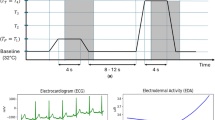
Abstract
Disease monitoring typically requires the acquisition of multiple physiological signals with different modalities, yet existing epidermal electronics rely on separate sensors for each modality, increasing device footprint, bandwidth and power consumption. Here we report a wearable electronic system that fuses multimodal physiological signals into a single cross-modal biosignal (X-Sig). Leveraging a cross-layered device architecture and in-sensor signal fusion strategy, the X-Sig sensor concurrently acquires biopotential signals (electrocardiography and electromyography) and biomechanical signals (force myography and radial pulse) through a single channel. This approach enables continuous monitoring of haemodynamic parameters with high accuracy, including heart rate, pulse arrival time, and diastolic and systolic blood pressure. In machine-learning-based gesture recognition, the sensor substantially reduced the decoding error rate compared with conventional electromyography. By fusing complementary modalities at the sensor level, the X-Sig sensor provides a versatile platform for designing bandwidth-efficient and low-power wearable electronics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the Article and its Supplementary Information. Source data are provided with this paper. Other data are available from the corresponding authors upon request.
Code availability
Code for implementing and experimenting with the proposed methodology is available via GitHub at https://github.com/ap152/X-Sig-sensor.
References
Li, D. et al. Motion-unrestricted dynamic electrocardiogram system utilizing imperceptible electronics. Nat. Commun. 16, 3259 (2025).
Chen, S. et al. Starfish-inspired wearable bioelectronic systems for physiological signal monitoring during motion and real-time heart disease diagnosis. Sci. Adv. 11, eadv2406 (2025).
Zhang, L. et al. Fully organic compliant dry electrodes self-adhesive to skin for long-term motion-robust epidermal biopotential monitoring. Nat. Commun. 11, 4683 (2020).
Tan, P. et al. Solution-processable, soft, self-adhesive, and conductive polymer composites for soft electronics. Nat. Commun. 13, 358 (2022).
Yang, S. et al. Stretchable surface electromyography electrode array patch for tendon location and muscle injury prevention. Nat. Commun. 14, 6494 (2023).
Xue, J. et al. A patterned mechanical–electrical coupled sensing patch for multimodal muscle function evaluation. InfoMat 7, e12631 (2025).
Wang, H. et al. High-performance hydrogel sensors enabled multimodal and accurate human–machine interaction system for active rehabilitation. Adv. Mater. 36, 2309868 (2024).
Meng, K. et al. Wearable pressure sensors for pulse wave monitoring. Adv. Mater. 34, e2109357 (2022).
Wang, J. et al. Wearable multichannel pulse condition monitoring system based on flexible pressure sensor arrays. Microsyst. Nanoeng. 8, 16 (2022).
Ates, H. C. et al. End-to-end design of wearable sensors. Nat. Rev. Mater. 7, 887–907 (2022).
Mahato, K. et al. Hybrid multimodal wearable sensors for comprehensive health monitoring. Nat. Electron. 7, 735–750 (2024).
Zhao, C., Park, J., Root, S. E. & Bao, Z. Skin-inspired soft bioelectronic materials, devices and systems. Nat. Rev. Bioeng. 2, 671–690 (2024).
Chung, H. U. et al. Binodal, wireless epidermal electronic systems with in-sensor analytics for neonatal intensive care. Science 363, eaau0780 (2019).
Zhao, Y. et al. Ultra-conformal skin electrodes with synergistically enhanced conductivity for long-time and low-motion artifact epidermal electrophysiology. Nat. Commun. 12, 4880 (2021).
Li, S. et al. Monitoring blood pressure and cardiac function without positioning via a deep learning–assisted strain sensor array. Sci. Adv. 9, eadh0615 (2023).
Min, S. et al. Wearable blood pressure sensors for cardiovascular monitoring and machine learning algorithms for blood pressure estimation. Nat. Rev. Cardiol. 22, 629–648 (2025).
Chun, K. Y., Seo, S. & Han, C. S. A wearable all-gel multimodal cutaneous sensor enabling simultaneous single-site monitoring of cardiac-related biophysical signals. Adv. Mater. 34, 2110082 (2022).
Song, Y. et al. Synergetic monitoring of both physiological pressure and epidermal biopotential based on a simplified on-skin-printed sensor modality. Small 19, 2303301 (2023).
Sharma, S. et al. Wearable multifunctional epidermal patch for continuous and reliable blood pressure monitoring with simultaneous tracking of physiological health states. Adv. Funct. Mater. 36, e16764 (2025).
Hua, Q. et al. Skin-inspired highly stretchable and conformable matrix networks for multifunctional sensing. Nat. Commun. 9, 244 (2018).
Hozumi, S., Honda, S., Arie, T., Akita, S. & Takei, K. Multimodal wearable sensor sheet for health-related chemical and physical monitoring. ACS Sens 6, 1918–1924 (2021).
Han, S. et al. Battery-free, wireless sensors for full-body pressure and temperature mapping. Sci. Transl. Med. 10, eaan4950 (2018).
Cho, S. et al. Wireless, multimodal sensors for continuous measurement of pressure, temperature, and hydration of patients in wheelchair. npj Flex. Electron. 7, 8 (2023).
Lee, S. et al. A fabric-based wearable sensor for continuous monitoring of decubitus ulcer of subjects lying on a bed. Sci. Rep. 13, 5773 (2023).
Yu, Y. et al. All-printed soft human-machine interface for robotic physicochemical sensing. Sci. Robot. 7, eabn0495 (2022).
Xu, C. et al. A physicochemical-sensing electronic skin for stress response monitoring. Nat. Electron. 7, 168–179 (2024).
Chung, H. U. et al. Skin-interfaced biosensors for advanced wireless physiological monitoring in neonatal and pediatric intensive-care units. Nat. Med. 26, 418–429 (2020).
Ryu, D. et al. Comprehensive pregnancy monitoring with a network of wireless, soft, and flexible sensors in high- and low-resource health settings. Proc. Natl Acad. Sci. USA 118, e2100466118 (2021).
Shin, J. H., Choi, J. Y., June, K., Choi, H. & Kim, T. Polymeric conductive adhesive-based ultrathin epidermal electrodes for long-term monitoring of electrophysiological signals. Adv. Mater. 36, 2313157 (2024).
Xiao, Y. et al. High-adhesive flexible electrodes and their manufacture: a review. Micromachines 12, 1505 (2021).
Derakhshankhah, H. et al. Conducting polymer-based electrically conductive adhesive materials: design, fabrication, properties, and applications. J. Mater. Sci. Mater. Electron. 31, 10947–10961 (2020).
Wang, S. et al. Wearable stretchable dry and self-adhesive strain sensors with conformal contact to skin for high-quality motion monitoring. Adv. Funct. Mater. 31, 2007495 (2020).
Ma, Y. et al. Relation between blood pressure and pulse wave velocity for human arteries. Proc. Natl Acad. Sci. USA 115, 11144–11149 (2018).
Tian, G. et al. Hierarchical piezoelectric composites for noninvasive continuous cardiovascular monitoring. Adv. Mater. 36, 2313612 (2024).
IEEE Standard for Wearable Cuffless Blood Pressure Measuring Devices IEEE Std 1708–2014 1–38 (IEEE, 2014).
Franklin, D. et al. Synchronized wearables for the detection of haemodynamic states via electrocardiography and multispectral photoplethysmography. Nat. Biomed. Eng. 7, 1229–1241 (2023).
Moin, A. et al. A wearable biosensing system with in-sensor adaptive machine learning for hand gesture recognition. Nat. Electron. 4, 54–63 (2020).
Luo, Y. et al. Learning human–environment interactions using conformal tactile textiles. Nat. Electron. 4, 193–201 (2021).
Kim, K. K. et al. A substrate-less nanomesh receptor with meta-learning for rapid hand task recognition. Nat. Electron. 6, 64–75 (2022).
Tang, L., Yang, S., Zhang, K. & Jiang, X. Skin Electronics from Biocompatible in situ welding enabled by intrinsically sticky conductors. Adv. Sci. 9, 2202043 (2022).
Yang, H. et al. Adhesive biocomposite electrodes on sweaty skin for long-term continuous electrophysiological monitoring. ACS Mater. Lett. 2, 478–484 (2020).
Wang, S. et al. Self-adhesive, stretchable, biocompatible, and conductive nonvolatile eutectogels as wearable conformal strain and pressure sensors and biopotential electrodes for precise health monitoring. ACS Appl. Mater. Interfaces 13, 20735–20745 (2021).
Cao, J. et al. Stretchable and self-adhesive PEDOT:PSS blend with high sweat tolerance as conformal biopotential dry electrodes. ACS Appl. Mater. Interfaces 14, 39159–39171 (2022).
Yang, X. et al. Ultrathin, stretchable, and breathable epidermal electronics based on a facile bubble blowing method. Adv. Electron. Mater. 6, 2000306 (2020).
Ma, C. et al. Compliant and breathable electrospun epidermal electrode towards artifactfree electrophysiological monitoring. Chem. Eng. J. 490, 151118 (2024).
Pan, L. et al. A compliant ionic adhesive electrode with ultralow bioelectronic impedance. Adv. Mater. 32, 2003723 (2020).
Yang, G. et al. Adhesive and hydrophobic bilayer hydrogel enabled on‐skin biosensors for high‐fidelity classification of human emotion. Adv. Funct. Mater. 32, 2200457 (2022).
Pan, S. et al. Mechanically interlocked hydrogel–elastomer hybrids for on‐skin electronics. Adv. Funct. Mater. 30, 1909540 (2020).
Zheng, Y. et al. Skin temperature-triggered switchable adhesive coatings for wearing comfortable epidermal electronics. Chem. Eng. J. 488, 150459 (2024).
Li, J. et al. Thin, soft, wearable system for continuous wireless monitoring of artery blood pressure. Nat. Commun. 14, 5009 (2023).
Chen, Y., Cheng, S., Wang, T. & Ma, T. Novel blood pressure estimation method using single photoplethysmography feature. In 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC, 2017).
Byfield, R., Miller, M., Miles, J., Guidoboni, G. & Lin, J. Towards robust blood pressure estimation from pulse wave velocity measured by photoplethysmography sensors. IEEE Sens. J. 22, 2475–2483 (2022).
Yang, S., Zhang, Y., Cho, S.-Y., Correia, R. & Morgan, S. P. Non-invasive cuff-less blood pressure estimation using a hybrid deep learning model. Opt. Quantum Electron. 53, 93 (2021).
Lin, M. et al. A fully integrated wearable ultrasound system to monitor deep tissues in moving subjects. Nat. Biotechnol. 42, 448–457 (2023).
Gong, S. et al. A gold nanowire-integrated soft wearable system for dynamic continuous non-invasive cardiac monitoring. Biosens. Bioelectron. 205, 114072 (2022).
Sel, K., Osman, D., Huerta, N., Edgar, A., Pettigrew, R. I. & Jafari, R. Continuous cuffless blood pressure monitoring with a wearable ring bioimpedance device. npj Digital Med. 6, 59 (2023).
Wang, L., Tian, S. & Zhu, R. A new method of continuous blood pressure monitoring using multichannel sensing signals on the wrist. Microsyst. Nanoeng. 9, 117 (2023).
Ibrahim, B. & Jafari, R. Cuffless blood pressure monitoring from a wristband with calibration-free algorithms for sensing location based on bio-impedance sensor array and autoencoder. Sci. Rep. 12, 319 (2022).
Kireev, D. et al. Continuous cuffless monitoring of arterial blood pressure via graphene bioimpedance tattoos. Nat. Nanotechnol. 17, 864–870 (2022).
Poon, C. C. Y. & Zhang, Y. T. Cuff-less and noninvasive measurements of arterial blood pressure by pulse transit time. In IEEE Engineering in Medicine and Biology 27th Annual Conference 5877–5880 (IEEE, 2005).
Ding, X.-R., Zhang, Y.-T., Liu, J., Dai, W.-X. & Tsang, H. K. Continuous cuffless blood pressure estimation using pulse transit time and photoplethysmogram intensity ratio. IEEE Trans. Biomed. Eng. 63, 964–972 (2016).
Acknowledgements
This study was supported by Sichuan Science and Technology Program (grant no. 2024YFFK0133), National University of Singapore Presidential Young Professorship Award (grant no. 22-4974-A0003), MTC Programmatic ‘BLISS’ (grant no. M24M9b0013), Wellcome Leap’s Dynamic Resilience Program jointly funded by Temasek Trust, MOE AcRF Tier 1 grant (grant no. 22-5402-A0001-0), A*STAR Manufacturing, Trade and Connectivity (MTC) MedTech Programmatic Seed grant (grant no. M24N9b0121), National Natural Science Foundation of China (grant no. 52203272), and Fundamental Research Funds for the Central Universities of China, Medical Interdisciplinary Research Key Project of Sichuan University (grant no. 2022).
Author information
Authors and Affiliations
Contributions
X.W. conceptualized the work. X.W., Z.L. and Y.L. developed the methodology. J.W., C.Z., L.Z., Y.S. and X.W. performed experiments. J.W., C.Z., L.Z., Y.S., X.Z., Z.Y., Z.L. and X.W. visualized data. Y.H., Z.W. and Z.J. provide supporting resources, invaluable expertise and insights. X.W., Z.L. and Y.L. acquired funding. X.W., Z.L. and Y.L. supervised the project. J.W., C.Z., L.Z., X.W., Z.L. and Y.L. wrote the paper. X.W., Z.L. and Y.L. edited the paper.
Corresponding authors
Ethics declarations
Competing interests
X.W., C.Z., L.Z. and Y.H. are inventors on a pending patent application in China (202410442845.X) pertaining to this research. The other authors declare no competing interests.
Peer review
Peer review information
Nature Sensors thanks Mengdi Han, Jinxing Li and Xing Sheng for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–10, Supplementary Figs. 1–51, Supplementary Tables 1–3, captions for Supplementary Videos 1–3 and Supplementary References 1–34.
Supplementary Video 1 (download MP4 )
Water-assisted peeling process of adhesive CARD electrodes from the skin surface.
Supplementary Video 2 (download MP4 )
Continuous monitoring of ECG signal with X-Sig sensor under different skin deformations.
Supplementary Video 3 (download MP4 )
Continuous monitoring of HR, SBP and DBP with X-Sig sensor.
Supplementary Data (download XLSX )
Source data for Supplementary Figs. 12b, 29a, 37b and 38–40.
Source data
Source Data Fig. 2 (download XLSX )
Source data for Fig. 2c–j.
Source Data Fig. 3 (download XLSX )
Source data for Fig. 3g–m,o.
Source Data Fig. 4 (download XLSX )
Source data for Fig. 4g–j,l.
Source Data Fig. 5 (download XLSX )
Source data for Fig. 5e–g.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, X., Zhu, C., Zheng, L. et al. A cross-modal epidermal sensor enables single-channel fusion of biopotential and biomechanical signals. Nat. Sens. (2026). https://doi.org/10.1038/s44460-026-00044-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44460-026-00044-0