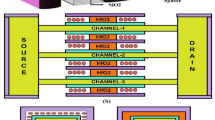
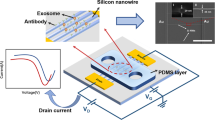
Abstract
Sensitive, reliable detection of molecular biomarkers in complex clinical samples through portable biosensors remains a central challenge for early cancer diagnostics. Field-effect transistor (FET) biosensors offer strong potential for miniaturized, low power sensing, yet their clinical translation is often constrained by probe instability, non-specific adsorption and Debye screening-limited signal transduction in serum. Here we present a site-specific anchored FET platform (SNAP-FET) that integrates genetic code expansion and click chemistry to immobilize nanobodies with a controlled, uniform, site-specific orientation in serum, within the Debye length for efficient signal transduction. This strategy yields attomolar-level sensitivity and stable electronic readout of endometrial cancer biomarkers directly in serum, overcoming longstanding limitations of biofluid FET sensing by coupling compact affinity probes with precision interface design. More broadly, the SNAP-FET and its portable implementation, ENDOCARE, provide a generalizable framework for next-generation biochemical sensing in point-of-care settings for early diagnostics in oncology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The data that support the findings of this study are available within this article and its Supplementary Information. Source data are provided with this paper.
References
Gu, B. et al. Variations in incidence and mortality rates of endometrial cancer at the global, regional, and national levels, 1990–2019. Gynecol. Oncol. 161, 573–580 (2021).
Nicholson, B. D. et al. Multi-cancer early detection test in symptomatic patients referred for cancer investigation in England and Wales (SYMPLIFY): a large-scale, observational cohort study. Lancet Oncol. 24, 733–743 (2023).
Jacobs, I. et al. Sensitivity of transvaginal ultrasound screening for endometrial cancer in postmenopausal women: a case–control study within the UKCTOCS cohort. Lancet Oncol. 12, 38–48 (2011).
Kinkel, K. et al. Radiologic staging in patients with endometrial cancer: a meta-analysis. Radiology 212, 711–718 (1999).
Clark, T. J. et al. Accuracy of outpatient endometrial biopsy in the diagnosis of endometrial cancer: a systematic quantitative review. BJOG 109, 313–321 (2002).
Gadducci, A., Cosio, S., Carpi, A., Nicolini, A. & Genazzani, A. R. Serum tumor markers in the management of ovarian, endometrial and cervical cancer. Biomed. Pharmacother. 58, 24–38 (2004).
Das, S., Dey, M. K., Devireddy, R. & Gartia, M. R. Biomarkers in cancer detection, diagnosis, and prognosis. Sensors 24, 37 (2023).
Hosseini, S., Vázquez-Villegas, P., Rito-Palomares, M. & Martinez-Chapa, S. O. in Enzyme-Linked Immunosorbent Assay (ELISA): From A to Z 67–115 (Springer, 2017)
Luppa, P. B., Müller, C., Schlichtiger, A. & Schlebusch, H. Point-of-care testing (POCT): current techniques and future perspectives. Trends Analyt. Chem. 30, 887–898 (2011).
Heng, W. et al. A smart mask for exhaled breath condensate harvesting and analysis. Science 385, 954–961 (2024).
Wu, J., Liu, H., Chen, W., Ma, B. & Ju, H. Device integration of electrochemical biosensors. Nat. Rev. Bioeng. 1, 346–360 (2023).
Balderston, S. et al. Discrimination of single-point mutations in unamplified genomic DNA via Cas9 immobilized on a graphene field-effect transistor. Nat. Biomed. Eng. 5, 713–725 (2021).
Macchia, E. et al. Point-of-care ultra-portable single-molecule bioassays for one-health. Adv. Mater. 36, 2309705 (2024).
Wang, T. et al. Development of nucleic acid aptamer-based lateral flow assays: a robust platform for cost-effective point-of-care diagnosis. Theranostics 11, 5174–5196 (2021).
Zhang, Z. et al. Advanced point-of-care testing technologies for human acute respiratory virus detection. Adv. Mater. 34, 2103646 (2022).
Choi, S., Goryll, M., Sin, L. Y. M., Wong, P. K. & Chae, J. Microfluidic-based biosensors toward point-of-care detection of nucleic acids and proteins. Microfluid. Nanofluidics 10, 231–247 (2011).
Zhang, Y. et al. Interface-engineered field-effect transistor electronic devices for biosensing. Adv. Mater. 37, 2306252 (2025).
Vacic, A. et al. Determination of molecular configuration by Debye length modulation. J. Am. Chem. Soc. 133, 13886–13889 (2011).
Guo, K. et al. Rapid single-molecule detection of COVID-19 and MERS antigens via nanobody-functionalized organic electrochemical transistors. Nat. Biomed. Eng. 5, 666–677 (2021).
Li, J. et al. A universal reagent for detection of emerging diseases using bioengineered multifunctional yeast nanofragments. Nat. Nanotechnol. 18, 1222–1229 (2023).
Janićijević, Ž & Baraban, L. Integration strategies and formats in field-effect transistor chemo-and biosensors: a critical review. ACS Sens. 10, 2431–2452 (2025).
Pandey, M. et al. Advanced materials for biological field-effect transistors (Bio-FETs) in precision healthcare and biosensing. Adv. Healthcare Mater. 14, 2500400 (2025).
Boukherroub, R. & Szunerits, S. The future of nanotechnology-driven electrochemical and electrical point-of-care devices and diagnostic tests. Annu. Rev. Anal. Chem. 17, 173–195 (2024).
Liu, M., Li, L., Jin, D. & Liu, Y. Nanobody—a versatile tool for cancer diagnosis and therapeutics. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 13, e1697 (2021).
Seo, M.-H. et al. Controlled and oriented immobilization of protein by site-specific incorporation of unnatural amino acid. Anal. Chem. 83, 2841–2845 (2011).
Chaparro Sosa, A. F., Bednar, R. M., Mehl, R. A., Schwartz, D. K. & Kaar, J. L. Faster surface ligation reactions improve immobilized enzyme structure and activity. J. Am. Chem. Soc. 143, 7154–7163 (2021).
Bednar, R. M. et al. Immobilization of proteins with controlled load and orientation. ACS Appl. Mater. Interfaces 11, 36391–36398 (2019).
Al Ghamdi, W. S. et al. A peptide nucleic acid-functionalized heterojunction thin film transistor as a scalable and reusable platform for label-free detection of microRNA. Adv. Funct. Mater. 36, e12490 (2025).
Zhang, H. et al. Gate-voltage-modulated electrostatic interaction between the probe and the channel material of FET biosensors for detecting telomerase. Chem. Commun. 59, 5209–5212 (2023).
Kim, J. et al. Fabrication of high-performance ultrathin In2O3 film field-effect transistors and biosensors using chemical lift-off lithography. ACS Nano 9, 4572–4582 (2015).
Yu, J. et al. Targeting and neutralizing human epididymis protein 4 by novel nanobodies to suppress ovarian cancer cells and attenuate cisplatin resistance. Int. J. Biol. Macromol. 199, 298–306 (2022).
Yong, K. W., Yuen, D., Chen, M. Z., Porter, C. J. & Johnston, A. P. Pointing in the right direction: controlling the orientation of proteins on nanoparticles improves targeting efficiency. Nano Lett. 19, 1827–1831 (2019).
Xu, X. et al. Tuning electrostatic gating of semiconducting carbon nanotubes by controlling protein orientation in biosensing devices. Angew. Chem. Int. Ed. 60, 20184–20189 (2021).
Gwyther, R. E. et al. Differential bio-optoelectronic gating of semiconducting carbon nanotubes by varying the covalent attachment residue of a green fluorescent protein. Adv. Funct. Mater. 32, 2112374 (2022).
Gwyther, R. E. et al. Optimising CNT-FET biosensor design through modelling of biomolecular electrostatic gating and its application to β-lactamase detection. Nat. Commun. 15, 7482 (2024).
Li, Y. et al. Computation-guided discovery of diazole monosubstituted tetrazines as optimal bioorthogonal tools. J. Am. Chem. Soc. 146, 26884–26896 (2024).
Hermanson, G. T. Bioconjugate Techniques (Academic Press, 2013).
He, S. et al. Interfacial water-dictated oil adhesion based on ion modulation. J. Am. Chem. Soc. 145, 24145–24152 (2023).
Lo Nostro, P. & Ninham, B. W. Hofmeister phenomena: an update on ion specificity in biology. Chem. Rev. 112, 2286–2322 (2012).
Simões, B. et al. Direct immobilization of engineered nanobodies on gold sensors. ACS Appl. Mater. Interfaces 13, 17353–17360 (2021).
Rios, F. & Smirnov, S. Biochemically responsive smart surface. ACS Appl. Mater. Interfaces 1, 768–774 (2009).
Rudawska, A. & Jacniacka, E. Analysis for determining surface free energy uncertainty by the Owen–Wendt method. Int. J. Adhes. Adhes. 29, 451–457 (2009).
Uhlén, M. et al. Tissue-based map of the human proteome. Science 347, 1260419 (2015).
Deng, L. et al. Expression and clinical significance of annexin A2 and human epididymis protein 4 in endometrial carcinoma. J. Exp. Clin. Cancer Res. 34, 96 (2015).
Kamat, A. A. et al. Clinical and biological significance of vascular endothelial growth factor in endometrial cancer. Clin. Cancer Res. 13, 7487–7495 (2007).
Arranz-Gibert, P. et al. Chemoselective restoration of para-azido-phenylalanine at multiple sites in proteins. Cell Chem. Biol. 29, 1046–1052 (2022).
Brennan, D. J. et al. Serum HE4 as a prognostic marker in endometrial cancer—a population based study. Gynecol. Oncol. 132, 159–165 (2014).
Li, J., Wang, X., Qu, W., Wang, J. & Jiang, S.-W. Comparison of serum human epididymis protein 4 and CA125 on endometrial cancer detection: a meta-analysis. Clin. Chim. Acta 488, 215–220 (2019).
Maurya, V., Singh, S., Singh, S., Singh, S. P. & Verma, M. K. Tumor biomarkers HE4 and CA125 alongside enhanced computed tomography imaging in assessing lymph node metastasis in endometrial cancer. Bioinformation 20, 1453–1460 (2024).
Bian, J., Sun, X., Li, B. & Ming, L. Clinical significance of serum HE4, CA125, CA724, and CA19-9 in patients with endometrial cancer. Technol. Cancer Res. Treat. 16, 435–439 (2017).
Jiang, T., Huang, L. & Zhang, S. Preoperative serum CA125: a useful marker for surgical management of endometrial cancer. BMC Cancer 15, 396 (2015).
Behrouzi, R., Barr, C. E. & Crosbie, E. J. HE4 as a biomarker for endometrial cancer. Cancers 13, 4764 (2021).
Tian, F. et al. A general approach to site-specific antibody drug conjugates. Proc. Natl Acad. Sci. USA 111, 1766–1771 (2014).
Jang, H. S., Jana, S., Blizzard, R. J., Meeuwsen, J. C. & Mehl, R. A. Access to faster eukaryotic cell labeling with encoded tetrazine amino acids. J. Am. Chem. Soc. 142, 7245–7249 (2020).
Li, L. et al. Methods to identify immunogenic peptides in SARS-CoV-2 spike and protective monoclonal antibodies in COVID-19 patients. Small Methods 5, 2100058 (2021).
Zhang, Q. et al. Machine learning-guided evolution of pyrrolysyl-tRNA synthetase for improved incorporation efficiency of diverse noncanonical amino acids. Nat. Commun. 16, 6648 (2025).
Frisch, M. et al. Gaussian 16, Revision B.01 (Gaussian, Inc., 2016).
Chai, J.-D. & Head-Gordon, M. Long-range corrected hybrid density functionals with damped atom–atom dispersion corrections. Phys. Chem. Chem. Phys. 10, 6615–6620 (2008).
Marenich, A. V., Cramer, C. J. & Truhlar, D. G. Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. B 113, 6378–6396 (2009).
Hu, L. et al. Designing artificial fluorescent proteins and biosensors by genetically encoding molecular rotor-based amino acids. Nat. Chem. 16, 1960–1971 (2024).
Acknowledgements
This work was supported by the National Key R&D Program of China (grant nos. 2020YFA0211200 and 2021YFA1200403), the National Natural Science Foundation of China (grant nos. 22090050, 22474131, U24A20502, 22522402, 22325701 and U22A20332). X.L. was supported by the Natural Science Foundation of Hubei Province (grant no. 2024AFA001) and the Natural Science Foundation of Shenzhen (grant nos. JCYJ20230807113706013). J.D. was supported by the Natural Science Foundation of Hubei Province (grant no. 2025AFA075). We thank the High-Performance Computing Cluster of the AI for Drug Discovery Platform at Peking University for providing computational resources. We also thank the Faculty of Materials Science and Chemistry at China University of Geosciences (CUG) for access to TEM facilities (Talos F200X) and M. Gong for his assistance with data acquisition and analysis. We also thank T. Zhai from the School of Materials Science and Engineering, Huazhong University of Science and Technology, for his technical guidance and support in the fabrication of In2O3 FET.
Author information
Authors and Affiliations
Contributions
Z.Z. and Y.L. contributed equally to this paper. X.L. supervised the project. X.L., Z.Z., J.D., F.X., T.L. and Y.L. conceived and designed the study. Z.Z. fabricated and characterized transistors and devices. Z.Z., Y.L. and Y.J. synthesized probes. Y.L. performed theoretical modelling of orthogonal-1G8-linker complexes. Z.Z., Q.W. and G.L. performed cell and animal experiments. D.Y., J.D. and G.L. provided clinical samples. Z.Z., Y.L. and Q.W. analysed the data and prepared figures. Z.Z., Y.L., T.L. and X.L. drafted the paper. Z.Z., Y.L., Q.W., Y.J., T.L. and X.L. revised the paper. All authors contributed to the writing of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Sensors thanks Ryan A. Mehl and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Schematic diagram of the functionalization process of SNAP-FET.
Schematic diagram of the functionalization process of SNAP-FET.
Extended Data Fig. 2 GCE engineering modification of the 1G8-S85Tet nanobody and its application in HE4 detection.
GCE engineering modification of the 1G8-S85Tet nanobody and its application in HE4 detection. (a) Schematic illustration of the introduction of Tet-v-3.0 at the S85 site via GCE technology. (b) Deconvoluted ESI-MS spectra confirm the precise molecular mass. (c) SDS-PAGE image of the 1G8-S85Tet nanobody. (d-e) Kinetic comparison of orthogonal conjugation efficiency for 1G8-S85AzF and 1G8-S85Tet probes. (d) Time-dependent responses of SNAP-FETs functionalized with 1G8-S85AzF and 1G8-S85Tet probes at different modification durations to 1×10-11 M HE4. Error bars indicate mean ± standard deviation (n = 3). (e) Quantitative kinetics comparison.
Extended Data Fig. 3 Selection of the insertion site for Orthogonal-NT2 and validation of its CA125 detection capability.
Selection of the insertion site for Orthogonal-NT2 and validation of its CA125 detection capability. (a) Schematic illustration of the insertion site for the non-natural amino acid N6-[(2-azidoethoxy)carbonyl]-L-lysine introduced into the nanobody NT2 via GCE technology. (b) Normalized current response (ΔI/I0) of SNAP-FET functionalized with Orthogonal-NT2 compared with four random immobilization strategies (via carboxyl and amino) upon exposure to 100 U/mL CA125. Dots represent individual independent samples, and bars indicate mean ± standard deviation (n = 3). (c) Normalized current response ΔI/I0 of NT2-Q13AzK-modified SNAP-FET and bare In2O3 at different CA125 concentrations. Error bars indicate mean ± standard deviation (n = 3).
Extended Data Fig. 4 Preparation and performance verification of SNAP-FET with Ag/AgCl side gates.
Preparation and performance verification of SNAP-FET with Ag/AgCl side gates. (a) Fabrication of In2O3 FET with integrated Ag/AgCl side-gate electrodes. Starting from conventional In2O3 FET devices, electron-beam lithography (EBL) was used to define a patterned area between the source and drain electrodes. A Cr/Au layer (3 nm / 40 nm) was deposited by thermal evaporation to form a gold pad for Ag/AgCl deposition. Subsequently, 0.1 μL of Ag/AgCl paste was drop-cast onto the distal end of the gold pad to create a prototype Ag/AgCl gate electrode, yielding In2O3 FET devices with integrated side-gate configuration. (b) Schematic diagram of the working principle of SNAP-FET with Ag/AgCl side gate. (c) Normalized signal response (ΔI/I0) measured at VGS = 215 mV (Ag/AgCl side gate) and VGS = 200 mV (Ag/AgCl external gate) of In2O3 FET biosensors. The biosensors were tested in PBS upon exposure to HE4 (1× 10-14 M) and CA125 (3.75 × 10-2 U/mL). Dots represent individual independent samples, and bars indicate mean ± standard deviation (n = 8). Statistical significance was assessed using a two-sided unpaired t-test. No significant difference was observed between the two gating configurations for either biomarker (HE4, P = 0.7718; CA125, P = 0.0808); ns indicates not significant.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–57, Notes 1–7, Tables 1–11, Data 1 and 2 and sequence.
Supplementary Video 1 (download MP4 )
EC biomarker detection by ENDOCARE POCT device.
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 4 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Z., Li, Y., Jing, Y. et al. Oriented nanobody–field-effect transistor interfaces enable ultrasensitive cancer biomarker detection. Nat. Sens. (2026). https://doi.org/10.1038/s44460-026-00040-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44460-026-00040-4