Abstract
Chronic kidney disease (CKD) affects 850 million people worldwide and is associated with significant cardiovascular morbidity and mortality. Routine laboratory tests do not reflect early stages of microcirculatory changes and vascular rarefaction that characterise kidney fibrosis, the common endpoint of CKD. Imaging techniques that detect CKD in early stages could promote timely treatment with new drugs like SGLT2 inhibitors, thus, decreasing CKD progression and the cardiovascular disease burden. Ultrasound is the most used imaging modality in CKD, as it is non-invasive and radiation free. Initially, ultrasound imaging was applied to assess kidney macro-morphology and to rule out ureteral obstruction. The development of higher frequency probes allowed for more detailed imaging of kidney parenchyma, and advances in Doppler ultrasound provided insights into segmental arterial flow patterns including resistive indices as an indirect measure of microcirculatory impedance, elevated values of which correlated with progressive organ failure and fibrosis. Today, low-flow detection methods and matrix probes better resolve organ parenchyma and smaller vascular beds, and contrast-enhanced ultrasound allows perfusion measurement. Particularly, super-resolution ultrasound imaging, a technology currently being in clinical translation, can characterise the microcirculation morphologically and functionally in unrivalled detail. This is accompanied by rapid developments in radiomics and machine learning supporting ultrasound image acquisition and processing, as well as lesion detection and characterisation. This perspective article introduces emerging ultrasound methods for the diagnosis of CKD and discusses how the promising technical and analytical advancements can improve disease management after successful translation to clinical application.
Similar content being viewed by others
Introduction
Chronic kidney disease is common, has a high cardiovascular mortality, and non-invasive diagnostic options for early disease stages are urgently needed
Chronic kidney disease (CKD) is a silent epidemic affecting nearly 10% of the population worldwide (an estimated 850 million people). It is more prevalent in people with diabetes, hypertension, obesity and in the elderly, and its incidence is increasing accordingly. Higher CKD stage correlates with higher cardiovascular risk and mortality1. Kidney function is vital; its loss can partly be replaced with dialysis several times a week, or kidney transplantation and subsequent immunosuppression. However, kidney donations are lower than the demand, and waitlisted patients spend years on dialysis with a high associated mortality, often not living long enough to receive a kidney transplant.
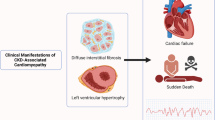
CKD is defined as either known kidney disease independent of kidney function (after biopsy, or inherited disease such as Alport syndrome) or impaired kidney function for more than three months independent of a known cause. CKD is graded according to the excretory function (measured by serum creatinine) and filter function (measured by albuminuria)2. Fibrosis, the replacement of functional tissue by scar tissue, associated with loss of small blood vessels (microvascular rarefaction)3, is the common pathological finding in later CKD stages, and is inversely correlated with residual kidney function (Fig. 1).
The correlation between creatinine and kidney function is not linear: a creatinine increase within the normal range can reflect a loss of >50% of kidney function. To adjust for differences in age and gender, an estimated glomerular filtration rate (eGFR) is calculated4 as proxy for excretory kidney function. While sensitive in low kidney function, serum creatinine is not sensitive enough to detect changes in higher or nearly normal kidney function, and definitive or early diagnosis still requires renal biopsy and histology.
An important prognostic parameter in CKD is the eGFR slope, the rate of loss of kidney function over time. Modern treatment options, such as SLGT2 inhibitors, GLP1 analogues, RAAS blockade and mineralocorticoid receptor antagonists, can slow the annual decline in kidney function and delay terminal disease requiring dialysis. Diagnosing CKD non-invasively in early stages would enable preservation of higher kidney function and thereby result in lower cardiovascular disease burden and mortality.
Acute kidney injury is a risk factor for CKD, but kidney function, measured by serum creatinine, can recover fully. This does not reflect lack of disease: In a mouse model of kidney injury using adenine diet, eGFR improved to normal after stop of adenine diet.However, the kidneys histologically showed marked fibrotic changes and no recovery5. Thus, early fibrotic changes can only be reliably assessed by organ biopsy and histology.
Recent advances in ultrasound technology, data processing and analysis seem to make it possible to predict organ microcirculatory function6,7,8,9,10,11,12. Hence, they could gain a high impact in the non-invasive diagnosis and better understanding of early-stage CKD.
Non-contrast-enhanced ultrasound techniques facilitate indirect and direct assessment of renal vasculature
Loss of microcirculation is a hallmark of kidney fibrosis; however, visualisation of small blood vessels in the kidney through ultrasound has been a challenge. Colour Doppler can visualise renal segmental and interlobar arteries13, but not smaller vessels. Pulsed-wave-Doppler can measure blood flow velocity changes in segmental and interlobar renal arteries. Peak systolic velocity and end-diastolic velocity are needed to calculate the resistive index (RI), which increases with age as well as in kidney disease14. The reason is that damage in small vessels (as in small vessel vasculitis) or microvascular rarefaction (as in fibrosis) decreases the cross-sectional area and vascular compliance. While values of 0.6–0.65 are considered normal in young adults, values over 0.75 are considered pathological and have been associated with faster CKD progression15 and worse allograft survival in kidney transplants16,17. However, RI is also dependent on systemic parameters such as pulse pressure; also, high arterial stiffness and organ oedema result in increased RI. Thus, although RI is a non-invasive and reproducible parameter, it is still only an indirect measure of microvascular impedance with numerous possible confounders.
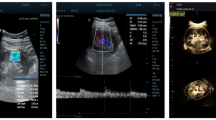
Recently, digital subtraction methods like B-flow imaging and low-flow Doppler (such as SMI (Superb Microvascular Imaging)18, microvascular imaging or “slow flow” (depending on the manufacturing company)) have shown higher sensitivity for direct detection of lower-flow and smaller vasculature in the kidney cortex (Fig. 2). For instance, in kidney tumours, SMI imaging performs almost as well as contrast-enhanced methods in identifying tumour microvasculature patterns to discern malignant lesions19. Furthermore, B-flow, a flow visualization method based on B-mode imaging, first patented by GE, is used in assessment of kidney transplants due to good resolution with short distance to the probe20,21. However, these methods can only resolve kidney structures above 300–500 µm in size, and their effectiveness depends on the used probe. Also, very low flow such as in glomeruli or capillaries cannot be resolved with these methods.
Another feature of kidney fibrosis is the deposition of extracellular matrix, resulting in increased organ stiffness. Shear Wave Elastography (SWE) or Acoustic Radiation Force Impulse (ARFI) can assess tissue elasticity, which may change with disease progression. While these methods are routinely used in liver ultrasound to grade liver fibrosis22, clinical utility of SWE/ARFI in kidney diseases has not yet been established. Albeit widely studied23, practical hurdles are the natural inhomogeneity of kidney tissue with cortex, medulla and papilla as well as large vessels and ureter structures in close proximity, allowing for only small ROIs; the organ-probe-distance is longer than in the liver context and dependent on patient BMI, and the pressure on the probe influences measurements in some elastography methods, resulting in a high inter-observer variability and low reproducibility of measurements. Possible applications include kidney tumour diagnostics. Particularly, angiomyolipomas, which are benign fat-containing tumours with softer than normal kidney tissue, thus showing differences in SWE24, can be identified. ARFI measurements also worked well in transplant kidneys as they are at shorter distance to the probe. In kidney transplant recipients, ARFI results correlated with the degree of fibrosis in transplant biopsies25. However, these protocols are difficult to integrate into standard care25 and the robust and reproducible measurement of mechanical tissue properties in the kidney requires further research.
In conclusion, non-contrast-enhanced ultrasound techniques facilitate the indirect and direct assessment of renal microvascular status, either by calculating RI or by imaging slow flow in small vessels. However, to improve our understanding of the mechanism of early CKD and to establish image-based biomarkers for non-invasive management of CKD progression, we need ultrasound imaging with higher sensitivity, resolution, and mechanistic expressiveness.
Contrast-enhanced ultrasound (CEUS) may let us see more
To increase sensitivity, ultrasound contrast agents have been developed and evolved from free gas bubbles to shell-coated microbubbles in which a polymer or phospholipid shell stabilises the gas core. When exposed to cycles of high and low pressure, microbubbles undergo asymmetrical compression and expansion, which allows microbubbles to be detected and differentiated from normal tissue, which express a more linear response in contrast to the characteristic non-linear response of microbubbles26. The size of microbubbles (1–10 µm) allows them to pass capillaries and makes them visible in the frequency range of clinical ultrasound devices (1–15 MHz)27.
Intravenously injected microbubbles can be detected in vessels using the Doppler effect. Here, they either act as scatterers (like blood cells but with stronger reflection) or their fragmentation by high-intensity ultrasound pulses is registered as characteristic Doppler shift. Both increase the sensitivity of ultrasound in detecting small vessels with low flow, such as capillaries. The administration of any exogenous agent, including microbubbles for contrast-enhanced ultrasound, carries the risk of allergic reactions. Moreover, in 2008, the U.S. Food and Drug Administration (FDA) issued a safety notification about ultrasound contrast agents due to concerns about their use in patients with severe cardiovascular conditions such as myocardial infarction or pulmonary hypertension, which arose from post-market surveillance data. The clinical community debated this warning extensively. There was no conclusive evidence of more than a temporal association28. Recently, a new study reported an increased incidence of severe adverse events associated with the use of sulphur hexafluoride microbubbles in echocardiography. This uptrend has been examined in the context of the concurrent rise of mRNA-based COVID19 vaccines - both share polyethylene glycol as a component, which is described for its allergenic potential29. However, more than two decades of clinical practice and numerous safety studies demonstrated the good tolerability of microbubbles. Serious adverse events were very rare and most reported adverse events were transient and mild to moderate in severity, presenting with symptoms resembling (pseudo)allergic or physiological reactions30,31,32.
In kidney CEUS, the arterial arrival of microbubbles and the cortical enhancement and subsequent filling of the medullary pyramids can be monitored. CEUS is superior to conventional Colour Doppler ultrasound in the differentiation of renal infarction33 and cortical necrosis, and it can characterize kidney lesions, which are indeterminate in CT and MR. The CEUS-Bosniak classification allows assessment of cystic masses34 and shows a high negative predictive value with close to 100% accuracy35. For diagnosis of renal cell carcinoma, CEUS shows sensitivity of 100% and specificity of 86–95%36.
In dynamic CEUS, time-intensity curves are recorded to quantify signal enhancement over time, enabling semi-quantitative assessment of organ perfusion. After continuous infusion or during the steady-state phase, the disruption-replenishment-analysis provides quantitative values. Here, a short destructive ultrasound pulse removes the signal enhancement in the image plane and microbubble replenishment from the surrounding tissue is recorded37,38. Using this quantitative analysis, operator dependency is reduced, and reproducibility improved. Additionally, the destruction replenishment evaluation does not have the challenge of adequately timed image acquisition (as in DCE MRI and CT), which can be difficult in CKD patients with hyper- or hypodynamic circulation. Dynamic CEUS was shown to be capable of depicting perfusion differences in acute kidney injury39, after renal transplantation40, and in fibrotic CKD kidneys by a delayed signal enhancement and reduced peak intensity41. However, correlation of eGFR to dynamic CEUS parameters did not give consistent results42,43, and further studies are needed.
The ups and downs of renal microvasculature imaging with super-resolution ultrasound
The resolution of conventional ultrasound imaging is not sufficient to spatially resolve the microvasculature. It is limited by diffraction and has a theoretical optimal resolution of half a wavelength which is in the range of ca. 50-250 µm for frequencies between 3 and 15 MHz. The ultrasound frequency related limits were challenged by super-resolution ultrasound methods based on ultrasound localization microscopy (ULM) that were proposed independently by Siepmann et al. 44. and Couture et al. 45. in 2011. ULM is a post-processing method that is based on localizing spatially isolated microbubbles. While a microbubble is typically only 1–2 µm large, an individual microbubble is shown in the ultrasound image as a bright spot with a much larger size determined by the system’s spatial resolution. However, if microbubbles are far enough from each other so that their spots in the image do not overlap, the position of the centre of the large spot image of an isolated microbubble can be determined with high precision. The microbubble positions can either be just accumulated or tracked from frame to frame, and the microbubble velocity can be measured with algorithms as used in RADAR processing10,44,46,7. The tracks are then shown in high resolution images and aggregated over time revealing the microvasculature (Fig. 3).
A B-mode images of a contrast bolus are acquired along with either contrast-enhanced imaging or as shown here with a slow-flow Doppler mode (SMI, Canon) to better detect microbubbles. B The image of tissue background is used for motion estimation and compensation. Here motion estimation was performed with the function imregdemons provided by Matlab (Mathworks, Natick, MA, USA). C Microbubbles are detected and localised in the contrast image. D Tracking of microbubbles and visualizing the tracks results in the images of super-resolved vessel. Here, an image of microbubble counts is shown. Images are from a study at RWTH Aachen University with ethical approval by the local review board.
ULM was applied preclinically44 and clinically47 to image the microvasculature in malignant tumours. Recent reviews10,7 gave an overview of further applications and the capability of ULM to show changes of the kidney microvasculature in CKD animal models (Fig. 4). ULM imaging of the kidney was first investigated by Foiret et al.48 in Wistar rats with a high-frame-rate research scanner (300 Hz). They demonstrated a resolution beyond the diffraction limit of 2.1 µm axially and 6.4 µm laterally, and vessels with flow velocities below 2 mm/s could be imaged. In this study, imaging was performed non-invasively and frames with out-of-plane motion were excluded while rigid in-plane motion was corrected. With a high-frame-rate research scanner (400 Hz) Qiu et al.49 showed a significant difference in the mean blood flow speed of interlobular arteries between hypertensive and healthy rats. The same group observed with ULM that the arterial and venous flow velocities in the renal cortex of a diabetic rat model went down compared to the control50. Søgaard et al.51 investigated changes in the density and tortuosity of kidney microvasculature in obese Zucker rats with a modified low-framerate (54 Hz) clinical scanner. They did not observe significant changes in the tortuosity, but the vessel density was reduced significantly. They pointed out that the measurement of the vessel density needs better normalization because of its dependence on acquisition time, flow rate and microbubble concentration. With the same setup Andersen et al.12 used ULM in kidney ischaemia/reperfusion experiments. Six blinded assessors could differentiate ULM images acquired at reperfusion after venal clamping from baseline images and 60 minutes after reperfusion no changes were seen. A further study of the same group demonstrated a decrease in the mean flow velocity under prazosin medication in the cortical arteries/arterioles and in the outer medulla descending vasa recta52. The use of a low-frame-rate clinical scanner in these studies led to long acquisition times of 10 minutes under constant microbubble infusion. Also, to reduce motion, the animal was opened, and the kidney was held in place with a metal retractor. Additionally, non-rigid in-plane motion correction was developed and applied53. It also has been observed that the detection of microbubbles in kidney images is strongly hampered by noise and different approaches for noise reduction were proposed54,55.
In humans, ULM of healthy kidneys was realized first with a high-frame-rate clinical scanner at 250 Hz framerate by Huang et al.56 and an acquisition time of only 10 s after bolus injection of Sonovue (Bracco). The study demonstrated the feasibility of ULM in patients for various organs and showed impressive detail of the kidney microvasculature56. However, such high framerates can currently only be achieved with few clinical scanners that are using plane-wave imaging instead of the conventional line-by-line imaging. Bodard et al.57 and Denis et al.58 investigated the microvasculature in human kidney allografts using a clinical scanner with a conventional low frame-rate between 14 Hz and 64 Hz. Both studies injected microbubbles intravenously and acquired data for 1–3 min. The authors did not implement motion compensation in their post-processing scheme. Therefore, in the studies by Denis et al.58 9 of 14 and by Bodard et al.57 7 of 14 patients had to be excluded from analysis due to breathing motion. This demonstrates the necessity to compensate for breathing motion or to reduce the acquisition times allowing breath holding. Bodard et al.57 stated that ULM identified vessels two to four times thinner than it is possible with Doppler modes. In the seven analysed patients, the mean diameter of smallest analysable vessel was reported to be 0.3 ± 0.2 mm and blood velocities were determined for the cortical region. Denis et al.58 additionally characterized the tracks of individual microbubbles in externalized rat kidneys and human kidney allografts. They calculated a measure for the straightness of a track being minimal for a straight linear path. Using this parameter, they identified regions with a local retention of microbubbles and hypothesized that these are locations of glomeruli. The statistical properties of their spatial distribution were analysed and compared with µCT, and results support this assumption.
While the preclinical and first clinical studies on ULM of the kidney vasculature are promising, several obstacles must be overcome for its clinical application. For instance, the degree of reconstruction of the microvessels depends on the observation time, the flow rates, and the contrast agent concentration8,9. In the literature, the measurement times vary from a few seconds to tenths of minutes7,10 resulting in a different amount of resolved microvessels9. Standardization of measurements is needed to derive reproducible quantitative parameters from ULM. Furthermore, for many preclinical studies the kidney was immobilized to avoid breathing motion and in clinical imaging often transplant kidneys are imaged, which also move less. To translate the results to clinical applications, it is necessary to tackle the artefacts caused by the movement that occurs due to breathing, heartbeat and transducer motion. Motion within the image plane can be estimated and corrected, and deep learning (DL) could help to better compensate for complex motion patterns. However, out-of-plane motion cannot be corrected in 2D imaging and thus frames with strong out-of-plane motion often are excluded.
Additionally, in 2D imaging in the elevational direction, perpendicular to the imaging plane, no super-resolution is achieved. This will project vessels within the image slice and makes the imaging of the dense parallel vessels in the kidney medulla and cortex difficult48. Moreover, it was shown by a comparison of ULM images of rat kidneys with µCT that several parallel vasa recta bundles could contribute to a single pixel in ULM and thus cannot be resolved60. In the future, this could be overcome by transducers also electronically focused in the elevational direction like 2D transducer arrays for 3D imaging or full matrix arrays for volume imaging.
The larger inter-frame time makes tracking of microbubbles more difficult and tracking methods based on motion models as proposed by Ackermann and Schmitz46 and Tang et al.61 must be applied. However, in the case of ultrasound kidney imaging, the large variation of velocities makes the application of motion models more difficult. It could be handled by a hierarchical Kalman tracker as proposed by Taghavi et al.59, which separates the tracking for different velocity ranges. In the future this may be solved by the availability of high frame-rate clinical scanners as well as advanced tracking using DL-based methods.
In summary, super-resolution ultrasound imaging of the kidney is evolving and holds a diagnostic potential to study microvascular changes in early stages of fibrosis non-invasively, improving our understanding of disease mechanism and allowing for early diagnosis and timely initiation of progression-delaying therapy. Also, by using the characteristic flow patterns of microbubbles passing through glomeruli, ULM could potentially identify and count nephrons non-invasively (related publications are summarized in Table 1), allowing for non-invasive estimation of perfused glomeruli in a defined cortex volume and extrapolation of kidney function. The perspective of introducing ULM in real-life clinical condition lays in a combination of technological advances of clinical ultrasound systems and artificial intelligence techniques. DL will be particularly useful for advanced motion compensation, tracking of the microbubbles and the extraction of diagnostic parameters.
Artificial intelligence can enrich the conventional and advanced ultrasound imaging of renal microvasculature
Recent developments in AI have significantly expanded the scope and scale of medical image analysis62,63. This offers the prospect of a solid and objective basis for standardizing kidney ultrasound imaging and its interpretation, with a direct impact on improving the management of CKD. Certainly, the risk of developing CKD or monitoring its progression can be assessed by building classification models based on clinical values alone64. However, using these features alone in a predictive model may lead to inconclusive results. This is because the outcomes of common tests, i.e. blood serum creatinine or urine protein, do not directly correlate with kidney function.
AI-based analysis of kidney ultrasound images can facilitate the discovery of non-invasive image-based biomarkers for the assessment of kidney function (Fig. 5). Kidney ultrasound imaging is a preferred method for predicting and monitoring CKD due to its safety, convenience and affordability65. However, this non-invasive assessment of kidney function is not without its pitfalls. Ultrasound imaging is highly operator-dependent, making conventionally derived parameters such as kidney length, parenchymal thickness or echogenicity subjective to the reader6. Therefore, it would be optimal to use other strategies to interpret kidney ultrasound images, and radiomics analysis is one of them.
Ultrasound data acquired by established (B-mode, colour Doppler, SMI) and emerging (ULM) ultrasound methods can be used for building AI-based decision models. First a region of interest (ROI) is selected. This is usually done by segmenting the image either automatically with e.g., deep learning or manually by delineating the ROI. Then, features are extracted from the selected ROI. Here, the features can be calculated automatically (deep learning) or by using predefined radiomics features (histogram, texture and wavelet). In the final step, the most descriptive features are selected and used to build a decision model that facilitates the diagnosis of a disease from the image.
Radiomics is a method that supports the conversion of image data into quantifiable features66. To calculate radiomics features, the region of interest must first be selected, and in the case of CKD monitoring, this is kidney parenchyma. Features are then obtained using first order (histogram) and second order (texture) statistics, but also wavelet transformation to obtain the relationships between pixels hidden to the human eye. For example, wavelet features are sensitive to the directionality of speckle pattern, though they can be useful in classifying fibrosis in CKD, as its presence in the kidney parenchyma can alter the speckle pattern in the b-mode ultrasound image67. Additionally, descriptive image-based features can be derived from filtered or fractal ultrasound images to achieve location-sensitive classification of CKD68,69.
Radiomics is certainly a powerful tool which could facilitate the ultrasound-based analysis of kidney function for CKD prediction and management. For instance, a preliminary clinical study demonstrated that ultrasound-based radiomics features allowed biopsy-free differentiation between membranous and IgA nephropathy70. Another study showed that combining radiomics with common clinical CKD biomarkers improves the predictive performance of a CKD risk assessment classification model71. Nevertheless, the limitation of radiomics lays in the predefined nature of the extracted features.
DL alleviates the bottlenecks of conventional radiomics analysis. It can be employed to guide medical image processing from segmentation to disease prediction without the need for user interaction. Recent studies showed that DL-based automatic segmentation of kidney in B-mode ultrasound images can be performed with high accuracy (94% similarity to radiologist’s delineations)72. In addition, DL-based assessment of kidney function can discriminate between healthy and CKD patients, as well as between stages of CKD itself, and it can also be used to estimate AI-based glomerular filtration rate from B-mode images62,73. However, DL-based image analysis is a data-driven approach and collecting and storing medical data is still challenging. Therefore, the prospect of standardizing DL for CKD management lies in using data augmentation, transfer learning, or federated learning62,64. Additionally, published studies relay mostly on static B-mode images, and using Doppler or contrast-enhanced data would already expand the scope of DL-based analysis62,72,73. Furthermore, combination of DL and super-resolution ultrasound imaging holds great promise for non-invasive visualization and assessment of micro-pathological changes in kidney tissue. Union of these two methods could result in providing information on glomerular number, perfusion and structural integrity of kidneys.
Conclusion
Ultrasound is already the most widely used non-invasive imaging modality for the assessment of CKD. It provides a quick and inexpensive assessment of macro-morphology and some relevant functional estimates from which the eGFR can be roughly estimated. However, as ultrasound is more user-dependent compared to other imaging modalities such as CT and MRI, and thus has low reproducibility, there is a strong demand for computer-based solutions that help the physician to re-identify a tissue slice and perform precise lesion detection and size measurements for clinical routine more reliably. These tools can be supported by AI-based algorithms and new hardware such as 3D matrix probes and help to compensate for tissue motion, which is currently the biggest challenge in translating new high-resolution ultrasound imaging techniques of the kidney into the clinic. The motion caused by the doctor’s hand and patient’s breathing, heartbeat and bowel movements, is quite complex, as it occurs in multiple directions and sometimes out of plane. If this is properly addressed, even glomeruli can become visible, as shown in a study using contrast-enhanced ultrasound localisation microscopy58.
From a clinical point of view, however, robustness, routine suitability and costs are essential for the acceptance of a method by physicians. Although ultrasound contrast agents are well tolerated by patients, their administration complicates examinations and increases costs. Whether super-resolution ultrasound imaging will be able to work without microbubbles in the future59, and whether it will add value to a comprehensive AI-based analysis of morphological and Doppler imaging, remains to be investigated. Nevertheless, it is likely that ultrasound will remain the method of choice for routine imaging in CKD and has enormous potential for further development.
References
Go, A. S., Chertow, G. M., Fan, D., McCulloch, C. E. & Hsu, C. Y. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N. Engl. J. Med. 351, 1296–1305 (2004).
National Kidney, F. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am. J. Kidney Dis. 39, S1–266 (2002).
Kramann, R., Tanaka, M. & Humphreys, B. D. Fluorescence microangiography for quantitative assessment of peritubular capillary changes after AKI in mice. J. Am. Soc. Nephrol. 25, 1924–1931 (2014).
Miller, W. G. et al. National Kidney Foundation Laboratory Engagement Working Group recommendations for implementing the CKD-EPI 2021 race-free equations for estimated glomerular filtration rate: Practical guidance for clinical laboratories. Clin. Chem. 68, 511–520 (2022).
Klinkhammer, B. M. et al. Current kidney function parameters overestimate kidney tissue repair in reversible experimental kidney disease. Kidney Int. 102, 307–320 (2022).
Kuo, C.-C. et al. Automation of the kidney function prediction and classification through ultrasound-based kidney imaging using deep learning. NPJ Digit. Med. 2, 29 (2019).
Christensen-Jeffries, K. et al. Super-resolution ultrasound imaging. Ultrasound Med. Biol. 46, 865–891 (2020).
Dencks, S. et al. Clinical pilot application of super-resolution US imaging in breast cancer. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 66, 517–526 (2019).
Dencks, S., Piepenbrock, M. & Schmitz, G. Assessing vessel reconstruction in ultrasound localization microscopy by maximum likelihood estimation of a zero-inflated poisson model. IEEE Trans. Ultrason., Ferroelectr., Frequency Control 67, 1603–1612 (2020).
Dencks, S. & Schmitz, G. Ultrasound localization microscopy. Zeitschrift für Medizinische Physik, https://doi.org/10.1016/j.zemedi.2023.02.004 (2023).
Porte, C. & Kiessling, F. [Super-resolution ultrasound imaging : Methods and applications]. Radiologie (Heidelb.) 62, 467–474 (2022).
Andersen, S. et al. Super-resolution imaging with ultrasound for visualization of the renal microvasculature in rats before and after renal ischemia: A pilot study. Diagnostics 10, 1–15 (2020).
Stock, K. F. [Ultrasound diagnostics of renal blood vessels and transplant kidney]. Radiologe 49, 1040–1047 (2009).
Petrucci, I., Clementi, A., Sessa, C., Torrisi, I. & Meola, M. Ultrasound and color Doppler applications in chronic kidney disease. J. Nephrol. 31, 863–879 (2018).
Parolini, C. et al. Renal resistive index and long-term outcome in chronic nephropathies. Radiology 252, 888–896 (2009).
Radermacher, J. & Haller, H. The role of the intrarenal resistive index in kidney transplantation. N. Engl. J. Med. 369, 1853–1855 (2013).
Radermacher, J. et al. The renal arterial resistance index and renal allograft survival. N. Engl. J. Med. 349, 115–124 (2003).
Gao, J., Thai, A. & Erpelding, T. Comparison of superb microvascular imaging to conventional color Doppler ultrasonography in depicting renal cortical microvasculature. Clin. Imaging 58, 90–95 (2019).
Mao, Y., Mu, J., Zhao, J., Yang, F. & Zhao, L. The comparative study of color doppler flow imaging, superb microvascular imaging, contrast-enhanced ultrasound micro flow imaging in blood flow analysis of solid renal mass. Cancer Imaging 22, 1–8 (2022).
Morgan, T. A., Jha, P., Poder, L. & Weinstein, S. Advanced ultrasound applications in the assessment of renal transplants: contrast-enhanced ultrasound, elastography, and B-flow. Abdom. Radiol. 43, 2604–2614 (2018).
Russo, E., Cerbone, V., Sciano, D., Valeriani, G. & De Rosa, P. In Transplantation proceedings. 1127–1129 (Elsevier).
Chen, S. et al. Assessment of liver viscoelasticity by using shear waves induced by ultrasound radiation force. Radiology 266, 964–970 (2013).
Zaffanello, M., Piacentini, G., Bruno, C., Brugnara, M. & Fanos, V. Renal elasticity quantification by acoustic radiation force impulse applied to the evaluation of kidney diseases: a review. J. Investigative Med. 63, 605–612 (2015).
Stock, K., Kübler, H., Maurer, T., Slotta-Huspenina, J. & Holzapfel, K. CEUS—diagnosis of solid renal tumors. Der Radiol. 58, 553–562 (2018).
Stock, K. et al. ARFI-based tissue elasticity quantification in comparison to histology for the diagnosis of renal transplant fibrosis. Clin. Hemorheol. Microcirculation 46, 139–148 (2010).
Köse, G., Darguzyte, M. & Kiessling, F. Molecular ultrasound imaging. Nanomaterials 10, 1935 (2020).
Frinking, P., Segers, T., Luan, Y. & Tranquart, F. Three decades of ultrasound contrast agents: A review of the past, present and future improvements. Ultrasound Med Biol. 46, 892–908 (2020).
Main, M. L. Ultrasound Contrast Agent Safety: From Anecdote to Evidence**Editorials published in JACC: Cardiovascular Imaging reflect the views of the authors and do not necessarily represent the views of JACC: Cardiovascular Imaging or the American College of Cardiology. JACC: Cardiovascular Imaging 2, 1057–1059 (2009). .
Ali, M. T. et al. Incidence of severe adverse drug reactions to ultrasound enhancement agents in a contemporary echocardiography practice. J. Am. Soc. Echocardiogr. 37, 276–284.e273 (2024).
Ignee, A., Atkinson, N. S., Schuessler, G. & Dietrich, C. F. Ultrasound contrast agents. Endoscopic ultrasound 5, 355–362 (2016).
Shang, Y. et al. Safety findings after intravenous administration of sulfur hexafluoride microbubbles to 463,434 examinations at 24 centers. Eur. Radiol. 33, 988–995 (2023).
Li, Q. et al. Safety analysis of adverse events of ultrasound contrast agent Lumason/SonoVue in 49,100 patients. Ultrasound Med. Biol. 49, 454–459 (2023).
Bertolotto, M. et al. Value of contrast-enhanced ultrasonography for detecting renal infarcts proven by contrast enhanced CT. A feasibility study. Eur. Radiol. 18, 376–383 (2008).
Herms, E. et al. Ultrasound-based “CEUS-Bosniak” classification for cystic renal lesions: an 8-year clinical experience. World J. Urol. 41, 679–685 (2023).
Alrashed, A. et al. Negative predictive value of contrast‐enhanced ultrasound in differentiating avascular solid‐appearing from vascularized masses: A retrospective consecutive study. J. Ultrasound Med. 37, 2935–2942 (2018).
Barr, R. G., Peterson, C. & Hindi, A. Evaluation of indeterminate renal masses with contrast-enhanced US: a diagnostic performance study. Radiology 271, 133–142 (2014).
Dietrich, C. et al. An EFSUMB introduction into dynamic contrast-enhanced ultrasound (DCE-US) for quantification of tumour perfusion. Ultraschall in der Medizin-Eur. J. Ultrasound 33, 344-351 (2012).
Selby, N. M., Williams, J. P. & Phillips, B. E. Application of dynamic contrast enhanced ultrasound in the assessment of kidney diseases. Curr. Opin. Nephrol. Hypertension 30, 138–143 (2021).
Harrois, A., Grillot, N., Figueiredo, S. & Duranteau, J. Acute kidney injury is associated with a decrease in cortical renal perfusion during septic shock. Crit. Care 22, 1–9 (2018).
Kazmierski, B. J., Sharbidre, K. G., Robbin, M. L. & Grant, E. G. Contrast‐enhanced ultrasound for the evaluation of renal transplants. J. Ultrasound Med. 39, 2457–2468 (2020).
Garessus, J. et al. Cortical perfusion as assessed with contrast-enhanced ultrasound is lower in patients with chronic kidney disease than in healthy subjects but increases under low salt conditions. Nephrol. Dialysis Transpl. 37, 705–712 (2022).
Jeong, S., Park, S. B., Kim, S.-H., Hwang, J. H. & Shin, J. Clinical significance of contrast-enhanced ultrasound in chronic kidney disease: a pilot study. J. ultrasound 22, 453–460 (2019).
Srivastava, A. et al. Association of contrast-enhanced ultrasound–derived kidney cortical microvascular perfusion with kidney function. Kidney360 3, 647 (2022).
Siepmann, M., Bzyl, J., Palmowski, M., Kiessling, F. & Schmitz, G. In 2011 IEEE International Ultrasonics Symposium. 1906-1909.
Couture, O., Besson, B., Montaldo, G., Fink, M. & Tanter, M. In 2011 IEEE International Ultrasonics Symposium. 1285-1287.
Ackermann, D. & Schmitz, G. Detection and tracking of multiple microbubbles in ultrasound B-mode images. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 63, 72–82 (2016).
Opacic, T. et al. Motion model ultrasound localization microscopy for preclinical and clinical multiparametric tumor characterization. Nat. Commun. 9, 1527 (2018).
Foiret, J. et al. Ultrasound localization microscopy to image and assess microvasculature in a rat kidney. Sci. Rep. 7, 13662 (2017).
Qiu, L. et al. In vivo assessment of hypertensive nephrosclerosis using ultrasound localization microscopy. Med. Phys. 49, 2295–2308 (2022).
Zhang, H. et al. Evaluation of early diabetic kidney disease using ultrasound localization microscopy: A feasibility study. J. Ultrasound Med. 42, 2277–2292 (2023).
Sogaard, S. B. et al. Super-resolution ultrasound imaging provides quantification of the renal cortical and medullary vasculature in obese zucker rats: A pilot study. Diagnostics (Basel) 12, https://doi.org/10.3390/diagnostics12071626 (2022).
Andersen, S. B. et al. “Super-Resolution Ultrasound Imaging Can Quantify Alterations in Microbubble Velocities in the Renal Vasculature of Rats”, Diagnostics, Vol. 12, Issue 5, Art. No. 5, May 2022, doi: 10.3390/diagnostics12051111.
Taghavi, I. et al. In vivo motion correction in super-resolution imaging of rat kidneys. IEEE Trans. Ultrason., Ferroelectr. Freq. Control 68, 3082–3093 (2021).
Lei, S. et al. In vivo ultrasound localization microscopy imaging of the kidney’s microvasculature with block-matching 3-D denoising. IEEE Trans. Ultrason., Ferroelectr., Frequency Control 69, 523–533 (2022).
Song, P. et al. Improved super-resolution ultrasound microvessel imaging with spatiotemporal nonlocal means filtering and bipartite graph-based microbubble tracking. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 65, 149–167 (2018).
Huang, C. et al. Super-resolution ultrasound localization microscopy based on a high frame-rate clinical ultrasound scanner: an in-human feasibility study. Phys. Med. Biol. 66, 08NT01 (2021).
Bodard, S. et al. Ultrasound localization microscopy of the human kidney allograft on a clinical ultrasound scanner. Kidney Int. 103, 930–935 (2023).
Denis, L. et al. Sensing ultrasound localization microscopy for the visualization of glomeruli in living rats and humans. eBioMedicine 91, https://doi.org/10.1016/j.ebiom.2023.104578 (2023).
Taghavi, I. et al. Ultrasound super-resolution imaging with a hierarchical Kalman tracker. Ultrasonics 122, 106695 (2022).
Andersen, S. B. et al. Evaluation of 2D super-resolution ultrasound imaging of the rat renal vasculature using ex vivo micro-computed tomography. Sci. Rep. 11, 24335 (2021).
Tang, S. et al. Kalman filter-based microbubble tracking for robust super-resolution ultrasound microvessel imaging. IEEE Trans. Ultrason., Ferroelectr., Frequency Control 67, 1738–1751 (2020).
Alnazer, I. et al. Recent advances in medical image processing for the evaluation of chronic kidney disease. Med Image Anal 69, 101960 (2021).
Zhao, D. et al. Current progress in artificial intelligence-assisted medical image analysis for chronic kidney disease: A literature review. Comput Struct Biotechnol J (2023).
Tangri, N. & Ferguson, T. W. Artificial Intelligence in the Identification, Management, and follow-up of CKD. Kidney360 3, 554-556 (2022).
Singla, R. K., Kadatz, M., Rohling, R. & Nguan, C. Kidney ultrasound for nephrologists: A review. Kidney Med. 4, 100464 (2022).
Gillies, R. J., Kinahan, P. E. & Hricak, H. Radiomics: Images are more than pictures, they are data. Radiology 278, 563–577 (2016).
Kim, D. H. & Ye, S. Y. Classification of chronic kidney disease in sonography using the GLCM and artificial neural network. Diagnostics (Basel) 11, https://doi.org/10.3390/diagnostics11050864 (2021).
Iqbal, F., Pallewatte, A. S. & Wansapura, J. P. In 2017 Seventeenth International Conference on Advances in ICT for Emerging Regions (ICTer). 1-5 (IEEE).
Acharya, U. R. et al. Automated detection of chronic kidney disease using higher-order features and elongated quinary patterns from B-mode ultrasound images. 32, 11163-11172 (2020).
Lee, S. H., Cho, H. H., Kwon, J., Lee, H. Y. & Park, H. Are radiomics features universally applicable to different organs? Cancer Imaging 21, 31 (2021).
Bandara, M. S. et al. Ultrasound based radiomics features of chronic kidney disease. Acad. Radiol. 29, 229–235 (2022).
Yin, S. et al. In 2019 IEEE 16th International Symposium on Biomedical Imaging (ISBI 2019). 1741-1744 (IEEE).
Kuo, C.-C. et al. Automation of the kidney function prediction and classification through ultrasound-based kidney imaging using deep learning. NPJ Digit Med 2, 29 (2019).
Acknowledgements
This study was funded by the German Research Foundation (DFG) [CRU 5011/445703531] and [233312120]. The funder played no role in study design, data collection, analysis and interpretation of data, or the writing of this manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
SF and ZAM contributed equally. Initial development of ideas: SF, ZAM; GS, FK. Drafting of the manuscript: all authors. Image acquisition: SF, SD, ZAM. Super resolution post-processing: SJ, GS; Interpretation of the content: all authors. Critical revisions of the manuscript: all authors. Final approval of the completed version: all authors. Overseeing the project: FK.
Corresponding author
Ethics declarations
Competing interests
Author SF has previously acted as a paid consultant for or received lecture fees from Stadapharm, Medice and AstraZeneca (not ultrasound related.) She is a member of the German Society for Ultrasound in Medicine (DEGUM) and a qualified ultrasound instructor “DEGUM II Internal Medicine”. FK and GS collaborate with Fujifilm Visualsonics on super-resolution ultrasound and FK additionally is an advisor of the company. FK is co-owner of the SonoMAC GmbH.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fleig, S., Magnuska, Z.A., Koczera, P. et al. Advanced ultrasound methods to improve chronic kidney disease diagnosis. npj Imaging 2, 22 (2024). https://doi.org/10.1038/s44303-024-00023-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44303-024-00023-5