Abstract
Objectives
The global increase in obesity predisposes individuals to prediabetes and type 2 diabetes, but data on their prevalence, temporal trends, and associated factors in children remain limited. We examined these issues in well-defined patient and population cohorts.
Methods
Data were collected from 602 patients aged 6–16 years, who were examined in healthcare for overweight/obesity once in 2002–2020. Controls comprised 483 population-representative children aged 7–16, who participated in 1–3 prospective visits. Prediabetes signified fasting glucose 5.6–6.9 mmol/L or 2h-post-challenge glucose 7.8–11.0 mmol/L, and diabetes as values ≥7.0 mmol/L or ≥11.1 mmol/L, respectively. Factors associated with prediabetes in patients were studied using logistic regression. The prevalence of prediabetes was compared between patients having their first healthcare visit in different years between 2002 and 2019.
Results
Altogether, 89.2% of patients and 3.3–4.7% of controls had obesity. The prevalence of prediabetes was 34.2% and of type 2 diabetes 1.3% among patients, and 6.9% and 0% in controls respectively, with prediabetes increasing significantly with age and stage of puberty. Both conditions were associated with presence of metabolic dysfunction-associated steatotic liver disease (OR 1.69, 95% CI 1.01–2.80) and acanthosis nigricans (1.83, 1.05–3.21), after adjusting for age. Prevalence of prediabetes increased steeply over time from 11.4% in patients examined in 2002–2004 to 50.0% in patients examined in 2017–2019 (OR 1.16, CI 1.10–1.21 p < 0.001) without concurrent changes in the degree of obesity, body mass index, other metabolic conditions, age, sex, or gestational/neonatal factors, except for an increase in maternal prepregnancy/pregnancy overweight (20.0–68.8%, OR 1.14, CI 1.08–1.21, p < 0.001).
Conclusions
Prediabetes was decidedly prevalent in pediatric patients with obesity and was associated particularly with steatotic liver disease. Its prevalence increased steeply over time, independent of the degree of obesity.
Similar content being viewed by others
Introduction
The prevalence of obesity has increased steeply across all geographical regions [1]. Obesity is associated with metabolic disorders, including type 2 diabetes (T2D), which nowadays imposes a considerable burden on society [2]. T2D is typically preceded by moderately impaired glucose metabolism, known as prediabetes. Besides predisposing individuals to T2D, prediabetes may also be associated with adverse cardiometabolic outcomes [3]. Although previously thought to mainly affect adults, these conditions also appear to be increasingly common among children [4, 5]. The rapid progression and poor prognosis of early-onset diabetes underscore the critical need for early intervention [6].
Although some research on this issue exists, particularly data from Nordic countries and long-term trends remain limited. In the few existing reports focusing on children with overweight, the prevalence has varied widely, showing an increasing trend [7,8,9]. The condition also appears to be more common in males, certain ethnic groups, and individuals with metabolic dysfunction-associated steatotic liver disease (MASLD) and other metabolic risk factors [10,11,12,13,14,15]. Interpreting these results is challenging due to the use of diverse cohorts, varying prevalences of obesity, inconsistent criteria, and the lack of control groups [5, 16, 17]. The relationship between obesity and prediabetes remains partly unclear, as it may also be influenced by factors like fat tissue distribution, muscle mass, physical inactivity and maternal disorders during pregnancy [18, 19].
The Finnish population is homogeneous, and systematic healthcare data have been available for an extended period. Leveraging these advantages, we investigated the prevalence, temporal trends, and associated factors of prediabetes and T2D in children examined due to overweight and obesity. A longitudinal, population-representative cohort was available for comparisons.
Methods
Patients and study design
The study was conducted at Tampere University and at the University of Eastern Finland. The patients included 1000 children aged 6–16 years who received an overweight- or obesity-related ICD-10 code (E65, E66 or R63.5) in 2002–2020. They were identified from the primary and tertiary care units in the Tampere area. Medical data were collected from patient records. In total, 398 patients were excluded due to incomplete data, incorrect diagnostic codes, unavailable glucose values, or comorbidities or medications that might affect glucose metabolism. After exclusions, 602 children were included (Supplementary Fig. S1). Patient data was collected from the first obesity-related healthcare visit.
For the population cohort, 736 children aged 7–9 who started first grade in primary schools were invited to participate in the prospective Physical Activity and Nutrition in Children (PANIC) study. They did not differ in age, sex, or body mass index (BMI) Z-scores from other children in the same area [20]. Altogether, 512 attended the baseline examinations in 2007–2009, but eight were excluded due to inability to participate or lack of motivation. Comprehensive health data, along with samples, were collected at each visit [20]. The study included baseline investigations at ages 7–9, a 2-year follow-up at ages 9–11, and an 8-year follow-up at ages 14–17. Children with incomplete data and those with any diseases or treatments potentially affecting glucose metabolism, including epilepsy, hypothyroidism, type 1 diabetes, Klinefelter syndrome, ulcerative colitis, juvenile rheumatoid arthritis, multiple sclerosis, or medication for depression or neuropathic pain, were excluded. This left 483 children at baseline, 420 at the 2-year visit, and 258 at the 8-year visit. Two patients reported occasional alcohol consumption, and one control subject previous amphetamine use. This information was deemed nonsignificant.
The collection of patient data was approved by Tampere University Hospital, Tampere Healthcare Services and the Social and Health Data Permit Authority Findata, while the recruitment of children for the PANIC was approved by the Research Ethics Committee of the Hospital District of Northern Savo. Informed consent was obtained from all children prospectively enrolled, as well as from their legal guardians. The study was conducted in accordance with the Declaration of Helsinki.
Data collection
The following data recorded during the study visit of PANIC or at the first obesity-related healthcare visit (patients) were collected: demographic and anthropometric data, stage of puberty, presence of acanthosis nigricans and chronic diseases and medications, blood pressure, use of alcohol or illicit drugs, presence of overweight, obesity and T2D in first-degree relatives, and laboratory parameters.
Growth data were converted into BMI Z-scores and weight-for-height percentages (WH%). Cut-off values for underweight, normal weight, overweight, obesity class I, and obesity class II were defined as BMI Z-scores using national cutoffs, corresponding to a BMI of <18.5, 18.5–24.9, ≥25.0, ≥30.0 and ≥35.0 kg/m2 at age 18 years, according to method recommended by the International Obesity Task Force (IOTF) [21, 22]. For 52 children without data of height or weight but WH% available, equivalent WH% values were used as follows: 10–20% for overweight, 20–40% for obesity class I, and >40% for obesity class II in children younger than 7 years of age; and 20–40%, 40–60%, and >60% for older children [23, 24]. These cutoffs are widely used in clinical practice in Finland [24] and, based on national data sources not publicly available, correspond to levels of body fat determined by DEXA scan, similarly to BMI Z-score thresholds. Pubertal stage was classified as prepubertal (Tanner M/G 1), pubertal (M/G 2–4), and postpubertal (M/G 5), while hypertension was defined as blood pressure exceeding the 95th percentile for age, sex, and height [25].
The laboratory parameters included alanine aminotransferase (ALT), fasting glucose and insulin, total cholesterol, high-density lipoprotein (HDL) cholesterol, low-density lipoprotein (LDL) cholesterol, triglycerides, and thyroid stimulating hormone (TSH). MASLD was defined as ALT >2× the upper limit of normal in patients with overweight or obesity [13, 26]. Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) was calculated as fasting insulin mIU/mL × glucose mmol/L/22.5 [27]
Perinatal data on the patients were retrieved to investigate potential temporal changes in these measures that might help explain the possible increase in prediabetes prevalence. Maternal data included presence of gestational hypertension, diabetes, pre-eclampsia, and abnormal weight gain as diagnosed by clinicians, as well as smoking and prepregnancy or gestational overweight or obesity [28]. Offspring data included gestational age and presence of macrosomia (birth weight >4.5 kg), large for gestational age (LGA), and small for gestational age (SGA). Data for analyzing temporal changes in gestational and neonatal risk factors between 2002 and 2019 were available for 344–380 patients, except for gestational diabetes and weight gain, for which data were available for 119 and 191 patients, respectively.
Prediabetes and type 2 diabetes
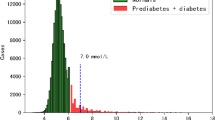
Prediabetes was defined as fasting glucose levels 5.6–6.9 mmol/L, also called impaired fasting glucose (IFG), or 2-h levels 7.8–11.0 mmol/L in the oral glucose tolerance test (OGTT), also known as impaired glucose tolerance (IGT). T2D was defined as fasting glucose ≥7.0 mmol/L or 2-h glucose >11.0 mmol/L as recommended by the American Diabetes Association [3]. Glycated hemoglobin (HbA1c) was unavailable for the controls and was therefore not included in the analyses.
Statistical methods
Categorical variables are reported as numbers and percentages. The distributions of some continuous variables were skewed, and they are therefore reported as medians and quartiles. The prevalences of prediabetes and T2D were calculated separately for ages <9, 9–11, and ≥12. The characteristics of patients with or without prediabetes and T2D were compared with the Mann–Whitney test for continuous variables and with the Chi-square test for dichotomous or categorical variables. Spearman’s coefficients for correlation were used to analyze the association between BMI Z-score and fasting glucose in the controls. The associations of prediabetes and T2D with patient characteristics were analyzed using logistic regression, both unadjusted and adjusted for age. ALT was included as a continuous variable in the logistic regression analysis; however, to enhance clarity, some of the results are presented per ten units, as one unit is too small to effectively display the findings. The exact characteristics of patients with T2D are not presented separately due to the low number of these cases and strict national data privacy regulations. The associations between the year of the first obesity-related visit (as a continuous variable) and the prevalence of prediabetes or T2D, female sex, obesity class I or II, hypertension, dyslipidemia, and MASLD were examined using logistic regression, while those between year of examination and median age, BMI-Z score, ALT, triglycerides, total-, LDL- and HDL cholesterol, fasting glucose and median insulin were examined by Pearson correlation among the patients. The association between the prevalence of prediabetes/T2D and year of examination was further analyzed using logistic regression models adjusted for median age, sex, BMI Z-score, ALT, hypertension, or dyslipidemia as a univariate model and also adjusted for age, sex and BMI z-score as a multivariable model. The analysis was not adjusted for perinatal factors due to the limited number of participants with available data. The association between the prevalence of prediabetes or T2D and year of examination was also analyzed separately in patients from primary or specialized healthcare. Temporal trends in prenatal variables were analyzed using Pearson correlation and logistic regression. The year 2020 was excluded from the analyzes due to the low number of participants. Statistical significance was defined as P-value < 0.05. Analyses were performed using SPSS, version 25.0 (IBM Corp., Armonk, NY, USA).
Results
Basic characteristics and prevalence of prediabetes and type 2 diabetes
The median age of the patients was 11.6, with 41.0% being girls (Table 1). The median ages of the controls were 7.6 at baseline, 9.8 at 2-year follow-up, and 15.8 at 8-year follow-up. Of the controls in these three study stages, 44.6–48.8% were girls, 3.3–4.7% had obesity, and 13.9–16.7% had overweight.
Of the patients, 34.2% had prediabetes and 1.3% T2D. IGT was diagnosed in 27.0% (89 tested with OGTT), and IFG was found in 32.7% based on data from 602 patients. The combined prevalence of prediabetes and T2D was similar in patients from primary healthcare and specialized care (32.7% vs. 35.0%, p = 0.572). Altogether, 6.9% of the controls had prediabetes, and none had T2D. The prevalence of prediabetes and T2D increased with age in both patients and controls (Fig. 1), as well as with pubertal stage in the patients (22.8% in prepuberty, 43.9% in puberty, 49.3% in postpuberty; p < 0.001) and controls (3.7%, 13.5% and 14.3%, p < 0.001). In the controls, fasting glucose correlated with BMI Z-score (r = 0.114, p < 0.001). The prevalence of prediabetes was 6.4% in controls with normal weight, 6.9% in those with overweight, and 18.2% in those with obesity (p = 0.010).
Temporal trends of glucose disturbances and early risk factors in patients
The prevalences of prediabetes or T2D and IFG were higher in patients making their first obesity-related healthcare visit in 2017–2019 compared to earlier years, while no significant changes were observed in the prevalence of obesity or MASLD (Fig. 2).
Prediabetes was defined based fasting glucose or 2 h oral glucose tolerance test, utilizing cutoffs by the American Diabetes Association [3].
Changes in sex distribution (OR for female sex 1.02, 95% CI 0.98–1.07, p = 0.349), median ages (r = −0.037, p = 0.373) and median BMI z-scores in different years of examination (2.47 [2002–4], 2.54 [2005–7], 2.36 [2008–10], 2.39 [2011–13], 2.46 [2014–16], 2.47 [2017–19], r = 0.005, p = 0.901) were not significant. Prediabetes was defined based fasting glucose or 2 h oral glucose tolerance test, utilizing cutoffs by the American Diabetes Association [3].
There was also a significant positive association between the year of the first obesity-related visit and the prevalence of prediabetes or T2D (OR 1.16, 95% CI 1.10–1.21, p < 0.001), level of fasting glucose (r = 0.191, p < 0.001) and median insulin (r = 0.211, p < 0.001). The increasing prevalence of prediabetes/T2D over time were observed in both primary (OR 1.18, 95% CI 1.06–1.33, p = 0.004) and specialized care (1.19, 1.12–1.26, p < 0.001), and remained significant after adjustment for age, sex, BMI z-score, ALT, hypertension and dyslipidemia (Supplementary Table S1). Significant association between the year of the examination and prediabetes/T2D prevalence persisted in a multivariable model consisting of examination year, children sex, age and BMI Z-score (OR 1.18, 95% CI 1.12–1.25, p < 0.001, Supplementary Table S1).
A significant negative association was observed between the year of the first obesity-related visit and the level of total cholesterol (r = –0.168, p < 0.001), and in the prevalence of hypertension (OR 0.940, 95% CI 0.894–0.988, p = 0.015). No changes over time were seen in child’s age at the obesity-related healthcare visit, BMI z-score, ALT, triglycerides, LDL, HDL, or in prevalence of female sex, obesity class I or II, dyslipidemia, or MASLD (Supplementary Table S2).
The prevalence of maternal overweight or obesity increased from 20.0% to 68.8% in patients making their obesity-related healthcare visit between 2002 and 2019 (OR 1.14, 95% CI 1.08–1.21, p < 0.001), while there were no changes in SGA, LGA, smoking, or hypertension (Supplementary Fig. S2, Supplementary Table S3). No changes were observed in the prevalence of gestational diabetes, pre-eclampsia, gestational weight gain, macrosomia, gestational age, or birth size (Supplementary Table S3). The prevalence of maternal overweight/obesity did not differ significantly between children with and without prediabetes (45.0% vs 38.8%, p = 0.240).
Associated factors for prediabetes/type 2 diabetes in the patient cohort
Patients with prediabetes or T2D were older, had higher levels of fasting insulin, HOMA-IR, triglycerides, and ALT, as well as lower HDL cholesterol level. They were also more likely to have acanthosis nigricans and MASLD compared to those without prediabetes or T2D (Table 1). Unadjusted and age-adjusted associations between prediabetes or T2D and other variables are presented in Table 2.
Patients with T2D had more advanced puberty, lower HDL cholesterol, a higher prevalence of acanthosis nigricans and MASLD, and a higher prevalence of T2D in first degree relatives compared to those without it (data not shown due to strict national data privacy regulations).
Discussion
We observed the prevalence of prediabetes of 34.2% and of T2D of 1.2% in patients with overweight or obesity, compared to 6.9% and 0%, respectively in controls. For comparison, a meta-analysis reported a global prediabetes prevalence of 8.8% in children, with the highest rates in Africa and the Americas and the lowest in Europe. The figure increased to 9.8% in children with overweight and to 14.3% in those with obesity [29]. Overall, there is substantial variation between studies. For example, Andes et al. observed a prevalence of 18.8% in the NHANES study, which increased to 25.7% in those with obesity [30]. Ghaddar et al. reported respective figures of 26.2% and 31% in 7–12-year-old US children [8]. In Europe, Brufani et al. detected glucose dysregulation in 12.4% of 3–18-year-old Italian children with overweight or obesity, while Hagman et al. reported this in 5.7% of 2–18-year-old German children and 17.1% of Swedish children with obesity [7, 31].
Obesity is a primary risk factor for glucose dysregulation; therefore, its prevalence and severity are likely to account for much of the variation in the prevalence of prediabetes. Obesity was substantially less common in our controls (3.3–4.7%) than in the US population (16.2–19.2%), likely partially explaining the similarly wide differences in prediabetes prevalence between the countries [8, 30]. Although many studies show an almost linear increase in the likelihood of glucose abnormalities with degree of obesity [8, 31, 32], this trend was not evident here. A potential explanation is the high proportion of children with obesity class II in this cohort, which may also account for the high prevalence of prediabetes compared to controls, who – by global comparison – tend to have relatively low rates of overweight and obesity.
Inconsistent definitions of prediabetes could also impact the comparisons. Here, it was defined based on IFG or IGT, whereas other studies may have used different criteria, such as combinations of IFG, IGT, and HbA1c [7, 8, 30, 31]. These measures reflect different aspects of glucose dysregulation and have varying prognostic significance and sources of error [30, 32]. The lack of HbA1c could be considered a weakness, although its utility in children is debated [33, 34]. Another controversial issue is the choice of the cut-off values, which have sometimes been criticized for being too low [35]. However, their usefulness as markers of disturbed glucose metabolism was demonstrated by the differences between patients and controls, as well as between controls with normal and increased weight. Moreover, early detection of prediabetes may enable timely interventions, reducing progression to T2D. Additional studies, for instance those using continuous glucose monitoring, could establish optimal cutoffs for identifying children at increased risk - without leading to unnecessary burden on families.
Besides obesity, other characteristics associated with prediabetes may also vary between cohorts. In line with earlier reports, we observed that the risk of prediabetes increased with age [7, 8, 30, 31]. Furthermore, both we and Brufani et al. identified an association with advanced pubertal stage, which aligns with the phenomenon of transient insulin resistance [7, 36]. Intriguingly, this may not fully resolve in adolescents with obesity, possibly explaining why prediabetes continued to increase from puberty to postpuberty [37]. Additionally, sex differences in the timing and magnitude of pubertal insulin resistance may contribute to the reported overrepresentation of prediabetes in boys [30, 31]. However, we, along with some others, found no sex difference in the prevalence, indicating the need for further research [7, 8].
Ethnicity partly explains the variation in the prevalence of prediabetes, with the risk gradually decreasing from non-Hispanic Blacks to non-Hispanic White [16, 38]. Consequently, the aforementioned high-prevalence US cohort predominantly consisted of Hispanic and non-Hispanic Blacks, among whom prediabetes was 2.8–4.8 times more common than among non-Hispanic Whites [8]. Conversely, Andes et al. included mostly non-Hispanic Whites, among whom the prevalence was lower as a whole even among those with obesity than in the study by Ghaddar et al. [8, 30]. Ethnicity data were lacking in the European studies, but they likely included mostly non-Hispanic White [7, 31]. However, the prevalence was markedly higher in Sweden than in Germany, and even higher in our cohort, which likely comprised mostly non-Hispanic White, despite comparable BMI [31]. This could be due to genetic variation and unidentified environmental factors. Altogether, glucose disturbances appear to result from a complex interaction between genetics, the quantity and function of fat and muscular tissue, physical activity, diet, and other factors [18, 19].
Additional factors associated with prediabetes included higher levels of triglycerides and ALT, and presence of acanthosis nigricans and MASLD, most of which were independent of age. This is in line with the reported clustering of glucose disturbances and other cardiometabolic risk factors already in adolescence [10, 11, 15]. Of particular interest is the association between MASLD, which, as also noted here, even occurs independent of the degree of obesity [14, 32, 39]. This, together with the key role of liver in glucose metabolism, indicates shared pathogenesis [40]. In line, the study by Putri et al. found that presence of MASLD markedly increased the risk of T2D, and a synergistic effect on risk was observed in children with obesity, MASLD, and prediabetes [41]. The risk of such a vicious circle further supports screening for MASLD in children with prediabetes, and vice versa.
We observed a striking increase in prediabetes over time, independent of changes in BMI z-score or other metabolic conditions. In fact, the prevalence of hypertension and levels of total cholesterol even decreased during the same period, while age, sex distribution, prevalence of obesity class I or II, and other metabolic disturbances remained unchanged. We cannot fully exclude lower-threshold testing or changes in referral practice, which may partly explain the observed increase in specialized care. However, we consider these factors unlikely to fully account for the observed changes of this magnitude. To support this conclusion, a similar increase was also observed in primary healthcare, which patients can access without a referral. Additionally, to the best of our knowledge, there was also no changes in the laboratory methods and reference ranges used. Nevertheless, it must be emphasized that these results were observed in a single population, and further studies are needed to better understand the trend.
Generally, an increase in prediabetes has been observed also previously, the main explanation being the obesity epidemic [9, 29]. Mayer-Davis et al. reported gradual increase in pediatric T2D in 2002–12, but noted that there was no similar change in obesity within the population [42]. Our novel finding of an obesity-independent change could be due to population-level changes, for example, in body composition, diet and physical activity. An additional contributing factor could be early-life programming, as maternal obesity tripled during the same period [43]. However, no differences were observed between children with and without prediabetes regarding birth size or prevalence of gestational diabetes, although data on the latter were limited. There was also a parallel, albeit non-significant, increase in MASLD, which is an important consideration due to its strong association with T2D. Of note, although these findings are likely generalizable to the homogenous Finnish population, further studies in more diverse populations and geographical areas are needed. Additionally, due to the short recruitment period for the baseline visit, it is not possible to determine whether a similar increase in prediabetes occurred also in the PANIC cohort. Overall, our findings add to concerns about the already alarming predictions regarding the future prevalence of T2D [44].
The study consisted of a large cohort of patients from different levels of healthcare and a population-representative control cohort. However, the somewhat higher proportion of children from tertiary care than primary care, along with the high prevalence of class II obesity, may have led to an overestimation of the prediabetes prevalence. In contrast, the lower prevalence of obesity in the PANIC cohort compared to the general Finnish population may have resulted in an underestimation of glucose disturbances [20, 45, 46]. A further strength was the careful consideration of confounding factors. Systematic recording of glucose measurements throughout the study period also enabled the assessment of temporal trends. Limitations include the retrospective design in the patient cohort, which led to incomplete OGTT measurements, and lack of HBA1c values for the controls. Furthermore, the statistical power was reduced in some subgroup analyses, including the prevalence of MASLD across different time intervals. Finally, while the availability of free public healthcare and the homogeneous study population increased internal validity, these factors may limit the generalizability of the results.
Conclusion
Disturbances in glucose metabolism were highly prevalent among pediatric patients with overweight or obesity and were particularly associated with MASLD. Additionally, the prevalence of prediabetes appears to be increasing rapidly, independent of changes in the degree of obesity or other metabolic conditions. This alarming trend warrants further confirmation, along with an in-depth investigation into its underlying causes.
Data availability
Due to privacy and confidentiality concerns, the data supporting the findings of this study are not publicly available. De-identified data are available from the corresponding author upon reasonable request.
References
Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014;384:766–81.
Zheng Y, Ley SH, Hu FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol. 2018;14:88–98.
American Diabetes Association Professional Practice Committee. Diagnosis and Classification of Diabetes: Standards of Care in Diabetes—2024. Diabetes Care. 2023;47:S20–42.
Type 2 diabetes in children and adolescents. American Diabetes Association. Diabetes Care. 2000;23:381-9.
Candler TP, Mahmoud O, Lynn RM, Majbar AA, Barrett TG, Shield J. Continuing rise of type 2 diabetes incidence in children and young people in the UK. Diabetic Med. 2018;35:737–44.
Bjornstad P, Chao LC, Cree-Green M, Dart AB, King M, Looker HC, et al. Youth-onset type 2 diabetes mellitus: an urgent challenge. Nat Rev Nephrol. 2023;19:168–84.
Brufani C, Ciampalini P, Grossi A, Fiori R, Fintini D, Tozzi A, et al. Glucose tolerance status in 510 children and adolescents attending an obesity clinic in Central Italy. Pediatric Diabetes. 2010;11:47–54.
Ghaddar R, Hudson EA, Jeans MR, Vandyousefi S, Landry MJ, Davis JN. Ethnicity/race, parent educational attainment, and obesity associated with prediabetes in children. Nutr Diabetes. 2023;13:15.
Liu J, Li Y, Zhang D, Yi SS, Liu J. Trends in prediabetes among youths in the US from 1999 through 2018. JAMA Pediatr. 2022;176:608–11.
Li C, Ford ES, Zhao G, Mokdad AH. Prevalence of pre-diabetes and its association with clustering of cardiometabolic risk factors and hyperinsulinemia among U.S. adolescents: National Health and Nutrition Examination Survey 2005–2006. Diabetes Care. 2009;32:342–7.
Casagrande SS, Menke A, Linder B, Osganian SK, Cowie CC. Cardiovascular risk factors in adolescents with prediabetes. Diabetic Med. 2018;35:1202–9.
Newton KP, Hou J, Crimmins NA, Lavine JE, Barlow SE, Xanthakos SA, et al. Prevalence of prediabetes and type 2 diabetes in children with nonalcoholic fatty liver disease. JAMA Pediatr. 2016;170:e161971.
European Society for Pediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN), European Association for the Study ofthe Liver (EASL), North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition (NASPGHAN), et al. Paediatric steatotic liver disease has unique characteristics: A multisociety statement endorsing the new nomenclature. J Ped Gastroenterol Nutr. 2024;78:1190–6..
Nobili V, Mantovani A, Cianfarani S, Alisi A, Mosca A, Sartorelli MR, et al. Prevalence of prediabetes and diabetes in children and adolescents with biopsy-proven non-alcoholic fatty liver disease. J Hepatol. 2019;71:802–10.
Di Bonito P, Licenziati MR, Corica D, Wasniewska MG, Di Sessa A, Del Giudice EM, et al. Phenotypes of prediabetes and metabolic risk in Caucasian youths with overweight or obesity. J Endocrinol Invest. 2022;45:1719–27.
Perng W, Conway R, Mayer-Davis E, Dabelea D. Youth-onset type 2 diabetes: the epidemiology of an awakening epidemic. Diabetes Care. 2023;46:490–9.
Rooney MR, Fang M, Ogurtsova K, Ozkan B, Echouffo-Tcheugui JB, Boyko EJ, et al. Global prevalence of prediabetes. Diabetes Care. 2023;46:1388–94.
Valaiyapathi B, Gower B, Ashraf AP. Pathophysiology of type 2 diabetes in children and adolescents. Curr Diabetes Rev. 2020;16:220–9.
Kantartzis K, Machann J, Schick F, Fritsche A, Häring HU, Stefan N. The impact of liver fat vs visceral fat in determining categories of prediabetes. Diabetologia. 2010;53:882–9.
Viitasalo A, Eloranta AM, Lintu N, Väistö J, Venäläinen T, Kiiskinen S, et al. The effects of a 2-year individualized and family-based lifestyle intervention on physical activity, sedentary behavior and diet in children. Prev Med. 2016;87:81–88.
Saari A, Sankilampi U, Hannila ML, Kiviniemi V, Kesseli K, Dunkel L. New Finnish growth references for children and adolescents aged 0 to 20 years: Length/height-for-age, weight-for-length/height, and body mass index-for-age. Ann Med. 2011;43:235–48.
Cole TJ, Lobstein T. Extended international (IOTF) body mass index cut-offs for thinness, overweight and obesity. Pediatr Obes. 2012;7:284–94.
Sorva R, Lankinen S, Tolppanen EM, Perheentupa J. Variation of growth in height and weight of children. II. After infancy. Acta Paediatr Scand. 1990;79:498–506.
Obesity. Current Care Guidelines. Working group set up by the Finnish Medical Society Duodecim, the Finnish Association for the Study of Obesity, and the Finnish Paediatric Society. Helsinki: The Finnish Medical Society Duodecim; 2025. www.kaypahoito.fi.
Flynn JT, Kaelber DC, Baker-Smith CM, Blowey D, Carroll AE, Daniels SR, et al. Clinical Practice Guideline for Screening and Management of High Blood Pressure in Children and Adolescents. Pediatrics. 2017;140:e20171904.
Eslam M, Alkhouri N, Vajro P, Baumann U, Weiss R, Socha P, et al. Defining paediatric metabolic (dysfunction)-associated fatty liver disease: an international expert consensus statement. Lancet Gastroenterol Hepatol. 2021;6:864–73.
Kurtoğlu S, Hatipoğlu N, Mazıcıoğlu M, Kendirici M, Keskin M, Kondolot M. Insulin resistance in obese children and adolescents: HOMA-IR cut-off levels in the prepubertal and pubertal periods. J Clin Res Pediatr Endocrinol. 2010;2:100–6.
FIGO Textbook of Pregnancy Hypertension. APEC International. 2016. https://apecint.org/professionals/education-and-training/figo-textbook-of-pregnancy-hypertension/.
Han C, Song Q, Ren Y, Chen X, Jiang X, Hu D. Global prevalence of prediabetes in children and adolescents: A systematic review and meta-analysis. J Diabetes. 2022;14:434–41.
Andes LJ, Cheng YJ, Rolka DB, Gregg EW, Imperatore G. Prevalence of prediabetes among adolescents and young adults in the United States, 2005-2016. JAMA Pediatr. 2020;174:e194498.
Hagman E, Reinehr T, Kowalski J, Ekbom A, Marcus C, Holl RW. Impaired fasting glucose prevalence in two nationwide cohorts of obese children and adolescents. Int J Obes. 2014;38:40–45.
Pedicelli S, Fintini D, Ravà L, Inzaghi E, Deodati A, Spreghini MR, et al. Prevalence of prediabetes in children and adolescents by class of obesity. Pediatr Obes. 2022;17:e12900.
Weiss R, Santoro N, Giannini C, Galderisi A, Umano GR, Caprio S. Prediabetes in youth – mechanisms and biomarkers. Lancet Child Adolesc Health. 2017;1:240–8.
Farmer A. Use of HbA1c in the diagnosis of diabetes. BMJ. 2012;345:e7293.
World Health Organization, International Diabetes Federation. Definition and diagnosis of diabetes mellitus and intermediate hyperglycaemia: report of a WHO/IDF consultation. 2006. https://iris.who.int/handle/10665/43588.
Moran A, Jacobs DR Jr, Steinberger J, Hong CP, Prineas R, Luepker R, et al. Insulin resistance during puberty: results from clamp studies in 357 children. Diabetes. 1999;48:2039–44.
Kelsey MM, Zeitler PS. Insulin resistance of puberty. Curr Diabetes Rep. 2016;16:64.
Lawrence JM, Divers J, Isom S, Saydah S, Imperatore G, Pihoker C, et al. Trends in prevalence of type 1 and type 2 diabetes in children and adolescents in the US, 2001-2017. JAMA. 2021;326:717–27.
Cali AM, De Oliveira AM, Kim H, Chen S, Reyes-Mugica M, Escalera S, et al. Glucose Dysregulation and hepatic steatosis in obese adolescents: Is there a link?. Hepatology. 2009;49:1896–903.
Perseghin G. Viewpoints on the way to a consensus session: where does insulin resistance start? The liver. Diabetes Care. 2009;32:S164–7.
Putri RR, Casswall T, Danielsson P, Marcus C, Hagman E. Steatotic Liver disease in pediatric obesity and increased risk for youth-onset type 2 diabetes. Diabetes Care. 2024;47:1–9.
Mayer-Davis EJ, Lawrence JM, Dabelea D, Divers J, Isom S, Dolan L, et al. Incidence Trends of type 1 and type 2 diabetes among youths, 2002-2012. N Eng J Med. 2017;376:1419–29.
Sauder KA, Hockett CW, Ringham BM, Glueck DH, Dabelea D. Fetal overnutrition and offspring insulin resistance and β-cell function: the Exploring Perinatal Outcomes among Children (EPOCH) study. Diabet Med. 2017;34:1392–9.
Imperatore G, Boyle JP, Thompson TJ, Case D, Dabelea D, Hamman RF, et al. Projections of type 1 and type 2 diabetes burden in the U.S. population aged <20 years through 2050: dynamic modeling of incidence, mortality, and population growth. Diabetes Care. 2012;35:2515–20.
Finnish Institute for Health and Welfare (THL). Child and adolescent overweight and obesity 2023. Helsinki: Finnish Institute for Health and Welfare; 2024. https://thl.fi/tilastot-ja-data/tilastot-aiheittain/lapset-nuoret-ja-perheet/lasten-ja-nuorten-ylipaino-ja-lihavuus.
De Jesus JM. Expert panel on integrated guidelines for cardiovascular health and risk reduction in children and adolescents: summary report. Pediatrics. 2011;5:213–56.
Acknowledgements
Funding and support for the study was given by: The Foundation for Pediatric Research, the Competitive State Research Financing of the Expert Area of Tampere University Hospital, the Maire Rossi Foundation, the Maud Kuistila Foundation, the Juho Vainio Foundation, the Finnish Cultural Foundation, the Emil Aaltonen Foundation, the Paulo Foundation, Orion Research Foundation, and the Sigrid Jusélius Foundation. The funders had no role in study design, data collection and analysis, decision to publish, or in the preparation of the present manuscript.
Funding
Open access funding provided by Tampere University (including Tampere University Hospital).
Author information
Authors and Affiliations
Contributions
Hanna Riekki and Linnea Aitokari were involved in study design, data collection and analysis drafting of the manuscript. Laura Kivelä, Antti Saari and Eero Haapala were involved in study design and critical revision of the manuscript. Anna Viitasalo and Sonja Soininen were involved in data collection and critical revision of the manuscript. Heini Huhtala was involved in study design, statistical analysis, and critical revision of the manuscript. Timo Lakka was involved in study design, data collection, and critical revision of the manuscript. Kalle Kurppa was involved in study design, study supervision, and critical revision of the manuscript; he is thus the guarantor of this work. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Riekki, H., Aitokari, L., Saari, A. et al. The prevalence of prediabetes is high and has rapidly increased, independent of the degree of obesity, in Finnish children with overweight or obesity. Int J Obes 50, 407–413 (2026). https://doi.org/10.1038/s41366-025-01950-y
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41366-025-01950-y