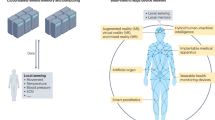
Abstract
Bioelectronic interfaces aim to seamlessly connect biological systems with electronics, enabling real-time monitoring and modulation of physiological activity. Organic electrochemical transistors (OECTs) offer operation relying on mixed ionic–electronic conduction suitable for multimodal biosensing, yet integrating chemical detection, electrophysiology and on-device processing within a single architecture has remained elusive. Here we introduce a monolithically fabricated, flexible OECT array that functions as a multimodal sensing platform. By combining electrolyte-specific regions across the array, we achieve dual-scale dopamine detection across physiological concentrations with picomolar sensitivity, high-bandwidth electroencephalography recording enabled by ion–gel gating, and hydrogel-gated neuromorphic computation capable of classifying seizure activity with 87.8% accuracy, comparable to inorganic neuromorphic hardware. This multifunctional integration demonstrates that a single organic semiconductor can support chemical sensing, signal amplification and on-device computation, extending OECTs beyond single-function electronics. This platform establishes a pathway towards compact, intelligent biointerface sensing systems for continuous monitoring and real-time signal interpretation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The EEG data used for simulated signal recordings, ANN model training and model testing are from the open-source CHB-MIT scalp EEG database at https://physionet.org/content/chbmit/1.0.0/. Figure source data in this study are available via GitHub at https://github.com/wskaust/Mono_BioInterfacing. Further data that support the plots within this paper and other findings of this study are available from the corresponding author upon request.
Code availability
The MATLAB code used to fit both the LTP and LTD data in this study is available via GitHub at https://github.com/wskaust/Mono_BioInterfacing.
References
Tang, X., Shen, H., Zhao, S. Y., Li, N. & Liu, J. Flexible brain–computer interfaces. Nat. Electron. 6, 109–118 (2023).
Remy, A., Lin, X. Y. & Liu, J. Materials for flexible and soft brain–computer interfaces, a review. MRS Commun. 14, 827–834 (2024).
Wang, S. et al. Dopamine detection and integration in neuromorphic devices for applications in artificial intelligence. Device 2, 100284 (2024).
Venkatraman, V. et al. Subthreshold operation of organic electrochemical transistors for biosignal amplification. Adv. Sci. 5, 1800453 (2018).
Melgari, J. M. et al. Alpha and beta EEG power reflects L-dopa acute administration in Parkinsonian patients. Front. Aging Neurosci. 6, 302 (2014).
Pezzetta, R. et al. Combined EEG and immersive virtual reality unveil dopaminergic modulation of error monitoring in Parkinson’s disease. NPJ Parkinsons Dis. 9, 3 (2023).
Waninger, S. et al. Neurophysiological biomarkers of Parkinson’s disease. J. Parkinsons Dis. 10, 471–480 (2020).
Chen, S., Jiang, D., Li, M., Xuan, X. & Li, H. Brain-computer interface and electrochemical sensor based on boron-nitrogen Co-doped graphene-diamond microelectrode for EEG and dopamine detection. ACS Sens. 10, 868–880 (2025).
Giordani, M. et al. Neuromorphic organic devices that specifically discriminate dopamine from its metabolites by nonspecific interactions. Adv. Funct. Mater. 30, 2002141 (2020).
Sharifshazileh, M., Burelo, K., Sarnthein, J. & Indiveri, G. An electronic neuromorphic system for real-time detection of high frequency oscillations (HFO) in intracranial EEG. Nat. Commun. 12, 3095 (2021).
Rivnay, J. et al. Organic electrochemical transistors. Nat. Rev. Mater. 3, 17086 (2018).
Roh, H., Cunin, C., Samal, S. & Gumyusenge, A. Towards organic electronics that learn at the body-machine interface: a materials journey. MRS Commun. 12, 565–577 (2022).
Wang, Y. et al. Designing organic mixed conductors for electrochemical transistor applications. Nat. Rev. Mater. 9, 249–265 (2024).
Zhong, Y., Saleh, A. & Inal, S. Decoding electrophysiological signals with organic electrochemical transistors. Macromol. Biosci. 21, e2100187 (2021).
Rashid, R. B., Ji, X. & Rivnay, J. Organic electrochemical transistors in bioelectronic circuits. Biosens. Bioelectron. 190, 113461 (2021).
Lee, W. et al. Transparent, conformable, active multielectrode array using organic electrochemical transistors. Proc. Natl Acad. Sci. USA 114, 10554–10559 (2017).
Ohayon, D. et al. Biofuel powered glucose detection in bodily fluids with an n-type conjugated polymer. Nat. Mater. 19, 456–463 (2020).
Guo, K. et al. Rapid single-molecule detection of COVID-19 and MERS antigens via nanobody-functionalized organic electrochemical transistors. Nat. Biomed. Eng. 5, 666–677 (2021).
Ling, H. F. et al. Electrolyte-gated transistors for synaptic electronics, neuromorphic computing and adaptable biointerfacing. Appl. Phys. Rev. 7, 011307 (2020).
Wang, S. J. et al. An organic electrochemical transistor for multi-modal sensing, memory and processing. Nat. Electron. 6, 281–291 (2023).
Torricelli, F. et al. Electrolyte-gated transistors for enhanced performance bioelectronics. Nat. Rev. Methods Primers 1, 66 (2021).
Lu, L. et al. Stretchable all-gel organic electrochemical transistors. Nat. Commun. 16, 3831 (2025).
Lee, Y., Park, S. J., Kwon, J., Song, K. I. & Jung, S. Addressing transconductance-bandwidth trade-off by three-dimensional electrolyte-surrounded organic electrochemical transistors. Sci. Adv. 11, eady0279 (2025).
Savva, A. et al. Influence of water on the performance of organic electrochemical transistors. Chem. Mater. 31, 927–937 (2019).
Armand, M., Endres, F., MacFarlane, D. R., Ohno, H. & Scrosati, B. Ionic-liquid materials for the electrochemical challenges of the future. Nat. Mater. 8, 621–629 (2009).
Zhong, Y. et al. Ionic liquid gated organic electrochemical transistors with broadened bandwidth. ACS Appl. Mater. Inter. 16, 61457–61466 (2024).
Melianas, A. et al. Temperature-resilient solid-state organic artificial synapses for neuromorphic computing. Sci. Adv. 6, eabb2958 (2020).
Quill, T. J. et al. Ion pair uptake in ion gel devices based on organic mixed ionic–electronic conductors. Adv. Funct. Mater. 31, 2104301 (2021).
Lee, H. et al. Ultrathin organic electrochemical transistor with nonvolatile and thin gel electrolyte for long-term electrophysiological monitoring. Adv. Funct. Mater. 29, 1906982 (2019).
Xie, Z. C. et al. All-solid-state vertical three-terminal N-type organic synaptic devices for neuromorphic computing. Adv. Funct. Mater. 32, 2107314 (2022).
Zhang, Y. et al. Adaptive biosensing and neuromorphic classification based on an ambipolar organic mixed ionic-electronic conductor. Adv. Mater. 34, e2200393 (2022).
Zhao, Y. F. et al. Side chain engineering enhances the high-temperature resilience and ambient stability of organic synaptic transistors for neuromorphic applications. Nano Energy 104, 107985 (2022).
Wang, Y. et al. Hybrid alkyl–ethylene glycol side chains enhance substrate adhesion and operational stability in accumulation mode organic electrochemical transistors. Chem. Mater. 31, 9797–9806 (2019).
Zhong, Y. et al. Eutectogels as a semisolid electrolyte for organic electrochemical transistors. Chem. Mater. 36, 1841–1854 (2024).
Kurian, M. A., Gissen, P., Smith, M., Heales, S. Jr & Clayton, P. T. The monoamine neurotransmitter disorders: an expanding range of neurological syndromes. Lancet Neurol. 10, 721–733 (2011).
Roelands, B. & Meeusen, R. Alterations in central fatigue by pharmacological manipulations of neurotransmitters in normal and high ambient temperature. Sports Med. 40, 229–246 (2010).
Okumura, T., Nakajima, Y., Matsuoka, M. & Takamatsu, T. Study of salivary catecholamines using fully automated column-switching high-performance liquid chromatography. J. Chromatogr. B 694, 305–316 (1997).
Shetty, S. R. et al. Salivary ascorbic acid levels in betel quid chewers: a biochemical study. South Asian J. Cancer 2, 142–144 (2013).
Lomonaco, T. et al. The effect of sampling procedures on the urate and lactate concentration in oral fluid. Microchem. J. 136, 255–262 (2018).
Bakhtiari, S., Toosi, P., Samadi, S. & Bakhshi, M. Assessment of uric acid level in the saliva of patients with oral lichen planus. Med. Princ. Pract. 26, 57–60 (2017).
Thanarajah, S. E. et al. Food intake recruits orosensory and post-ingestive dopaminergic circuits to affect eating desire in humans. Cell Metab. 29, 695–706 e694 (2019).
Liao, C., Zhang, M., Niu, L., Zheng, Z. & Yan, F. Organic electrochemical transistors with graphene-modified gate electrodes for highly sensitive and selective dopamine sensors. J. Mater. Chem. B 2, 191–200 (2014).
Gualandi, I. et al. Selective detection of dopamine with an all PEDOT:PSS Organic Electrochemical Transistor. Sci. Rep. 6, 35419 (2016).
Qing, X. et al. Wearable fiber-based organic electrochemical transistors as a platform for highly sensitive dopamine monitoring. ACS Appl. Mater. Inter. 11, 13105–13113 (2019).
Ji, W. et al. Carbonized silk fabric-based flexible organic electrochemical transistors for highly sensitive and selective dopamine detection. Sens. Actuator B 304, 127414 (2020).
Park, S. J. et al. High-performance conducting polymer nanotube-based liquid-ion gated field-effect transistor aptasensor for dopamine exocytosis. Sci. Rep. 10, 3772 (2020).
Tang, K. et al. Organic electrochemical transistor with molecularly imprinted polymer-modified gate for the real-time selective detection of dopamine. ACS Appl. Polym. Mater. 4, 2337–2345 (2022).
Tseng, H. S., Chen, Y. L., Zhang, P. Y. & Hsiao, Y. S. Additive blending effects on PEDOT:PSS composite films for wearable organic electrochemical transistors. ACS Appl. Mater. Interfaces 16, 13384–13398 (2024).
Gamboa, J. et al. Carbon quantum dots composite for enhanced selective detection of dopamine with organic electrochemical transistors. Microchim. Acta 191, 639 (2024).
Ito, K. et al. Development of biosignal recording board system with agile control of circuit characteristics for various biosignals. Electr. Commun. Jpn 101, 47–54 (2018).
Goldberger, A. L. et al. PhysioBank, PhysioToolkit and PhysioNet: components of a new research resource for complex physiologic signals. Circulation 101, E215–E220 (2000).
Shoeb, A. H. Application of Machine Learning to Epileptic Seizure Onset Detection and Treatment. PhD thesis, Massachusetts Institute of Technology (2009).
Guttag, J. CHB-MIT Scalp EEG Database (version 1.0.0) (PhysioNet, 2010).
Seneviratne, U., Cook, M. & D’Souza, W. The electroencephalogram of idiopathic generalized epilepsy. Epilepsia 53, 234–248 (2012).
Wustoni, S. et al. MXene improves the stability and electrochemical performance of electropolymerized PEDOT films. APL Mater. 8, 121105 (2020).
Spyropoulos, G. D., Gelinas, J. N. & Khodagholy, D. Internal ion-gated organic electrochemical transistor: a building block for integrated bioelectronics. Sci. Adv. 5, eaau7378 (2019).
Ji, X. et al. Mimicking associative learning using an ion-trapping non-volatile synaptic organic electrochemical transistor. Nat. Commun. 12, 2480 (2021).
Liu, R. et al. Hardware-feasible and efficient N-type organic neuromorphic signal recognition via reservoir computing. Adv. Mater. 37, e2409258 (2025).
Kumar, D. et al. Highly efficient back-end-of-line compatible flexible Si-based optical memristive crossbar array for edge neuromorphic physiological signal processing and bionic machine vision. Nanomicro Lett. 16, 238 (2024).
Zhong, Y. An organic electrochemical transistor integrated photodetector for high quality photoplethysmogram signal acquisition. Adv. Funct. Mater. 33, 2211479 (2023).
Zhong, Y. et al. Monitoring blood pressure through a single hybrid hemodynamic signal with a flexible optoelectronic patch. Device 3, 100778 (2025).
Wang, Y. et al. An optoelectrochemical synapse based on a single-component n-type mixed conductor. Nat. Commun. 16, 1615 (2025).
Koklu, A. et al. Convection driven ultrarapid protein detection via nanobody-functionalized organic electrochemical transistors. Adv. Mater. 34, e2202972 (2022).
Long, G. L. & Winefordner, J. D. Limit of detection. A closer look at the IUPAC definition. Anal. Chem. 55, 712A–724A (1983).
Schulz, P. E., Cook, E. P. & Johnston, D. Changes in paired-pulse facilitation suggest presynaptic involvement in long-term potentiation. J. Neurosci. 14, 5325–5337 (1994).
Higuchi, T. Approach to an irregular time series on the basis of the fractal theory. Physica D 31, 277–283 (1988).
Acknowledgements
We acknowledge H. Ombao and his team (Computer, Electrical and Mathematical Sciences and Engineering Division, KAUST), including I. I. Gauran and F. A. Gomawi, for the design of the clinical study involving saliva, and A. Hama (Biological and Environmental Science and Engineering Division, KAUST) for his help with the design and fabrication of the PCB used in this work. We thank N. El-Atab and D. Kumar (Smart Advanced Memory Devices and Applications Laboratory, KAUST) for providing the inorganic neuromorphic device. We acknowledge M. Amad and P. L. Sanchez (Analytical Chemistry Core Lab, KAUST) for their assistance with dopamine sensing using ELISA. This publication is based on work supported by KAUST under awards ORA-2021-CRG10-4650 and OFP2023-5522 and the KAUST Center of Excellence for Smart Health (KCSH) under award 5932. We acknowledge funding by the European Union as part of the IONBIKE 2.0 project (grant agreement no. 101129945; https://doi.org/10.3030/101129945). Participants of this study provide their consent for the publication of the manuscript detailed above, including any accompanying images or data contained within the manuscript that indirectly disclose their identities.
Author information
Authors and Affiliations
Contributions
Y.Z. fabricated the device, performed OECT characterization, electrochemical characterization, dopamine-sensing measurements and electrophysiological monitoring measurements. W.S. prepared the [BMIM][TFSI] IL gel, performed the neuromorphic computing measurements and developed the machine learning methods. Y.W. provided the semiconducting polymer PIBET-AO. H.A. performed the UV–vis spectroscopy measurements. D.A. helped with the design of the dopamine-sensing measurements and contributed to the sensing mechanism investigations. N.L.-L. and D.M. provided the hydrogel. S.I. conceived the research, designed the experiments, and supervised the work. Y.Z. and S.I. wrote the paper and interpreted the results with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Sensors thanks Keehoon Kang, Shiming Zhang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Details of flexible OECT array.
a The exploded-view schematics showing the components of the flexible OECT array. b The interconnects on the flexible OECT array. c Microscope image of all 64 channels on the flexible OECT array. d A close-up view of four channels, with the dimensions of L = 10 μm, W = 100 μm, and d = 73 nm. e The photograph of the PCB housing the flexible OECT array, enabling communication from the OECT array to the external analyzing instrument. f Hardware setup for connecting the flexible OECT array to the external instrument for all applications demonstrated in this work.
Extended Data Fig. 2 Electrochemical characterization of PIBET-AO film.
a 50 successive CV curves of a PIBET-AO film recorded in PBS or IL. The scan rate was 50 mV/s. b The Nyquist plot of a PIBET-AO film recorded in PBS or IL. Insets show the data points at low impedance values and the equivalent circuit model used to fit the impedance spectra, consisting of the electrolyte resistance (Rs), the polymer capacitance (Cp), the charge transfer resistance (Rct), and the Warburg impedance (ZW). c The Bode plot of a PIBET-AO film recorded in PBS or IL (solid lines and triangle symbols correspond to the magnitude and the phase of the impedance, respectively). d Volumetric capacitance (C*) and Rct of the PIBET-AO film recorded in PBS or IL. Data are presented as mean values ± s.d., calculated from 3 PIBET-AO film samples.
Extended Data Fig. 3 The electrochemical potential of OECT terminals.
a The schematic of multichannel potentiostat setup to determine the electrochemical potential of transistor terminals with respect to the Ag/AgCl reference electrode during device operation. b Real-time electrochemical potential changes at the OECT terminals (gate potential, EG, drain potential, ED, and source potential, ES) were measured in PBS with dopamine. VD was at −0.2 V, and VG changed from 0 to −0.5 V with a step of 0.1 V.
Extended Data Fig. 4 Dopamine oxidation at the PIBET-AO film.
a The real-time OCP change of the PIBET-AO thin film as dopamine or oxidized dopamine are added to the PBS at various concentrations. The arrows mark the addition of each concentration. The OCP shift resulting from blank PBS addition is also plotted. b The change in OCP when correcting by the change caused by PBS pipetting (ΔOCP) of as a function of dopamine concentrations.
Extended Data Fig. 5 Scan rate dependent CV measurements.
Five successive CV curves of a PIBET-AO film recorded in PBS and dopamine-containing PBS with different concentrations (1 pM, 20 pM, 1 nM, 20 nM, 500 nM, 10 μM, 500 μM, and 10 mM). The scan rates were 25 mV/s, 50 mV/s, and 100 mV/s for each measurement.
Extended Data Fig. 6 EEG frequency spectra comparison.
Comparison of EEG frequency spectra between simulator output (red) and OECT-acquired signals (blue) across all eight recordings, processed using a fast Fourier transform. Spectra were normalized to the peak amplitude within the 0.5 to 10 Hz range.
Extended Data Fig. 7 The flexible OECT array configuration to acquire EEG from the volunteer.
a The configuration includes attaching OECT array on the subject’s forehead and setting other gadgets (polymer electrodes, FFC, and PDMS). Photographs of b polymer electrode coated by PEDOT:PSS and c FFC.
Extended Data Fig. 8 Assessment of the flexible OECT array for concurrent multimodal applications.
a The real-time change in OECT | ID| in response to dopamine at concentrations of 10 μM, 500 μM, and 10 mM, measured at VD = -0.2 V and VG = -0.25 V. b The LR calculated from the data in a. c The concurrent simulated real-time EEG signal recorded during the dopamine sensing. d Comparison of the LR results with the calibration curve obtained from Supplementary Fig. 14. e Comparison of EEG signals in the frequency domain from the beginning and end sections of the real-time measurement.
Extended Data Fig. 9 Assessment of the interfacial stability between the gels and the PIBET-AO thin film.
a Schematic of interfacial impedance measurement using the 3-electrode configuration. b Attachment of flexible OECT array on a chicken breast to simulate a wearable condition. c The Nyquist plot of a PIBET-AO film with the hydrogel. d The Bode plot of the PIBET-AO film with the hydrogel. e The Nyquist plot of a PIBET-AO film with the IL gel. f The Bode plot of the PIBET-AO film with the IL gel. The data in c-f were recorded right after the simulated wearable configuration (0 h) and 1, 2, 4, 8, 24 hours after the first measurement.
Extended Data Fig. 10 LTP/LTD operation of the hydrogel-gated OECT.
ΔID measured over 500 pulsing cycles and using time intervals of tp = Δt = 2 ms. Measurements were conducted: a after initial contact with the hydrogel and after b 1, c 2, d 4, and e 8 hours, respectively. VG pulses alternated between 0 and -0.6 V, with VD fixed at -0.6 V.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–33, Tables 1–3, Notes 1–8 and references.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhong, Y., Shan, W., Wang, Y. et al. Monolithic design of an organic electrochemical transistor array for multimodal bioelectronic interfacing. Nat. Sens. (2026). https://doi.org/10.1038/s44460-026-00052-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44460-026-00052-0