Abstract
The need for spatially-confined electrical stimulation is growing in biomedical applications, for example intracortical stimulation and retinal implant, for enhancement of stimulating resolution. Local grounding techniques have been widely explored to suppress undesired current spread. However, in conventional microneedle arrays like the Utah array, grounding is typically achieved by assigning neighboring electrodes as ground or employing grounding wall around stimulating electrode, which compromises spatial efficiency. In this work, we introduce, for the first time, a bipolar microneedle electrode array (BMEA) that integrates two electrically-independent electrodes within each three-dimensional microneedle structure. The microtip electrode, located at the apex of the microneedle, delivers electrical stimulation, while the local ground electrode, embedded on the sidewall below the microtip, serves to locally confine the spread of current. COMSOL Multiphysics simulations and ex vivo experiments using isolated mouse retina demonstrated that activating the local ground electrode effectively restricts current diffusion, enabling more focused and localized stimulation. This approach offers a compact and efficient solution for focal electrical stimulation with enhanced spatial resolution, providing a promising platform for advanced neural interfacing systems in various biomedical fields.

Similar content being viewed by others
Introduction
Electrical stimulation has become a foundational technique in modern biomedical engineering, playing a crucial role in the development of neural prosthetics and neuromodulation strategies. Clinical applications include intracortical neural interfaces for functional restoration1,2, cochlear implants for hearing rehabilitation3,4, and retinal prostheses that restore visual perception in patients with photoreceptor degeneration5,6. These systems function by delivering precisely controlled electrical pulses through implanted multi-electrode arrays (MEAs), which are often engineered with three-dimensional architectures that physically penetrate neural tissue to access specific target regions. The applied current modulates the activity of nearby excitable cells, enabling artificial modulation or restoration of sensory functions. Achieving high-resolution stimulation is one of the central goals in advancing the performance of neural prosthetics. Toward this end, significant efforts have been focused on increasing the spatial density of electrodes7, optimizing electrode geometry8, and incorporating novel materials with enhanced electrical or mechanical properties6,9. These strategies aim to improve the selectivity and focality of stimulation, thereby ultimately enhancing perceptual outcomes for prosthetic users.
Despite substantial progress in electrode design and material innovation, improving the spatial resolution of neural stimulation remains a critical challenge. A primary limiting factor is the unintended spread of current through biological tissue, which results in the inadvertent activation of neurons in non-target regions. This effect significantly compromises the precision of stimulation and poses a substantial barrier to enhancing the functional outcomes of neural prosthetic systems. To mitigate this issue, increasing attention has been directed toward strategies that confine electrical fields within a localized region. These approaches range from electrode design optimization to emerging grounding-free methods using ferroelectric materials10. Among these, incorporating a local ground (return) electrode placed in close proximity to the stimulation electrode has emerged as an effective approach for shaping and restricting the current distribution11,12,13. This configuration enables more precise control of the electric field and reduces unintended stimulation of adjacent tissue regions. Numerous studies across diverse applications such as retinal prostheses have demonstrated that local grounding significantly enhances the focality of stimulation11,12,13. Consequently, local ground electrodes have become a promising key design feature in the development of neural interfaces for modulating neural activities in precisely targeted spatial regions.
To enable spatially-confined stimulation through local grounding, an additional ground electrode must be positioned in close proximity to the stimulation site. For in-plane neural interfaces such as planar MEAs and Michigan-type neural probes, the local ground integration could be easily achieved with minimal design complexity because planar microfabrication techniques allow for multiple electrodes to be patterned on the same plane14,15. In contrast, however, out-of-plane neural interfaces such as the Utah array present substantial fabrication challenges in implementing local grounding. Prior efforts have typically addressed this fabrication issue by designating adjacent microneedle electrodes as ground16 or by creating another conductive wall structure around each stimulation site17. While effective in reducing current spread, these methods compromise spatial efficiency—an increasingly critical tradeoff as the demand grows for ultra-high-density out-of-plane neural interfaces7 for applications requiring large-area brain stimulation or implantation into anatomically complex or curved tissues such as the retina or cochlea18.
In this study, we introduce a bipolar microneedle electrode array (BMEA) in which each individual microneedle incorporates two distinct electrodes: a stimulation microelectrode at the tip and a local ground electrode embedded along the sidewall beneath it. This vertically positioned bipolar configuration enables localized current confinement without sacrificing spatial efficiency, thereby overcoming the limitations of conventional microneedle electrode designs that rely on neighboring electrodes for grounding. To evaluate the efficacy of our design, we conducted Multiphysics simulations, which confirmed that activating the integrated local ground electrode effectively suppresses undesired current spread. Furthermore, we validated the focal stimulation capability of the fabricated BMEA in ex-vivo experiments using isolated mouse retina—a representative model system for confined neural tissues requiring high-precision stimulation. These results collectively demonstrate the feasibility and potential of the proposed BMEA as a next-generation bioelectronic interface for compact, high-density, and spatially-selective neuromodulation.
Results and discussion
Design of a bipolar microneedle electrode array and system integration
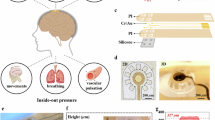
Scanning electron microscopy (SEM) images of the BMEA at key fabrication stages are presented in Fig. 1. For example, Fig. 1a shows the formation of the silicon microneedle structures via isotropic wet etching. The wet etching was performed for 60 min, with optical microscope monitoring at 10-minute intervals. The etching rate was measured to be approximately 1.2 μm/min in the upper region of the silicon pillar, while the lower region exhibited a slower average etching rate of 0.5 μm/min, indicating a more than twofold difference. This differential etching rate resulted in the gradual formation of a tapered microneedle profile.
a A cross-sectional schematic (ai) and a SEM image (aii) after the formation of the microneedle array. b Same as a but after selective exposure of the upper insulation layer. c Same as a but after the final fabrication step. d A magnified view of a single microneedle highlighting the microtip electrode, local ground electrode, and insulation layers (di). Two white dashed lines indicate the microtip electrode and local ground electrode, with two insulation layers are positioned between two electrodes and beneath the local ground electrode. Yellow dashed boxes indicate the locations of cross-sectional images for (dii) and (diii) obtained with FIB etching process. e A microscope image of the fabricated bipolar electrodes showing through-glass vias. f A microscope image of metal pads and bonding wires. See Figures S2 and S3 for additional details. g Averaged impedance-frequency curve from randomly chosen bipolar electrodes (n = 10). Red dot indicates the impedance at 1 kHz which is considered to be crucial for neural signal recordings. Error bars indicate standard deviation
Figure 1b presents a SEM image of the microneedle electrode following the first self-aligned etching process. During spin-coating, the photoresist layer was deposited non-uniformly, resulting in a thinner coating near the microneedle tip (Fig. S1a). Consequently, the photoresist near the tip of each microneedle was removed earlier during reactive ion etching (RIE), selectively exposing the SiO2 upper insulation layer at the tip. Notably, the height of the exposed local ground electrode location on the side of the microneedle decreased proportionally with longer RIE duration.
Figure 1c shows the final fabricated structure of the BMEA. After the second self-aligned etching process, in which the SiO2 upper insulation layer was selectively removed to expose the Au microtip electrode by precisely controlling the etching time16, Parylene-C was, firstly, deposited over the entire surface to form the lower insulation layer. Subsequently, RIE was conducted to selectively remove the Parylene-C at the tip with the optimized condition (Fig. S1b), resulting in the well-defined formation of the microtip electrode, local ground electrode, and two insulation layers (Fig. 1d). Cross-sectional analysis of the fabricated microtip electrode was performed using focused ion beam (FIB) etching. FIB milling, which employs ion beams for material removal, enables high-resolution analysis at nanometer to micrometer scales. However, due to the relatively large vertical dimension of the microtip electrode and its broad structural diameter, the cross-sectional study was conducted by segmenting the device into upper and lower regions, as illustrated in Fig. 1dii and 1diii. To prevent damage during FIB milling, the outermost surfaces of both regions were coated with protective platinum and carbon layers prior to etching. In the upper section near the apex, structural observation revealed a gold microtip electrode conformally deposited on a silicon substrate, consistent with the intended device architecture. In the lower section near the base, a multilayered configuration was identified, consisting of an upper insulation layer (SiO2), a local ground electrode (Cr/Au), and a lower insulation layer (Parylene-C), sequentially formed atop the silicon body. These results confirm the successful fabrication of the bipolar electrode structures with well-defined layer separation in each microneedle.
To evaluate the uniformity of fabricated BMEAs, we measured the dimensions of randomly selected ten microneedle electrodes: the average height of the microneedles was 120.0 ± 4.6 μm. The sidewall exposure lengths of the microneedle electrode, the upper insulation layer, and the local ground electrode were measured to be 32.7 ± 3.1 μm, 20.5 ± 2.2 μm, and 16.9 ± 1.8 μm, respectively. The standard deviations in these dimensions confirm the high structural uniformity achieved across the array. Given that all processes were based on standard CMOS-compatible fabrication techniques, further improvements in dimensional consistency are expected to be attainable through optimization of tool parameters and process control.
Top electrodes, designated for stimulation or recording, were positioned at the tip of each microneedle and connected to the electrode pads through TGVs penetrating the glass substrate (Fig. 1e). Then, individual microtip electrodes were electrically connected to corresponding printed circuit board (PCB) metal pads via wire bonding to the Cr/Au metal pads formed on the backside of the device substrate (Fig. 1f; see also Fig. S2a and S2b). Additionally, two Omnetics connectors were used (Fig. S2c) to interface the PCB signal lines with 16-channel amplifiers for neural signal recording from individual electrodes. The local ground electrode was connected to its designated PCB pad using silver paste, which was further extended to an external ground wire. For both electrochemical characterization and ex vivo retina experiments, a polydimethylsiloxane (PDMS) well was integrated onto the PCB (Fig. S3a). Also, to protect the bonding wires and ensure sufficient spacing from the fluidic environment, a wire bonding guard was installed on the backside of the PCB (Fig. S3b). Average impedance of the microtip electrodes was measured to be 176.0 ± 51.6 kΩ at 1 kHz (Fig. 1g; see Materials and methods for details). The variations in impedance may partially result from differences in the exposed lengths of the microtip electrodes. Also, the impedance variations can be attributed to multiple contributing factors, such as the electrical contact conditions between the BMEA and the PCB, and the spatial alignment of the electrodes during the measurement process. Despite this variability, impedance values below 2 MΩ are generally considered acceptable for neural signal acquisition. The impedance variation can be further reduced, along with impedance itself, by introducing additional layer such as Pt black and carbon nanotube (CNT)19 on top of our gold electrode surface. Taken together, the measured impedance values indicate that the BMEA maintains adequate electrical performance and is suitable for use as a neural interface electrode.
The novelty of our work lies in the unique geometry of the BMEA, but electrode material is also an important factor to be considered in neural interface application. In this study, bare Cr/Au electrodes were utilized for acute electrical stimulation. Gold itself continues to serve as a widely used base material for stimulation electrodes owing to its excellent electrical conductivity, chemical stability, and biocompatibility20. Indeed, several studies have demonstrated the successful use of gold electrodes for stimulation in various tissues, including the retina21 and other neural targets such as limb22,23. This ongoing interest highlights that Au remains a valuable and reliable electrode material for acute testing, particularly when combined with appropriate device design and stimulation protocols. For example, a previous study demonstrated the electroplated Au in a 3D shape can improve the charge injection capacity by ~30% than the 2D Au electrode24. However, for future chronic applications, additional materials (e.g., Pt black, IrOx, TiN, and PEDOT:PSS)25 need to be incorporated on top of gold to improve the electrode–electrolyte interface performance. Also, the magnitude of interfacial electrochemical reactions is strongly influenced by the uncompensated solution resistance (iR drop)26, which increases with the distance between the working and reference (or return) electrodes27. Placing the reference/return closer to the working electrode lowers the iR drop, stabilizes the local potential, and thereby reduces unwanted (irreversible) interfacial reactions. Therefore, our BMEAs decrease the interfacial electrochemical reaction due to the use of proximal local returns.
Computational simulation of current spreading
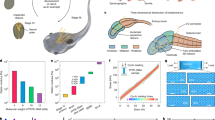
Multiphysics simulations were performed using a model geometry replicating the fabricated BMEA structure (see Materials and methods), incorporating the assumption of retina tissue existing between the electrodes and ground8, in order to more realistically assess the effectiveness of local ground electrodes in confining current spread (Fig. 2ai). The array consisted of seven microneedles arranged in a hexagonal pattern with a 200 μm pitch, which also equals the structure of the fabricated BMEA (Fig. 2aii). Analyses were conducted based on the cross-sectional view shown in Fig. 2aiii, which includes three central electrodes in the array. Current density distributions were evaluated at three different vertical planes: (1) 5 μm above the microtip electrode (Fig. 2bii and 2cii), (2) at the level of the local ground electrode (Fig. 2biii and 2ciii), and (3) 5 μm above the bottom substrate (Fig. 2biv and 2civ).
a Computational model of a bipolar electrode array, including the structure of a single bipolar electrode (ai), an array of seven bipolar electrodes within a simulation domain (aii), and a cross-sectional view of three bipolar electrodes.Simulated current flow and density distribution without (b) and with (c) local ground activation. Each result indicates the cross-sectional current flow and density (bi, ci); and three-dimensional current density heat map of 5 μm above the microtip electrode (bii, cii), at the middle of the local ground electrode (biii and ciii), and 5 μm above the substrate (biv, civ) corresponding to a black dashed line in aiii
Figure 2bi and 2ci illustrate the effect of local ground electrode presence on the direction of current flow. In the absence of a local ground electrode (Fig. 2bi), the current disperses omnidirectionally from the stimulation site. Although a global ground was included in the simulation to fulfill boundary conditions, such a return path does not exist in the actual physiological environment. Therefore, in practical scenarios, the current would likely exhibit a more symmetric dispersion around the stimulation site. In contrast, when the local ground electrode is employed (Fig. 2ci), the current is preferentially directed toward the local ground, demonstrating its effectiveness in guiding and localizing the stimulation current.
Subsequently, three-dimensional current density heatmaps revealed that at a plane 5 μm above the microtips, the spatial distribution remained relatively consistent regardless of local ground activation (Fig. 2bii and 2cii) suggesting that the stimulation performance near the microtip remains comparable in both conditions. However, analysis at different depths demonstrated distinct differences: with local ground activation, current was largely confined to the local ground electrode layer (Fig. 2biii and 2ciii), whereas without local grounding, current spread more extensively and reached the bottom substrate (Fig. 2biv and 2civ, see Figure S4 for more detail).
Collectively, the computational results confirm that the incorporation of a local ground electrode effectively modulates the current flow path. While the stimulation efficiency at the microtip electrode site was comparably preserved the presence of the local ground significantly altered the broader spatial distribution. Specifically, lateral current dispersion is restricted as the current is redirected toward the local ground electrode, rather than dispersing omnidirectionally. Moreover, vertical spreading into surface area, such as the bottom substrate, is notably suppressed, demonstrating enhanced confinement of the stimulation within the targeted depth. These results suggest that the local ground electrode enables localized and focal stimulation while minimizing unintended activation of non-target horizontal and vertical region.
Ex vivo experiments using mouse retina
To demonstrate the capability of electric stimulation and neural signal recording, we performed ex-vivo experiments using an isolated mouse retina tissue placed onto the fabricated BMEA. The BMEA successfully recorded retinal neural signals evoked by the applied electric stimulation (Fig. 3). From the raw extracellular recordings (Fig. 3a and 3b), single-unit spike waveforms were extracted for each electrode (Fig. 3c and 3d), and then raster plots were created (Fig. 3e). To define cells responsive to electrical stimulation, we focused our analysis on cells showing more significant (>50 Hz)28 evoked spike rates than the baseline rate (during the 50 ms pre-stimulus baseline period). The raster plots presented in Fig. 3e and 3f were obtained from recordings of the same retinal sample using the same electrodes, and cell numbering was consistently maintained across both conditions. Crucially, the cells displayed correspond to those that exceeded the 50 Hz threshold in at least one of the two conditions—either with or without local ground activation. These raster plots clearly demonstrate that activation of the local ground significantly reduces the number of stimulation-evoked spikes, as evident from the comparison between the two conditions. Also, it is worth to note that, when the local ground was activated, the baseline firing rate beyond 300 ms post-stimulus was significantly lower than that without local ground activation: baseline spike rates averaged across all trials and all cells were 16.00 ± 7.54 Hz without local ground and 5.50 ± 3.89 Hz with local ground, respectively (compare top and bottom raster plots of Fig. 3e and 3f for without and with local ground activation, respectively). This phenomenon is likely due to the narrower spread of stimulation charges across the retina when local ground was activated, probably resulting in less noisy signals from cells located in the surrounding area.
a Representative raw recordings after an electrical stimulation with a 4-msec-long 50 μA biphasic pulse. b Magnified views of a raw recording before (upper) and after (lower) electrical artifact reduction. c Preprocessed recordings from the electrode adjacent to the stimulating electrode (corresponding to the region marked by the purple dashed circle in a). d Spike waveforms of detected neurons from different electrodes (C1–C8). Gray traces indicate overlays of spike waveforms, and each red trace represents the average spike waveform in a given cell. e Raster plots from all recorded cells without (top, red) and with (bottom, blue) local ground activation. An identical electric stimulus (4-msec-long 100 μA biphasic pulse) was repeated 10 times; stimulus onset timings are marked with vertical red dashed lines. Each black tick represents an individual spike. The assigned cell numbers reflect their distance from the stimulating electrode; smaller numbers denote cells closer to the stimulation site, whereas large numbers represent cells farther away. Cell numbering is consistent between the two without and with local ground conditions. f Enlarged raster plots corresponding to the region highlighted by the bold box in (e), with the time scale expanded for clearer visualization for 1 sec, the duration of recordings after each of the 10 repeated stimuli
Using the criterion—a minimum maximum firing rate of 50 Hz above baseline—the numbers of effectively evoked channels were quantified as 15 out of 22 total channels (68.2%) without local ground, and 7 out of 22 total channels (31.8%) with local ground. Notably, in with local ground conditions, the evoked channels were confined within 314 µm of the stimulation site, whereas without local ground, responsive channels extended out to 714 µm. This comparison strongly supports that including the local ground in the electrode design significantly improves focality and efficacy by restricting activation to a smaller spatial range.
Figure 4 illustrates the effects of local ground activation on spiking activity across three different stimulation current amplitudes. To investigate how the spiking patterns of individual cells changed with or without local ground, we further analyzed cells. Paired t-tests were applied to maximum firing rate, mean firing rate, and spike count across the two conditions. The cells included in this analysis were those that met the 50 Hz maximum firing rate criterion in at least one condition. To minimize the influence of baseline firing, the maximum and mean firing rates were calculated after subtracting the baseline firing rate measured during the 50 ms pre-stimulus period. All major spiking metrics—maximum firing rate (p = 0.0146 for 50 µA; p = 0.0111 for 80 µA; p = 0.0007 for 100 µA), mean firing rate (p = 0.0004 for 50 µA; p = 0.0002 for 80 µA; p < 0.0001 for 100 µA), and spike count (p = 0.0005 for 50 µA; p < 0.0001 for 80 µA; p < 0.0001 for 100 µA)—were significantly reduced within the 300 msec post-stimulus window under local ground activation (Fig. 4a). As stimulation current increased, more distant neurons were activated in the absence of local ground, whereas their activity was markedly reduced when the local ground was enabled. This resulted in lower spiking metrics with increasing current under local ground conditions (the blue box-whisker plots in Fig. 4a), reflecting improved spatial selectivity by limiting activation to neurons near the electrode and reducing off-target stimulation. Furthermore, the spatial distribution of the mean firing rates was visualized as heatmaps for each electrode (Fig. 4b). In the absence of local grounding, strong firing activity was observed even in distant electrodes, indicating a broader spatial spread of applied current. In sharp contrast, with local ground activation, spiking activity was markedly suppressed in distal regions, suggesting that the local ground effectively confined stimulation to the target area in an ex vivo biological tissue.
a Comparison of spiking metrics (maximum firing rate, mean firing rate and spike count) of electrically evoked responses without and with local ground activation across different stimulation current amplitudes (50, 80, and 100 μA). The number of cells exhibiting a significant response at each stimulation intensity was as follows: 14 cells for 50 µA, 16 cells for 80 µA, and 26 cells for 100 µA. The box denotes the interquartile range (IQR; 25th to 75th percentile), and the whiskers extend to the most extreme data points (defined as within 1.5×IQR from the quartiles. The central red lines represent the median. Red bars indicate results without local ground activation, while blue bars represent results with local ground activation. *p < 0.05, **p < 0.01 and ***p < 0.001. b Heatmaps showing the spatial distribution of mean firing rate for each condition. The left column represents firing activities without local ground, the middle column shows activities with local ground, and the right column presents the percentage of difference between the two conditions (mean firing rates with local ground activation compared to those without local ground. Each row corresponds to different stimulation intensities (50, 80, and 100 μA). The color legend applies to both the mean firing rate in Hz (for left and middle columns) and the percentage decrease in percentage (for right column). Black dots indicate electrode positions, and a star shape indicates the stimulation electrode
To further quantify this spatial effect, we also plotted the mean firing rate as a function of electrode distance from the stimulation site (Fig. 5). Without local grounding, the mean firing rates remained relatively stable across distances. On the other hand, when the local ground was activated, the mean firing rates decayed more steeply with increasing distance (Fig. 5b). The stronger reduction in firing activities recorded from distant electrodes is more clearly visible when the firing rate difference between the two conditions was plotted as a function of distance (Fig. 5c). As shown in Fig. 5c, the suppression effect of local ground becomes increasingly prominent with increasing distance from the stimulation site, as evidenced by a significant positive correlation between firing rate difference and electrode distance at higher current levels (e.g., 80 and 100 μA). This indicates that local ground activation primarily limits the spatial spread of stimulation, rather than merely reducing overall excitability. Representative PSTHs and raster plots from two electrodes located 200 µm and 529 µm away from the stimulation site (Fig. 5d and 5e) also confirmed that the local ground activation more effectively suppressed neural responses at distant electrodes than at nearby ones.
a Electrode map outlining the locations of a stimulation electrode (yellow), adjacent electrodes (pink, 200 µm away), distant electrodes (green, 529 µm away), and other recording electrodes (gray, recorded no signal). b Mean firing rate comparison between without local ground (red) and with local ground (blue) conditions at different distances (200, 346, 400, 529, 600, 692, 714, and 871 µm) from the stimulation site across three stimulation intensities (50, 80, and 100 µA). Error bars, one standard deviation. c Percentage difference in mean firing rates between the two conditions (i.e., without and with local ground activation), plotted against distance from the stimulating electrode. Each data point represents the percent difference of mean firing rates in each cell. Black solid line indicates a linear fit. Both linear regression (ρ) and Pearson’s correlation (p) are shown in each panel. d PSTHs (upper) and corresponding raster plots (lower) of a representative cell recorded from adjacent (200 µm) electrodes in responses to ten repeats of 100 µA stimulation. Red and blue tick represent without local ground and with local ground conditions, respectively. e Same as d but for cell recorded from distant (529 µm) electrodes
Confocal imaging of retinal tissue for penetration verification
Retinal tissue penetration by the BMEA was verified using confocal microscopy. Low-magnification high-resolution images revealed a distinct hexagonal array of penetration holes spaced 200 μm apart, precisely corresponding to the layout of the fabricated BMEA (Fig. 6ai and 6aii). Cross-sectional imaging further confirmed that the electrodes successfully reached inner layer of the retina, especially around the bipolar cell (BC) layer at multiple insertion sites (Fig. 6aiii and 6aiv). To examine individual insertion sites in greater detail, high-magnification imaging of a single penetration hole was performed (Fig. 6bi). Z-stack scanning allowed for visualization of electrode penetration across different retinal layers, enabling a comprehensive assessment of electrode positioning. Top-view images of the retinal ganglion cell (RGC) layer (Fig. 6bii), BC layer (Fig. 6biii), and photoreceptor layer (Fig. 6biv) revealed clearly defined penetration holes in both the RGC and BC layers, while the photoreceptor layer showed no noticeable perforations. Additionally, cross-sectional images confirmed that the microneedle tips were accurately positioned within the BC layer (Fig. 6bv and 6bvi), indicating that the sharpness of the BMEA was sufficient to penetrate the soft retinal tissue without damaging deeper structures.
a Low-magnification confocal imaging and three-dimensional reconstruction of the electrode inserted retina (ai), top view (aii), and cross-sectional views along the planes marked in aii (aiii, aiv). White dashed circles and triangles denote the positions of bipolar electrodes. b High-magnification confocal imaging and three-dimensional reconstruction of a single penetration site (bi), top-view images at the interface between the retinal ganglion cell and bipolar cell layers (bii), in the middle of the bipolar cell layer (biii), and in the middle of the photoreceptor layer (biv), which align with the layers appeared in the simulation results in Fig. 7d, 7c, and 7b, respectively. Cross-sectional view showing the depth of electrode penetration across different retinal layers (bv). Schematic cross-sectional illustration of the retinal layers, corresponding to bv (bvi). All scale bars: 50 μm
Comprehensive advantages of the BMEA
Building upon the results presented above, we now discuss the structural advantages and broader implications of the proposed BMEA. The fabricated BMEA incorporates multiple layers, including two electrodes and two insulation layers onto each three-dimensional microneedle structure, while maintaining a sufficiently sharp geometry to allow insertion into soft retinal tissue. Moreover, computational simulation and ex-vivo experiment ensured the performance of BMEA in electric spread controlling. Here are the notable advantages shown in this work:
Firstly, a major structural innovation of the BMEA lies in the coaxial integration of the stimulating and local ground electrodes within a single three-dimensional microneedle. As introduced earlier in the 1. Introduction section, traditional designs of MEA often require spatially-separate electrodes arranged on different planes to accommodate local grounding techniques16,17, which reduces spatial efficiency and limits electrode density. In contrast, our vertically stacked configuration minimizes the physical footprint required for local grounding, thereby enabling denser packing of bipolar microneedle electrodes without compromising focality or selectivity. By integrating with high-density microneedle fabrication7 and self-aligned etching techniques for electrodes of varying heights16, the concept of BMEA offers a transformative platform with exceptional potential to enhance the spatial resolution of electrical stimulation across a broad range of neural interfaces including intracortical, cochlear, and retinal applications, ultimately paving the way for next-generation neural prosthetic technologies.
Second, despite undergoing multiple deposition and etching steps, BMEA retained sufficiently sharp geometry capable of penetrating dense and elastic biological tissue. This capability was experimentally validated through the successful perforation of the retina (see Fig. 6 for details), which exhibits a similar mechanical stiffness (0.94–1.8 kPa)29 with other applicable neural tissues such as the brain (0.2–2.0 kPa)30. The ability to form well-aligned insertion holes in such a mechanically stiff tissue not only confirms the structural integrity of the BMEA but also suggests strong potential for application in other neural interfaces beyond retinal implants. Furthermore, the high penetration capability is particularly advantageous for chronic implantation, as it minimizes tissue dimpling, bleeding, and mechanical trauma, which are key factors that influence long-term immune response31,32. Therefore, with further miniaturization, the BMEA design may offer a promising platform for long-term in vivo applications with reduced inflammatory response in various applications.
Third, a key structural advantage lies in the Utah array-inspired three-dimensional configuration, which facilitates large-area stimulation targeting inner layer of the neural tissues. Unlike Michigan-type neural probes that require complex multi-shank packaging to cover large area33,34, the spatial coverage of our design can be conveniently scaled by modifying the photomask layout. This planar scalability greatly simplifies fabrication while supporting high-density integration. Additionally, the microneedle height can be precisely tuned by controlling the thicknesses of the silicon and glass substrates, allowing device customization based on application-specific anatomical/mechanical constraints. This level of design flexibility broadens the applicability of the BMEA to diverse biomedical interfaces – ranging from intracortical neural probes to retinal prostheses requiring deep-layer penetration of delicate tissues.
Last key advantage of the BMEA lies on its current-confining capabilities validated through both computational simulations and ex vivo electrophysiological recordings. In both cases, significant lateral confinement of the electric current was observed. In particular, the ex-vivo experiments demonstrated that activation of the local ground electrode substantially reduced the neuronal activation area, as evidenced by decreased maximum and mean firing rates, as well as a lower total spike count across all stimulation intensities (50, 80, and 100 μA) (Fig. 4a). Spatial heatmaps revealed that neural activation became more localized around the stimulation site when the local ground was engaged, with reduced firing detected in more distant regions (Fig. 4b) A distance-dependent analysis of the firing rate further emphasized this effect, showing a relatively uniform distribution without local ground activation, and a pronounced attenuation with increasing distance when it was applied. Beyond lateral confinement of electric spreading, the simulations also demonstrated effective vertical confinement of current spread. Specifically, current density was highly localized at the level of the local ground electrode, and minimal current influx was observed near the substrate when the local ground was activated. These results confirm that the integration of local ground electrodes within each microneedle enables precise current shaping in both lateral and vertical dimensions, offering a robust strategy for spatially-selective neuromodulation.
Taken together, these structural and functional innovations—including the scalable Utah array-like configuration, mechanically robust and sharp microneedle geometry, coaxially-integrated local grounding, and lateral, vertical confinement of current spreading—highlight the potential of the proposed BMEA as a versatile platform for delivering high-density, large-area, and spatially-confined neural stimulation in a wide range of neural engineering and bioelectronic applications.
Materials and methods
Bipolar electrode formation and packaging
We selected materials that are widely recognized for their biocompatibility and frequent use in biomedical microdevices: Gold as the conductive metal22, and SiO2 and Parylene-C as the insulating materials35,36. The fabrication process of the BMEA is schematically illustrated in Fig. 7. A heavily doped, low-resistance silicon (Si) wafer (p-type <100 > , 0.001–0.003 Ω·cm, ~525 μm in thickness) was used as a substrate. A 1 μm-thick SiO2 layer was deposited via plasma-enhanced chemical vapor deposition (PECVD) and patterned as an etching mask. The Si wafer was then etched using deep reactive ion etching (DRIE) to form conductive Si pillars (Fig. 7a and 7b). The remaining SiO2 layer was subsequently removed by immersing the wafer in a diluted hydrofluoric acid (HF) solution in the ratio of 10:1 with deionized water.
a–c Patterning of Si wafer using deep reactive ion etching (DRIE) and electrical isolation of individual electrodes using a reflow process of glass wafer anodically bonded to the Si wafer. d, e Formation of micro-pillars and microneedles via DRIE and wet etching. f–h Sequential depositions of a metal layer (Cr/Au) for stimulation electrodes, an upper insulation layer (SiO₂), and another metal layer (Cr/Au) for local ground electrode. i, j) Partial exposure of the upper insulation layer and the stimulation electrode using self-aligned patterning processes. k, l Deposition of a Parylene-C insulation layer followed by tip exposure using reactive ion etching (RIE) to define a lower insulation layer
To prevent crystallization during the subsequent glass reflow process, the borosilicate glass substrate underwent plasma treatment for 3 min using Cl2 gas (50 sccm, 55 mTorr, 150 W RF power). The treated glass substrate was anodically bonded to the silicon wafer containing the etched pillars. The bonding was conducted under low pressure (10 mTorr) to facilitate effective glass infiltration into the cavities between the silicon pillars during the reflow process. The bonded wafer was heated at 850°C for 5 h, allowing the melted glass to reflow and fully encapsulate the silicon pillars. Following the reflow process, the wafer was subjected to chemical-mechanical polishing (CMP) to planarize the top surface and expose the silicon pillars on the glass substrate surface. In the final structure, the low-resistance silicon pillars embedded in the glass layer functioned as through-glass vias (TGVs) for independent electrical connections to the microtip electrodes (Fig. 7c).
The Si wafer was then cleaned using a piranha solution composed of sulfuric acid (H2SO4) and hydrogen peroxide (H2O2). To define the microneedle structures, a 1.5 μm-thick SiO2 layer was deposited via PECVD on the opposite side of the wafer and patterned as an etching mask. DRIE was performed to etch the cylindrical silicon pillars (Fig. 7d). Subsequently, the silicon pillars were wet-etched in a solution of 5% HF and 70% HNO3 at 55 °C without stirring, allowing the differential etch rates at the top and bottom of the structures to gradually shape them into sharp microneedle (Fig. 7e). Therefore, the overall height and base diameter of the microneedle structure can be controlled by adjusting the diameter and height of the silicon pillar formed in the process step shown in Fig. 7d, as well as the etching duration in Fig. 7e. Given that the microtip array is vertically protruded from the substrate, further increases in tip height are feasible within the constraints of the substrate thickness. In addition, finer control over the base diameter can be achieved through precise tuning of the pillar geometry and etch parameters. In our previous studies, much narrower microneedles were fabricated7 and microtip electrodes with heights approaching 200 μm were fabricated16 or neural interface application.
A Cr/Au (200/2000 Å) layer was then deposited and patterned via a lift-off process to form the stimulation electrode (Fig. 7f). A SiO2 insulation layer was deposited via PECVD to electrically isolate the first (microtip) and second (local ground) electrode layers (Fig. 7g). A second Cr/Au (200/2000 Å) layer was sputter-deposited to establish the ground electrode on top of the SiO2 insulation layer (Fig. 7h).
To selectively expose the electrode layers at the microneedle structure, a three-step self-aligned etching process was performed. Due to the high aspect ratio and sharp geometry of the microneedle, the photoresist layer at the tip end was significantly thinner than that in other regions. Consequently, during the dry etching process, the thinner photoresist at the tip end was removed first, enabling selective exposure of the insulating and electrode layers. This self-aligned process facilitated precise layer exposure without the need for additional photolithography alignment.
In the first etching step, the second electrode layer (Cr/Au, local ground electrode) was selectively removed at the microneedle tip (Fig. 7i). In the second step, the SiO2 insulation layer at the tip was etched to expose the underlying first electrode layer (Cr/Au, stimulation electrode) (Fig. 7j). To provide insulation for the base of ground electrode, a 3000 Å-thick Parylene-C layer was deposited over the whole structure (Fig. 7k). Finally, in the third etching step, the Parylene-C layer was selectively removed to expose both the tip electrode and a top portion of the ground electrode via RIE under the following conditions: RF power of 200 W, O2 gas flow rate of 100 sccm, Ar gas flow rate of 5 sccm, and chamber pressure of 100 mTorr. This process ensured precise exposure of all the layers on the proposed bipolar microneedle structure (Fig. 7l).
Electrochemical characterization of bipolar microneedle electrode array
After packaging the fabricated BMEA on a PCB by backside wire bonding, electrochemical impedance spectroscopy (EIS) measurements were performed on randomly selected electrodes (n = 10) using a potentiostat (PalmSens 4, PalmSens, Houten, Utrecht, Netherlands). The BMEA was immersed in the phosphate-buffered saline (PBS), with an Ag/AgCl reference electrode and a platinum counter electrode. A titanium working electrode was connected to each selected bipolar electrode via connectors on the PCB. Impedance was recorded across a frequency range of 1 Hz to 100 kHz using a given software (PSTrace 5.9, PalmSens, Houten, Utrecht, Netherlands).
Computational modeling and finite element simulation
Computational models were developed to analyze the electric field distribution and current conduction behavior of the BMEA using finite element method (FEM) simulations (COMSOL Multiphysics 5.2, COMSOL AB, Stockholm, Sweden) in the Electric Currents (EC) module to solve the current conservation equation based on Ohm’s law8. This approach enabled the modeling of volumetric electric potential conduction under the assumption of a steady-state electric current. The simulated electrode structure consisted of a microtip electrode, upper insulation, local ground electrode, and lower insulation (see Results and Discussion section for more details). In this work, we characterized the electric stimulation properties of the BMEA penetrated the retina tissue to simulate a retinal implant as one of the applications of the fabricated BMEA. To optimize computational efficiency, the background setup of the simulation was set as a single-layer of the retina, following previous studies37. While the actual human retina has a radius exceeding 18,000 μm38, the simulation model was designed with a reduced radius of 9,000 μm and a height of 1.5 cm. The electrical conductivity of the retinal tissue was set to 0.1 S/m, in accordance with the value reported in the prior literature37. Since the primary objective was to analyze current flow within the retina tissue rather than within the electrode, the electrode itself was excluded from the simulation domain to define the computational model.
Given the intricate geometry of the sharp bipolar electrodes, the mesh was manually refined to ensure accurate simulation results. The initial mesh setting was configured as extremely fine, with a maximum element size of 300, a growth rate of 1.3, and a minimum element size of 1. The curvature factor was set to 0.2, and the resolution of narrow regions was defined as 1 to better capture fine structural details.
To evaluate the effect of local grounding, simulations were conducted under two distinct conditions: one with only a global ground (i.e., local ground electrode deactivated), and the other with only the local ground electrode activated. Given the comparative focus of this analysis, emphasis was placed on evaluating the relative differences in potential distribution rather than on the absolute magnitude of current.
Ex vivo experiment setup and neural signal acquisition
All animal experiment procedures were conducted in accordance with institutional ethical guidelines and were approved by the Institutional Animal Care and Use Committee (KIST-IACUC-2023-081-5). Prior to experiments, artificial cerebrospinal fluid (ACSF) was prepared by dissolving following reagents in deionized water to a final volume of 1 L: sodium chloride (NaCl,124 mM), potassium chloride (KCl, 5 mM), monobasic potassium phosphate (KH2PO4, 1.15 mM), magnesium sulfate (MgSO4, 1.15 mM), glucose (10 mM), sodium bicarbonate (NaHCO3, 25 mM), and calcium chloride (CaCl2, 2.5 mM). The solution was continuously bubbled with a gas mixture of 95% O2 and 5% CO2 for at least 30 min, until it reached room temperature and a pH of 7.3–7.4.
For ex-vivo experiments, 8–10 weeks old male wild-type mice (C57BL/6J, DBL, Eumseong-gun, Chungcheongbuk-do, Republic of Korea) were used. Mice were anesthetized with isoflurane, and both eyes were enucleated for retinal extraction. The anterior segment, including the cornea and lens, was carefully removed, followed by gentle removal of the vitreous humor. The isolated retina was trimmed into a flat sheet and positioned on the BMEA with the epiretinal side facing downward (Fig. 8a and 8b). A fine mesh was placed over the retina, and any excess fluid was removed using a cotton swab to promote tissue-electrode adhesion. To prevent retinal detachment due to ACSF perfusion, a custom-designed horseshoe-shaped Teflon anchor was placed on top of the mesh (Fig. 8c). The Ag/AgCl global ground ball was immersed in the ACSF solution during recordings in which the local ground electrode was deactivated, and removed when the local ground electrode was activated.
a Schematic illustration showing the insertion of the bipolar microneedle electrode array (BMEA) into the degenerated retina (black dashed box) from the epiretinal surface (i.e., retinal ganglion cell side). The inset shows a cross-sectional view of a single bipolar electrode within the degenerated retinal tissue. Red dashed arrows indicate the direction of electric current flow, and the red-shaded region denotes the stimulated area. b A microscope image of a mouse retina placed on the BMEA. The isolated mouse retina is outlined with a white dashed line, and dark dots covered by the retina are images of bipolar microneedle electrodes, while white dashed circled bipolar microneedle electrodes that worked as the stimulation electrodes. c A photograph of the experimental setup, showing an anchor to press down a retina tissue onto the BMEA, perfusion components (solution inlet and outlet), and two amplifiers connected to a main printed circuit board. The white dashed outline indicates the anchor to stably fix the retina tissue. d Schematic representation of the ex vivo experiment system, including a current generator for electrical stimulation, an artificial cerebrospinal fluid (ACSF) perfusion system, and an Intan RHD recording system for measuring spiking activities
Once the retinal tissue was secured onto the electrode array, continuous perfusion with fresh ACSF was maintained using a peristaltic pump (PPS2, Multichannel Systems, Reutlingen, Germany) at a flow rate of ~2.0 mL/min. The perfusate temperature was regulated at 35–36°C using an in-line solution heater (PH01, Multichannel Systems, Reutlingen, Germany). Electrical stimulation was delivered via a programmable current generator (STG4002, Multichannel Systems, Reutlingen, Germany), which was connected to the stimulation line on the PCB. Both the amplitude and duration of the stimulation were controlled using the manufacturer’s dedicated software.
Neural signals were recorded using two 16-channel Intan amplifiers (RHD2132, Intan Technologies, Los Angeles, CA, USA), interfaced with Omnetics connectors (A79038-001, Omnetics Connector Corporation, Minneapolis, MN, USA) mounted on both sides of the PCB. The amplified signals were then transmitted to a recording system controller (RHD 512-channel recording controller, Intan Technologies, Los Angeles, CA, USA), with the signal bandwidth set to 300–3000 Hz via the Intan Technologies software. A synchronization signal from the current generator was also simultaneously recorded by the Intan controller to precisely log stimulation timing (Fig. 8d).
Electrical stimulation was applied concurrently with extracellular neural activity recording. Cathodic-first biphasic pulses (a fixed pulse width of 4 msec for each phase without inter-phase interval) in amplitudes of 50, 80, or 100 µA were delivered for charge balance39.
Data analysis
Extracellular recordings were preprocessed using customized Python scripts incorporating the Scikit-learn and SciPy libraries40,41. Electrical stimulation artifacts were detected using synchronized digital trigger signals and removed by zero-padding within a 5-msec window before and after stimulation onset. To minimize ringing effects caused by abrupt signal transitions due to zero-padding, we identified the post-stimulus time point at which the signal amplitude returned closest to baseline (within 10 msec) and extended the zero-padding window accordingly (see Fig. 3b for details). Following artifact removal, a fourth-order Butterworth band-pass filter (500–3000 Hz) was applied to suppress low-frequency drift and high-frequency noise.
To improve the reliability of spike detection in noisy extracellular recordings, we applied the Smoothed Teager Energy Operator (STEO) to the band-pass filtered neural signals42. The STEO enhances the energy characteristics of neural spikes by amplifying the unique, high-energy features of spike waveforms – such as their sharpness and transient nature – while suppressing background noise. The threshold for spike detection was determined as follow:
\({STEO}\) represents the full STEO value of each electrode, while \({STE}{O}_{{background}}\) refers to the STEO value of each electrode during the pre-stimulus period (~150 msec). To automate the threshold determination while considering the noise level of the signal, we utilized the standard deviation (SD) of the background STEO signal from the pre-stimulus period. Detected spike waveforms were further processed using Principal Component Analysis (PCA) and Hierarchical Density-Based Spatial Clustering of Applications with Noise (HDBSCAN) to isolate single-unit activities43,44.
Peri-stimulus time histograms (PSTHs) were generated to examine time-locked neural responses following stimulation, using a 20-msec time bin with a 5-msec overlapping window. Baseline PSTHs from the pre-stimulation period (-50 to 0 msec) for every stimulation were subtracted to account for spontaneous activity. After calculating the PSTHs, retinal ganglion cells exhibiting maximum firing rate below 50 Hz were excluded from further analysis. Statistical significance of the maximum firing rate, mean firing rate, and spike count within 300 msec post-stimulation was evaluated using Student’s t-test. Additionally, the percentage decrease in mean firing rates with local ground activation, relative to the condition without local grounding, was calculated as follows:
where MFRwithout and MFRwith are mean firing rates without and with local ground activation, respectively.
Whole-mount retinal fluorescent staining and imaging
Fluorescence imaging was performed to verify the ability of the BMEA to penetrate soft biological tissue. Excised retinal tissue was selected as a representative model due to its delicate and fragile structure, making it an ideal substrate for assessing the mechanical suitability of microneedle electrode insertion. Because effective penetration into such tissue requires sufficient sharpness, the retinal model provided a critical testbed for evaluating mechanical performance. Following enucleation and retinal isolation, the tissue was positioned onto a fabricated BMEA, secured using the mesh and anchor system described in the 2.4 Ex-vivo Experimental Setup and Neural Signal Acquisition section. The prepared retinal tissue was then fixed in 4% paraformaldehyde (PFA) at 4°C for approximately 12 h. After fixation, the sample was washed three times with PBS, each wash lasting for 20 min. For nuclear visualization, the tissue was stained with 4′,6-diamidino-2-phenylindole (DAPI; two drops diluted in 1 mL of PBS) for 2 h.
Following staining, the retina was carefully detached from the BMEA and washed three additional times with PBS (20 min per wash). The sample was subsequently mounted on a slide glass using Vectashield mounting medium (H-1000; Vector Laboratories, Burlingame, CA, USA), and a cover glass was gently placed on top. The prepared slide was allowed to dry in a dark environment at room temperature for 24 h.
Three-dimensional imaging of the whole-mounted retina was performed using a confocal inverted microscope (LSM800, Zeiss, Baden-Württemberg, Germany) and its operating software (ZEN 3.3 Blue Edition, Zeiss, Baden-Württemberg, Germany). Under 450 nm excitation, dark circular regions corresponding to the microneedle electrode penetration sites were observed in top-view fluorescence images (see Fig. 6 for details). Z-stack imaging was additionally conducted to reconstruct microneedle penetration tracks through the retinal layers, providing detailed spatial visualization of tissue-electrode interactions.
Conclusion
In this study, we developed a novel bipolar microneedle electrode array (BMEA) that integrates both stimulation and local ground electrodes within a single three-dimensional microneedle structure. This vertically-aligned bipolar configuration enables spatially-confined neural stimulation while preserving mechanical penetrability. The fabrication process involved three times of depositions and two times of self-alignment patternings (Cr/Au–SiO₂–Cr/Au) combined with conformal Parylene-C insulation and etching steps for selective tip exposure. This approach enabled precise layer alignment and reliable electrical isolation between the two electrodes. The proposed electrode platform provides multiple advantages over conventional planar or shank-type designs, including tunable microneedle height, simplified array scaling via photomask redesign, and compact spatial integration of bipolar stimulation components. These structural features support a wide range of applications requiring precise and localized neuromodulation in confined anatomical environments.
Our BMEA demonstrated reliable insertion into delicate retinal tissue while maintaining sharpness and structural integrity. COMSOL Multiphysics simulations and ex vivo neural signal recordings confirmed that activating the integrated local ground electrode effectively suppresses current spread, resulting in enhanced stimulation focality and reduced off-target activation. Future work will focus on in vivo validation of the BMEA platform in animal models, long-term biocompatibility assessment, and closed-loop interface development with integrated sensing modules. Additionally, optimization of microneedle geometries for different tissue targets (e.g., brain, spinal cord, or peripheral nerves) will be pursued to extend the applicability of the platform. Clinically, the proposed BMEA design has strong relevance for next-generation neural prosthetics, particularly in retinal implants for restoring vision in patients with photoreceptor degeneration, as well as for high-resolution neuromodulation in cortical or subcortical regions. The compact form factor and electrical selectivity enabled by the bipolar configuration may contribute to more effective and safer clinical neurostimulation systems in the future. In parallel, integrating emerging grounding-free approaches such as ferroelectric bioelectronics10 with our electrode-based design could create hybrid systems that combine wireless convenience with precise current confinement.
Data availability
The raw and analyzed datasets generated during the study are available for research purposes from the corresponding authors on reasonable request.
Code availability
Custom-written codes used to analyze data from this study are available from the corresponding authors on request.
References
Fernández, E. et al. Visual percepts evoked with an intracortical 96-channel microelectrode array inserted in human occipital cortex. J Clin Invest 131, (2021).
Chen, X., Wang, F., Fernandez, E. & Roelfsema, P. R. Shape perception via a high-channel-count neuroprosthesis in monkey visual cortex. Science (1979) 370, 1191–1196 (2020).
Isaakidou, A., Apachitei, I., Fratila-Apachitei, L. E. & Zadpoor, A. A. High-precision 3D printing of microporous cochlear implants for personalized local drug delivery. J Funct Biomater 14, 494 (2023).
Arora, K., Plant, K., Dawson, P. & Cowan, R. Effect of reducing electrical stimulation rate on hearing performance of Nucleus® cochlear implant recipients. Int J Audio 64, 173–182 (2025).
Fried, S. I., Lasker, A. C. W., Desai, N. J., Eddington, D. K. & Rizzo, J. F. 3rd Axonal sodium-channel bands shape the response to electric stimulation in retinal ganglion cells. J Neurophysiol 101, 1972–1987 (2009).
Chung, W. G. et al. Liquid-metal-based three-dimensional microelectrode arrays integrated with implantable ultrathin retinal prosthesis for vision restoration. Nat Nanotechnol 19, 688–697 (2024).
Roh, H. et al. Fabrication of high-density out-of-plane microneedle arrays with various heights and diverse cross-sectional shapes. Nanomicro Lett 14, 24 (2022).
Son, Y., Chen, Z. C., Roh, H., Lee, B. C. & Im, M. Effects on retinal stimulation of the geometry and the insertion location of penetrating electrodes. IEEE Trans Neural Syst Rehabil Eng. 31, 3803–3812 (2023).
Eleftheriou, C. G. et al. Carbon nanotube electrodes for retinal implants: A study of structural and functional integration over time. Biomaterials 112, 108–121 (2017).
Wang, F., Wang, L., Zhu, X., Lu, Y. & Du, X. Neuron-inspired ferroelectric bioelectronics for adaptive biointerfacing. Adv Mater 2416698 (2025).
Fan, V. H. et al. Epiretinal stimulation with local returns enhances selectivity at cellular resolution. J Neural Eng. 16, 025001 (2019).
Seo, H. W. et al. Focal stimulation of retinal ganglion cells using subretinal 3D microelectrodes with peripheral electrodes of opposite current. Biomed Eng Lett 14, 355–365 (2024).
Flores, T., Goetz, G., Lei, X. & Palanker, D. Optimization of return electrodes in neurostimulating arrays. J Neural Eng 13, 036010 (2016).
HajjHassan, M., Chodavarapu, V. & Musallam, S. NeuroMEMS: neural probe microtechnologies. Sensors 8, 6704–6726 (2008).
Wang, T. et al. Advanced neural probe sensors toward multi-modal sensing and modulation: design, integration, and applications. Adv Sens Res 4, 2400142 (2025).
Shin, S.-B. et al. Fabrication of a transparent array of penetrating 3D microelectrodes with two different heights for both neural stimulation and recording. Sens Actuators B Chem. 393, 134184 (2023).
Flores, T. et al. Honeycomb-shaped electro-neural interface enables cellular-scale pixels in subretinal prosthesis. Sci Rep 9, 10657 (2019).
Normann, R. A. & Fernandez, E. Clinical applications of penetrating neural interfaces and Utah Electrode Array technologies. J Neural Eng 13, 061003 (2016).
Sim, S. et al. Neural probe integrated with low-impedance electrodes implemented using vertically aligned carbon nanotubes for three-dimensional mapping of neural signals. Sens Actuators B Chem. 393, 134124 (2023).
Merrill, D. R., Bikson, M. & Jefferys, J. G. R. Electrical stimulation of excitable tissue: design of efficacious and safe protocols. J Neurosci Methods 141, 171–198 (2005).
Shpun, G. et al. Optimizing the fabrication of a 3D high-resolution implant for neural stimulation. J Biol Eng. 17, 55 (2023).
Lam, D. V. et al. Evaluation of gold helical microwire structure electrode for long-term rodent nerve stimulation. J Neural Eng 22, 036042 (2025).
Ionescu, O. N. et al. System of implantable electrodes for neural signal acquisition and stimulation for wirelessly connected forearm prosthesis. Biosensors 14, 31 (2024).
Lee, S., Ahn, J. H., Seo, J.-M., Chung, H. & Cho, D.-I. “Dan”. Electrical characterization of 3D Au microelectrodes for use in retinal prostheses. Sensors 15, 14345–14355 (2015).
Cogan, S. F. Neural stimulation and recording electrodes. Annu Rev Biomed Eng 10, 275–309 (2008).
Zheng, W. iR compensation for electrocatalysis studies: considerations and recommendations. ACS Energy Lett 8, 1952–1958 (2023).
Zhang, F. et al. Reference and counter electrode positions affect electrochemical characterization of bioanodes in different bioelectrochemical systems. Biotechnol Bioeng. 111, 1931–1939 (2014).
Quiroga, R. Q., Mukamel, R., Isham, E. A., Malach, R. & Fried, I. Human single-neuron responses at the threshold of conscious recognition. Proc Natl Acad Sci 105, 3599–3604 (2008).
Franze, K. et al. Spatial mapping of the mechanical properties of the living retina using scanning force microscopy. Soft Matter 7, 3147–3154 (2011).
Antonovaite, N., Beekmans, S. V., Hol, E. M., Wadman, W. J. & Iannuzzi, D. Regional variations in stiffness in live mouse brain tissue determined by depth-controlled indentation mapping. Sci Rep 8, 12517 (2018).
Prodanov, D. & Delbeke, J. Mechanical and biological interactions of implants with the brain and their impact on implant design. Front Neurosci 10, 11 (2016).
Xiang, Y. et al. Implantable neural microelectrodes: how to reduce immune response. ACS Biomater Sci Eng 10, 2762–2783 (2024).
Wise, K. D., Anderson, D. J., Hetke, J. F., Kipke, D. R. & Najafi, K. Wireless implantable microsystems: high-density electronic interfaces to the nervous system. Proc IEEE 92, 76–97 (2004).
Zhang, B. et al. Development of 3D-Stacked Multi-Shank Si Neural Probe Array (1)-Fabrication of Embedded Multiple Optical Fibers in Multi-Shank. in JSAP Annual Meetings Extended Abstracts The 65th JSAP Spring Meeting 2018 2922 (The Japan Society of Applied Physics, 2018).
Wang, Y., Yang, X., Zhang, X., Wang, Y. & Pei, W. Implantable intracortical microelectrodes: reviewing the present with a focus on the future. Microsyst Nanoeng. 9, 7 (2023).
Yue, L., Weiland, J. D., Roska, B. & Humayun, M. S. Retinal stimulation strategies to restore vision: Fundamentals and systems. Prog Retin Eye Res. 53, 21–47 (2016).
Werginz, P. & Rattay, F. The impact of calcium current reversal on neurotransmitter release in the electrically stimulated retina. J Neural Eng. 13, 046013 (2016).
Kolb, H., Fernandez, E., Jones, B. & Nelson, R. Webvision: the organization of the retina and visual system [Internet]. (1995).
Im, M. & Fried, S. I. Indirect activation elicits strong correlations between light and electrical responses in ON but not OFF retinal ganglion cells. J Physiol 593, 3577–3596 (2015).
Pedregosa, F. et al. Scikit-learn: Machine learning in Python. J Mach Learn Res 12, 2825–2830 (2011).
Virtanen, P. et al. SciPy 1.0: fundamental algorithms for scientific computing in Python. Nat Methods 17, 261–272 (2020).
Semmaoui, H., Drolet, J., Lakhssassi, A. & Sawan, M. Setting adaptive spike detection threshold for smoothed TEO based on robust statistics theory. IEEE Trans Biomed Eng. 59, 474–482 (2011).
Eggermont, J. J., Epping, W. J. M. & Aertsen, A. Stimulus dependent neural correlations in the auditory midbrain of the grassfrog (Rana temporaria L.). Biol Cyber 47, 103–117 (1983).
Ester, M., Kriegel, H.-P., Sander, J. & Xu, X. A density-based algorithm for discovering clusters in large spatial databases with noise. In KDD vol. 96 226–231 (1996).
Acknowledgements
Seung-Han Chung, Chaesung Kim, and Minju Kim contributed equally to this work. This work was supported in part by KIST (Korea Institute of Science and Technology) institutional grants (Nos. 2E33881 and 2E33682), and in part by the National R&D Program through the National Research Foundation (NRF) of Korea, funded by the Ministry of Science and ICT (Nos. 2023R1A2C2003786, RS-2023-00302397, RS-2025-25465381, and RS-2025-00514523).
Author information
Authors and Affiliations
Contributions
Conceptualization: J.-H. Park, M. Im. Fabrication: S.-H. Chung, D. Choi. Simulation: Y. Son, B. C. Lee. Ex vivo experiment: C. Kim, M. Kim, H. Roh. Data analysis: C. Kim, M. Kim, M. Im. Supervision: H.-M. Lee, Y.-K. Kim, S.-K. Lee, J.-H. Park, M. Im. Writing—original draft: C. Kim, M. Kim, J.-H. Park, M. Im. Writing—review and editing: C. Kim, M. Kim, J.-H. Park, M. Im.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chung, SH., Kim, C., Kim, M. et al. A three-dimensional bipolar microneedle electrode array with local ground integrated at each sidewall for enhanced focal electric stimulation. Microsyst Nanoeng 11, 230 (2025). https://doi.org/10.1038/s41378-025-01093-0
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41378-025-01093-0