment Method with Carrageenan and Polyvinyl Alcohol
Arabian Journal for Science and Engineering (2025)