Abstract
Many notable applications have been described for magnetic nanoparticles in delivery of diverse drugs and bioactive compounds into cells, magnetofection for the treatment of cancer, photodynamic therapy, photothermal therapy, and magnetic particle imaging (MPI). In response to the growing demand for magnetic nanoparticles for drug delivery or biomedical imaging applications, more effective and eco-friendly methodologies are required for large-scale biosynthesis of this nanoparticles. The major challenge in the large-scale biomedical application of magnetic nanoparticles lies in its low efficiency and optimization of nanoparticle production can address this issue. In the current study, a prediction model is suggested by the fractional factorial designs. The present study aims to optimize culture media components for improved growth and iron uptake of this strain. The result of optimization for iron uptake by the strain ASFS1 is to increase the production of magnetic nanoparticles by this strain for biomedical applications in the future. In the present study, design of experiment method was used to probe the effects of some key medium components (yeast extract, tryptone, FeSO4, Na2-EDTA, and FeCl3) on Fe content in biomass and dried biomass of strain ASFS1. A 25−1 fractional factorial design showed that Na2-EDTA, FeCl3, yeast extract-tryptone interaction, and FeSO4-Na2-EDTA interaction were the most parameters on Fe content in biomass within the experimented levels (p < 0.05), while yeast extract, FeCl3, and yeast extract-tryptone interaction were the most significant factors within the experimented levels (p < 0.05) to effect on dried biomass of strain ASFS1. The optimum culture media components for the magnetic nanoparticles production by strain ASFS1 was reported to be 7.95 g L−1 of yeast extract, 5 g L−1 of tryptone, 75 μg mL−1 of FeSO4, 192.3 μg mL−1 of Na2-EDTA and 150 μg mL−1 of FeCl3 which was theoretically able to produce Fe content in biomass (158 μg mL−1) and dried biomass (2.59 mg mL−1) based on the obtained for medium optimization. Using these culture media components an experimental maximum Fe content in biomass (139 ± 13 μg mL−1) and dried biomass (2.2 ± 0.2 mg mL−1) was obtained, confirming the efficiency of the used method.
Similar content being viewed by others
Introduction
Nanotechnology is an interdisciplinary field at the forefront of scientific research, encompassing chemistry, fundamental physics, biology, medicine, and material science1,2,3. The concept of nanoscience was first presented by Nobel prize-winning physicist Richard Feynman to construct different materials at the nanoscale level in 19593,4. Nanomaterials comprise various categories of materials if those have at least one dimension smaller than 100 nm3,4,5. Among them, magnetic nanoparticles (MNPs) are an intriguing group of nanoparticles that have been considerably utilized in diverse fields because of their magnetic properties such as targeted drug delivery, sensing technologies, magnetic labeling, engineering, magnetic separation, and environment applications6,7,8,9,10,11,12. Among MNPs, iron oxide nanoparticles (IONPs) are promising nanoparticles due to their superior biocompatibility for biomedical applications such as magnetic hyperthermia6,13,14. The comprehensive investigation efforts are underway to commercialize IONPs for progressive biomedical applications15. The superparamagnetic magnetite (Fe3O4) is the most common iron oxide compared to α-Fe2O3 (hematite) and γ-Fe2O3 (maghemite) due to its superior magnetic properties, higher biocompatibility, and lower cytotoxicity6,16. Several techniques for preparing nanoparticles have been described, including physical, chemical, and biological methods3. The biological synthesis of nanoparticles is being performed by various macro and microscopic organisms such as bacteria, fungi, viruses, yeast, and plant extracts3,17. The biosynthesis of nanoparticles has received considerable attention in recent years due to their environment-friendly behavior, lower toxicity, more biocompatibility, and more potential for pharmaceutical and biomedical applications than conventional synthesis including chemical and physical methods18,19. Biogenic nanoparticles are remarkably stable and can be made mono-dispersed by changing various factors such as temperature, shaking speed, pH, and incubation duration20. Furthermore, magnetic nanoparticles synthesized by biological methods are pharmacologically better than those produced with physical or chemical techniques21. However, microbial-assisted Synthesis is slow and needs long stages such as culturing, nanoparticle extraction, and purification20. The efficacy of biosynthesis techniques is yet limited to the laboratory step because biosynthesis methods are not still capable of producing large-scale homogeneous nanoparticles19. Thus, there is a critical necessity for stable, efficient, scalable, and eco-friendly fabrication procedures19. Although there have been significant advancements in the fabrication of nanoparticles using various methods such as hydrothermal reactions, laser pyrolysis, co-precipitation, sol–gel method, electrochemistry, flow injection syntheses, as summarized by Laurent et al13. developing an ideal synthetic method for IONP still poses a challenge. This optimal synthesis should meet certain criteria, such as high production yield and avoiding the use of harmful chemicals22. Design of experiments (DoE) is described as a mathematical model that can be employed to design and investigate experiments23. In addition, this method significantly reduces the number of experiments required to screen a large number of variables (more than three variables) simultaneously24. In a DoE experiment, the various factors are discretized into a collection of values that are presented as levels. These levels experiment in various assortments, and a model predicting the response of the system is created based on input data and data collected via multiple iterations of investigation25. Fractional Factorial Design (FFD) supplies details on potential high-order interactions, something that is difficult utilizing a full factorial approach – measuring one factor at a time24. Another advantage of FFD over full factorial is fewer experimental runs due to decreasing the time needed to obtain data and reducing experiment costs24,26. In this study, we carried on our previous microbial synthesis approach using the thermophilic strain ASFS1, producing intracellular superparamagnetic Fe3O4 nanoparticles. Utilizing fractional factorial design (FFD), as a collection of mathematical and statistical techniques, we determined the optimal medium composition and ideal productive conditions for cultivating the strain ASFS. Additionally, we employed the dry weight of biomass and iron in the cell plate to monitor the corresponding biomineralization process.
Material and method
Materials and reagents
Tryptone, ferrous sulfate (FeSO4), yeast extract, NaCl, ethylenediaminetetraacetic acid (EDTA), and ferric chloride (FeCl3) were provided by Merck Chemicals (Darmstadt, Germany).
Bacterial strain
Bacillus tequilensis strain ASFS1 (GenBank accession number is MZ669973.1), which magnetic behavior and synthesis of superparamagnetic Fe3O4 nanoparticles had proven by Satarzadeh et al27. in 2024. This strain was used in this study to optimize the growth of bacteria and the iron uptake.
Biosynthesis of superparamagnetic Fe3O4 nanoparticles
To produce magnetite nanoparticles by strain ASFS1, 2 mL of fresh inoculum (equal to 0.5 McFarland) was added to 500 mL Erlenmeyer flasks containing 200 mL of Luria–Bertani medium (containing yeast extract 0.5 g, tryptone 1 g, and NaCl 1 g in 100 ml distilled water) supplemented with FeSO4 (200 μg mL−1) and FeCl3 (400 μg mL−1) and incubated at 37 °C and shaken for 72 h (120 rpm) and then 24 h not shaken according to previous report. After incubation, the bacterial cells were removed from the culture medium by centrifugation (12,800 g for 5 min). The cell pellets were transmitted to a mortar and disrupted by freeze–thaw cycles (5 cycles). Magnetic nanoparticles were separated using a magnetic and reproducible method previously described. In summary, magnetite nanoparticles were separated from disrupted cells using passing via a magnetic separation with a syringe placed near a neodymium-iron-boron magnet and set on solid phase extraction (SPE) Manifolds. Subsequently, the syringe was washed with 1.5 M Tris/HCl buffer (pH 8.3), 1% SDS, and deionized water respectively. Finally, the magnet was separated from the syringe to elute magnetite nanoparticles with deionized water. The obtained magnetite nanoparticles were studied by field-emission scanning electron microscopy (FESEM, Sigma VP model, ZEISS, Germany). This study was conducted to optimize the components of the culture medium to increase the number of cells and subsequently increase the amount of magnetite nanoparticles. The amount of magnetite nanoparticles was analyzed using an indirect method as previously described28. In summary, one ml aliquots (96 h incubated) were centrifuged (9600 g for 5 min) and the supernatant was discarded. The cell pellets were washed with sterile 0.9% NaCl solution mixed with 69% nitric acid (1 ml) and incubated at 98 °C for 2 h. The amount of iron in the cell pellets were measured using an atomic absorption spectrometer (AA670, Shimadzu, Japan). To measure the biomass dry weight, one ml of the samples was centrifuged and then dried and weighed.
Experimental design
A 25−1 FFD, was employed to screen the most effective variables in superparamagnetic Fe3O4 nanoparticles production by strain ASFS1 and for developed a model for optimal conditions. Luria–Bertani medium was used as the basal medium, and then FeSO4, FeCl3, and ethylenediaminetetraacetate disodium salt dihydrate (Na2-EDTA) were added to the basal media. The parameters were selected yeast amount (X1), tryptone amount (X2), FeSO4 concentration (X3), Na2-EDTA concentration (X4), and FeCl3 concentration (X5). The range and the levels of the factors showed in Table 1.
A 25−1 FFD with three center points consisting of 19 factorial runs were designed by Design Expert version 6.0.4 statistical software (Table 2). The coding of the factors was accomplished according to the following equation:
In this study, \({x}_{i}\) refers to the coded value of an independent variable, \({X}_{i}\) represents the real value of the independent variable, \({X}_{i;0}\) denotes the real value of the independent variable at the center point, and \(\Delta {X}_{i}\) represents the step change value. The dry weight of biomass (one ml of bacterial culture) and iron content in biomass (one ml of bacterial culture) were measured as the dependent variable or response. The runs were conducted randomly in an attempt to minimize the influence of extraneous variables. The selected factors can be linked to the response through a model, which is presented in the following equation:
In this equation, \({x}_{i}\) (i = 1–5) represents the variables of the experiment. Y denotes the response function, which in this study is Fe content in biomass (Response 1) and dried biomass (Response 2). The variable e represents the experiment's error. \({b}_{0}\) showed the constant coefficient, while bi (i = 1–5) refers to the linear coefficient. \({b}_{ij}\) (i ≠ j) represents the second-order coefficient, and \({b}_{ii}\) (i = 1–4) signifies the second-order interaction coefficient. The independent variables denoted by x. In the present study, the independent variables were coded as A, B, C, D, and E. Eventually, the predicted model was confirmed for variables used in current design.
Statistical analysis
The statistical analysis of the outcomes was conducted by Design Expert@ version 7.0.0. The qualities of fitted models assessed based on the coefficient of determination (R2). To assess the significance of each term, the ANOVA combined with the F-test was employed to analyze the iron concentration in the cell plate and dry weight of biomass, with a significance level of p ≤ 0.05. The location of the optimum variables was estimated using solving the fitted model.
Results and discussion
Biosynthesis of nanoparticles
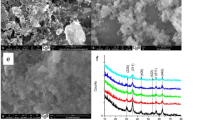
As demonstrated in Fig. 1A (the FESEM image of the purified superparamagnetic Fe3O4 nanoparticles), biogenic magnetite nanoparticles well dispersed and spherical shape. The particle size distribution of biogenic magnetite nanoparticles based on the FESEM image is demonstrated in Fig. 1B. The particle size histogram of magnetite nanoparticles indicates that the particle size ranges from 10 to 110 nm which may be due to the magnetic property that they attach. The frequency distribution revealed that almost 50% of the particles are in the range of 10–40 nm.
Optimization of biogenic magnetite nanoparticles production
The first step towards optimization is to identify the components of the medium that have a notable effect on magnetite nanoparticles biosynthesized by strain ASFS1. Five medium components were evaluated at specified levels (Table. 1). The color change of the culture medium to a brownish-black hue after 96 h showed in 19 runs, which indicates the production of magnetite nanoparticles (Fig. 2). The Fe content in biomass and dried biomass results in an augmented 25−1 FFD with the levels in coded units described in Table 2. The response range were from 25.6 to 160.8 μg mL−1 for Fe content in biomass (response 1) and 1.0–3.3 mg mL−1 for dried biomass (response 2). To the analysis of the response effects and assess the significance of the model terms, the ANOVA tables were designed (Table 3 for response 1 and response 2).
Changing the color of the culture medium in 19 different runs designed with different components of the culture medium. The change in the color of the culture medium to brownish-black indicates an increase iron uptake by strain ASFS1. The result of increase for iron uptake by this strain is to increase the synthesis of magnetic nanoparticles. The result of increase for iron uptake by this strain is to increase the synthesis of magnetic nanoparticles.
The normality of the data for both responses examined using plotting a normal probability plot of the residuals. According to Fig. 3, the data points on the plot fall fairly near to the straight line, which indicates the data follows a normal distribution with a relatively consistent variance throughout the responses range.
In response 1, X4, X5, X1 X2, X3 X4 are significant model terms. The p-value of Na2-EDTA (X4) was less than 0.05 (Table 3), thus this parameter had a significant effect on superparamagnetic Fe3O4 nanoparticles production strain ASFS1. Alphandéry et al29. reported addition of iron-chelating agents (specially EDTA) to the bacterial growth medium enables the enhance of the growth of Magnetospirillum magneticum strain AMB-1, an increase production yield of the magnetosomes size and chain length and yielded improved magnetosome heating properties. The kinetics of magnetosome synthesis in Magnetospirillum magneticum strain AMB-1 probably happens after the bacteria have reached the stationary phase29,30. Adding iron-chelating agents to the growth medium improves the rate of production of magnetic nanoparticles strain AMB-129. Nevertheless, in the strain MSR-1, magnetosomes are produced before the bacteria have reached the stationary phase, possibly due to various mechanisms of magnetosome formation or iron uptake29,31. Thus, the effects of the presence of iron-chelating agents on the rate of magnetosome formation may be less noticeable for strain MSR-1 than strain AMB-129,31. Therefore, EDTA as an iron-chelating factor has a positive effect on the growth and iron uptake in the strain ASFS1 of this study, similar to the AMB-1strain29, due to probably the mechanism of formation of magnetic nanoparticles in both strains being similar and different from the MSR1 strain31. Similar to our study, Heyen et al31. and Alphandéry et al29. optimized the components of the culture medium for strains MSR-1 and AMB-1 respectively, unlike the present study, none of them used the experimental design method. The use of statistical methods such as Design Expert to design optimization experiments enhanced the efficiency and precision, and it provides investigation of several factors simultaneously. Finally, it improved the comprehensiveness of optimization results in bioprocessing applications25. Iron is a significant factor influencing the production of magnetic nanoparticles using magnetotactic bacteria32,33. Fe (III) deposited directly inside the bacterial cells comprising magnetosomes32. However, magnetotactic bacteria can utilize different iron sources, but the association between growth bacteria and iron uptake is not so clear33. Analysis of various concentrations of FeCl3 (X5) exhibited that this factor had considerable effect on magnetite nanoparticles production by strain ASFS1 (p < 0.05), while different concentration of FeSO4 had not significant effects (p < 0.05). Similar to our study, kabary et al33. reported that FeSO4 were less significant effects for bacterial growth and iron uptake by Pseudomonas aeruginosa Kb1 (confirmed as magnetotactic bacteria). Some studies have used FeSO4 as a single source of iron34,35. Berny et al22. used FeSO4 and FeCl3 as iron sources for the production of magnetic nanoparticles by magnetotactic bacteria, similar to our study. Although, various concentrations of FeSO4 had not significant effects, the interaction effect of X3 X4 is significant (p < 0.05) in the present study. Table 3 revealed that the yeast concentration (X1) and tryptone concentration (X2) not significant separately, while the interaction of X1X2 is significant. Yang et al36. showed yeast extract is not significant in magnetic nanoparticle production by bacteria. Their results are similar to our study, whereas peptone only improves the final bacterial cell density. Furthermore, adding only yeast extract and polypeptide as a culture medium produces a small number of magnetosomes37, similar to the current study.
Figure 4A, B exhibited one factor affecting Fe content in biomass within their experimental range while holding four factors constant. Furthermore, Fig. 4C, D illustrate the interaction of two factors, which the other three factors are kept constant. In Fig. 4A, tryptone (10 g L-1), yeast extract (5 g L-1), FeSO4 (50 μg mL−1), and FeCl3 (100 μg mL−1) are constant, and with increasing Na2-EDTA concentration, the amount of iron in the biomass significantly increased. Analysis of response at the different levels of FeCl3 when tryptone (10 g L-1), yeast extract (5 g L-1), FeSO4 (50 μg mL−1), and Na2-EDTA (100 μg mL−1) are constant, indicating that FeCl3 considerably increased iron in the biomass (Fig. 4B). The interaction of tryptone and yeast extract on iron in the biomass (Fig. 4C) exhibited that by raising tryptone, the amount of iron in biomass not significantly raised, while using increasing yeast extract, the amount of iron in biomass considerably increased (constant factors: 50 μg mL−1 of FeSO4, 100 μg mL−1 of Na2-EDTA and 100 μg mL−1 of FeCl3). Thus, yeast extract alone has no effect on the amount of iron in biomass, but its interaction with tryptone has a significant effect on increasing the amount of iron in biomass. In Fig. 4D, the interaction of FeSO4 and Na2-EDTA shows that the amount of iron in biomass does not increase with the increase level of FeSO4, while increase level of Na2-EDTA lead to the amount of iron in biomass increases significantly).
In response 2, X1, X5, X1 X2 are significant model terms. The p-value of yeast extract (X1) and FeCl3 (X5) was less than 0.05 (Table 4). Therefore, both parameters have significant effects on magnetite nanoparticles production strain ASFS1. Contrary to Yang et al36. study that polypeptone increases the biomass dry weight and yeast extract has no significant, the yeast extract increases biomass dry weight in the present study. Also, the interaction effects of yeast extract and tryptone (X1X2) is significant (p < 0.05).
Figure 5A, B displayed one factor affecting dried biomass within their testing range and keeping four factors’ constants. In addition, Fig. 5C revealed the interaction of two factors, which the further three factors are held constant. The results show that increasing the level of yeast extract and FeCl3 separately, has led to a significant increase in dry biomass (Fig. 5A, B, respectively). In Fig. 5C, the interaction of yeast extract and tryptone displays that dry biomass does not significantly rise with the raised level of tryptone, while the enhanced level of yeast extract leads to dry biomass enhancing considerably (constant factors: 100 μg mL−1 of FeCl3, 100 μg mL−1 of Na2-EDTA and 50 μg mL−1 of FeSO4).
The fitted model (in the terms of coded values) for estimating Fe content in biomass (3) and dried biomass from 1 cc culture (4) by the selected strain were, respectively:
The effects of X1, X3, X4, X5, and X3 X4 were positive (synergistic), while X2 and X1 X2 had negative (antagonism) effects on Fe content in biomass. In response 2, the effects of X1, X2, X5 were synergistic and only X1 X2 had antagonism effects on Dried Biomass.
The Model F-value of 8.42 and 8.57 for response 1 and response 2 respectively, implies the models are significant. Furthermore, values of "Prob > F" less than 0.0500 for both responses indicate model terms are significant. There is only a 0.16% and a 0.13% chance that a "Model F-value" this large could occur due to noise for responses 1 and 2, respectively. The relatively high value of R2 = 0.8549 (response 1) and R2 = 0.7251 (response 2) indicated that the models were significantly fitted. The higher the R2 value (closer to the number one) indicates better the model fits, as it explains more of the data's variance. In this study, R2 = 0.8549 (response 1) and R2 = 0.7251 (response 2) indicate a good quality model that explains 85% and 72% of the data variance, respectively.
The location of optimum levels by the selected model for Fe content in biomass (158 μg mL−1) and dried biomass (2.59 mg mL−1) were predicted to be 7.95 g L−1 of yeast extract, 5 g L−1 of tryptone, 75 μg mL−1 of FeSO4, 192.3 μg mL−1 of Na2-EDTA and 150 μg mL−1 of FeCl3.
To confirm the model adequacies for predicting Fe content in biomass and dried biomass, one additional experiment (repeated three times) by the same medium composition were performed. In the event that the average of the validation outcomes was within the limits of the confidence interval (CI), then the significant parameters and suitable levels for acquiring the desired outcomes were correctly selected38. The outcomes of three validation experiments demonstrated that there are good agreements between the predicted (The Fe content in biomass and dried biomass were 158 μg mL−1 and 2.59 mg mL−1, respectively) and experimental (The Fe content in biomass and dried biomass were 139 ± 13 μg mL−1 and 2.2 ± 0.2 mg mL−1, respectively) outcomes that revealed the chosen model was able to navigate within the design space.
Conclusion
The present study was aimed to optimize culture media components for strain ASFS1 growth and magnetite nanoparticles formation by fractional factorial designs. Yeast extract, tryptone, and FeSO4 were confirmed not to be very significant factors considering to Fe content in biomass assisted by strain ASFS1, while Na2-EDTA, FeCl3, interaction yeast extract-tryptone, and interaction FeSO4- Na2-EDTA were illustrated the significant effect on Fe content in biomass. Tryptone was proved not significant parameter considering to dried biomass, while yeast extract, FeCl3, and interaction yeast extract-tryptone were demonstrated a significant effect on dried biomass of strain ASFS1. In both responses 1 and 2, the factors of FeCl3 and interaction effects yeast extract-tryptone are significant (p < 0.05). The maximum fe content in biomass, along with the maximum dried biomass, were achieved at 7.95 g L−1 of yeast extract, 5 g L−1 of tryptone, 75 μg mL−1 of FeSO4, 192.3 μg mL−1 of Na2-EDTA and 150 μg mL−1 of FeCl3. Nevertheless, further studies must be accomplished to find about the related pathway involved in magnetite nanoparticles biosynthesis and the potential application of this strain for large-scale production of superparamagnetic Fe3O4 nanoparticles.
Data availability
All data generated or analyzed during current study included in this manuscript.
References
Kirya, P. et al. Biomimicry of Blue Morpho butterfly wings: An introduction to nanotechnology through an interdisciplinary science education module. J. Soc. Inform. Disp. 29, 896–915 (2021).
Satarzadeh, N. et al. Facile microwave-assisted biosynthesis of arsenic nanoparticles and evaluation their antioxidant properties and cytotoxic effects: a preliminary in vitro study. J. Clust. Sci. 34(4), 1831–1839 (2022).
Satarzadeh, N. et al. An insight into biofabrication of selenium nanostructures and their biomedical application. 3 Biotech 13, 79 (2023).
Abdullah, N. H. Optimization of magnetic nano-iron production by Aspergillus flavipes MN956655. 1 using response surface methodology and evaluation of their dye decolorizing and antifungal activities. Sci. Rep. 12, 21059 (2022).
Khan, I., Saeed, K. & Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 12, 908–931 (2019).
Nguyen, M. D., Tran, H.-V., Xu, S. & Lee, T. R. J. A. S. Fe3O4 nanoparticles: Structures, synthesis, magnetic properties, surface functionalization, and emerging applications. Appl. Sci. 11, 11301 (2021).
Singamaneni, S., Bliznyuk, V. N., Binek, C. & Tsymbal, E. Y. Magnetic nanoparticles: recent advances in synthesis, self-assembly and applications. J. Mater. Chem. 21, 16819–16845 (2011).
Lisjak, D. & Mertelj, A. Anisotropic magnetic nanoparticles: A review of their properties, syntheses and potential applications. Progr. Mater. Sci. 95, 286–328 (2018).
Koh, I. & Josephson, L. Magnetic nanoparticle sensors. Sensors 9, 8130–8145 (2009).
Chen, Y.-T., Kolhatkar, A. G., Zenasni, O., Xu, S. & Lee, T. R. J. S. Biosensing using magnetic particle detection techniques. Sensors 17, 2300 (2017).
Bilal, M., Zhao, Y., Rasheed, T. & Iqbal, H. M. N. Magnetic nanoparticles as versatile carriers for enzymes immobilization: A review. Int. J. Biol. Macromol. 120, 2530–2544 (2018).
Wu, K. et al. Magnetic-nanosensor-based virus and pathogen detection strategies before and during COVID-19. ACS Appl. Nano Mater. 3, 9560–9580 (2020).
Laurent, S. et al. Magnetic iron oxide nanoparticles: Synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chem. Rev. 108, 2064–2110 (2008).
Ling, D. & Hyeon, T. J. S. Chemical design of biocompatible iron oxide nanoparticles for medical applications. Small 9, 1450–1466 (2013).
Bobo, D., Robinson, K. J., Islam, J., Thurecht, K. J. & Corrie, S. R. Nanoparticle-based medicines: a review of FDA-approved materials and clinical trials to date. Pharmaceut. Res. 33, 2373–2387 (2016).
Teja, A. S. & Koh, P.-Y. Synthesis, properties, and applications of magnetic iron oxide nanoparticles. Prog. Cryst. Growth Charact. Mater. 55, 22–45 (2009).
Kuppusamy, P., Yusoff, M. M., Maniam, G. P. & Govindan, N. Biosynthesis of metallic nanoparticles using plant derivatives and their new avenues in pharmacological applications–an updated report. Saudi Pharmaceut. J. 24, 473–484 (2016).
Bhardwaj, B., Singh, P., Kumar, A., Kumar, S. & Budhwar, V. J. A. P. B. Eco-friendly greener synthesis of nanoparticles. Adv. Pharmaceut. Bull. 10, 566 (2020).
Ahmed, S. F. et al. Green approaches in synthesising nanomaterials for environmental nanobioremediation: Technological advancements, applications, benefits and challenges. Environ. Res. 204, 111967 (2022).
Singh, P., Kim, Y.-J., Zhang, D. & Yang, D.-C. Biological synthesis of nanoparticles from plants and microorganisms. Trends Biotechnol. 34, 588–599 (2016).
Rana, A., Yadav, K. & Jagadevan, S. A comprehensive review on green synthesis of nature-inspired metal nanoparticles: Mechanism, application and toxicity. J. Clean. Prod. 272, 122880 (2020).
Berny, C. et al. A method for producing highly pure magnetosomes in large quantity for medical applications using Magnetospirillum gryphiswaldense MSR-1 magnetotactic bacteria amplified in minimal growth media. Front. Bioeng. Biotechnol. 8, 16 (2020).
Moon, S., Saboe, A. & Smanski, M. J. Using design of experiments to guide genetic optimization of engineered metabolic pathways. J. Ind. Microbiol. Biotechnol. 51, 010 (2024).
Khaw, K.-Y. et al. Factorial design-assisted supercritical carbon-dioxide extraction of cytotoxic active principles from Carica papaya leaf juice. Sci. Rep. 9, 1716 (2019).
Weissman, S. A. & Anderson, N. G. Design of experiments (DoE) and process optimization. A review of recent publications. Org. Process Res. Dev. 19, 1605–1633 (2015).
Jankovic, A., Chaudhary, G. & Goia, F. Designing the design of experiments (DOE)–An investigation on the influence of different factorial designs on the characterization of complex systems. Energy Build. 250, 111298 (2021).
Satarzadeh, N., Shakibaie, M., Forootanfar, H. & Amirheidari, B. Purification, characterization, and assessment of anticancer activity of iron oxide nanoparticles biosynthesized by novel thermophilic Bacillus tequilensis ASFS1. J. Basic Microbiol. https://doi.org/10.1002/jobm.202400153 (2024).
Rosenfeldt, S. et al. Towards standardized purification of bacterial magnetic nanoparticles for future in vivo applications. Acta Biomater. 120, 293–303 (2021).
Alphandéry, E., Amor, M., Guyot, F. & Chebbi, I. The effect of iron-chelating agents on Magnetospirillum magneticum strain AMB-1: Stimulated growth and magnetosome production and improved magnetosome heating properties. Appl. Microbiol. Biotechnol. 96, 663–670. https://doi.org/10.1007/s00253-012-4199-5 (2012).
Yang, C.-D. et al. Synthesis of bacterial magnetic particles during cell cycle of Magnetospirillum magneticum AMB-1. Appl. Biochem. Biotechnol. 91, 155–160 (2001).
Heyen, U. & Schüler, D. Growth and magnetosome formation by microaerophilic Magnetospirillum strains in an oxygen-controlled fermentor. Appl. Microbiol. Biotechnol. 61, 536–544 (2003).
Moisescu, C., Bonneville, S., Staniland, S., Ardelean, I. & Benning, L. G. Iron uptake kinetics and magnetosome formation by Magnetospirillum gryphiswaldense as a function of pH, temperature and dissolved iron availability. Geomicrobiol. J. 28, 590–600 (2011).
Kabary, H., Eida, M. F., Attia, M., Awad, N. & Easa, S. M. Optimization of growth and Fe uptake by Pseudomonas aeruginosa Kb1 for biosynthesis of magnetic nanoparticles. Middle East J. 7, 1503–1513 (2018).
Yan, L. et al. Optimization of magnetosome production by Acidithiobacillus ferrooxidans using desirability function approach. Mater. Sci. Eng. C 59, 731–739 (2016).
Zhang, S., Yan, L., Li, H. & Liu, H. J. Optimal conditions for growth and magnetosome formation of Acidithiobacillus ferrooxidans. Afr. J. Microbiol. Res. 6, 6142–6151 (2012).
Yang, C.-D., Takeyama, H., Tanaka, T., Matsunaga, T. J. E. & Technology, M. Effects of growth medium composition, iron sources and atmospheric oxygen concentrations on production of luciferase-bacterial magnetic particle complex by a recombinant Magnetospirillum magneticum AMB-1. Enzyme Microb. Technol. 29, 13–19 (2001).
Basit, A., Wang, J., Guo, F., Niu, W. & Jiang, W. Improved methods for mass production of magnetosomes and applications: A review. Microb. Cell Fact. 19, 1–11 (2001).
Anderson, M. J. & Whitcomb, P. J. DOE simplified: practical tools for effective experimentation (CRC Press, 2017).
Acknowledgements
The present study was financially supported using grant NO: BIODC-34975-35555.2 and 99000469 of the Biotechnology Development Council of the Islamic Republic of Iran and Faculty of Pharmacy, Kerman University of Medical Sciences, Kerman, Iran, respectively.
Author information
Authors and Affiliations
Contributions
N.S. Methodology, Validation, Investigation, Resources, Writing—Original Draft, Writing—Review & Editing. B.A. Methodology, Data Curation, Resources, Writing—Review & Editing, Supervision, Project administration. M.Sh. Methodology, Validation, Formal analysis, Writing—Review & Editing, Supervision, Project administration. H.F. Methodology, Writing—Review & Editing, Supervision.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Satarzadeh, N., Amirheidari, B., Shakibaie, M. et al. Medium optimization to improve growth and iron uptake by Bacillus tequilensis ASFS1 using fractional factorial designs. Sci Rep 14, 20141 (2024). https://doi.org/10.1038/s41598-024-70896-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-70896-4
Keywords
This article is cited by
-
Comparative Analysis of Biogenic and Chemically Synthesized Iron Oxide Nanoparticles and Interaction with Human Intestinal Epithelial Cells
BioNanoScience (2025)
-
Biosynthesized Nanoparticles for the Dermaceutical Applications: A Review
BioNanoScience (2025)
-
Biosynthesized Metal Nanoparticles as a High-Potential Tool in Bone Regenerative Medicine: A Review
Biological Trace Element Research (2025)