Abstract
There is increasing clinical evidence that pancreatic dysfunction in diabetes needs to be viewed in the context of crosstalk with the liver as well as other organs. Our goal for this study was to develop a pancreas-liver co-culture system suited for mechanistic and therapy testing studies in the context of multi-organ cross talk. To achieve this goal, we developed a co-axial flow-focusing microfluidic device to fabricate multi-compartment hydrogel microcapsules. Each microcapsule contained two aqueous compartments or cores surrounded by poly(ethylene glycol) (PEG) hydrogel. Each microcapsule had pancreatic β-cells loaded into one compartment and hepatic cells into another compartment. Individual encapsulated cells assembled into pancreatic and hepatic cell spheroids over time. Characterization of microcapsules revealed enhanced hepatic and pancreatic function in microcapsules containing pancreas-liver co-cultures compared to microcapsules with one cell type only. Multicompartment microcapsules represent a novel microphysiological system type and hold the promise of increasing experiment throughput for mechanism discovery and drug development studies.
Similar content being viewed by others
Introduction
Diabetes and metabolic dysfunction-associated steatohepatitis (MASH) are increasingly viewed as interconnected conditions fitting under the umbrella of metabolic syndrome1. It is well appreciated for example that patients with MASH are twice as likely to develop type 2 diabetes2. Given these clinical observations and given that the pancreas and the liver are connected anatomically in the body, there is an increasing interest in establishing in vitro culture systems with both organs represented. Microphysiological systems are well suited for integrating multiple cell types into a common fluidic circuit. There have been several reports of microphysiological systems recapitulating pancreas-liver interactions. For example, Bauer et al. developed a microfluidic system featuring distinct liver and pancreas compartments connected into a closed-loop fluidic circuit driven by an on-chip micropump3. Their co-culture system demonstrated glucose responsiveness, including an initial decrease in glucose levels followed by a reduction in insulin secretion. Shafagh et al. developed another microfluidic system that used pneumatic actuation to drive flow through channels with varying resistance, presenting a different way of achieving inter-organ signaling4. Their study demonstrated that glucose challenge led to a rapid insulin secretion, which in turn activated hepatic AKT phosphorylation to inhibit gluconeogenesis, a well-known hepatic response to insulin exposure. More comprehensive microphysiological systems described by Ronaldson-Bouchard and colleagues included six organs bathing in common media5. While possessing numerous advantages, microphysiological systems relying on microfluidics are often complex which limits testing throughput.
Microcapsules represent an interesting alternative to microfluidic devices. They may be produced at rates of 10 to 1000/s and may contain cells and bioactive polymers. While a number of encapsulation strategies have been reported6,7, droplet microfluidics has gained popularity because this approach allows to precisely tune geometry/dimensions of microcapsules and is relatively gentle on the cells8,9,10,11. There have been several reports describing encapsulation of pancreatic β-cells or liver cells in microgels comprised of alginate or other materials12,13,14. Our team has a long-standing interest in microencapsulation and has demonstrated encapsulation of pluripotent stem cells as well as primary hepatocytes15,16,17,18. The microcapsules used by us previously were comprised of a hydrogel shell and an aqueous core – the latter being particularly well-suited for rapid cell aggregation and spheroid formation. Both pancreatic β-cells and hepatocytes are epithelial cells that require cell-cell contact for survival and function19,20. Both cell types have been shown to benefit from 3-dimension (3D) organization and to remain functional as spheroid cultures for days to weeks21,22,23,24,25.
The spatial organization of distinct cell types (e.g. micropatterned co-cultures or microfluidic co-cultures) has proven valuable for understanding specific roles individual cell or tissue types play in disease progression or drug response26,27. While the use of microencapsulation for culturing cells has been documented extensively15,16,28, only few reports describe spatially patterning of encapsulated cell types29,30. To the best of our knowledge, no report has demonstrated microcapsule-based patterned co-cultures of cell spheroids where encapsulated cells interact in a synergistic manner.
Materials and methods
Materials
100 mm silicon wafer was purchased from University Wafer (South Boston, MA, USA). SU-8-2050 and SU-8-2025 were purchased from Kayaku Advanced Materials (Westborough, MA, USA). NaHCO₃ was purchased from Fisher Scientific (Pittsburgh, PA, USA). Urea kit was purchased from BioAssay Systems (Hayward, CA, USA). Penicillin-Streptomycin (10,000 U/mL), 1 M TEA, DMEM, and 2-mercaptoethanol were purchased from Thermo Fisher Scientific (Waltham, MA, USA). ACA-PEG-ACA (10k) and cell culture grade water were purchased from Creative PEGWorks (Chapel Hill, NC, USA). Ethanol was purchased from Brenntag (Reading, PA, USA). 8arm-PEG-maleimide (10k) was purchased from Jenkem Technology (Plano, TX, USA). Dithiothreitol (DTT), mineral oil, Span-80, FITC-labeled dextran (500,000 kDa), dextran (500,000 kDa), trypsin, NaOH, bovine serum albumin, (D)-+-glucose, HEPES, KCl, CaCl₂, NaCl, chlorotrimethylsilane, and Triton™ X-100 were purchased from Sigma-Aldrich (St. Louis, MO, USA). Fluorescent labeled beads were purchased from Spherotech (Lake Forest, IL, USA). Polydimethylsiloxane (PDMS) elastomer and base were purchased from Dow Corning (Ringwood, IL, USA). PBS and FBS were purchased from Corning (Manassas, NY, USA). OptiPrep densifier was purchased from STEMCELL Technologies (Vancouver, Canada). Live/dead staining kit, MgCl₂, Donkey anti-goat IgG Alexa Fluor Plus 488, and Vybrant multicolor cell-labeling kit were purchased from Invitrogen (Waltham, MA, USA). Goat anti-Human Albumin Antibody and Albumin ELISA Kit were purchased from Bethyl Laboratories Inc. (Montgomery, TX, USA). Rabbit anti-mouse Insulin Antibody was purchased from Cell Signaling Technology Inc. (Danvers, MA, USA). Ultrasensitive Insulin ELISA was purchased from American Laboratory Products Company (ALPCO) (Salem, NH, USA). Medical grade polyethylene micro tubing was purchased from Scientific Commodities Inc. (Lake Havasu City, AZ, USA). EasyStrainer 100 μm was purchased from Greiner Bio-One North America Inc. (Monroe, NC, USA).
Fabrication and operation of the co-axial flow focusing microfluidic device
The co-axial flow-focusing microfluidic device was designed in AutoCAD and fabricated via multilayer soft lithography. Detailed fabrication protocols are described in a previous study17. Briefly, mirrored and non-mirrored master molds were created by photolithography using SU-8-2025 and SU-8-2050 photoresists to produce channel heights of 60, 100, and 150 μm. Then, PDMS in 10:1 base to curing agent was poured over each mold, cured, and bonded following oxygen plasma treatment. The final square cross-sectional geometry of the microfluidic device is as follows: (1) 120 μm—core; (2) 200 μm—shell; (3) 300 μm—oil. Microcapsule fabrication was performed using a four-phase system consisting of core, shell, shielding oil, and crosslinking oil streams described as follows: (1) Core composed of 15% (w/v) 500 kDa dextran, 17% (v/v) Optiprep densifier, and 83% (v/v) DMEM/F12; (2) Shell composed of 10% (w/v) 8ARM-PEG-MAL, 5% (w/v) ACA-PEG-ACA, 90% (v/v) 15 mM TEA, and 10% (v/v) absolute ethanol; (3) Shielding oil composed of mineral oil with 0.5% (v/v) Span-80; (4) Crosslinking oil composed of 60 mM DTT and 3 mM NaOH emulsified in mineral oil with 3% (v/v) Span-80.
All solutions were transferred into syringes with a 27G needle and delivered to the microfluidic device via syringe pumps (Harvard Apparatus, Holliston, MA, USA) through tubing. Different flow rates were tested for core, shell, and shielding oil. Green and red fluorescent beads (1 μm) were incorporated into the core solution to visualize the architecture of the dual-core. Capsules were monitored under an inverted fluorescent microscope (IX83; Olympus, Center Valley, PA, USA) for morphology and core separation through brightfield and fluorescent images. Microcapsule and core diameters were measured via ImageJ software.
Fluorescent dextran diffusion studies
Microcapsules were loaded with 500 kDa FITC-dextran by incubating overnight in 5 mg/ml solution in a 12-well plate, then transferred to phosphate-buffered saline (PBS) in a 6-well plate. Fluorescence intensity inside the microcapsule compartments was monitored by acquiring images at 0, 5, 10, 20, 30, 60, 120, 180, and 240 min, followed by every 4 h of acquisition for a total of 24 h using Olympus IX83 fluorescence microscope. The average fluorescence intensity was measured and normalized with the value from 0 min via ImageJ software.
Cell maintenance and encapsulation
Hepatic cells (HepG2) were maintained in Dulbecco’s modified Eagle medium (DMEM; Gibco, 10-013-CV), supplemented with 10% (v/v) fetal bovine serum, and 0.5% penicillin-streptomycin. Pancreatic cells (MIN6) were maintained in Dulbecco’s modified Eagle medium (10-013-CV), supplemented with 15% (v/v) fetal bovine serum (FBS), 0.5 mM 2-mercaptoethanol (2-ME), and 0.5% penicillin-streptomycin (PS). For monoculture conditions, each cell line was maintained in its respective medium. Liver-pancreas co-cultures were maintained in pancreatic cell media described above.
Encapsulation was performed at the flow rates as follows: (1) Core—0.5 μL/min; (2) Shell—5 μL/min; (3) Shielding oil—20 μL/min; (4) Crosslinking Oil—60 μL/min. For HepG2 encapsulation, cells were detached and resuspended in a core solution at a concentration of 3 × 107 cells/ml. For MIN6 encapsulation, cells were resuspended at a concentration of 5 × 107 cells/ml. Capsules were collected for 10 minutes in a 15 ml conical tube containing 10 ml of culture media. The capsules were transferred to 100 μm filter and washed with the media to remove the oil residues. They were subsequently transferred to a 6-well plate with 2 ml of media per well. The capsules were cultured in an incubator at 37 °C with 5% CO2. Media was collected and renewed every 48 hours.
Cell viability and spheroid formation
For live/dead staining, microcapsules containing cells or spheroids were incubated with calcein-AM (green) and ethidium bromide (red) for 30 minutes at 37 °C with 5% CO2. They were imaged with the inverted fluorescence microscope. Cell viability was measured by the live/dead ratio represented by green/red fluorescent intensity. Quantification was done by ImageJ software. Detailed protocol to assess viability is described in a previous study16.
Glucose-stimulated insulin secretion (GSIS) assay
Two buffer solutions (500 ml) were prepared with final concentrations of 16 mM and 4 mM D-(+)-glucose, respectively. Both buffers contained the following components: 5 M NaCl, 3 M KCl, 1 M CaCl2·2H2O, 1 M MgCl2·6H2O, 1 M HEPES, 1.25 g bovine serum albumin, and 1 g NaHCO3.
On day 5, 20 capsules were collected from the batch for each replicate from each group. They were then initially incubated in 750 μL of 4 mM glucose buffer at 37 °C for 1 hour for washing. The capsules were then transferred to a fresh 750 μL aliquot of 4 mM glucose buffer and incubated for an additional hour at 37 °C. After incubation, the buffer was collected for analysis. Subsequently, the capsules were transferred to 750 μL of 16 mM glucose buffer and incubated at 37 °C for 1 hour. This buffer was also collected. All collected buffer samples were analyzed for insulin concentration using an ELISA kit, according to the manufacturer’s instructions.
Analysis of hepatic function
Secretion of albumin and urea was analyzed via their respective kits, following the manufacturer’s instructions. Briefly, for the albumin assay, culture media was collected and diluted as necessary to fall within the linear range of the standard curve with the absorbance measured at 450 nm. For the urea assay, samples were similarly processed and reacted with the provided reagents, with absorbance measured at 520 nm. Concentrations of albumin and urea were calculated based on standard curves generated from known concentrations prepared.
Figure generation
Schematics and figures were created using Adobe Illustrator 2025 (Adobe Inc., San Jose, CA, USA), with a micropipette-multi icon from Servier (https://smart.servier.com/), licensed under CC-BY 3.0 Unported (https://creativecommons.org/licenses/by/3.0/)
Statistical analysis
Data are presented as mean ± standard deviation from a minimum of three independent experimental replicates. GSIS result, however, is presented as mean ± standard error of mean. Student’s t test was employed to determine the statistical significance between groups. Significance levels were denoted as follows: *p <0.05, **p <0.01, ***p <0.001, ****p <0.0001.
Results and discussion
Fabricating multicompartment hydrogel microcapsules with aqueous cores
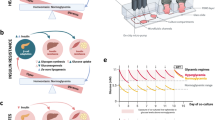
The objective of this study was to create 3D pancreas-liver co-cultures for disease modeling and drug development. The study builds on previous work from our team and others fabricating microcapsules with hydrogel shell and aqueous core. When cells are loaded into such microcapsules, they retain viability and function by rapidly aggregating into 3D structures or spheroids. While there are multiple reports describing hydrogel microcapsules with one aqueous core9,15,16,31,32, microcapsules carrying multiple aqueous cores have not been reported to the best of our knowledge. Figure 1 describes the microfluidic strategy developed by us to fabricate microcapsules with multiple cores. As shown in Fig. 1a–c, the microfluidic encapsulation device consisted of two aqueous core and two aqueous shell streams that converged on shielding oil junction where droplet generation occurs. Droplets then experienced another oil stream carrying a di-thiol crosslinker, DTT. The two oil junction design allowed for shell to fully wrap around the cores prior to cross-linking reaction that created a hydrogel. The crosslinking reaction involved a combination of Click chemistry reaction of maleimide with thiol moieties as well as Michael type addition reaction with acrylamide moieties (see Fig. 1e). In the past studies describing microcapsules with a single core, we deployed crosslinkable PEG prepolymer in the shell stream and noncrosslinkable densifying components in the core stream. We found this strategy to be suboptimal in the multicompartment format due to mixing of core and shell flow streams that resulted in cross-contamination of the cores. To remedy this, we implemented an aqueous two-phase system (ATPS) strategy where core solutions contained dextran (DEX) while shell stream still carried crosslinkable PEG molecules. The core and shell phases were formulated with 15% (w/v) DEX (500 kDa) and 10% (w/v) PEG (10 kDa), respectively—concentrations known to reliably induce phase separation16,33. Poor miscibility of PEG and DEX phases allowed us to create distinct compartments with minimal cross-contamination. This point is highlighted by Fig. 1d which shows a train of 300 µm diameter droplets, each droplet containing two distinct cores (120 µm diameter) carrying red and green dye.
a A schematic demonstration of encapsulation with specific dimensions for each domain of channels. b An image of microfluidic device with food dyes incorporated in core and shell channels. c Visual representation of microcapsule formation with each channel labeled. d Visual representation of microcapsule focusing on the droplet formation region. e Schematic that describes the chemistry of the hydrogel-forming crosslinking reaction between prepolymer molecules in the shell and the crosslinking agent
In the downstream collection phase, the geometry of the oil channel was also optimized to preserve core compartmentalization. In earlier designs, a serpentine channel was positioned immediately downstream of the droplet formation zone to enhance mixing and promote homogeneous shell gelation30. However, this configuration introduced turbulence, which increased the likelihood of core coalescence. To address this issue, the serpentine channel was relocated to the terminal segment of the device, allowing sufficient time for shell stabilization while minimizing shear-induced disturbances to the encapsulated cores.
To enable shell gelation and effective core isolation within each microcapsule, we incorporated 8-arm polyethylene glycol-maleimide (8ARM-PEG-Mal) and acrylamide-PEG-acrylamide (ACA-PEG-ACA) into the shell solution. This polymer mixture rapidly crosslinks into a hydrogel upon exposure to DTT via thiol–Michael addition chemistry (Fig. 1e). The shell solution was further optimized by adjusting the pH with 1 M triethanolamine (TEA) and adding ethanol to reduce surface tension, thus facilitating uniform crosslinking across the droplet interface.
Control of flow parameters allows to tune microcapsule geometry
After converging on the shell and core composition, we systematically characterized the flow parameters within the microfluidic device to identify optimal conditions for generating uniform multicompartment microcapsules. We focused specifically on evaluating the flow rates of the core, shell, and shielding oil phases, as these directly influence capsule geometry, compartmentalization, and droplet formation dynamics. The flow rate of the crosslinking oil was fixed at 60 μL/min as referenced in previous studies33.
For the shielding oil phase, we observed an inverse relationship between flow rate and capsule diameter (p < 0.05) within the range of 20–60 μL/min (Fig. 2a). At lower flow rates (e.g. 10 μL/min), the shear force was not enough to break the core and shell streams, resulting in a jetting phase. The dripping phase began consistently at 20 μL/min for the shielding oil. The average capsule diameter ranged from 375 ± 24 µm at 10 µL/min to 280 ± 21 µm at 60 μL/min. Increasing the shell flow rate produced larger capsules with diameters ranging from 358 ± 20 µm at 2 μL/min to 434 ± 21 µm at 8 μL/min (Fig. 2b). Similarly, for the core phase, we observed a direct relationship between flow rate and core diameter which ranged from 87 ± 22 µm for 0.125 µL/min to 132 ± 24 for 0.5 µL/min (Fig. 2c).
a The diameter of microcapsules under varying oil flow rates; the size of microcapsules decreases with an increasing oil flow rate (n ≥ 3). The red region and blue region indicate jetting and dripping phase, respectively. b The diameter of microcapsules under varying shell flow rates; the size of microcapsules increases with increasing shell flow rate (n = 3). c The diameter of core under varying core flow rates; the core size increases with increasing core flow rate (n ≥ 3). The yellow and green region indicate dual and single core, respectively. d Images microcapsules fabricated with varying ratios of core 1 to core 2 flow rates. e The core volumes for varying core flow rates (n = 3). Data for a, b, c, and e are presented as mean ± standard deviation and statistically evaluated by unpaired, one-tailed, equal-variance Student’s t test, with each group compared to the preceding experimental group. (NS: not significant, *p <0.05, **p <0.01, ***p <0.001)
An important optimization parameter was the flow rate at which two discrete cores could be produced. We found that multicompartment capsules could be fabricated at flow rates of up to 0.5 μL/min for core 1 and core 2. Exceeding this threshold (where sum of core 1 and core 2 flow rates was >1 μL/min) led to mixing of the two cores and production of single-core capsule (see Fig. 2c).
After testing these flow parameters, we calculated the coefficient of variation (CV) to determine the mono-dispersity of microcapsules. Shielding oil flow rates ranging between 20 and 40 μL/min yielded CVs below 5%, indicating uniform capsule sizes. Similarly, shell flow rates from 4 to 8 μL/min maintained CVs under 5%. These demonstrate the consistent performance of our microfluidic design across a range of flow parameters. However, core size yielded greater variability of CV around 15%. This is likely a consequence of ATPS, which has been reported for its ultra-low interfacial tension hindering droplet uniformity34. Despite this, we decided to continue with our strategy, as core flow rates gave us significant control over core size (p < 0.05) (Fig. 2c).
Based on these results, we decided to use the following flow rates for subsequent experiments: 5 μL/min (core), 0.5 μL/min (shell), 20 μL/min (shielding oil), and 60 μL/min (crosslinking oil). At a fixed shielding oil and shell flow rate, the core size is determined by the flow rate of the core. We incorporated red and green-fluorescent beads into separate core solutions and then tested the following ratios of core 1 to core 2 flow rate: 0.7:0.3, 0.5:0.5, and 0.3:0.7 μL/min (red:green). Fluorescent images demonstrate that by controlling the core flow rates, the volumes of core 1 and core 2 could be adjusted within each microcapsule (Fig. 2d). Quantification of core volume from the capsule images showed that 0.3, 0.5, and 0.7 μL/min of core flow rate corresponded to 0.44 ± 0.10, 0.83 ± 0.12, and 1.36 ± 0.14 nL, respectively (Fig. 2e). This highlights the tunability of core volume within each microcapsule. Such capability may be valuable for asymmetric loading of the two cell types into microcapsules where one may ask how local population size affects heterotypic crosstalk.
Evaluating cell loading, viability and functionality in multicompartment microcapsules
Following the optimization of flow parameters, we evaluated cell loading, survival, and function in multicompartment microcapsules. Given that the cores of our microcapsules contained high molecular weight dextran, we first wanted to assess how fast these macromolecules diffuse out for the core to become aqueous.
The capsules were incubated overnight in 500 kDa FITC-dextran, then transferred into PBS solution and monitored over time using a fluorescence microscope. This experiment revealed that fluorescence intensity due to dextran in the core decreased 2-fold with 6 h and that dextran was replaced by water molecules within 24 h (Fig. S1).
Next, we encapsulated hepatic cells (HepG2—human hepatoma cells) in multicompartment microcapsules and cultured them for 7 days. Live/dead staining revealed that encapsulated cells were viable, with 84.4 ± 7.2% viability at day 0 and 93.8 ± 6.3% viability at day 5 (Fig. 3a, b). Importantly, cells aggregated into spheroids within 72 h of encapsulation and maintained tight spheroids throughout the culture period (see Fig. 3f). This highlighted the fact that cells were free to move within the core and supported the observation of dextran diffusion and establishment of aqueous cores inside each microcapsule. In fact, as shown in Fig. 3c, spheroid diameter increased from 56.6 ± 1.60 μm on day 1 to 123.0 ± 4.68 μm on day 7. The hepatic function of encapsulated cells was assessed by analyzing albumin, a serum protein produced by the liver, and urea, a byproduct of ammonia detoxification performed by the liver23,35. The results, presented in Fig. 3d and Fig. 3e demonstrate that cells remained functional over the course of 7 days.
a Live/dead fluorescence image at day 0 and 5. b Quantification of cell viability at day 0 and 5. c Cell spheroid diameter increases over time in microcapsules (n = 3). d Urea production by encapsulated hepatic cells remains stable over time, pointing to hepatic phenotype maintenance (n = 3). e Albumin secretion increases over time, likely due to increasing cell number/spheroid size (n = 3). f Two groups of hepatic cells were labeled with either red or purple cell tracker dyes and then included in the core 1 and core 2 solutions. The image shows individual microcapsules carrying spatially distinct groups of red and purple cells at day 3 of culture. Data for (b), (c), (d), and (e) are presented as mean ± standard deviation. c, d and e are statistically evaluated by unpaired, one-tailed, equal-variance Student’s t test, with each group compared to the preceding experimental group for c and day 1 compared to day 3, 5, and 7 for (d) (NS: not significant, *p <0.05, **p <0.01, ***p <0.001, and ****p <0.001)
For spatial control of co-cultures to be successful, the contents of individual cores need to remain distinct during the encapsulation process. This was shown to be the case for microbeads that were labeled with different fluorophores and loaded into distinct cores of a microcapsule (see Fig. 2d). Here we wanted to test whether the same will hold true for cells. Hepatic cells were divided into two groups, one group labeled with red cell tracker dye and another with purple cell tracker dye. Cells were loaded into syringes driving flow streams for core 1 and core 2 in the encapsulation device. Fluorescence images collected at day 3 of culture revealed that each capsule contained distinct spheroids emitting red and purple fluorescence (Fig. 3F). This confirmed that core materials indeed remain segregated during encapsulation, allowing independent formation of spheroids of each cell population.
Collectively, these results demonstrate that our multicompartment hydrogel microcapsules support high cell viability, sustained hepatic functionality, and effective spatial segregation of distinct cell populations within a single capsule.
Creating spatially patterned liver-pancreas co-cultures in microcapsules and assessing their crosstalk
As the next step we proceeded to encapsule pancreatic β-cells (MIN6 – a mouse insulinoma cell line) alongside hepatic cells in the same multicompartment microcapsules. In addition to creating co-cultures, we also wanted to evaluate whether presence of hepatic cells in microcapsules improves functionality of neighboring pancreatic cells and vice versa. To achieve this goal, we established three experimental groups: 1) liver-pancreas co-culture microcapsules, 2) hepatic mono-cultures (one hepatic spheroid per capsule), and 3) pancreatic mono-cultures (one islet spheroid per capsule). All groups of microcapsules were cultured for 5 days in pancreatic culture media (Fig. 4a).
a Schematic describing experiment setup. b Live/dead fluorescence images on day 0 and 5 for pancreatic monoculture and coculture groups. c Quantification of cell viability on day 5 for each cell type. d Albumin synthesis in hepatic monoculture and coculture groups over time (n = 3). e Insulin secretion analysis for co-culture and pancreatic mono-culture microcapsule groups (n ≥ 6). f Stimulation index calculated based on insulin production at low and high glucose concentrations (n ≥ 6). Data for c and d are presented as mean ± standard deviation. Data for e and f are presented as mean ± standard error of mean. Values were statistically evaluated by unpaired, two-tailed, equal-variance Student’s t test. Day 5 data for (d) was compared between mono and co-culture groups (NS: not significant, *p <0.05, **p <0.01)
Cell viability was assessed via live/dead staining immediately after encapsulation and on day 5. To distinguish between cell populations, hepatic cells were labeled with a purple cell tracker dye (Cy5 emission) prior to encapsulation (Fig. S2). This way, hepatic spheroids could be visually distinguished from pancreatic spheroids. The cell tracker dye did not interfere with ethidium homodimer staining (dead stain emitted at 617 nm, compared to the cy5 tracker dye emitting at 665 nm). Shown in Fig. 4b, viability was high immediately after encapsulation and remained above 95% for pancreatic monoculture and co-culture microcapsules at day 5 (see Fig. 4b, c).
As the next step, we evaluated hepatic and pancreatic function in microcapsules. First, we investigated how the presence of pancreatic β-cells affects functionality of liver cells. Anatomically, the pancreas is directly connected to the liver via portal circulation and signals to the liver to store or release glucose with pancreatic hormones (e.g. insulin and glucagon). ELISA analysis revealed that microcapsules containing pancreatic as well as hepatic spheroids produced 2-fold higher levels of albumin (hepatic function indicator produced by hepatic cells but not pancreatic cells) after 5 days of culture compared to microcapsules carrying only hepatic spheroids (Fig. 4d). It is worth noting that both mono-culture and co-culture microcapsules were cultured in the same media (see “Materials and Methods” for details). This result suggested that hepatic cells benefited from presence of pancreatic cells and aligned with prior reports of hepatic albumin production being positively regulated by pancreatic hormones such as insulin36,37.
Next, we evaluated pancreatic β-cell function in liver-pancreas co-culture microcapsules as well as in pancreas mono-culture capsules. The function was evaluated using glucose stimulated insulin secretion (GSIS) assay, where cells are sequentially exposed to low (4 mM) and high (16 mM) glucose with concomitant insulin measurements. The GSIS assay was carried out after 5 days of culture. The results revealed that insulin secretion at the basal glucose concentration (4 mM) was more than two times lower in the co-culture condition compared to the pancreatic monoculture (p < 0.01) (Fig. 4e). This may mean that hepatic spheroids play a role in modulating basal insulin secretion of pancreatic spheroids. This regulation is as important as the insulin response to high glucose, as proper β-cell function requires minimal insulin secretion under normoglycemic level (i.e. 4 mM) followed by robust secretion when exposed to elevated glucose level (i.e. 16 mM).
Moreover, Fig. 4f showed that pancreas-liver co-cultures produced significantly higher levels of insulin and had 2.4-fold higher stimulation index compared to microcapsules containing only pancreatic cells.
Our findings point to enhanced function of hepatic and pancreatic spheroids co-encapsulated and cultured in common media. These findings are exciting as they show that each cell type contributes to improved function of another, highlighting the possibility for creating functional and physiological synergisms through co-encapsulation of multiple cell types.
Multiple avenues may be pursued in the future to further enhance technological aspects of the multicompartment capsules. The hydrogel composition around each compartment may be tuned to control exchange of secreted signals based on size, through porosity, or by incorporating ligand traps to eliminate specific signals38. It may also be beneficial to detect secretion of signals locally, inside microcapsules. This may be achieved by incorporating bead-based immune-sensing strategies into one compartment39 and cells into others. This development would enable local detection of signaling molecules within each microcapsule.
Conclusions
In this work, we describe a droplet microfluidic-based strategy for fabricating hydrogel microcapsules carrying multiple compartments or cores. By optimizing flow parameters, we achieved precise and stable generation of capsules with distinct cores where different cell types could be incorporated. With these microcapsules, we successfully encapsulated hepatic and pancreatic cells, demonstrating successful loading, high viability, and rapid spheroid formation inside multicompartment microcapsules. Importantly, we demonstrate that liver-pancreas microcapsules exhibit better function (both hepatic and pancreatic) compared to mono-culture microcapsules. This points to functional synergism arising from co-encapsulating of both cell types. Microcapsules carrying spatially organized co-cultures may represent an exciting alternative to microfluidics-based microphysiological systems and may, in the future, be used for applications ranging from disease modeling to development biology and drug testing. We also foresee that microcapsule complexity may be increased further by incorporating addition compartments or cores for co-culture of three or more cell types.
References
Ferdous, S.-E. & Ferrell, J. M. Pathophysiological relationship between type 2 diabetes mellitus and metabolic dysfunction-associated steatotic liver disease: novel therapeutic approaches. Int. J. Mol. Sci. 25, https://doi.org/10.3390/ijms25168731 (2024).
Mantovani, A., Byrne, C. D., Bonora, E. & Targher, G. Nonalcoholic fatty liver disease and risk of incident type 2 diabetes: a meta-analysis. Diab. Care 41, 372–382, https://doi.org/10.2337/dc17-1902 (2018).
Bauer, S. et al. Functional coupling of human pancreatic islets and liver spheroids on-a-chip: towards a novel human ex vivo type 2 diabetes model. Sci. Rep. 7, 14620–14620, https://doi.org/10.1038/s41598-017-14815-w (2017).
Zandi Shafagh, R. et al. Bioengineered pancreas–liver crosstalk in a microfluidic coculture chip identifies human metabolic response signatures in prediabetic hyperglycemia. Adv. Sci. 9, https://doi.org/10.1002/advs.202203368 (2022).
Ronaldson-Bouchard, K. et al. A multi-organ chip with matured tissue niches linked by vascular flow. Nat. Biomed. Eng. 6, 351–371, https://doi.org/10.1038/s41551-022-00882-6 (2022).
Alkayyali, T., Cameron, T., Haltli, B., Kerr, R. G. & Ahmadi, A. Microfluidic and cross-linking methods for encapsulation of living cells and bacteria - a review. Anal. Chim. Acta 1053, 1–21, https://doi.org/10.1016/j.aca.2018.12.056 (2019).
Kang, S.-M., Lee, J.-H., Huh, Y. S. & Takayama, S. Alginate microencapsulation for three-dimensional in vitro cell culture. ACS Biomater. Sci. Eng. 7, 2864–2879, https://doi.org/10.1021/acsbiomaterials.0c00457 (2021).
Mundel, P., Reiser, J. & Kriz, W. Induction of differentiation in cultured rat and human podocytes. J. Am. Soc. Nephrol. 8, 697–705 (1997).
Agarwal, P. et al. One-step microfluidic generation of pre-hatching embryo-like core–shell microcapsules for miniaturized 3D culture of pluripotent stem cells. Lab Chip 13, 4525–4533 (2013).
Bah, M. G., Bilal, H. M. & Wang, J. Fabrication and application of complex microcapsules: a review. Soft Matter 16, 570–590, https://doi.org/10.1039/c9sm01634a (2020).
Headen, D. M., García, J. R. & García, A. J. Parallel droplet microfluidics for high throughput cell encapsulation and synthetic microgel generation. Microsyst. Nanoeng. 4, 17076. https://doi.org/10.1038/micronano.2017.76 (2018).
Chen, T. et al. Alginate encapsulant incorporating CXCL12 supports long-term allo- and xenoislet transplantation without systemic immune suppression. Am. J. Transpl. 15, 618–627, https://doi.org/10.1111/ajt.13049 (2015).
Bochenek, M. A. et al. Alginate encapsulation as long-term immune protection of allogeneic pancreatic islet cells transplanted into the omental bursa of macaques. Nat. Biomed. Eng. 2, 810–821, https://doi.org/10.1038/s41551-018-0275-1 (2018).
Dhawan, A. et al. Alginate microencapsulated human hepatocytes for the treatment of acute liver failure in children. J. Hepatol. 72, 877–884, https://doi.org/10.1016/j.jhep.2019.12.002 (2020).
Kim, M., Lee, J. Y., Jones, C. N., Revzin, A. & Tae, G. Heparin-based hydrogel as a matrix for encapsulation and cultivation of primary hepatocytes. Biomaterials 31, 3596–3603, https://doi.org/10.1016/j.biomaterials.2010.01.068 (2010).
Gwon, K. et al. Bioactive hydrogel microcapsules for guiding stem cell fate decisions by release and reloading of growth factors. Bioact. Mater. 15, 1–14, https://doi.org/10.1016/j.bioactmat.2021.12.008 (2022).
Siltanen, C. et al. One step fabrication of hydrogel microcapsules with hollow core for assembly and cultivation of hepatocyte spheroids. Acta Biomater. 50, 428–436, https://doi.org/10.1016/j.actbio.2017.01.010 (2017).
Fattahi, P. et al. Core–shell hydrogel microcapsules enable formation of human pluripotent stem cell spheroids and their cultivation in a stirred bioreactor. Sci. Rep. 11, 7177. https://doi.org/10.1038/s41598-021-85786-2 (2021).
Klee, P. et al. Connexins protect mouse pancreatic β cells against apoptosis. J. Clin. Investig. 121, 4870–4879, https://doi.org/10.1172/JCI40509 (2011).
Vinken, M. et al. Involvement of cell junctions in hepatocyte culture functionality. Crit. Rev. Toxicol. 36, 299–318, https://doi.org/10.1080/10408440600599273 (2006).
Wu, F. J. et al. Efficient assembly of rat hepatocyte spheroids for tissue engineering applications. Biotechnol. Bioeng. 50, 404–415, https://doi.org/10.1002/(sici)1097-0290(19960520)50 (1996). 4<404::Aid-bit7>3.0.Co;2-p.
Brophy, C. M. et al. Rat hepatocyte spheroids formed by rocked technique maintain differentiated hepatocyte gene expression and function. Hepatology 49, 578–586, https://doi.org/10.1002/hep.22674 (2009).
Choi, J. H. et al. Microfluidic confinement enhances phenotype and function of hepatocyte spheroids. Am. J. Physiol.-Cell Physiol. 319, C552–C560, https://doi.org/10.1152/ajpcell.00094.2020 (2020).
Taroncher, M. et al. Using microfluidic hepatic spheroid cultures to assess liver toxicity of T-2 mycotoxin. Cells 13, https://doi.org/10.3390/cells13110900 (2024).
Diane, A., Mohammed, L. I. & Al-Siddiqi, H. H. Islets in the body are never flat: transitioning from two-dimensional (2D) monolayer culture to three-dimensional (3D) spheroid for better efficiency in the generation of functional hPSC-derived pancreatic β cells in vitro. Cell Commun. Signal. 21, 151–151, https://doi.org/10.1186/s12964-023-01171-8 (2023).
Mi, S. et al. Microfluidic co-culture system for cancer migratory analysis and anti-metastatic drugs screening. Sci. Rep. 6, 35544–35544, https://doi.org/10.1038/srep35544 (2016).
March, S. et al. Micropatterned coculture of primary human hepatocytes and supportive cells for the study of hepatotropic pathogens. Nat. Protoc. 10, 2027–2053, https://doi.org/10.1038/nprot.2015.128 (2015).
Weber, L. M., Hayda, K. N. & Anseth, K. S. Cell-matrix interactions improve beta-cell survival and insulin secretion in three-dimensional culture. Tissue Eng. Part A 14, 1959–1968, https://doi.org/10.1089/ten.tea.2007.0238 (2008).
Zheng, Y., Wu, Z., Lin, J.-M. & Lin, L. Imitation of drug metabolism in cell co-culture microcapsule model using a microfluidic chip platform coupled to mass spectrometry. Chin. Chem. Lett. 31, 451–454, https://doi.org/10.1016/j.cclet.2019.07.036 (2020).
Chairez-Cantu, K., González-González, M. & Rito-Palomares, M. Generation of polyethylene glycol-dextran aqueous two-phase system droplets using different culture media under in vitro conditions. Food Bioprod. Process. 139, 157–165, https://doi.org/10.1016/j.fbp.2023.03.010 (2023).
Watanabe, T., Motohiro, I. & Ono, T. Microfluidic formation of hydrogel microcapsules with a single aqueous core by spontaneous cross-linking in aqueous two-phase system droplets. Langmuir ACS J. Surf. colloids 35, 2358–2367, https://doi.org/10.1021/acs.langmuir.8b04169 (2019).
Nguyen, D. K., Son, Y. M. & Lee, N. E. Hydrogel encapsulation of cells in core-shell microcapsules for cell delivery. Adv. Healthc. Mater. 4, 1537–1544, https://doi.org/10.1002/adhm.201500133 (2015).
Seo, H., Nam, C., Kim, E., Son, J. & Lee, H. Aqueous two-phase system (ATPS)-based polymersomes for particle isolation and separation. ACS Appl. Mater. Interfaces 12, 55467–55475, https://doi.org/10.1021/acsami.0c16968 (2020).
Mastiani, M., Firoozi, N., Petrozzi, N., Seo, S. & Kim, M. Polymer-salt aqueous two-phase system (ATPS) micro-droplets for cell encapsulation. Sci. Rep. 9, 15561–15561, https://doi.org/10.1038/s41598-019-51958-4 (2019).
de Hoyos-Vega, J. M. et al. Microfluidic 3D hepatic cultures integrated with a droplet-based bioanalysis unit. Biosens. Bioelectron. 248, 115896. https://doi.org/10.1016/j.bios.2023.115896 (2024).
Chen, Q., Lu, M., Monks, B. R. & Birnbaum, M. J. Insulin is required to maintain albumin expression by inhibiting forkhead box O1 protein. J. Biol. Chem. 291, 2371–2378, https://doi.org/10.1074/jbc.M115.677351 (2016).
Flaim, K. E. et al. Direct effect of insulin on albumin gene expression in primary cultures of rat hepatocytes. Am. J. Physiol.-Endocrinol. Metab. 249, E447–E453, https://doi.org/10.1152/ajpendo.1985.249.5.E447 (1985).
Patel, D. et al. Microfluidic co-cultures with hydrogel-based ligand trap to study paracrine signals giving rise to cancer drug resistance. Lab Chip 15, 4614–4624, https://doi.org/10.1039/c5lc00948k (2015).
Son, K. J., Gheibi, P., Stybayeva, G., Rahimian, A. & Revzin, A. Detecting cell-secreted growth factors in microfluidic devices using bead-based biosensors. Microsyst. Nanoeng. 3, https://doi.org/10.1038/micronano.2017.25 (2017).
Acknowledgements
We thank Dr. Daheui Choi and Dr. Diana Cedillo Alcantar for their assistance with imaging and photolithography. We also acknowledge Eugene Kruger from the Microscopy and Microfluidic Core of the Mayo Center for Cell Signaling in Gastroenterology (C-SiG) for support with imaging. We are grateful to Roberto Leon-Gutierrez and Dr. Naureen Javeed for providing the MIN6 cell line. This work was supported in part by the NIH grants P30DK084567 and R01DK137231 and by Mayo Clinic Foundation.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
A.R. is an Editor for the journal; no other author declares competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cho, S., Nguyen, Q.H., de Hoyos-Vega, J.M. et al. Multicompartment hydrogel microcapsules for creating spatially patterned cell co-cultures. Microsyst Nanoeng 12, 25 (2026). https://doi.org/10.1038/s41378-025-01140-w
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41378-025-01140-w