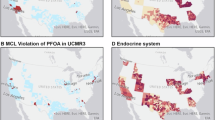
Abstract
Background
Studies from areas with high level per- and polyfluoroalkyl substance (PFAS) contamination have reported significant contributions of drinking water to overall exposure but few studies have focused on general populations.
Objective
To examine the association between PFAS in public water system drinking water and concentrations in serum among a general population of California adults.
Methods
The CARE (California Regional Exposure) biomonitoring study measured PFAS in serum of Southern and Eastern California adults between 2018 and 2020. Participant addresses were geocoded and geographically assigned to public water systems (PWS). Between 2019 and 2022, the California State Water Resources Control Board issued investigative PFAS monitoring orders to potentially impacted PWS, focusing primarily on source wells. PFAS detections above the required reporting limit (4 ng/L for PFAS included in the final analysis) were used to assess associations with serum PFAS levels.
Results
Out of 563 participants in Southern California included in the final analysis, 314 (56%) lived in a PWS service area with at least one PFAS detected in their untreated source water and/or treated drinking water. Serum perfluorohexane sulfonic acid (PFHxS) geometric mean concentrations were 31.9% (95% CI: 11.1–56.6%) higher among participants whose PWS had at least one PFHxS detection compared to those without detectable levels. For participants with post-treatment drinking water data (n = 235), serum PFAS geometric mean concentrations were higher compared to those without detections for all modeled PFAS: PFHxS 79.9% (38.3–133.9%), perfluorooctanoic acid (PFOA) 30.4% (5.5–61.2%), perfluorooctane sulfonic acid (PFOS) 31.2% (0.7–70.9%), and ∑5 PFAS 42.0% (14.5–76.1%).

Impact
-
This study examined the association between public water system PFAS detections and serum concentrations among a general population of Southern California adults. We found that PFAS detections in public water systems were associated with higher serum PFAS concentrations. Our findings suggest PFAS contamination in drinking water may be a significant contributor to serum PFAS levels, even among communities without high level contamination from industrial manufacturing. These results support drinking water monitoring initiatives in California to understand PFAS contamination and mitigate exposure.
Similar content being viewed by others
Introduction
Per- and polyfluoroalkyl substances (PFAS) are a class of thousands of environmental chemical pollutants that have been widely used in industrial and consumer applications for their stain-and water-resistant capabilities [1]. Due to their resistance to degradation and long biological half-lives, PFAS have persisted in the environment, accumulated in drinking water and living organisms [2,3,4], and have been detected in human blood around the world for many years [5,6,7]. Ingestion of contaminated water and food is a primary exposure route in humans, though other pathways of exposure include use of PFAS-containing personal care products, inhalation of contaminated dust or volatile PFAS, and dermal absorption [8,9,10,11,12,13]. Exposure to some PFAS have been associated with a variety of adverse health outcomes including endocrine disruption [14], immune suppression [15, 16], lower birth weight [17], and cancers such as testicular [18, 19] and kidney [20, 21].
Elevated levels of PFAS drinking water contamination have been found in areas surrounding fluorochemical manufacturing plants [22] and in surface and ground water near sources that use aqueous film-forming foams (AFFF), such as military training bases and airports [23, 24]. Contaminated groundwater near major PFAS exposure sources have reported PFAS concentrations up to thousands of parts per trillion [25,26,27,28,29,30,31]. However, recent data have also shown that lower-level drinking water contamination by PFAS is widespread and not restricted to these specific sites. The Environmental Protection Agency (EPA) has estimated that 4100–6700 water systems in the U.S., serving a population of 83–105 million individuals, likely have contamination by at least one PFAS exceeding the maximum contaminant levels (MCLs) finalized in April 2024 [32]. These MCLs, which represent the primary drinking water regulation for six PFAS, range from 4 to 10 parts per trillion. MCLs were set for five PFAS as individual contaminants (perfluorooctanoic acid [PFOA], perfluorooctane sulfonic acid [PFOS], perfluorohexane sulfonic acid [PFHxS], perfluorononanoic acid [PFNA], and hexafluoropropylene oxide dimer acid [HFPO-DA)]) and for PFAS mixtures containing at least two or more of PFHxS, PFNA, HFPO-DA, and perfluorobutane sulfonic acid [PFBS] [32]. After initial submission of this manuscript, in May 2025, the EPA announced plans to keep the MCLs for PFOA and PFOS but to rescind and reconsider regulatory determinations for PFHxS, PFNA, HFPO-DA, and PFBS [33].
Although the State of California does not have PFAS industrial manufacturing from fluorochemical plants, other PFAS contamination sources are present. These include direct sources such as airports, military facilities, chrome platers, and refineries/bulk fuel terminals, as well as indirect sources such as wastewater treatment plants, land application of sewage sludge, and landfills. Starting in 2018, the California State Water Resources Control Board (SWRCB) Division of Drinking Water began instituting health-based advisory drinking water thresholds (‘notification levels’) at which consumer notification of exceedances is required and concentrations at which removal of or treatment of a drinking water source (‘response levels’) is recommended for four PFAS: PFOS, PFOA, PFBS, and PFHxS [34]. From 2019 to 2022, the SWRCB sent investigative orders to 388 public water systems in California to monitor drinking water PFAS concentrations, focusing on groundwater near potential PFAS contamination sources.
Recent literature has identified associations between PFAS concentrations in drinking water and in individuals. However, studies have typically focused on areas with high levels of drinking water contamination due to industrial manufacturing or AFFF usage. The few studies that have assessed the contribution of PFAS drinking water contamination on PFAS levels in general populations have either focused on tap water data from several decades ago, specific sub-populations, or the U.S. EPA’s Unregulated Contaminant Monitoring Rule 3 (UCMR 3) PFAS sampling data with high minimum reporting levels (up to 10 times higher than current MCLs). More insight into the relationship between drinking water PFAS contamination and serum levels among populations without widespread high-level contamination is needed. In the current analysis we linked PFAS biomarker data to the SWRCB’s drinking water monitoring data to assess the association between PFAS detections in public water system drinking water and PFAS concentrations in serum among a general population of adults in Southern California. This study is among the first few studies to use high resolution public water system boundary mapping to link participant serum levels to PFAS monitoring data.
Methods
Study population/CARE study
The California Regional Exposure (CARE) study was a biomonitoring surveillance study designed to evaluate the extent of environmental chemical exposure among the California population. Detailed methods have been described previously [35]. Briefly, the CARE study was a cross-sectional biomonitoring study implemented regionally in Southern and Eastern California. The CARE study used a quota sampling approach with quotas based on race/ethnicity, gender, and geography to represent specific regions of California. Participants were recruited in Los Angeles County in 2018 (CARE-LA, n = 430), Riverside, San Bernardino, Imperial, Mono, and Inyo counties in 2019 (CARE-2, n = 359), and San Diego and Orange counties (CARE-3, n = 90) in 2020. CARE-3 recruitment was stopped prematurely due to the COVID-19 pandemic. CARE-LA, CARE-2, and CARE-3 were combined resulting in a total of 879 CARE participants.
Participant eligibility criteria included being 18 years or older and a resident in the region for at least 12 months. Participants completed demographic and exposure questionnaires and provided blood and urine samples. Full participant demographics and biomarker data are available in the initial report of CARE study findings [35]. Blood samples were collected by licensed phlebotomists typically within three weeks after enrollment. After blood samples were centrifuged, serum samples were stored in a –20 °C freezer and shipped on dry ice to the Biomonitoring California laboratory.
Serum PFAS analysis
Twelve PFAS in serum (Table 1) were analyzed by the Environmental Chemistry Laboratory, California Department of Toxic Substances Control using online solid-phase extraction ultra-high performance liquid chromatography tandem mass spectrometry (SPE-HPLC-MS/MS), based on a method by Wang et al. [36]. Serum samples were placed into the online SPE-HPLC system (Symbiosis Pharma system with Mistral CS Cool, Spark Holland Inc., Princeton, NJ, USA) and loaded onto a C18 cartridge (HySphere C18 HD, 7 μm, 10 × 2 mm, Spark Holland Inc., Princeton, NJ, USA). After washing, the target analytes were eluted to a C8 HPLC column (BETASIL C8 column, Thermo Fisher Scientific, Waltham, MA USA) for separation. The eluate was then introduced to the MS/MS (4000 QTrap, Sciex, Framingham, MA, USA) for multiple-reaction-monitoring (MRM) analysis. The area of the Q1/Q3 ion pairs were used in the analysis. Duplicate samples of National Institute of Standards and Technology Standard Reference Materials 1958 (National Institute of Standards and Technology, Gaithersburg, MD, USA), certified for four PFAS compounds PFOA, PFOS, PFHxS, and PFNA, were analyzed with each batch of samples to verify method accuracy. Duplicates of two in-house quality control samples containing all compounds of interest were also analyzed with each batch to monitor method recovery and precision. Additionally, the Environmental Chemistry Laboratory participates in external proficiency testing at least three times per year to monitor method accuracy. Duplicate blank samples (bovine calf serum) were also processed with each batch of samples to monitor background levels.
Drinking water PFAS analysis
Beginning in 2019, the California SWRCB Division of Drinking Water sent investigative orders to public water systems in California to monitor PFAS concentrations in drinking water [37], focusing on untreated raw source wells in areas with known contamination or adjacent to suspected PFAS sources, including airports, chrome platers, bulk fuel terminals and refineries, and those impacted secondarily by PFAS – landfills and wastewater treatment plants. While the focus of sampling was on source wells, some water systems elected to sample treated water. Monitoring of source wells allows for management of drinking water quality at the origin of PFAS contamination (Supplementary Fig. S1). Sampling coverage varied among water systems. Public water system monitoring data from the first three phases of investigative orders (2019–2022) were the focus of this work as described in Supplementary Table S1. Eighteen PFAS analytes (Table 2) were measured by regional laboratories using EPA method 537.1 [38], and results of testing were included in the Division of Drinking Water’s database of record, the Safe Drinking Water Information System (SDWIS) [39]. This analysis includes SDWIS PFAS monitoring data from community public water systems until July 2022.
The 2019–2022 investigative order statewide reporting limits (known as the Consumer Confidence Report Detection Limits [CCRDLs]) were determined by the SWRCB based on a review of method detection limits from regional laboratories throughout the state for monitoring conducted between August and December 2019 (Supplementary Table S1). During the 2019–2022 investigative orders, if a water system confirmed PFAS concentrations above the CCRDLs, the water system was required to report the detection in their annual drinking water quality report, also known as the Consumer Confidence Report. For our analysis, a uniform cutoff limit was applied to all public water system PFAS monitoring data by censoring the drinking water PFAS concentrations in the SDWIS dataset to the SWRCB determined CCRDLs from their investigative orders. Thus a ‘detection’ in this analysis refers to a drinking water concentration above the CCRDLs (2–4 ng/L).
Matching CARE participants to drinking water data
CARE participant home addresses were geocoded using ArcGIS and spatially joined to the System Area Boundaries shapefile published by the SWRCB (downloaded December 23, 2023) [40] to match participants to a single public water system service area. When participants matched to more than one water system service area, the water system service area was confirmed by contacting water systems directly or through reference of public water system websites. One large water system service area covering the Los Angeles area, the Los Angeles Department of Water and Power, had a substantial number of participant residences within its service area boundary (n = 184). Therefore, we divided this large water system into its five local water quality service area boundaries [41]. Well data were matched to the local service area by well assignments provided by the water system. Public water system drinking water data from the SWRCB’s SDWIS dataset was then linked to participants, where two of the five local service areas had PFAS monitoring data and were included in this analysis. For the purposes of this analysis, these two local service areas are treated as distinct water systems.
Statistical analysis
Participants were excluded from the original dataset of 879 individuals if geocoded addresses were invalid, outside of the water system service area boundaries, or did not have PFAS monitoring data. Participants missing serum samples or key sociodemographic variables, or who listed a private well as their primary drinking water source were also excluded for a final dataset of 563 participants (Supplementary Fig. S2). In order to provide regional weighted serum analyte distributions that are representative of the underlying population, missing gender, race-ethnicity, education, and income data for CARE-LA and CARE-2 participants had been previously imputed, as described in the initial report of CARE study findings [35]. Missing data were imputed by Hot-Deck SAS imputation (SAS version 9.4; SAS Institute Inc., Cary, NC, USA), a technique that uses observed values from the sample to impute missing values. Gender, race-ethnicity, and education (n = 10 or 2%) were first imputed, indexed by age and geography, and then the resulting values used to impute missing values of income (n = 59 or 10%). Since CARE-3 recruitment was not completed and only included 90 participants, CARE-3 data were not weighted and missing data not imputed.
We calculated descriptive statistics (detection frequencies, geometric means, confidence intervals, percentiles, and maximums) for serum PFAS and drinking water PFAS to describe the distribution. Serum PFAS concentrations below the method detection limit (MDL) were replaced with MDL/sqrt(2). Geometric means and 95% confidence intervals were suppressed for serum analytes with ≤60% detection frequency to avoid unreliable estimates due to high proportions of non-detects. For drinking water, N and detection frequencies were calculated by water system while percentile and maximum concentrations represent averages by sampling location after replacing concentrations below the CCRDL with CCRDL/sqrt(2). To assess the relationships among serum PFAS with detection frequency ≥60% we calculated Spearman rank correlations.
In order to create drinking water exposure indicators, we categorized the public water system drinking water data in three ways (Supplementary Table S2). This approach was chosen because the SWRCB sampled primarily from source wells (i.e., untreated), and data necessary to estimate finished water PFAS concentrations that enter the distribution system such as blending rates, removal due to treatment, and water volume were not available. First, drinking water data were divided into a binary category of water systems with no PFAS detections among sampling locations and water systems with at least one detection among sampling locations. Second, drinking water data were categorized into a three-tiered category after calculating the detection frequency for each water system as the number of sampling locations in a water system with at least one detection divided by the total number of sampling locations tested for PFAS. The three-tiered categorization included water systems with no PFAS detections, water systems with <50% detection frequency, and water systems with ≥50% detection frequency. The cutoff of 50% was chosen after determining that median detection frequencies among water systems were all ~50%. Third, while the 2019–2022 PFAS monitoring orders sampled primarily from raw water source wells, 26% of the drinking water was sampled at treatment plants or distribution systems. As post-treatment drinking water PFAS concentrations are more likely to be comparable to finished drinking water concentrations, our third portion of this analyses applied the binary drinking water exposure indicator categorization but only included the subgroup of participants matched to water systems with post-treatment drinking water PFAS data.
Of the 12 PFAS analyzed in serum samples and the 18 measured in water samples, many analytes had high proportions of non-detect data, so we prioritized chemicals for analysis if they were detected both in ≥60% of serum samples and at higher frequencies in drinking water data. Among the analytes detected in ≥60% of serum samples (N-methyl-perfluorooctane sulfonamido acetic acid [Me-PFOSA-AcOH], perfluorodecanoic acid [PFDeA], PFHxS, PFNA, PFOA, PFOS, perfluoroundecanoic acid [PFUnDA]), drinking water detection frequencies clustered into higher and lower groups: ≥47.1% for PFHxS, PFOA, and PFOS, and ≤11.4% for Me-PFOSA-AcOH, PFDeA, PFNA, and PFUnDA. PFOA, PFOS, and PFHxS met these criteria and were included in the final analysis. We also created a mass sum of the five PFAS (∑5 PFAS) that had at least one detection in drinking water and were detected in at least 60% of serum samples: PFOA, PFOS, PFHxS, PFNA, and PFDeA. Due to low exposure category counts, PFNA and PFDeA were included only in the binary categorization models. Serum PFAS concentrations were natural log transformed to normalize the data distribution. Separate multivariable regression models were run to examine the association between drinking water detection or detection frequency categories and serum concentrations.
Covariates were chosen based on a directed acyclic graph (DAG) conceptual model, and included age, sex, parity, race-ethnicity, education, income, and nativity (Supplementary Fig. S3). Race/ethnicity was categorized as Asian (single identification), Black (single identification), Hispanic or Latino (any race), non-Hispanic White (single identification), or Multi-racial and Other (non-Hispanic multi-racial, American Indian or Alaskan Native, Native Hawaiian or Other Pacific Islander). All analyses except for missing data imputation were carried out using R statistical software (R version 4.4.2, R Core Team 2024, Vienna, Austria). We report adjusted percent change for serum PFAS, calculated by exponentiating the beta estimate, subtracting 1, and multiplying by 100.
Sensitivity analyses
To investigate whether the primary type of drinking water consumed (tap water, bottled water, or other) modified the association between drinking water detection categories and serum concentrations, we repeated our analysis of the binary drinking water exposure indicator, stratifying regression models by participants who reported primarily drinking tap water (n = 302) and participants who reported primarily drinking bottled water or other water types (n = 258). The bottled or other water type group mostly consisted of participants who reported drinking primarily store-bought water including bottled water and water from a water cooler (n = 242 or 94%), and some participants who reported other type of water (n = 16 or 6%).
Since water systems sometimes purchase water from other water systems, we also ran a sensitivity analysis including water purchasing data in the models. Data on 2019 water purchasing behavior was obtained from the Office of Environmental Health Hazard Assessment (OEHHA), which employs this data in the CalEnviroScreen 4.0, a mapping tool identifying pollution burdens in California [42]. The binary drinking water exposure indicator was adjusted to include water purchasing behavior using the same SWRCB SDWIS drinking water quality dataset where PFAS data were available: a water system was considered to have no detections if that water system and the water systems it purchased water from did not have any PFAS detections, while a water system was considered to have at least one detection if that water system or any water systems it purchased water from had at least one PFAS detection.
Results
Population characteristics
Of the original 879 CARE participants, 563 participants in Southern California met inclusion criteria and matched to a public water system with PFAS testing data (Table 3; Supplementary Fig. S2). These participants’ ages ranged from 19 to 93 (mean 48.5 ± 15.6) years old, and 61% were female. Forty percent of participants were Hispanic, followed by 36% non-Hispanic White. Over three quarters of participants (82%) had attended college, a trade school, or reported receiving a graduate degree, while 69% reported household income in the lower income categories below $75,000. Most participants were U.S.-born (74%) and either lived in the U.S. for their whole life or more than 25 years (85%). Tap water was the primary drinking water type for 54% of participants, and 43% reported primarily drinking store-bought bottled water or water from a water cooler. Over half of participants (52%) had lived in their home for longer than 5 years at the time of the study.
PFAS concentrations in serum
Serum PFAS were detected in over 99% of study participants (Table 1). Among the twelve PFAS analytes measured in the CARE study, PFHxS, PFOA, PFOS, PFNA, Me-PFOSA-AcOH, PFUnDA, and PFDeA were detected in ≥60% of participants’ serum. PFOS was found in the highest concentration (geometric mean of 2.08 ng/mL), followed by PFOA and PFHxS (geometric means of 0.983 and 0.670 ng/mL). Spearman correlations between serum PFAS ranged from 0.12 to 0.75, with the highest correlations for PFOS and PFHxS (0.75), PFOA and PFHxS (0.71), PFOS and PFNA (0.71), PFOA and PFNA (0.7), and PFNA and PFDeA (0.7) (Supplementary Fig. S4). Overall, serum geometric mean PFAS concentrations as well as weighted CARE-LA and weighted CARE-2 serum geometric mean PFAS concentrations were lower than that of national levels from the 2017–2020 cycles of the National Health and Nutrition Examination Survey (NHANES) (Supplementary Fig. S5). Serum concentrations of CARE participants included in the final dataset (n = 563) were very similar to those excluded from the final analysis (n = 309) (Supplementary Table S3).
PFAS concentrations in drinking water
Of the 388 community public water systems statewide that conducted monitoring for 18 PFAS during 2019–2022, 70 water systems matched to CARE participants (Supplementary Fig. S6); 314 (56%) of participants matched to a water system with at least one detection. The 70 water systems included in this study were primarily large water systems, with 67 out of 70 (96%) serving water to more than 10,000 people (Supplementary Table S4) in primarily urban areas of Southern California. Among the 70 water systems in the study area, drinking water was sampled from a total of 788 sampling locations, 26% (204) of which were post-treatment sampling locations (Table 2, Supplementary Table S5).
Within the study area, each of the 18 PFAS included in the SWRCB monitoring was detected at least once in 0–48.6% of water systems (Table 2). California-wide, detection frequencies for these individual PFAS ranged from 0–36.3% of water systems (Supplementary Table S6). Overall, detection frequencies were higher in the study area compared to all of California. PFBS, PFOA, PFOS, PFHxS, and perfluorohexanoic acid (PFHxA) were detected the most among water systems and had detection frequencies in the study area 15–20% higher than the detection frequencies measured in all of California (PFBS: 47.1 vs 27.4%, PFOA: 48.6 vs 32.7%, PFOS: 47.1 vs 36.3%, PFHxS: 48.6 vs 31.5%, and PFHxA: 43.5 vs 28.4%). Among both the study area and California-wide, detectable concentrations were comparable and in the 10–300 ng/L range. Drinking water concentration exceedances above the SWRCB notification levels and response levels were also more concentrated in Southern California compared to the rest of the state (Supplementary Fig. S7, Supplementary Table S7).
Association between PFAS in drinking water and serum
Binary categorization
Participants living in water system service areas with any PFHxS detections had 31.9% higher serum PFHxS levels (95% CI: 11.1%, 56.6%) compared to participants living in water system service areas without PFHxS drinking water detections (Fig. 1, Supplementary Table S8). Participants living in water system service areas with PFOA, PFOS, PFNA, PFDeA, or ∑5 PFAS detections did not have statistically significant differences in serum concentrations [PFOA: −3.4% (95% CI: −15.9%, 11.0%); PFOS: 5.4% (95% CI: −11.9%, 26.0%); PFNA: 4.1% (95% CI: −12.7%, 24.1%); PFDeA: 0.2% (95% CI: −17.6%, 21.9%); ∑5 PFAS: 5.6% (95% CI: −8.5%, 21.8%)] compared to participants living in water system service areas without detections (Supplementary Table S8).
∑5 PFAS includes PFHxS, PFOA, PFOS, PFDeA, and PFNA. Due to one participant missing serum PFOS results and one water system missing PFDeA results, the PFOS model includes 562 participants and the ∑5 PFAS model includes 555 participants. Multivariable linear regression models were run separately for each analyte and adjusted for age, sex, parity, race/ethnicity, education, income, and nativity.
Three-tiered categorization
Participants whose water systems had PFHxS detected in over 50% of sampling locations had 64.0% higher serum PFHxS levels compared to participants residing in water systems without detections (95% CI: 32.4%, 103.3%) (Fig. 2, Supplementary Table S8). The adjusted percent change for the remaining exposure categories and models varied from −11.7% to 15.2% but were not statistically significant.
∑5 PFAS includes PFHxS, PFOA, PFOS, PFDeA, and PFNA. Due to one participant missing serum PFOS results and one water system missing PFDeA results, the PFOS model includes 562 participants and the ∑5 PFAS model includes 555 participants. Multivariable linear regression models were run separately for each analyte and adjusted for age, sex, parity, race/ethnicity, education, income, and nativity.
Subgroup of participants matched to water systems with post-treatment drinking water sampling
For a third analysis, we included only participants matched to water systems with post-treatment drinking water sampling (235 participants whose water came from 29 water systems). The adjusted percent change in serum PFAS concentrations were higher and statistically significant for all four PFAS models (Fig. 3, Supplementary Table S8). PFHxS had the largest effect size, with participants living in water system service areas with detections having 79.9% higher serum levels compared to participants without detections (95% CI: 38.3%, 133.9%), followed by ∑5 PFAS at 42.0% (95% CI: 14.5%, 76.1%), PFOS at 31.2% (95% CI: 0.7%, 70.9%), and PFOA at 30.4% (95% CI: 5.5%, 61.2%).
∑5 PFAS includes PFHxS, PFOA, PFOS, PFDeA, and PFNA. Due to one participant missing serum PFOS results and one water system missing PFDeA results, the PFOS model includes 234 participants and the ∑5 PFAS model includes 227 participants. Multivariable linear regression models were run separately for each analyte and adjusted for age, sex, parity, race/ethnicity, education, income, and nativity.
Sensitivity analyses
Stratification by primary drinking water type
Three hundred and two participants reported drinking primarily tap water, and 258 participants reported primarily drinking bottled or other water types (Supplementary Fig. S8). Serum concentrations were generally higher among the participants who primarily drank tap water compared to participants who primarily drank bottled or other water types (Supplementary Table S9). The adjusted percent change in PFHxS serum levels comparing participants whose water had detections vs no detections was 10.8% higher for participants who drank tap water (adjusted percent change: 41.1%, 95% CI: 14.0%, 74.5%) compared to bottled or other water types (adjusted percent change: 27.3%, 95% CI: −4.1%, 69.1%), and was statistically significant only for participants who drank tap water. Although not statistically significant, the effect estimates for PFOS and ∑5 PFAS were about 20% higher for bottled or other water drinkers compared to tap water drinkers.
Incorporating purchased water data
Since water systems often blend water purchased from wholesale or other water systems before distributing water to customers, the quality of purchased water might impact PFAS exposures. We examined whether incorporating 2019 water purchasing data impacted our results (Supplementary Fig. S9). The adjusted percent changes in serum PFAS were similar to the primary model.
Discussion
In this large, geographically diverse general population of Southern California adults without remarkably high serum PFAS concentrations relative to national averages, we found significant associations between drinking water PFAS detections and serum concentrations. Serum PFHxS concentrations were higher among participants living in water system service areas with detected PFAS, and when water systems had a higher detection frequency. When restricted to post-treatment PFAS drinking water data, serum levels of all modeled PFAS (PFHxS, PFOA, PFOS, and ∑5 PFAS) were also higher and had larger effect sizes.
Serum PFAS concentrations found in this study were lower than national levels and also considerably lower than studies of populations with major PFAS contamination of their drinking water. When comparing to reports from areas in the U.S. and Europe with high level drinking water contamination due to fluorochemical manufacturing or military AFFF use, the geometric mean and median serum PFAS concentrations found in this study were up to one thousand-fold lower, suggesting that the CARE population overall was not highly exposed [2, 25,26,27,28,29,30,31, 43,44,45,46]. However, according to 2022 PFAS clinical follow-up guidance from the National Academies of Sciences, Engineering, and Medicine (NASEM), 86% of CARE participants still had serum concentrations that represent a potential for adverse health effects (the sum of Me-PFOSA-AcOH, PFHxS, PFOA, PFDeA, PFUnDA, PFOS, and PFNA ≥ 2 ng/ml) and for which exposure reduction is encouraged, especially in sensitive populations (Supplementary Table S10) [47].
Drinking water concentrations from the SWRCB’s PFAS monitoring data were mostly below the CCRDL (up to 51% below the CCRDL by analyte) or in the low parts per trillion range when detected. The SWRCB monitored for PFAS at raw source wells, allowing for identification of the contamination origin so targeted mitigation efforts can be applied. Comparison of raw source well drinking water concentrations from the current study to other studies can be challenging due to other reports relying upon data from finished water samples. Nonetheless, detectable drinking water concentrations found in this study were approximately similar in magnitude to drinking water concentrations from other general population studies [48, 49] and up to one hundred-fold lower than drinking water concentrations impacted by major PFAS sources such as highly exposed communities near fluoropolymer production plants or military facilities [25, 27, 28, 31].
We found that participants living in water system service areas with PFHxS detections had higher serum concentrations compared to participants living in water system service areas with no detections in all of our models, while results were not consistent for PFOA, PFOS, and ∑5 PFAS. The persistent effect for PFHxS may be influenced by the long serum half-life of PFHxS, which is estimated to be 4.7–35 years, compared to 2.1–10.1 for PFOA, 3.3–27 for PFOS, 2.5–4.3 for PFNA, and 4.5–12 for PFDeA [2, 43, 50]. Other literature investigating the contribution of drinking water PFAS to blood PFAS levels in the general population have also found significant associations for PFHxS. Pennoyer et al. 2025, also utilizing CARE study biomonitoring data, demonstrated associations between UCMR 3 detections and serum PFHxS and PFOA [51], and Boronow et al. 2019, in a California cohort from the Child Health and Development Studies, found associations between UCMR 3 detections and PFHxS, PFOA, and PFNA [52]. Li et al. 2024, focusing on overweight or obese primarily Hispanic children and young adults aged 8–23 in eastern Los Angeles, California utilized both SWRCB monitoring data and UCMR 3 data [53]. Although PFOS was missing from their SWRCB PFAS monitoring analysis, they similarly observed significant associations between SWRCB PFHxS detections and plasma PFAS, as well as UCMR 3 PFOA and PFOS detections and plasma PFAS. Other legacy PFAS observed in drinking water have also been reported to contribute to serum in general populations: Hurley et al. 2016 found associations for PFOA and PFOS in the California Teacher’s Study [49] and Hu et al. 2019 for PFOA and PFNA in the Nurses’ Health Study [48]. However, these studies utilized UCMR 3 data with high method detection limits or tap water sampled several decades prior to the current study.
We found weaker associations for PFOA, PFOS, and ∑5 PFAS compared to PFHxS, which may be influenced by additional factors. PFOS and PFOA were extensively utilized for many decades and are still frequently detected at higher concentrations than other PFAS compounds across various environmental media. PFOS, PFOA, and PFHxS production in the U.S. and Europe has been phased out since the early 2000’s [54, 55], resulting in declining serum concentrations [56]. NHANES biomonitoring data have demonstrated that the average rate of decrease for serum PFOA and PFOS is higher than for PFHxS (74% and 87% decrease for PFOA and PFOS between the 1999–2000 cycle and the 2017–2020 cycle, compared to 52% for PFHxS) [56, 57]. The historical widespread use of PFOS and PFOA resulting in various exposure sources and shorter half-lives has led to faster declining serum concentrations, which may have obscured the association for PFOS and PFOA found in this analysis. Additionally, while the PFOS precursors Me-PFOSA-AcOH and Et-PFOSA-AcOH had no or very low detection frequencies in this study, we cannot rule out the potential impact of other PFAS precursors that can biotransform to terminal PFAS, which may have further influenced the observed associations [58, 59].
All modelled PFAS analytes in drinking water were significantly associated with serum levels when restricting to testing of post-treatment drinking water. While we did not have data on the type of drinking water treatment applied, post-treatment drinking water represents water that has likely been combined from upstream raw water sources and is more similar to finished water than source wells. Although this portion of our analysis included less than half of the original sample size (235 out of 563) as well as less than half of the total water systems (29 out of 70), these results suggest that even with the lower drinking water PFAS concentrations seen in this study, there is a strong association between PFAS in drinking water and serum PFAS concentrations. Comparison of results from post-treatment water alone to the results from both source and treated water illustrate both the utility and caveats of source well data for the purposes of assessing contribution to exposure. The relative strength of associations followed a consistent pattern between both methods, and while source well data identified the strongest PFHxS association, its effectiveness in detecting weaker associations was limited.
Among participants who primarily drank tap water, we found a larger difference in serum PFHxS concentrations compared to participants who drank primarily bottled or other water types. This stronger effect suggests that the contribution of PFHxS drinking water detections on serum PFHxS is likely associated with tap water distributed by public water systems. Interestingly, although overall serum PFAS concentrations were lower among participants who primarily drank bottled or other water types, the adjusted percent change in serum PFOS and ∑5 PFAS were about 20% higher. Limited data exist on PFAS composition in bottled water in the U.S. Bottled water can come from various sources including municipal water systems [60]. While the few bottled water reports show no PFOA or PFOS concentrations above EPA MCLs, Chow et al. 2025 found four bottled water products (4%) with ∑32 PFAS > 4 ng/L, and Consumer Reports testing from 2020 found three noncarbonated or carbonated products (6%) with ∑30 PFAS > 4 ng/L [60,61,62]. Other water-based products, such as tea and beer, may also contribute to PFAS exposure [63, 64]. Importantly, since even bottled water drinkers showed evidence of elevated PFAS, exposures from water-based products, occasional tap water consumption, and other uses should be considered when trying to reduce exposures.
The PFAS found in drinking water are influenced by the particular PFAS profiles from contamination sources. While California does not have industrial manufacturing of PFAS, drinking water contamination can occur from industrial sources that use AFFF including airports, military facilities, refineries, and bulk fuel terminals [65, 66]. From 2019, the SWRCB also monitored industrial site groundwater likely to be PFAS contamination sources including airports, landfills, chrome plating facilities, wastewater treatment plants, bulk fuel terminals, and refineries. This industry monitoring indicated that groundwater from airports and bulk fuel terminals/refineries had the highest concentrations of PFAS analytes, with nearly 20% of samples having concentrations above 1000 ng/L and 30% of samples in the 100–1000 ng/L range (Supplementary Fig. S10). AFFF point sources have been shown to contribute to groundwater contamination even for point sources that have been inactive for decades, though this can be dependent on PFAS chain length and head group, distance to prominent contamination source, well depth, and hydrogeological conditions [24, 67]. Up until the early 2000’s, U.S. manufactured AFFF was largely dominated by perfluoroalkyl sulfonates such as PFHxS and PFOS, while some legacy fluorotelomer AFFF manufactured until the early 2010’s also contained polyfluorinated precursors that break down into PFOA [68]. Consistent with these reports, communities highly exposed to AFFF have documented elevated PFHxS and PFOS in both drinking water and serum [29, 31]. The widespread historical industrial and military AFFF use across California may have contributed to the PFHxS association found in this analysis.
Limitations
There are several limitations to this study. Drinking water detections from raw water sources do not necessarily represent concentrations delivered to households, leading to potential exposure misclassification. To address this, we did not use concentration values but classified detection patterns into binary and three-tiered categorization. Another issue was that the drinking water data employed measured PFAS levels at concurrent or somewhat later dates to the serum samples. However, detections were likely to be indicative of earlier presence of PFAS in source wells. In the future, drinking water sampling data of finished water may allow for more precise estimates of the contribution of drinking water PFAS concentration to serum concentrations, but will be harder to link with historical serum data since water systems may have already begun treating or reducing use of particular source wells. EPA’s UCMR 5 monitoring, for which sampling had not been completed at the time of this publication, has started sampling finished water from water systems throughout the U.S., and will be a crucial future data source. Despite these limitations, higher detection frequencies of PFHxS as well as higher PFHxS intake in tap water was more strongly associated with elevated PFHxS serum levels, supporting a dose-dependent relationship between drinking water PFAS concentrations and overall exposure.
While drinking water is an established exposure pathway, this study did not address other routes of exposure, such as diet and indoor dust, which can also contribute to overall PFAS body burden [31]. The covariates selected in our DAG were included to reduce confounding by these alternative exposure pathways. Additionally, Pennoyer et al. 2025, also focusing on the CARE cohort, found minimal evidence of confounding between drinking water and diet in this large, geographically diverse population [51].
Another limitation to the data was the particular focus of the SWRCB’s 2019–2022 PFAS monitoring orders, which targeted wells in areas with previous PFAS drinking water detections, or near potential PFAS contamination sources. Drinking water sampling was not universal across water systems, and groundwater wells have been shown to be more likely to have PFAS detections compared to surface water [65]. Sampling of treated drinking water was more likely among larger water systems that are both more likely to have PFAS detections [69] and have greater resources to do additional testing. Therefore, the drinking water sampling was not inclusive of all public water systems in Southern California, introducing selection bias of water systems and wells chosen for analysis that may be more likely to have PFAS contamination. This selection bias has the potential to overestimate the association between drinking water PFAS detections and serum concentrations. These findings nonetheless point to an important association between drinking water contamination and overall PFAS exposure even among a population without remarkably high serum PFAS concentrations.
Conclusions
Identifying PFAS exposure sources and their source contribution is important for exposure mitigation strategies and informing public health policies. Our results indicate that PFAS drinking water contamination may be a significant contributor to serum concentrations, especially for PFHxS, even among communities without high level contamination from industrial manufacturing. Addressing drinking water treatment for PFAS contamination can be resource-intensive and presents significant challenges for water systems. Federal regulations were established for six PFAS in April 2024. However, in May 2025, the regulations for four of these PFAS were rescinded, and the compliance timeline for PFOA and PFOS MCLs was delayed. Our results support enforcement of regulations to reduce PFAS body burden in the general population.
Looking forward, data not yet available from current PFAS sampling initiatives enacted after the initiation of this study will be critical in addressing existing data gaps. Since 2022, the SWRCB has expanded upon the 2019–2022 investigative orders to sample more wells in the vicinity of contamination sources and has also implemented a state-funded PFAS monitoring program that includes nearly 4000 wells in water systems serving disadvantaged communities throughout the state, with state funding available to assist water systems with few resources in addressing PFAS drinking water contamination [70, 71]. Data collected from these SWRCB efforts and EPA’s UCMR 5 will be critical for deepening our understanding of drinking water PFAS contamination and the relative source contribution in the general population. Future studies should also investigate other PFAS analytes beyond legacy and long-chain compounds, consider other exposure sources including diet, consumer products, and indoor dust, and include drinking water sampling from rural areas.
Data availability
Complete data supporting the findings of this study are not publicly available due to IRB restrictions prohibiting sharing of information that could compromise research participant privacy. Some de-identified data may be available upon request.
References
Buck RC, Franklin J, Berger U, Conder JM, Cousins IT, de Voogt P, et al. Perfluoroalkyl and polyfluoroalkyl substances in the environment: terminology, classification, and origins. Integr Environ Assess Manag. 2011;7:513–41.
Li Y, Fletcher T, Mucs D, Scott K, Lindh CH, Tallving P, et al. Half-lives of PFOS, PFHxS and PFOA after end of exposure to contaminated drinking water. Occup Environ Med. 2018;75:46–51.
Kwiatkowski CF, Andrews DQ, Birnbaum LS, Bruton TA, DeWitt JC, Knappe DRU, et al. Scientific basis for managing PFAS as a chemical class. Environ Sci Technol Lett. 2020;7:532–43.
Domingo JL, Nadal M. Human exposure to per- and polyfluoroalkyl substances (PFAS) through drinking water: a review of the recent scientific literature. Environ Res. 2019;177:108648.
Kato K, Wong LY, Jia LT, Kuklenyik Z, Calafat AM. Trends in exposure to polyfluoroalkyl chemicals in the U.S. Population: 1999-2008. Environ Sci Technol. 2011;45:8037–45.
Bjerregaard-Olesen C, Bossi R, Liew Z, Long M, Bech BH, Olsen J, et al. Maternal serum concentrations of perfluoroalkyl acids in five international birth cohorts. Int J Hyg Environ Health. 2017;220:86–93.
International Pollutants Elimination Network (IPEN). PFAS Pollution across the Middle East and Asia. 2019. Available from: https://ipen.org/documents/pfas-pollution-across-middle-east-and-asia.
Fromme H, Tittlemier SA, Völkel W, Wilhelm M, Twardella D. Perfluorinated compounds–exposure assessment for the general population in Western countries. Int J Hyg Environ Health. 2009;212:239–70.
Gustafsson Å, Bergman Å, Weiss JM. Estimated daily intake of per- and polyfluoroalkyl substances related to different particle size fractions of house dust. Chemosphere. 2022;303:135061.
Dewapriya P, Chadwick L, Gorji SG, Schulze B, Valsecchi S, Samanipour S, et al. Per- and polyfluoroalkyl substances (PFAS) in consumer products: current knowledge and research gaps. J Hazard Mater Lett. 2023;4:100086.
Fraser AJ, Webster TF, Watkins DJ, Nelson JW, Stapleton HM, Calafat AM, et al. Polyfluorinated compounds in serum linked to indoor air in office environments. Environ Sci Technol. 2012;46:1209–15.
Haug LS, Huber S, Schlabach M, Becher G, Thomsen C. Investigation on per- and polyfluorinated compounds in paired samples of house dust and indoor air from Norwegian homes. Environ Sci Technol. 2011;45:7991–8.
Poothong S, Papadopoulou E, Padilla-Sánchez JA, Thomsen C, Haug LS. Multiple pathways of human exposure to poly- and perfluoroalkyl substances (PFASs): from external exposure to human blood. Environ Int. 2020;134:105244.
Jensen AA, Leffers H. Emerging endocrine disrupters: perfluoroalkylated substances. Int J Androl. 2008;31:161–9.
Looker C, Luster MI, Calafat AM, Johnson VJ, Burleson GR, Burleson FG, et al. Influenza vaccine response in adults exposed to perfluorooctanoate and perfluorooctanesulfonate. Toxicol Sci. 2014;138:76–88.
Stein CR, McGovern KJ, Pajak AM, Maglione PJ, Wolff MS. Perfluoroalkyl and polyfluoroalkyl substances and indicators of immune function in children aged 12-19 y: National Health and Nutrition Examination Survey. Pediatr Res. 2016;79:348–57.
Padula AM, Ning X, Bakre S, Barrett ES, Bastain T, Bennett DH, et al. Birth outcomes in relation to prenatal exposure to per- and polyfluoroalkyl substances and stress in the environmental influences on child health outcomes (ECHO) program. Environ Health Perspect. 2023;131:37006.
Vieira VM, Hoffman K, Shin HM, Weinberg JM, Webster TF, Fletcher T. Perfluorooctanoic acid exposure and cancer outcomes in a contaminated community: a geographic analysis. Environ Health Perspect. 2013;121:318–23.
Barry V, Winquist A, Steenland K. Perfluorooctanoic acid (PFOA) exposures and incident cancers among adults living near a chemical plant. Environ Health Perspect. 2013;121:1313–8.
Shearer JJ, Callahan CL, Calafat AM, Huang WY, Jones RR, Sabbisetti VS, et al. Serum concentrations of per- and polyfluoroalkyl substances and risk of renal cell carcinoma. J Natl Cancer Inst. 2021;113:580–7.
Mastrantonio M, Bai E, Uccelli R, Cordiano V, Screpanti A, Crosignani P. Drinking water contamination from perfluoroalkyl substances (PFAS): an ecological mortality study in the Veneto Region, Italy. Eur J Public Health. 2018;28:180–5.
Gebbink WA, van Asseldonk L, van Leeuwen SPJ. Presence of emerging per- and polyfluoroalkyl substances (PFASs) in river and drinking water near a fluorochemical production plant in the Netherlands. Environ Sci Technol. 2017;51:11057–65.
Milley SA, Koch I, Fortin P, Archer J, Reynolds D, Weber KP. Estimating the number of airports potentially contaminated with perfluoroalkyl and polyfluoroalkyl substances from aqueous film forming foam: a Canadian example. J Environ Manag. 2018;222:122–31.
Ruyle BJ, Thackray CP, Butt CM, LeBlanc DR, Tokranov AK, Vecitis CD, et al. Centurial persistence of forever chemicals at military fire training sites. Environ Sci Technol. 2023;57:8096–106.
Hoffman K, Webster TF, Bartell SM, Weisskopf MG, Fletcher T, Vieira VM. Private drinking water wells as a source of exposure to perfluorooctanoic acid (PFOA) in communities surrounding a fluoropolymer production facility. Environ Health Perspect. 2011;119:92–7.
Hall SM, Zhang S, Tait GH, Hoffman K, Collier DN, Hoppin JA, et al. PFAS levels in paired drinking water and serum samples collected from an exposed community in Central North Carolina. Science Total Environ. 2023;895:165091.
McDonough CA, Choyke S, Barton KE, Mass S, Starling AP, Adgate JL, et al. Unsaturated PFOS and Other PFASs in human serum and drinking water from an AFFF-impacted community. Environ Sci Technol. 2021;55:8139–48.
Johanson G, Gyllenhammar I, Ekstrand C, Pyko A, Xu Y, Li Y, et al. Quantitative relationships of perfluoroalkyl acids in drinking water associated with serum concentrations above background in adults living near contamination hotspots in Sweden. Environ Res. 2023;219:115024.
Barton KE, Starling AP, Higgins CP, McDonough CA, Calafat AM, Adgate JL. Sociodemographic and behavioral determinants of serum concentrations of per- and polyfluoroalkyl substances in a community highly exposed to aqueous film-forming foam contaminants in drinking water. Int J Hyg Environ Health. 2020;223:256–66.
Pitter G, Da Re F, Canova C, Barbieri G, Zare Jeddi M, Daprà F, et al. Serum levels of perfluoroalkyl substances (PFAS) in adolescents and young adults exposed to contaminated drinking water in the Veneto Region, Italy: a cross-sectional study based on a health surveillance program. Environ Health Perspect. 2020;128:27007.
Agency for Toxic Substances and Disease Registry. PFAS Exposure Assessments Final Report. Agency for Toxic Substances and Disease Registry; 2022.
US EPA. 40 CFR Parts 141 and 142 PFAS National Primary Drinking Water Regulation. 2024. Available from: https://www.govinfo.gov/content/pkg/FR-2024-04-26/pdf/2024-07773.pdf.
US EPA. EPA Announces It Will Keep Maximum Contaminant Levels for PFOA, PFOS. 2025. Available from: https://www.epa.gov/newsreleases/epa-announces-it-will-keep-maximum-contaminant-levels-pfoa-pfos.
State Water Resources Control Board (SWRCB). Drinking Water Notification Levels. 2024. Available from: https://www.waterboards.ca.gov/drinking_water/certlic/drinkingwater/NotificationLevels.html.
Attfield K, Kauffman D, D’Amico A, Dobraca D, Andrews F, Wu N. The California Regional Exposure (CARE) Study, 2018 – 2020. Richmond, CA: California Department of Public Health, Environmental Health Investigations Branch, Biomonitoring California; 2023.
Wang M, Park JS, Petreas M. Temporal changes in the levels of perfluorinated compounds in California women’s serum over the past 50 years. Environ Sci Technol. 2011;45:7510–6.
Division of Drinking Water State Water Resources Control Board. Order DW 2020-0003-DDW General Order Requiring Monitoring For Per and Polyfluoroalkyl Substances. 2020. Available from: https://www.waterboards.ca.gov/board_decisions/adopted_orders/drinking_water/2020/pfas_go_2020_0003_ddw/pfas_go_2020_0003_ddw.pdf.
Shoemaker J, Tettenhorst D. Method 537.1: Determination of Selected Per- and Polyfluorinated Alkyl Substances in Drinking Water by Solid Phase Extraction and Liquid Chromatography/Tandem Mass Spectrometry (LC/MS/MS). Washington, DC: U.S. Environmental Protection Agency, Office of Research and Development, National Center for Environmental Assessment; 2018.
Division of Drinking Water State Water Resources Control Board. Water Quality Database Files, Water Quality Analyses Data and Download Page. 2024. Available from: https://www.waterboards.ca.gov/drinking_water/certlic/drinkingwater/EDTlibrary.html.
Division of Drinking Water State Water Resources Control Board. California Drinking Water System Area Boundaries. 2024. Available from: https://gispublic.waterboards.ca.gov/portal/home/item.html?id=fbba842bf134497c9d611ad506ec48cc.
Los Angeles Department of Water and Power. Drinking Water Quality Report 2020. 2020. Available from: https://www.ladwp.com/sites/default/files/documents/2020_Drinking_Water_Quality_Report.pdf.
August L, Bangia K, Plummer L, Prasad S, Ranjbar K, Slocombe A, et al. CalEnviroScreen 4.0. Office of Environmental Health Hazard Assessment; 2021.
Olsen GW, Burris JM, Ehresman DJ, Froehlich JW, Seacat AM, Butenhoff JL, et al. Half-life of serum elimination of perfluorooctanesulfonate,perfluorohexanesulfonate, and perfluorooctanoate in retired fluorochemical production workers. Environ Health Perspect. 2007;115:1298–305.
Steenland K, Jin C, MacNeil J, Lally C, Ducatman A, Vieira V, et al. Predictors of PFOA levels in a community surrounding a chemical plant. Environ Health Perspect. 2009;117:1083–8.
Bartell SM, Calafat AM, Lyu C, Kato K, Ryan PB, Steenland K. Rate of decline in serum PFOA concentrations after granular activated carbon filtration at two public water systems in Ohio and West Virginia. Environ Health Perspect. 2010;118:222–8.
Cuffney M, Wilkie AA, Kotlarz N, Knappe D, Lea CS, Collier DN, et al. Factors associated with per- and polyfluoroalkyl substances (PFAS) serum concentrations in residents of New Hanover County, North Carolina: the GenX exposure study. Environ Res. 2023;237:117020.
National Academies of Sciences Engineering and Medicine. Guidance on PFAS exposure, testing, and clinical follow-up. Washington, DC: The National Academies Press; 2022. Report No.: 978-0-309-48244-8.
Hu XC, Tokranov AK, Liddie J, Zhang X, Grandjean P, Hart JE, et al. Tap water contributions to plasma concentrations of poly- and perfluoroalkyl substances (PFAS) in a nationwide prospective cohort of U.S. women. Environ Health Perspect. 2019;127:67006.
Hurley S, Houtz E, Goldberg D, Wang M, Park J-S, Nelson DO, et al. Preliminary associations between the detection of perfluoroalkyl acids (PFAAs) in drinking water and serum concentrations in a sample of California women. Environ Sci Technol Lett. 2016;3:264–9.
Agency for Toxic Substances and Disease Registry. Toxicological Profile for Perfluoroalkyls. 2021. Available from https://www.atsdr.cdc.gov/toxprofiles/tp200.pdf.
Pennoyer EH, Fillman T, Heiger-Bernays W, Attfield KR, Baek HG, Gao S, et al. Exposure to legacy per- and polyfluoroalkyl substances from diet and drinking water in California Adults, 2018-2020. Environ Sci Technol. 2025;59:9896–906.
Boronow KE, Brody JG, Schaider LA, Peaslee GF, Havas L, Cohn BA. Serum concentrations of PFASs and exposure-related behaviors in African American and non-Hispanic white women. J Expo Sci Environ Epidemiol. 2019;29:206–17.
Li S, Goodrich JA, Costello E, Walker DI, Cardenas-Iniguez C, Chen JC, et al. Examining disparities in PFAS plasma concentrations: Impact of drinking water contamination, food access, proximity to industrial facilities and superfund sites. Environ Res. 2024;264:120370.
US EPA. EPA and 3M announce phase out of PFOS. 2000. Available from: https://www.epa.gov/archive/epapages/newsroom_archive/newsreleases/33aa946e6cb11f35852568e1005246b4.html.
US EPA. Fact Sheet: 2010/2015 PFOA Stewardship Program. 2024. Available from: https://www.epa.gov/assessing-and-managing-chemicals-under-tsca/fact-sheet-20102015-pfoa-stewardship-program.
National Center for Environmental Health Center for Disease Control and Prevention (US CDC). National Report on Human Exposure to Environmental Chemicals. 2022.
Botelho JC, Kato K, Wong LY, Calafat AM. Per- and polyfluoroalkyl substances (PFAS) exposure in the U.S. population: NHANES 1999-March 2020. Environ Res. 2025;270:120916.
Gebbink WA, Berger U, Cousins IT. Estimating human exposure to PFOS isomers and PFCA homologues: the relative importance of direct and indirect (precursor) exposure. Environ Int. 2015;74:160–9.
Vestergren R, Cousins IT, Trudel D, Wormuth M, Scheringer M. Estimating the contribution of precursor compounds in consumer exposure to PFOS and PFOA. Chemosphere. 2008;73:1617–24.
Chow SJ, Ojeda N, Jacangelo JG, Schwab KJ. Detection of ultrashort-chain and other per- and polyfluoroalkyl substances (PFAS) in U.S. bottled water. Water Res. 2021;201:117292.
Felton R. What’s really in your bottled water? Consumer Reports; 2020. Available from: https://www.consumerreports.org/water-quality/whats-really-in-your-bottled-water-a5361150329/.
US Food and Drug Administration. FDA Shares Testing Results for PFAS in Bottled Water. 2025. Available from: https://www.fda.gov/food/hfp-constituent-updates/fda-shares-testing-results-pfas-bottled-water#:~:text=Two%20of%20the%20PFAS%20detected,PFAS%20in%20bottled%20water%20samples.
Hoponick Redmon J, DeLuca NM, Thorp E, Liyanapatirana C, Allen L, Kondash AJ. Hold my beer: the linkage between municipal water and brewing location on PFAS in popular beverages. Environ Sci Technol. 2025;59:8368–79.
Hampson HE, Costello E, Walker DI, Wang H, Baumert BO, Valvi D, et al. Associations of dietary intake and longitudinal measures of per- and polyfluoroalkyl substances (PFAS) in predominantly Hispanic young Adults: a multicohort study. Environment Int. 2024;185:108454.
Hu XC, Andrews DQ, Lindstrom AB, Bruton TA, Schaider LA, Grandjean P, et al. Detection of poly- and perfluoroalkyl substances (PFASs) in U.S. drinking water linked to industrial sites, military fire training areas, and wastewater treatment plants. Environ Sci Technol Lett. 2016;3:344–50.
Prevedouros K, Cousins IT, Buck RC, Korzeniowski SH. Sources, fate and transport of perfluorocarboxylates. Environ Sci Technol. 2006;40:32–44.
Weber AK, Barber LB, LeBlanc DR, Sunderland EM, Vecitis CD. Geochemical and hydrologic factors controlling subsurface transport of poly- and perfluoroalkyl substances, Cape Cod, Massachusetts. Environ Sci Technol. 2017;51:4269–79.
Place BJ, Field JA. Identification of novel fluorochemicals in aqueous film-forming foams used by the US military. Environ Sci Technol. 2012;46:7120–7.
Guelfo JL, Adamson DT. Evaluation of a national data set for insights into sources, composition, and concentrations of per- and polyfluoroalkyl substances (PFASs) in U.S. drinking water. Environ Pollut. 2018;236:505–13.
Division of Drinking Water State Water Resources Control Board. Order DW 2022-0001-DDW general order requiring monitoring for per and polyfluoroalkyl substances 2022. Available from: https://www.waterboards.ca.gov/drinking_water/certlic/drinkingwater/pfas_ddw_general_order/docs/pfas_go_2022-0001-DDW.pdf.
Division of Drinking Water State Water Resources Control Board. Order DW 2024-0002-DDW general order requiring monitoring for per and polufluoroalkyl substances. 2024. Available from: https://www.waterboards.ca.gov/drinking_water/certlic/drinkingwater/pfas_ddw_general_order/docs/pfas-general-order-dw-2024-0002-ddw.pdf.
Acknowledgements
We would like to thank the CARE study participants and field work staff. We are appreciative of the Los Angeles Department of Water and Power staff for their assistance in providing data on source well assignment to local service areas. We are grateful for Komal Bangia of OEHHA for providing CalEnviroScreen water purchasing data. We are thankful for Biomonitoring California staff in OEHHA, CDPH, and the Department of Toxic Substances Control for ongoing helpful feedback for this project. The findings and conclusions in this article are those of the author(s) and do not necessarily represent the views or opinions of the California Department of Public Health or the California Health and Human Services Agency.
Funding
The California Regional Exposure Study was supported by the Centers for Disease Control and Prevention Cooperative Agreement U88EH001148.
Author information
Authors and Affiliations
Contributions
Toki Fillman contributed to data curation, methodology, formal analysis, visualization, writing – original draft, and writing – review & editing. Scott Coffin contributed to conceptualization, data curation, methodology, and writing – review & editing. Brandon Ta contributed to the conceptualization, data curation, methodology, and writing – review & editing. Emily Beglarian contributed to the visualization, writing – original draft, and writing – review & editing. Wendy Linck contributed to the data curation, visualization, and writing – review & editing. Hyoung-Gee Baek contributed to investigation, validation, methodology, and writing – review & editing. Songmei Gao contributed to investigation, validation, methodology, and writing – review & editing. Sabrina Smith contributed to investigation, validation, methodology, and writing – review & editing. June-Soo Park contributed to investigation, validation, methodology, and writing – review & editing. Nerissa Wu contributed to investigation, funding acquisition, resources, and writing – review & editing. Kathleen Attfield contributed to supervision, conceptualization, data curation, methodology, and writing – review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All study materials and activities were approved by the California Health & Human Services Agency Committee for the Protection of Human Subjects. Informed consent was obtained from all subjects involved in the CARE studies. All methods were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Fillman, T., Coffin, S., Ta, B. et al. Associations between PFAS in public water system drinking water and serum among Southern California adults. J Expo Sci Environ Epidemiol 36, 311–321 (2026). https://doi.org/10.1038/s41370-025-00817-8
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41370-025-00817-8