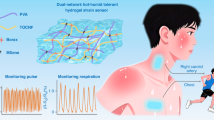
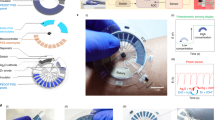
Abstract
Exhaled breath condensate (EBC) offers a non-invasive window into respiratory and systemic metabolism, yet wearable EBC systems are constrained by unstable moisture harvesting, short sensor lifetimes in humidity and battery dependence. Here we present EBClite, a battery-free smart mask for multiday EBC biomarker monitoring, using lactate as a model analyte. The platform integrates a regenerable antidrying porous hydrogel that achieves sustained and reactivatable breath condensation over several days, extending operational lifetime while reducing material cost. EBClite further incorporates a long-term stable electrochemical lactate sensor and an ultrathin quasi-two-dimensional perovskite solar cell with a power-to-weight ratio of 10 W g−1 and a power conversion efficiency exceeding 30% under ambient indoor illumination, enabling autonomous operation across diverse lighting conditions. Human studies demonstrate strong correlations between EBC and blood lactate levels during exercise and carbohydrate intake, enabling real-time tracking of metabolic fluctuations. EBClite provides a sustainable, user-friendly platform for continuous respiratory and metabolic monitoring.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The main data supporting the results of this study are available within the article and its Supplementary Information. Source data are provided with this paper.
References
Heng, W. et al. A smart mask for exhaled breath condensate harvesting and analysis. Science 385, 954–961 (2024).
Horváth, I., Hunt, J. & Barnes, P. J. Exhaled breath condensate: methodological recommendations and unresolved questions. Eur. Respir. J. 26, 523–548 (2005).
Heng, W., Yin, S., Chen, Y. & Gao, W. Exhaled breath analysis: from laboratory test to wearable sensing. IEEE Rev. Biomed. Eng. 18, 50–73 (2024).
Fahy, J. V. & Dickey, B. F. Airway mucus function and dysfunction. N. Engl. J. Med. 363, 2233–2247 (2010).
Ma, J. et al. Airborne biomarker localization engine for open-air point-of-care detection. Nat. Chem. Eng. 2, 321–333 (2025).
Li, X. et al. Rapid and on-site wireless immunoassay of respiratory virus aerosols via hydrogel-modulated resonators. Nat. Commun. 15, 4035 (2024).
Li, H. et al. Wireless, battery-free, multifunctional integrated bioelectronics for respiratory pathogens monitoring and severity evaluation. Nat. Commun. 14, 7539 (2023).
Brasier, N. et al. Applied body-fluid analysis by wearable devices. Nature 636, 57–68 (2024).
Kim, J., Campbell, A. S., de Ávila, B. E.-F. & Wang, J. Wearable biosensors for healthcare monitoring. Nat. Biotechnol. 37, 389–406 (2019).
Yang, Y. et al. A laser-engraved wearable sensor for sensitive detection of uric acid and tyrosine in sweat. Nat. Biotechnol. 38, 217–224 (2020).
Ates, H. C. & Dincer, C. Wearable breath analysis. Nat. Rev. Bioeng. 1, 80–82 (2023).
Nguyen, P. Q. et al. Wearable materials with embedded synthetic biology sensors for biomolecule detection. Nat. Biotechnol. 39, 1366–1374 (2021).
Maier, D. et al. Toward continuous monitoring of breath biochemistry: a paper-based wearable sensor for real-time hydrogen peroxide measurement in simulated breath. ACS Sens. 4, 2945–2951 (2019).
Wang, R., Du, Y., Wan, X., Xu, J. & Chen, J. On-mask magnetoelastic sensor network for self-powered respiratory monitoring. ACS Nano 19, 26862–26870 (2025).
Maranhao, B. et al. Probability of fit failure with reuse of N95 mask respirators. Br. J. Anaesth. 125, e322–e324 (2020).
Lai, C. & Lu, L. Hydrogel-based thermal regulation strategies for passive cooling: a review. Energy Built Environ. https://doi.org/10.1016/j.enbenv.2024.10.002 (2024).
Wang, M. et al. Printable molecule-selective core–shell nanoparticles for wearable and implantable sensing. Nat. Mater. 24, 589–598 (2025).
Gao, M. et al. Power generation for wearable systems. Energy Environ. Sci. 14, 2114–2157 (2021).
Fan, X. et al. Flexible and wearable power sources for next-generation wearable electronics. Batteries Supercaps 3, 1262–1274 (2020).
Bravo Diaz, L. et al. Review—meta-review of fire safety of lithium-ion batteries: industry challenges and research contributions. J. Electrochem. Soc. 167, 090559 (2020).
Park, C. et al. Adaptive electronics for photovoltaic, photoluminescent and photometric methods in power harvesting for wireless wearable sensors. Nat. Commun. 16, 5808 (2025).
Song, Y. et al. Wireless battery-free wearable sweat sensor powered by human motion. Sci. Adv. 6, eaay9842 (2020).
Kishore, R. A., Nozariasbmarz, A., Poudel, B., Sanghadasa, M. & Priya, S. Ultra-high performance wearable thermoelectric coolers with less materials. Nat. Commun. 10, 1765 (2019).
Min, J. et al. An autonomous wearable biosensor powered by a perovskite solar cell. Nat. Electron. 6, 630–641 (2023).
Kaltenbrunner, M. et al. Flexible high power-per-weight perovskite solar cells with chromium oxide–metal contacts for improved stability in air. Nat. Mater. 14, 1032–1039 (2015).
Hailegnaw, B. et al. Flexible quasi-2D perovskite solar cells with high specific power and improved stability for energy-autonomous drones. Nat. Energy 9, 677–690 (2024).
Kantareddy, S. N. R. et al. Perovskite PV-powered RFID: enabling low-cost self-powered IoT sensors. IEEE Sens. J. 20, 471–478 (2020).
Han, W. et al. All irradiance-applicable, perovskite solar cells-powered flexible self-sustaining sensor nodes for wireless internet-of-things. Adv. Funct. Mater. 35, 2425697 (2025).
Best research-cell efficiency chart. NREL https://www.nrel.gov/pv/cell-efficiency (2025).
Culik, P. et al. Design and cost analysis of 100 MW perovskite solar panel manufacturing process in different locations. ACS Energy Lett. 7, 3039–3044 (2022).
He, X. et al. 40.1% record low-light solar-cell efficiency by holistic trap-passivation using micrometer-thick perovskite film. Adv. Mater. 33, 2100770 (2021).
Zhang, J., Zhang, W., Cheng, H.-M. & Silva, S. R. P. Critical review of recent progress of flexible perovskite solar cells. Mater. Today 39, 66–88 (2020).
Shirayama, M. et al. Degradation mechanism of CH3NH3PbI3 perovskite materials upon exposure to humid air. J. Appl. Phys. 119, 115501 (2016).
Kore, P. B., Jamshidi, M. & Gardner, M. J. The impact of moisture on the stability and degradation of perovskites in solar cells. Mater. Adv. 5, 2200–2217 (2024).
Mikkelsen, M. E. et al. Serum lactate is associated with mortality in severe sepsis independent of organ failure and shock. Crit. Care Med. 37, 1670–1677 (2009).
Ouyang, J., Wang, H. & Huang, J. The role of lactate in cardiovascular diseases. Cell Commun. Signal. 21, 317 (2023).
Hirschhaeuser, F., Sattler, U. G. A. & Mueller-Klieser, W. Lactate: a metabolic key player in cancer. Cancer Res. 71, 6921–6925 (2011).
Chen, H. et al. NBS1 lactylation is required for efficient DNA repair and chemotherapy resistance. Nature 631, 663–669 (2024).
Kraut, J. A. & Madias, N. E. Lactic acidosis. N. Engl. J. Med. 371, 2309–2319 (2014).
Beneke, R., Leithäuser, R. M. & Ochentel, O. Blood lactate diagnostics in exercise testing and training. Int. J. Sports Physiol. Perform. 6, 8–24 (2011).
Spurway, N. C. Aerobic exercise, anaerobic exercise and the lactate threshold. Br. Med. Bull. 48, 569–591 (1992).
Poole, D. C., Burnley, M., Vanhatalo, A., Rossiter, H. B. & Jones, A. M. Critical power: an important fatigue threshold in exercise physiology. Med. Sci. Sports Exerc. 48, 2320–2334 (2016).
Leija, R. G. et al. Enteric and systemic postprandial lactate shuttle phases and dietary carbohydrate carbon flow in humans. Nat. Metab. 6, 670–677 (2024).
Rabinowitz, J. D. & Enerbäck, S. Lactate: the ugly duckling of energy metabolism. Nat. Metab. 2, 566–571 (2020).
Toffaletti, J. G. Blood lactate: biochemistry, laboratory methods, and clinical interpretation. Crit. Rev. Clin. Lab. Sci. 28, 253–268 (1991).
Gao, W. et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529, 509–514 (2016).
Kim, J. et al. Non-invasive mouthguard biosensor for continuous salivary monitoring of metabolites. Analyst 139, 1632–1636 (2014).
Tehrani, F. et al. An integrated wearable microneedle array for the continuous monitoring of multiple biomarkers in interstitial fluid. Nat. Biomed. Eng. 6, 1214–1224 (2022).
Arwani, R. T. et al. Stretchable ionic–electronic bilayer hydrogel electronics enable in situ detection of solid-state epidermal biomarkers. Nat. Mater. 23, 1115–1122 (2024).
Zhang, S. et al. Rapid measurement of lactate in the exhaled breath condensate: biosensor optimization and in-human proof of concept. ACS Sens. 7, 3809–3816 (2022).
Lillis, B., Grogan, C., Berney, H. & Lane, W. A. Investigation into immobilisation of lactate oxidase to improve stability. Sens. Actuators B 68, 109–114 (2000).
Minagawa, H., Nakayama, N., Matsumoto, T. & Ito, N. Development of long life lactate sensor using thermostable mutant lactate oxidase. Biosens. Bioelectron. 13, 313–318 (1998).
Zhou, L. et al. Highly efficient and stable planar perovskite solar cells with modulated diffusion passivation toward high power conversion efficiency and ultrahigh fill factor. Sol. RRL 3, 1900293 (2019).
Jiang, Q. et al. Surface passivation of perovskite film for efficient solar cells. Nat. Photon. 13, 460–466 (2019).
Hamadani, B. H. Understanding photovoltaic energy losses under indoor lighting conditions. Appl. Phys. Lett. 117, 043904 (2020).
Qamar, M. Z. et al. Advancement in indoor energy harvesting through flexible perovskite photovoltaics for self- powered IoT applications. Nano Energy 129, 109994 (2024).
Khenkin, M. V. et al. Consensus statement for stability assessment and reporting for perovskite photovoltaics based on ISOS procedures. Nat. Energy 5, 35–49 (2020).
Strawbridge, R. & Young, A. H. Lithium: how low can you go? J. Bipolar Disord. 12, 4 (2024).
Reaven, G. M., Hollenbeck, C., Jeng, C.-Y., Wu, M. S. & Chen, Y.-D. I. Measurement of plasma glucose, free fatty acid, lactate, and insulin for 24 h in patients with NIDDM. Diabetes 37, 1020–1024 (1988).
Hailegnaw, B. et al. Inverted (p–i–n) perovskite solar cells using a low temperature processed TiOx interlayer. RSC Adv. 8, 24836–24846 (2018).
De France, K. J., Xu, F. & Hoare, T. Structured macroporous hydrogels: progress, challenges, and opportunities. Adv. Healthc. Mater. 7, 1700927 (2018).
Acknowledgements
This work was supported through the Heritage Medical Research Institute (W.G.), the Horizon 2020 research and innovation programme of the European Union under grant agreement number 101016411 ‘Soft Milli-robots-SOMIRO’ (M.K.). The funders had no role in the study design, data collection and analysis, decision to publish or preparation of the manuscript. We acknowledge N. Dalleska for help with ICP-MS.
Author information
Authors and Affiliations
Contributions
W.G., M.K., W.H. and C.P. initiated the concept and designed the studies; W.H., C.P. and W.T. led the experiments and collected the overall data; W.H., W.T., J.M., G.K., C.W., M.-J.K. and Y.C. contributed to EBClite characterization, validation and sample analysis. C.P., L.E.L, B.H., S.D. and M.S. contributed to solar module development, fabrication and characterization. C.P. and E.R. contributed to experimental design and characterization of VOC test for the solar module. W.G., M.K. and Z.F. supervised the studies. W.H., C.P., L.E.L., W.G. and M.K. cowrote the paper. All authors contributed to the data analysis and provided the feedback on the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Sensors thanks Jingcheng Ma and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Methods, Figs. 1–44, Tables 1–7 and References.
Source data
Source Data Figs. 2–5 (download XLSX )
All source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Heng, W., Putz, C., Tang, W. et al. A battery-free smart mask for long-term exhaled breath biochemical sensing. Nat. Sens. (2026). https://doi.org/10.1038/s44460-026-00041-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44460-026-00041-3