Abstract
Dysregulated protein synthesis has been implicated in multiple neurodevelopmental, neurodegenerative, and neuropsychiatric diseases. Protein synthesis or mRNA translation is critically regulated through phosphorylation of eukaryotic elongation factor 2 (eEF2) by its kinase eEF2K. Increased eEF2K activity leads to elevated phosphorylation and inhibition of eEF2 and inhibits the elongation phase of protein synthesis. Recent studies suggest a link between eEF2 hyper-phosphorylation and several neuronal diseases characterized by cognitive impairments. Phosphorylation of eEF2 by eEF2K has also been implicated as a molecular mechanism for the rapid antidepressant effect of ketamine. Whether there exists a causal relationship between overactive eEF2K/eEF2 signaling and impaired synaptic and cognitive function remains unknown. To fill this critical knowledge gap, we generated a transgenic mouse model (eEF2K-cKI) overexpressing eEF2K in excitatory neurons to investigate how eEF2K/eEF2 signaling can impact cognitive functions and neuropsychiatric behaviors. We assessed hippocampal-dependent learning and memory, as well as multiple neuropsychiatric domains associated with a depressive phenotype including despair, anhedonia, apathy, anxiety, and sociability. The eEF2K-cKI mice exhibit learning and memory impairments, and robust apathy-like phenotype without other despair/depression-like behaviors. We also found impaired long-term potentiation and altered dendritic spine and synaptic morphology in the hippocampus of the eEF2K-cKI mice. Proteomic analysis revealed changes in levels of proteins associated with neuropsychiatric and neurodegenerative disorders. Our findings present direct evidence supporting the pathophysiological role of aberrant eEF2K/eEF2 signaling in brain function and help provide insight into novel mechanisms and therapeutic avenues for neuronal diseases characterized by dementia and neuropsychiatric symptoms.
Similar content being viewed by others
Introduction
Dysregulated mRNA translation (protein synthesis) has been implicated in multiple neurodevelopmental, neurodegenerative, and neuropsychiatric diseases such as Down Syndrome (DS), Alzheimer’s disease (AD), and depression [1,2,3,4,5,6]. Protein synthesis is a highly regulated process that involves numerous signaling pathways and translation factors particularly regulating the initiation and elongation stages [7, 8]. This regulation allows for complex changes to the proteome and increases in specific mRNAs in response to different cellular environments [9,10,11,12]. While the initiation phase is considered as the “rate-limiting step” and draws the most attention in studying translational control, accumulating evidence indicates that regulation of elongation plays a critical role in the protein synthesis process during cellular responses to deficiency of energy and nutrients [13]. Furthermore, the energy and amino acids used in protein synthesis are mostly ( > 95%) consumed during the elongation phase [14, 15]. One critical mechanism for elongation regulation is through the eukaryotic elongation factor 2 kinase (eEF2K). eEF2K is a Ca2+/calmodulin-dependent α-kinase and acts specifically to phosphorylate its only known substrate eukaryotic elongation factor 2 (eEF2) at T56 and inhibit its activity and thus protein synthesis [15]. eEF2 is a GTPase that catalyzes the translocation of the tRNA from the A site to the P site in the ribosome facilitating the growing peptide chain and regulates the elongation step of protein synthesis [15]. Activated eEF2K increases eEF2 phosphorylation and inhibits the elongation step of protein synthesis [15]. This process is associated with the nutrient and energy state of the cell via its regulation by the mammalian target of rapamycin complex 1 (mTORC1) and AMP-activated protein kinase (AMPK) [16]. mTORC1 is a nutrient sensor and is inhibited when nutrients like amino acids, the building blocks of proteins, are low [17]. mTORC1 signaling is responsible for phosphorylation of multiple inhibitory sites on eEF2K either directly by mTORC1 or by its downstream substrates such as ribosomal protein S6 kinase 1 [15, 16]. AMPK is an energy sensory that is activated by low energy levels [18, 19]. It interacts with the mTORC1 pathway at multiple points to affect protein synthesis. AMPK can activate TSC2, an upstream inhibitor of mTORC1 [20]. AMPK inhibits Raptor, an mTORC1 constituent, leading to inactivation of mTORC1 signaling [21]. AMPK can also directly phosphorylate and activate eEF2K [22,23,24]. Overall, eEF2K is activated under conditions of cellular stress [11, 25,26,27,28].
Regulation of protein synthesis may be a crucial aspect of major depressive disorder (MDD) and other mood disorders. A recent study found that anisomycin, a general protein synthesis inhibitor, induced depressive-like behaviors in mice [29]. Additionally, increased eIF2α phosphorylation and decreased eIF2B, i.e. inhibition of translation initiation, was also linked to depressive-like behaviors in humans and mice [29]. Another recent study reported altered tRNA dynamics in suicide (MDD associated) brain [30]. Notably, recent studies have implicated a role of eEF2K signaling regulation in mediating the antidepressant effects of ketamine [6, 31]. Ketamine may exert its antidepression effects by inhibiting eEF2K and eEF2 phosphorylation, leading to an increase in translation [6, 31]. Genetic deletion of eEF2K abolishes ketamine’s antidepressant effect [31]. In agreement with these findings, eEF2K overactivation and eEF2 hyper-phosphorylation, as well as decreased protein synthesis, have been linked to MDD [1, 32]. Therefore, we hypothesize that increased eEF2K expression and eEF2 phosphorylation could promote a depression/despair-like phenotype as well as learning and memory deficits.
Here, we generated a novel transgenic mouse model with neuron-specific eEF2K overexpression to investigate whether and how overactive eEF2K/eEF2 signaling in the mature brain can impact psychological and cognitive functions. We assessed multiple neuropsychiatric domains associated with a depression-like phenotype including despair, anhedonia, apathy, anxiety, and sociability. We also assessed hippocampal-dependent learning and memory and evaluated the role of eEF2K in synaptic plasticity and synaptic function and structure. In addition, we performed a proteomic analysis to identify proteins dysregulated by eEF2K overexpression to better understand how eEF2K signaling might contribute to the synaptic and behavioral changes.
Results
Generation of the eEF2K overexpression mouse model
To understand the role of eEF2K and eEF2 phosphorylation in mammalian neurons in regulating synaptic and cognitive function, we generated a conditional eEF2K knock-in model (referred as eEF2K-cKI hereafter). Briefly, we first created a line of transgenic mouse with Eef2K knock-in flanked by a loxSTOPlox cassette using the CRISPR/Cas9-mediated genome editing combined with a Rosa26 knock-in strategy [33] (Supplemental Fig. 1A). We then crossed these mice with the transgenic mice expressing a neuron-specific Cre recombinase (Camk2a-cre) [34] to remove the STOP cassette and thus induce conditional overexpression of eEF2K in excitatory neurons in the forebrain and hippocampus (Supplemental Fig. 1B). Overexpression of eEF2K was confirmed by both the mRNA and protein levels measured by RT-PCR and western blot respectively (Supplemental Figs. 1C and D). As expected, an increased eEF2K expression resulted in a significant increase in eEF2 phosphorylation at the T56 site (Supplemental Fig. 1E). Both male and female mice, 3-5 months old, were used for all experiments.
eEF2K overexpression does not lead to despair-like behavior but instead elicits a robust apathy-like phenotype
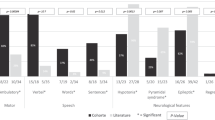
Abnormal translational control is linked to neuropsychiatric disorders and neurodegenerative diseases that are characterized or accompanied by multiple neuropsychiatric symptoms (NPS) [16, 35,36,37]. In preclinical models, depression-like phenotypes are often determined by assessing despair-like behavior (DLB) and anhedonia [38]. We first evaluated the DLB in the eEF2K-cKI mice using the forced swim test (FST) and tail suspension test (TST) [31]. FST immobility is the time spent immobile during the last 4 minutes of the trial, while latency is the first instance of immobility after being placed in the water. In contrast to our hypothesis, there was no difference between the groups in either FST immobility or latency (Fig. 1A, B). The number and duration of bouts was also recorded where each bout is defined as the conversion between mobility and immobility [39]. No differences were noted between groups for FST bouts and the longest bout (Supplemental Figs. 2A and B). Consistent with the FST results, analysis of the TST revealed no differences between the two groups in immobility (Fig. 1C), latency (Fig. 1D), and duration of the longest bout (Supplemental Fig. 2D). However, the eEF2K-cKI mice did exhibit more bouts in TST than the Cre controls (Supplemental Fig. 2C). Z-scores were calculated separately for each behavioral assay for each mouse [37]. A composite z-score was calculated as the average z-score for each mouse from multiple tests for each behavioral domain (i.e. despair-like behavior, apathy-like behavior, anxiety-like behavior). The composite score was used for assessment of overall despair-like behavior and showed no difference between the eEF2K-cKI mice and Cre controls (Fig. 1E). We assessed anhedonia using the sucrose preference test [40]. There was no difference in sucrose preference between eEF2K-cKI mice and Cre controls (Fig. 1F) and both groups had similar total consumption amounts (Supplemental Fig. 2E). Analysis of the z-score demonstrates no anhedonia-like behavior in the eEF2K-cKI mice (Fig. 1G). Therefore, in contrast to our hypothesis, these findings suggest that overactive eEF2K signaling and eEF2 hyper-phosphorylation are not associated with DLB or anhedonia.
(A) Total immobility time in FST was not different between groups. Cre, n = 10; eEF2K-cKI, n = 11. p = 0.3079. (B) Latency to immobility in FST was not significantly different between groups. Cre, n = 10; eEF2K-cKI, n = 11. p = 0.1094. (C) Total immobility time in TST was not different between groups. Cre, n = 10; eEF2K-cKI, n = 10. p = 0.6726. (D) Latency to immobility in TST was not significantly different between groups. Cre, n = 10; eEF2K-cKI, n = 10. p = 0.8638. (E) No significant difference between groups in the combined despair z-score. Cre, n = 10; eEF2K-cKI, n = 11. p = 0.4985. (F) Percent sucrose preference on day 3 was not different between groups. Cre, n = 10; eEF2K-cKI, n = 11. p = 0.2413. (G) There was no significant difference in the anhedonia z-score. Cre, n = 10; eEF2K-cKI, n = 11. p = 0.2413. Error bars represent ± SEM. Student’s T-test.
Anhedonia and DLB are often co-occurring with apathy, such as the case in neurodegenerative diseases [41]. We assessed apathy-like behavior (ALB) using rodent-typical behaviors as previously described [37, 42]. Briefly, mice were subjected to 5 behavioral tasks including nestlet shredding (NS), nest building (NB), marble burying (MB), and burrowing (2 h and overnight). First, we looked at nestlet shredding and nest building behavior. There was a significant decrease in the amount of nestlet shredded in a 30-minute period in the eEF2K-cKI mice (Fig. 2A). Similarly, in the nest building task, the eEF2K-cKI mice made poorer nests with a lower nest score [37, 43], and left more of the nestlet unshredded after an overnight period (Fig. 2B-2D). In the marble burying task, the eEF2K-cKI mice buried fewer marbles than Cre controls (Fig. 2E). We did not observe significant differences between groups in 2 hour and overnight burrowing behavior tests (Figs. 2F and 2G). Finally, analysis of the composite z score revealed that the eEF2K-cKI mice showed a significantly higher apathy composite z-score indicating apathy-like behavior (Fig. 2H). These findings indicate that overactive eEF2K signaling results in apathy-like behavior.
(A) The percent of nestlet shredded in a 30 min period was decreased in the eEF2K-cKI mice. Cre, n = 10; eEF2K-cKI, n = 11. p = 0.0060. (B) Representative images of nests for nest score (C) Nest building score was decreased in the eEF2K-cKI mice. Cre, n = 10; eEF2K-cKI, n = 11. p = 0.0047. (D) The percent of nestlet left unshredded during the nest building task was increased in the eEF2K-cKI mice. Cre, n = 10; eEF2K-cKI, n = 11. p = 0.0160. (E) The number of marbles buried in a 30 min period was decreased in the eEF2K-cKI mice. Cre, n = 10; eEF2K-cKI, n = 11. p = 0.0017. (F) There was no difference in the percent burrowed in the 2 hr burrowing task. Cre, n = 10; eEF2K-cKI, n = 11. p = 0.4232. (G) There was no difference in the percent burrowed in the overnight burrowing task. Cre, n = 10; eEF2K-cKI, n = 11. p = 0.1077. (H) The eEF2K-cKI mice had significantly higher combined apathy z-score compared to cre controls. Cre, n = 10; eEF2K-cKI, n = 11. p = 0.0003. Error bars represent ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001; Student’s T-test.
Changes in social behavior can also be associated with ALB, as well as a depressive phenotype [38, 44]. We evaluated the preference for social interaction and social novelty using the 3-chamber sociability task (3-CST) [45]. The eEF2K-cKI mice did not show any significant differences in the preference for mouse (stranger 1) over an object or in the total interaction time for the sociability portion of the test (Fig. 3A, B). However, when presented with a novel stranger mouse (stranger 2), eEF2K-cKI mice had a lower preference for Stranger 2 than Cre controls (Fig. 3C). The total interaction time for the social novelty portion of the test was not significantly different between groups and both groups spent similar amount of time interacting with a stranger mouse throughout both portions of the test (Figs. 3D and 3E). Such findings suggest that overactive eEF2K signaling and eEF2 phosphorylation leads to a decrease for social novelty preference but not in general sociability.
(A) No change in preference for stranger 1 over an object in the sociability portion of the task. Cre, n = 10; eEF2K-cKI, n = 10. p = 0.4248. (B) No change in total interaction time for both stranger 1 and object in the sociability portion of the task. Cre, n = 10; eEF2K-cKI, n = 10. p = 0.4453. (C) Decrease in stranger 2 preference over stranger 1 in eEF2K-cKI mice in the social novelty portion of the task. Cre, n = 10; eEF2K-cKI, n = 9. p = 0.0195. (D) No change in total interaction time for both stranger 1 and stranger 2 in the social novelty portion of the task. Cre, n = 10; eEF2K-cKI, n = 9. p = 0.6400. (E) No change in the total mouse interaction time (excluding time interacting with object) for both the sociability and social novelty portions of the task. Cre, n = 10; eEF2K-cKI, n = 9. p = 0.4895. Error bars represent ± SEM. *p < 0.05; Student’s T-test.
Moreover, as a series of control experiments, we investigated anxiety-like behavior, which may be sensitive to eEF2K signaling and protein synthesis dysregulation [46, 47]. Anxiety-like behaviors may also confound ALB. In the open field (OF) task there was no significant difference between the eEF2K-cKI mice and the Cre controls for the percent time spent in the periphery (Fig. 4A, B). We performed two additional behavioral tests for evaluation of anxiety-like behavior including the novelty-induced hypophagia (NIH) and novelty suppressed feeding task (NSFT) [40, 48] For NIH, there was no difference in the latency to consume food or the amount consumed between groups in the home cage or in the novel environment, though both groups demonstrated the expected increase in anxiety in the novel environment (Figs. 4C and 4D; Supplemental Figs. 3A and 3B). There was also no difference in the latency to consume in the NSFT between groups (Fig. 4E). In addition, there was no difference in the amount consumed in the NSFT or in the weight change before and after food deprivation (Supplemental Figs. 3C and 3D). Finally, analysis of the composite anxiety z-score demonstrated no difference in anxiety-like behavior between the Cre and eEF2K-cKI mice (Fig. 4F). These data suggest that neuronal activation of eEF2K/eEF2 signaling does not result in anxiety-like behavior.
(A) Representative heatmaps for OF. (B) Percent time spent in the periphery during OF was not significantly different between groups. Cre, n = 10; eEF2K-cKI, n = 11. Student’s T-test. p = 0.1955. (C) Latency to drink in the home cage (day 4) and in the novel cage (day 5) in the NIH task. There was no difference between groups in the novel cage. Cre, n = 10; eEF2K-cKI, n = 11. 2-way ANOVA. Cre Home vs Cre Novel p < 0.0001; eEF2K-cKI Home vs eEF2K-cKI Novel p = 0.0037; Cre Home vs eEF2K-cKI Home p = 0.8920; Cre Novel vs eEF2K-cKI Novel p = 0.0790. (D) Amount consumed as percent body weight in the home cage and novel cage in the NIH task. Both groups had decreased consumption in the novel cage but did not significantly differ from each other. Cre, n = 10; eEF2K-cKI, n = 11. 2-way ANOVA. Cre Home vs Cre Novel p < 0.0001; eEF2K-cKI Home vs eEF2K-cKI Novel p = 0.0001; Cre Home vs eEF2K-cKI Home p = 0.2563; Cre Novel vs eEF2K-cKI Novel p = 0.4342. (E) No difference in the latency to consume the food pellet in the NSFT. Cre, n = 10; eEF2K-cKI, n = 11. Student’s T-test. p = 0.7989. (F) There was no difference in the combined anxiety z-score between groups. Cre, n = 10; eEF2K-cKI, n = 11. Student’s T-test. p = 0.7376. Error bars represent ± SEM. **p < 0.01, ***p < 0.001, ****p < 0.0001; Student’s T-test and 2-way ANOVA.
eEF2K-cKI mice exhibit learning and memory impairments
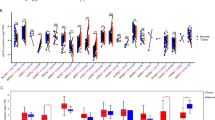
Previous studies have indicated that eEF2K signaling may play an important role in learning and memory [16, 49,50,51]. We carried out a battery of behavioral assays to investigate the effects of eEF2K overexpression on learning and memory performance. We first evaluated long-term recognition memory using the novel object recognition (NOR) task where the mice distinguish between a familiar and novel object 24 h after familiarization [52, 53]. The Cre mice exhibited normal cognition and spent more time with the novel object with a positive discrimination index (Fig. 5A–C). In comparison, the eEF2K-cKI mice failed to distinguish the familiar object from the novel object with a significantly lower, negative discrimination index compared to the control group (Fig. 5A-C). eEF2K-cKI mice also spent less time interacting with either object compared to Cre controls (Supplemental Fig. 4C). Additionally, we performed the open field (OF) task to determine if there were any locomotor abnormalities and did not observe any differences between groups in distance and velocity moved (Supplemental Figs. 4A and B). These findings suggest impairments of recognition memory in the eEF2K-cKI mice.
(A) Representative heatmaps during the NOR testing phase. “F” indicates familiar object. “N” indicates novel object. (B) Ratio of time spent with familiar (empty) and novel (stripped) objects in the NOR task during the testing phase. Preference of less than 0.5 for novel object indicates cognitive impairment as seen in the eEF2K-cKI mice. Cre Familiar vs Cre Novel p < 0.0001; eEF2K-cKI Familiar vs eEF2K-cKI Novel p = 0.0390 (C) Discrimination index, calculated as (time interacting with novel object – time interacting with familiar object) / total interaction time, was significantly decreased in eEF2K-cKI mice. Cre, n = 11; eEF2K-cKI, n = 9. p < 0.0001. (D) Percent of spontaneous alternations in the Y-maze task was decreased in eEF2K-cKI mice. % Alternation = (the number of correct alternations / total number of arm entries – 2) * 100. Cre, n = 9; eEF2K-cKI, n = 13. p < 0.0001. (E) Representative heatmaps for MWM. “P” indicates platform location. (F) Escape latency over 4 trials/day for 5 days of training in MWM. eEF2K-cKI mice had higher average escape latencies during the training phase. (G) Percent of time spent in target quadrant during MWM Probe trial was reduced in eEF2K-cKI mice. p < 0.0001. (H) Trending decrease in the number of platform crossings during MWM Probe trial in eEF2K-cKI mice. Cre, n = 11; eEF2K-cKI, n = 9. p = 0.0686 (I) Escape latency over 4 trials/day for 2 days of VP was not different between groups. Cre, n = 11; eEF2K-cKI, n = 9. Error bars represent ± SEM. ***p < 0.001, ****p < 0.0001; Student’s T-test.
Spatial working memory was evaluated in the Y-maze task. The eEF2K-cKI mice had a significantly lower percentage of correct arm alternations compared to Cre controls, suggesting impaired spatial working memory (Fig. 5D). Interestingly, the number of total entries was higher in the eEF2K-cKI mice (Supplemental Fig. 4D). We also assessed hippocampal-dependent spatial learning and memory using the hidden platform Morris Water Maze (MWM) task [53, 54]. During the training phase, the eEF2K-cKI mice took significantly longer to find the hidden platform compared to the Cre mice, suggesting impaired learning ability (Figs. 5E and 5F). The probe trial results showed that the eEF2K-cKI mice spent significantly less time in the target quadrant where the platform was located and had a trending decrease in the number of “platform” crossings compared to Cre controls (Figs. 5G and 5H). To determine potential memory-independent effects that may confound the above findings such as swimming ability and vision, we performed the visible platform (VP) task and found no difference between the Cre and eEF2K-cKI mice in the escape latency for the visible platform (Fig. 5I) or in the average swimming velocity (Supplemental Fig. 4E). These results indicate that overactive eEF2K signaling can impair learning and memory.
Functional and structural synaptic deficits in eEF2K-cKI mice
Protein synthesis is essential for maintenance of long-term potentiation (LTP), a major form of synaptic plasticity and putative cellular basis of learning and memory [55]. Previous studies have shown that eEF2K/eEF2 signaling is associated with expression of plasticity-related proteins [56,57,58]. eEF2K is a calcium/calmodulin-dependent kinase, also known as CAMKIII, and is involved in multiple mechanisms of synaptic plasticity [56, 59]. LTP deficits have also been associated with neuropsychiatric disorders such as MDD and schizophrenia [60,61,62,63]. To determine if eEF2K overexpression affected synaptic plasticity, we performed ex vivo electrophysiology experiments to evaluate protein synthesis-dependent LTP at the CA3-CA1 synapses of acute hippocampal slices [64,65,66]. Compared to normal sustained LTP in the control Cre mice, the eEF2K-cKI mice exhibited LTP failure (Fig. 6A–C). Analysis of the synaptic responses with various stimulation intensities (input-output relationships) did not reveal any difference between the groups, indicating unaltered basal synaptic transmission in the eEF2K-cKI mice (Fig. 6D). We also investigated presynaptic function by measuring paired pulse facilitation (PPF), a form of short-term presynaptic plasticity [67, 68]. There was no significant difference in PPF between groups, suggesting normal presynaptic function (Fig. 6E). These findings demonstrate that synaptic plasticity is impaired with overactive eEF2K signaling.
(A) Acute hippocampal slices were stimulated with HFS to induce LTP. Arrows indicate HFS. eEF2K-cKI failed to show sustained LTP. Cre, n = 7 slices; eEF2K-cKI, n = 8 slices. (B) Representative fEPSP traces. (C) fEPSP slope 90 min after HFS stimulation was significantly decreased in the eEF2K-cKI mice. Cre, n = 7 slices; eEF2K-cKI, n = 8 slices. (D) Input/output curve assessing basal synaptic transmission was not significantly different between groups. Cre, n = 7 slices; eEF2K-cKI, n = 8 slices. p < 0.0001. (E) Curve of Paired-pulse ratio with varying interval time as a measure of presynaptic transmission was not different between groups. Cre, n = 7 slices; eEF2K-cKI, n = 8 slices. (F) Representative images from Golgi-Cox stain of CA1 dendritic spines. Original magnification x100. Scale bar = 10μm (G) Total spine density per 10μm was not significantly different between groups. p = 0.9800. (H) Immature spine density per 10μm was increased in eEF2K-cKI mice. Filopodia and thin spines were classified as immature. p = 0.0003. (I) Mature spine density per 10μm was decreased in eEF2K-cKI mice. Mushroom, stubby, and branched spines were classified as mature. p < 0.0001. (J) The ratio of mature to immature spines was reduced in eEF2K-cKI mice. n = 3 mice per group with 15 images per mouse, approximately 100μm of dendrite analyzed per image. p < 0.0001. (K) Representative TEM images of PSDs in CA1. Red arrows indicate PSDs. Scale bar = 1μm. Original magnification x9.8k. (L) eEF2K-cKI mice had fewer PSDs per an area of 100μm2. p < 0.0001. (M) Trending decrease in average PSD area in eEF2K-cKI mice. p = 0.0131. (N) PSD length was significantly increased in eEF2K-cKI mice. p = 0.2809. (O) PSD width was decreased in eEF2K-cKI mice. p = 0.0012. n = 4 mice per group with 15-20 images (ROIs) per mouse. Error bars represent ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001; Student’s T-test.
The structural plasticity and dynamics of dendritic spines requires protein synthesis and is associated with synaptic plasticity and memory formation [69, 70]. Dendritic spine changes have also been associated with neuropsychiatric diseases [71,72,73,74]. We used the rapid Golgi-Cox stain approach [34] to assess dendritic spine density and morphology in apical dendrites in the CA1 region of the hippocampus based on published guidelines [75]. We did not observe any significant differences in total spine density between groups. However, further analysis revealed a significant increase in the number of immature spines (thin, filopodia), and a significant decrease in the number of mature spines (mushroom, stubby, branched) in the eEF2K-cKI mice compared to Cre controls (Fig. 6F-I). Accordingly, there was a decrease in the mature/immature spine ratio in the eEF2K-cKI mice compared to the control group (Fig. 6J). Using transmission electron microscopy (TEM) approach, we also evaluated postsynaptic densities (PSDs) which are specialized synaptic structures located on dendritic spines that are critical to synaptic function [76, 77]. We found that eEF2K-cKI mice had significantly reduced PSD density compared to Cre controls (Figs. 6K and 6L). Further morphological analysis revealed that PSDs of the eEF2K-cKI mice were significantly smaller and thinner than those in the Cre control mice (Fig. 6M-O). Taken together, overactive eEF2K signaling and eEF2 hyper-phosphorylation are associated with functional and structural synaptic deficits.
Proteomic analysis of eEF2K-cKI mice
Phosphorylation of eEF2 at T56 by eEF2K inhibits the elongation phase of protein synthesis. We assessed de novo protein synthesis using the surface sensing of translation (SUnSET) assay where acute hippocampal slices were incubated with a subthreshold dose of puromycin, a tRNA analogue, and then a western blot performed with an anti-puromycin antibody [78]. Levels of de novo protein synthesis, as indicated by puromycin measurement, were significantly decreased in eEF2K-cKI mice (Fig. 7A). Consistently, imaging analysis using TEM revealed that the eEF2K-cKI mice had fewer polyribosomes, which are sites of active translation [50, 53, 79], near synapses in the CA1 area of the hippocampus compared to Cre controls (Fig. 7B). These data demonstrate that eEF2K overexpression and subsequent eEF2 hyper-phosphorylation leads, as expected, to a decrease in de novo protein synthesis.
(A) Western blot analysis of puromycin incorporation in the SUnSET assay to measure de novo protein synthesis shows decreased puromycin incorporation in the eEF2K-cKI mice. Cre, n = 14; eEF2K-cKI, n = 10. p = 0.0316. (B) Representative TEM images for CA1 polyribosomes near synapses. Polyribosomes within axons and somas were excluded from quantification. Red arrows indicate polyribosomes. Original magnification x9.8k. Scale bar = 1μm. eEF2K-cKI mice had fewer polyribosomes per an area of 100μm2. n = 4 mice per group with 15-20 images (ROIs) per mouse. p < 0.0001. (C) PCA plot of Cre and eEF2K-cKI mice used for proteomics. (D) Heatmap of the 133 proteins significantly dysregulated between the eEF2K-cKI and Cre mice. (E)Volcano plot showing the distribution of differently regulated proteins. Gray circles are the proteins that were not significantly altered. Green circles represent proteins that had a fold change greater that 20% but were not significant. Blue circles represent proteins that were significantly changed but had a fold change less than 20%. Red circles represent the proteins that were significant and had a fold change greater than 20%. (F) Gene ontology enrichment analysis of all 59 proteins that met the q-value and fold change requirements. n = 7 per group. (G) Proteomic analysis of the Log2 abundance of Shank1 normalized to Cre. n = 4 per group. p = 0.0020. (H) Western blot confirming an increase in Shank1 in eEF2K-cKI mice. n = 6 per group. p = 0.0097. (I) Proteomic analysis of the Log2 abundance of GFAP normalized to Cre. n = 4 per group. p = 0.0027. (J) Western blot confirming an increase in GFAP in eEF2K-cKI mice. n = 6 per group. p = 0.0009. Error bars represent ± SEM. **p < 0.01, ***p < 0.001; Student’s T-test.
To determine how eEF2K-driven changes in protein synthesis affected the proteome, we performed mass spectrometry-based proteomics to determine changes in the hippocampal proteome associated with eEF2K overexpression. In brief, 16 age and sex matched samples underwent LC-MS/MS analysis in 2 batches. Two samples, one from each batch, were determined to be outliers via hierarchical clustering and were excluded from further analysis (Supplemental Fig. 5). The resulting 14 samples (n = 7 for each genotype) were corrected for batch effects using the sva package and principal component analysis shows that samples cluster based primarily on genotype (Fig. 7C) [80]. 1815 proteins were detected across 14 samples. After statistical analysis, we observed that 131 proteins were significantly changed and 59 met the criteria for a fold change greater than 20% with the expression pattern shown in a heatmap (Fig. 7D). Within this group, 44 proteins were significantly upregulated while 15 proteins were found to be downregulated as shown in Tables 1 and 2 and the volcano plot (Tables 1 and 2, Fig. 7E). To further determine the potential interactions and functions, GO enrichment analysis was performed. The majority of the proteins that were differentially expressed were associated with mRNA translation, gene expression, and ribosomal assembly and biogenesis (Fig. 7F). eEF2 showed the highest fold change among upregulated proteins. Of the upregulated proteins, 10 were 60 s ribosomal proteins and 8 were 40 s ribosomal proteins. Two proteins of interest that were significantly upregulated, Shank1 and GFAP, were verified via western blot (Fig. 7G-I). Additionally, several downregulated proteins were synaptic membrane components (Flot1, Flot2, SNAP25, Prrt1, and Marcksl1).
Given the significant increase in GFAP and C1qa, we wanted to determine if there were glial changes associated with eEF2K overexpression. 60μm thick sections were stained with GFAP (green), Iba1 (red), and Dapi (blue). The eEF2K-cKI had significantly more GFAP positive cells compared to Cre controls while there was no significant difference in Iba1 positive cells (Supplemental Fig. 6). Additionally, changes in astrocyte morphology were observed with an increase in soma size with thicker processes and decreased ramification (Supplemental Fig. 6).
Discussion
Overactive eEF2K signaling i.e. eEF2 hyper-phosphorylation has been implicated in neuronal diseases such as Alzheimer’s disease and depression [16, 31, 49, 50, 53]. Recent studies have shown that the anti-depressant drug, Ketamine, may act through the inhibition of eEF2K [6, 31]. Genetic deletion of eEF2K abolishes ketamine’s antidepressant effect suggesting that eEF2K hyperactivity may play a role in depression [6, 31]. Our lab has previously shown that eEF2 is hyperphosphorylated in the hippocampus of AD patients and in AD model mice [53]. Reducing eEF2 phosphorylation by genetic or pharmacologic means ameliorates learning and memory deficits in AD mouse models and improves synaptic function [49, 50]. This has led us to hypothesize that eEF2K over expression and hyper phosphorylation of eEF2 will cause depression-like behavior and cognitive deficits in mice.
We thoroughly and robustly characterized multiple neuropsychiatric-like behaviors by using domain specific z-scores. Using the z-score method of analysis allows for greater sensitivity and reliability in behavioral testing, especially when multiple behavioral paradigms are necessary to elucidate complex phenotypes like anxiety, depression, or apathy [81, 82]. This approach allows for the summary and comparison of multiple behavior tasks. Many of the behavior tests that we performed are commonly used in the study of depression [38]. However, we chose to separate these tests into multiple domains in order to better understand the role of eEF2K in these complex phenotypes. Additionally, while apathy, anxiety, and depression are often co-occurring, they are clinically separate and distinct disorders [83]. We also chose to separate depression-associated behaviors into multiple sub-domains depending on the specific aspect measured in the behavioral paradigm. For example, FST and TST were summarized as despair-like behavior while SPT was separated as anhedonia. We ultimately found that eEF2K overexpression resulted in a profound apathetic phenotype but did not lead to any significant changes in anxiety, despair, anhedonia, or social behaviors. Additionally, the effect of eEF2K on depressive phenotypes may only be visible under stress conditions. It may also be a question of acute vs. chronic eEF2K activation and future studies are needed to better understand this complex relationship.
Apathy, dementia, and eEF2K
Apathy, defined by decreased motivation and goal-directed behavior, is often associated with dementia syndromes such as AD. Indeed, the most common NPS in AD patients is apathy, which causes greater caregiver burden, lower quality of life, faster cognitive and functional decline, and increased morbidity [84,85,86,87,88,89,90,91,92]. Apathy has also been associated with other diseases such as Parkinson’s disease, Huntington’s disease, amyotrophic lateral sclerosis, and geriatric depression [93,94,95]. Apathy commonly accompanies cognitive dysfunction and is a predictor of cognitive decline in many disease states [95,96,97,98,99,100,101,102]. Currently, little is known about the underlying mechanisms of apathy.
Historically, apathy is understudied with very few studies in preclinical models. There is currently no “gold standard” behavior test to measure apathy-like behavior. For our study, we chose 4 different rodent-typical behaviors that did not require training since eEF2K overexpression induces learning and memory deficits that could confound results. These tests (nestlet shredding, nest building, marble burying, and burrowing) were recently used to assess apathy-like behavior in mouse models of AD and down syndrome (DS) [37, 42]. Understanding the molecular underpinnings of apathy and other NPS is important to the treatment of AD and other neurological disorders. We demonstrate here, for the first time, that overactive eEF2K signaling is associated with an apathy-like phenotype. Such findings could provide insights into novel therapeutic strategies for treatment of NPS in AD and other neuronal diseases.
While eEF2K overexpression did not lead to a deficit in sociability, there was a deficit in the preference for social novelty. This decreased preference could be due to deficits in working memory as eEF2K-cKI displayed spatial working memory deficits in the Y-maze task. Additionally, several studies have found a connection between apathy and olfaction deficits which may contribute to the lack of preference for a novel mouse [103, 104].
eEF2K overexpression is associated with synaptic and memory deficits
We first showed that eEF2K overexpression resulted in increased phosphorylation of eEF2 at T56. This phosphorylation site is phosphorylated by eEF2K and prevents eEF2 from binding to the ribosome [15]. Other phosphorylation sites on eEF2 may affect eEF2 activity by modulating T56 phosphorylation or dephosphorylation [105, 106]. The overall decrease in de novo protein synthesis agrees with the increased eEF2 phosphorylation at T56. Interestingly, total eEF2 was also increased in eEF2K-cKI mice. eEF2 mRNA contains 5’ terminal oligopyrimidine (TOP) motif [107]. TOP mRNAs have increased expression when eEF2 is phosphorylated [11]. Under conditions of cellular stress, when the rate of translation slows, the rate of TOP mRNAs translation remains constant leading to a relative increase in the proportion of TOP mRNAs [11]. This supports the findings of increase in total eEF2 in the eEF2K-cKI mice.
eEF2K signaling has also been linked to dendritic spine alterations [108]. Structural changes in dendritic spines are essential for normal neurodevelopment and brain function [72, 109]. Dendritic spine alterations also accompany activity changes and respond to synaptic plasticity mechanisms (LTP, LTD, etc.) [109,110,111]. They are also thought to underlie memory formation [112, 113]. Synaptic plasticity and these morphological changes are associated with protein synthesis and may involve eEF2K/eEF2 signaling [16, 59, 108]. We observed impairments in LTP, the process considered the cellular basis of learning and memory. LTP impairment was accompanied by changes in dendritic spine and PSD morphology. As mentioned, dendritic spines are closely linked to synaptic plasticity with mature spines (mushroom, branched, and stubby) usually considered “memory” spines [114]. Here we found a distinct decrease in the number of mushroom spines in eEF2K-cKI mice and a corresponding increase in the number of immature thin spines. eEF2K signaling controls expression of many plasticity-related proteins such as CAMKII, BDNF, and Arc, all of which have been implicated in synaptic plasticity and dendritic spine dynamics [59, 108, 115]. Increased eEF2K activity has previously been linked to learning and memory impairments [16, 51]. We found that eEF2K overexpression resulted in robust learning and memory impairments in the MWM, Y-maze, and NOR behavior paradigms. While dendritic spines and synaptic plasticity are most closely associated with learning and memory, evidence has accumulated for a significant role of dendritic spines in neuropsychiatric disorders as well [72, 116]. Dendritic spine changes have been linked to motivational impairments in schizophrenia [73, 74]. Dendritic spines and synaptic alterations have been implicated in the pathophysiology of MDD [61]. Further studies are needed to define the association between apathy-like behavior and dysregulated synaptic plasticity and changes in dendritic spine and synaptic morphology.
Proteomic analysis of eEF2K overexpression
We investigated the effects of eEF2K overexpression in mature excitatory neurons on the proteome in the hippocampus. We performed mass spectrometry-based proteomics as well as gene ontology enrichment analysis. The majority of upregulated proteins were associated with translation and largely consisted of ribosomal proteins and translation factors. The subset of mRNAs whose translation is upregulated by eEF2K includes those with 5’ terminal oligopyrimidine (TOP) motif, many of which encode ribosomal proteins and translation factors, as seen in our results [11]. Additionally, eEF2 is an established TOP mRNA and was found to be upregulated in our proteomics data and this was further confirmed with western blot [107].
Several proteins that were differentially regulated are associated with dendritic spine morphology, synaptic function, and cognitive impairments. Shank1 is involved in dendritic spine morphology and has been associated with cognitive deficits [117, 118]. In schizophrenia, decreased Map2 has been associated with fewer small dendritic spines [119]. In concordance, our data suggests that increased Map2 may be associated with increased numbers of small dendritic spines. Neurabin-2, also known as spinophilin, is a synaptic scaffolding protein that has been implicated in dendritic spine morphology and synaptic plasticity [120, 121]. Additionally, Chl1 is a cell adhesion molecule that has been associated with neurite outgrowth and synaptic plasticity [122].
A number of upregulated proteins in eEF2K-cKI mice have been linked to neuropsychiatric and neurodegenerative diseases that are often accompanied by apathy symptoms. Shank1 is a synaptic scaffold protein that interacts with the PSD and has been implicated in neuropsychiatric and neurodegenerative disorders such as autism spectrum disorder, schizophrenia, and AD [123, 124]. Additionally, Shank1 hypomethylation was associated with depressive episodes in adolescents [125]. Map2 is a neuronal cytoskeleton protein that has been implicated in schizophrenia, AD, and mood disorders [126, 127]. FAAH is an endocannabinoid hydrolase and its upregulation is implicated in Major depressive disorder (MDD), AD, and schizophrenia [128,129,130]. Neurabin-2 has also been linked to schizophrenia [131]. Another protein of interest is SNAP25, a SNARE complex member. SNAP25 is decreased in patients with mood disorders and schizophrenia [132, 133]. GFAP is an inflammatory marker that has been implicated in numerous neuropsychiatric and neurodegenerative disorders. Increased GFAP has been proposed as a useful biomarker for AD [134]. GFAP levels are negatively correlated with cognitive measures in AD and increased GFAP is associated with worse cognition [135, 136]. MDD and cognitive decline was associated with higher GFAP levels in older adults [137]. However, GFAP expression in MDD is somewhat mixed with some studies finding increased GFAP or no change in GFAP, while many show decreased GFAP expression [138, 139]. Similarly, GFAP may be increased in schizophrenia, but many studies show different results [138]. Furthermore, several studies have correlated increased GFAP with apathy [140,141,142]. Increased GFAP may explain the robust apathy-like phenotype with a lack of despair-like behavior. More work is needed to determine a direct relationship between these implicated proteins, eEF2K signaling, and disease pathogenesis.
It is important to note that proteomics method in the current study does not account for nascent changes in the proteome. Multiple studies have shown that changes in elongation rate affect the specific mRNAs being translated [143, 144]. eEF2 is directly responsible for the translocation of the ribosome down the mRNA [14, 15]. Phosphorylation of eEF2 may affect the rate of cap-dependent translation of certain mRNAs while also promoting the cap-independent translation of TOP mRNAs. In this way, it is possible that there are differences in the nascent as well as the total proteome. Further studies using alternative approaches to determine how p-eEF2 upregulation affects the nascent proteome are warranted.
In conjunction with the proteomic analysis, immunofluorescence staining revealed an increase in GFAP+ cells with altered morphology in the eEF2K-cKI mice. eEF2K overexpression lead to more astrocytes as well as more reactive astrocytes compared to controls. This is interesting and could be a response of glia to neuronal stress. Given the decrease in the number of PSDs, this might also suggest an increase in synaptic engulfment in eEF2K-cKI mice. Further studies are needed to determine the underlying mechanism of neuronal eEF2K expression and glial expression and reactivity.
Lastly, this study focused on the thorough characterization of hippocampal changes and their association with neuropsychiatric behaviors. Several studies have shown that the hippocampus, while most widely known for its role in learning and memory, is also involved in emotional regulation and has been implicated in mood disorders [145,146,147]. Apathy has also been associated with decreased hippocampal volume [148]. The hippocampus is also connected to other emotion-related brain regions such as the anterior cingulate cortex (ACC) and the amygdala [63]. However, the prefrontal cortex and other brain regions, such as the ACC, are also known to play a role in apathy and mood disorders [149]. Future work is necessary to determine how altered protein synthesis and synaptic function in different brain regions contribute to behavioral phenotypes and apathy.
In summary, overexpression of eEF2K resulted in dysregulation of multiple proteins that are known to be involved in neuropsychiatric and neurodegenerative disorders which highlights the potential critical role of eEF2 hyper-phosphorylation in their pathogenesis.
eEF2K has recently been studied for its contributions to AD pathogenesis and its potential role in MDD and other neuropsychiatric disorders. In this study, we found that contrary to our hypothesis based on eEF2K’s role in the rapid anti-depressant action of ketamine, eEF2K overexpression did not result in despair-like behavior. However, we determined a novel role of eEF2K in apathy-like behavior, which is common to many neurodegenerative and neuropsychiatric disorders. In addition, we demonstrated that eEF2K overexpression and subsequent hyperphosphorylation of eEF2 lead to a marked memory deficit. This was accompanied by impaired synaptic plasticity and structural synaptic changes. In summary, eEF2K may play a larger role in apathy-like behavior and learning and memory impairments. More work is needed to better understand these complex relationships.
Materials and methods
Mice
All mice were housed at the Wake Forest School of Medicine barrier facility under the supervision of Institutional Animal Care and Use Committee. The facility operates in accordance with standards and policies of the US Department of Agriculture’s Animal Welfare Information Center (AWIC) and the NIH Guide for Care and Use of Laboratory Animals. Mice adhered to a 12-hour light/12-hour dark cycle, with regular feeding, cage cleaning, and 24-hour food and water access. All genotyping was done by polymerase chain reaction (PCR).
Rosa26-CAG-LoxP-STOP-LoxP-Eef2k knock-in mice were generated by the University of North Carolina at Chapel Hill Animal Models Core using CRISPR/Cas9-mediated genome editing. Mice were bred with CAMKII-cre mice [150] to produce eEF2K conditional overexpression (eEF2K-ckI) mice and cre control littermates. Male and female mice 3-5 months old were used for all experiments. For each behavioral experiment, multiple cohorts of mice were used. Mice with significant injury or evidence of seizure activity were excluded from analysis. No randomization was used in determining groups.
RNA isolation and quantitative RT-PCR
The hippocampus was dissected out in ice cold PBS and total RNA was isolated with Trizol (Invitrogen, catalog #15596026). The first strand cDNA was generated using qScript cDNA SuperMix (Quanta bio, catalog #95048-025). The genes were amplified using quantitative PCR with PerfeCTa SYBR Green FastMix (Quanta bio, catalog #95072-250). The following PCR protocol was used: 95 °C for 2 min, then 40 cycles of 95 °C for 10 s, 60 °C for 30 s. To determine relative gene expression, ΔΔCt was calculated. GAPDH was used as the normalization control. Forward eEF2K primer sequence: 5’- CCACTTGGAGCACTACATTGAGG -3’. Reverse eEF2K primer sequence: 5’- ATAAAGGTCACCCACACCCTGG -3’.
Western Blot
Mouse hippocampal tissue was dissected and flash frozen on dry ice. Lysis and gel electrophoresis were performed as previously described [151]. Briefly, the tissue was sonicated in lysis buffer with protease and phosphatase inhibitors. Protein quantification was determined using the Bradford colorimetric Assay (Thermo Fischer Scientific, catalog #23227). Equal amounts of protein lysate from each sample were loaded on 4 - 12% Tris-glycine SDS-PAGE gels (Bio-Rad 18-well gel, catalog #5670184) followed by standard gel electrophoresis. After transfer, membranes were blocked for 10 min with SuperBlock TBS Blocking Buffer (Thermo Fischer Scientific, catalog #37535). All primary and secondary antibodies were diluted in 5% milk/TBST or 5% BSA/TBST. Blots were incubated with the following primary antibodies overnight at 4 °C: eEF2K (1:500, Cell Signaling Technology, catalog 3692), phospho-eEF2 (Thr56) (1:1000, Cell Signaling Technology, catalog 2331); eEF2 (1:1000, Cell Signaling Technology, catalog 2332); β-Actin (1:5000, Sigma Aldrich, catalog A2228); GFAP (1:1000, Cell Signaling Technology, catalog 12389S); Shank1 (1:500, Santa Cruz, catalog sc-398630). Blots were then incubated in secondary antibodies for 2 h at room temperature. The ChemiDoc Imaging System (Bio-Rad) was used for protein visualization. Densitometric analysis was performed using ImageJ software (NIH). Phospho-proteins were normalized to the levels of total protein; total proteins were normalized to the housekeeping protein β-Actin.
Acute Hippocampal Slice Preparation
A Leica VT200S vibratome was used to prepare 400 μm transverse acute hippocampal slices as previously described [64]. Slices were maintained in ACSF bubbled with 95% O2 / 5% CO2 at room temperature for 2 hr before experiment. ACSF contained the following: 118 mM NaCl, 3.5 mM KCl, 2.5 mM CaCl2, 1.3 mM MgSO4, 1.25 mM NaH2PO4, and 15 mM glucose.
Surface Sensing of Translation (SUnSET) Assay
Surface Sensing of translation was performed as previously described [53, 78]. Briefly, acute hippocampal slices were incubated in 1 μg/mL puromycin for 1 hr at 32 °C in bubbling ACSF. Slices were flash frozen on dry ice, lysed, and standard gel electrophoresis run. The puromycin-labeled proteins were identified by antibody (1:10,000, Millipore, catalog #MABE343). Total lane density was used to determine protein synthesis levels and densitometry was done using ImageJ (NIH).
Transmission Electron Microscopy (TEM)
Electron microscopy was performed as previously described [53]. Briefly, CA1 was dissected from acute 1 mm transverse hippocampal slices and fixed in 1% PFA + 2.5% glutaraldehyde in 0.1 M Millonig’s phosphate buffer (pH 7.3) overnight. Samples were washed and postfixed in 1% osmium tetroxide in PBS for 1 hr. Samples were then dehydrated through a graded ethanol series and incubated in propylene oxide twice for 15 min each. Samples were infiltrated with Spurr’s resin and cured overnight at 70 °C. 90 nm sections were made using a Reichert-Jung Ultracut E Ultramicrotome. Sections were stained with lead citrate and uranyl acetate. Samples were imaged on FEI Tecnai Bio Twin Transmission Electron Microscope (120 keV) with a 2 Vu AMT camera at x9.8k magnification. 4 mice were used per genotype and 15-20 images of the stratum radiatum of area CA1 were taken. ImageJ was used to analyze images and analysis was performed blinded.
Electrophysiology
Acute hippocampal slices were prepared as described above. Following the 2 h incubation period, slices were maintained at 32 °C, and monophasic current stimuli of 100μs were delivered with a bipolar silver electrode in the stratum radiatum of area CA3. Field excitatory postsynaptic potentials (fEPSPs) were recorded using a glass microelectrode from the stratum radiatum of CA1. LTP was induced using a high-frequency stimulation (HFS) comprised of two 1 s, 100 Hz trains, with a 60 s interval, delivered at 60-70% of evoked spike intensity.
Golgi Staining
The Rapid Golgi Kit (FD NeuroTechnologies, catalog PK401) according to the manufacturer’s instructions. Transverse 100μm hippocampal slices were made after impregnation with a Leica VT1200S vibratome and mounted on gelatin-coated slides. Sections were developed and fixed according to kit instructions. Imaging was performed with a Keyence BZ-X710 All-in-one Fluorescent Microscope with a x100/NA 1.45 oil immersion lens. 5 images within the stratum radiatum of area CA1 were taken per each hippocampal slice. Spines were classified based on published guidelines [75]. Imaging and spine analysis were performed blinded.
Behavioral Assays
For all behavioral assays, mice were handled prior to behavioral testing and were habituated to the testing room for 1 hr before start of experiments. Experiments were performed during the 12-hr light cycle. Behavioral assays were ordered from least stressful to most stressful. Experimenter was blinded to all genotypes during experiments and analysis.
Morris Water Maze (MWM)
A 5-day MWM protocol was performed with 4 trials a day (60 s maximum per trial, 15 min interval between trials) as previously described [53]. Escape latency was measured for each trial. Two hours after training on the 5th day, a probe trial was performed. EthoVision XT Tracking Software (Noldus Information Technology) was used to track trajectories, time spent in maze quadrants, distance and velocity.
Visible Platform (VP)
VP was performed after MWM with a 2-day protocol consisting of 4 trials a day (60 s maximum per trial, 15 min interval between trials). The platform was marked by a visual cue and moved randomly among 4 locations. Escape Latency was measured for each trial.
Novel Object Recognition (NOR)
Mice were habituated to the experiment chamber 1 day before start of experiments. A 2-day familiarization protocol was used. On the first 2 days, mice were placed in a white plastic chamber (40 cm x 40 cm x 40 cm) with 2 identical objects and allowed to explore for 5 min. Twenty-four hours after familiarization, mice were tested in the chamber with one object replaced with a novel object and allowed to explore for 5 min. Objects were randomly assigned to each mouse and location of the novel object was counterbalanced. Time spent exploring objects was measured manually and by EthoVision XT Tracking Software (Noldus Information Technology). Mice with <10 s total interaction time were excluded from analysis. Discrimination index was calculated as the novel object interaction time minus the familiar object interaction time divided by the total interaction time.
Y-maze
Y-maze as a measure of spatial working memory was performed as previously described [152]. The Y-maze apparatus consisted of 3 opaque arms of equal length at 120° angles from each other. The mouse was placed in the center of the maze and allowed to explore freely for 8 min. The entries into each arm, defined as all four limbs within an arm, were recorded. An alternation was defined as consecutive entries into all three arms. Percent alternation was calculated as (the number of alternations / total number of arm entries – 2) * 100. High percent alternation is indicative of good spatial working memory.
Nestlet Shredding (NS)
All mice were habituated in the testing room for 1 hour prior to start of testing. Group-housed mice were placed individually into a clean mouse cage with bedding and one piece of cotton fiber nestlet (5 cm×5 cm, 5 mm thick) that was pre-weighed and placed on top of the bedding in each cage. Each mouse was left undisturbed in the cage with the nestlet for 30 minutes. After the test, the nestlet was left in open space overnight to dry and was weighed again. The last weight and the starting weight were used to calculate percentage of nestlet shredded, a smaller number will indicate more severe apathy-like behavior [153].
Nest Building (NB)
Nest building was performed after the nestlet shredding and marble burying tests. The group-housed mice were habituated with a cotton fiber nestlet (5 cm×5 cm, 5 mm thick) in their home-cage overnight before the day of testing. On the test day, mice were individually housed in a new cage with fresh, unscented bedding with a piece of clean and pre-weighed nestlet. The mice were allowed to behave freely overnight, and the nests were evaluated the following morning. The nest from each mouse was photographed and the nests were graded using scores ranging from 1 (very poor/no nest building) to 5 (optimal nest building) with half-point scores for nests falling between categories according to a previously published scoring scheme by Deacon [43]. Similar to the nestlet shredding test, the amount of nestlet left unshredded was weighed and used to calculate the percentage of unshredded nestlet. A lower nest score and a higher percentage of unshredded nestlet indicates more severe apathy-like behavior.
Marble Burying Test (MBT)
Group-housed Mice were habituated in the testing room 1 hour before the. A standard rat cage (26 cm×48 cm x 20 cm) was used with 5 cm layer of unscented mouse bedding material. 15 standard glass toy marbles (assorted styles and colors/15 mm diameter, 5.2 g in weight) were gently put on the surface of the bedding in 3 rows of 5 marbles. Each mouse was gently placed in the cage in a corner away from marbles and allowed to behave freely, undisturbed for 30 minutes. The percent of total marble volume buried for each marble was recorded and the number of those with > 2/3 total volume buried were counted as buried. Fewer marbles buried indicates more severe apathy-like behavior [153].
Burrowing
Group-housed mice were habituated with a burrowing tube filled with food pellets in their home-cage overnight before the day of testing. On the testing day, the mice were individually housed in a new cage with fresh bedding. A clean burrowing tube was filled with 200 grams of food pellets and placed in each cage. For the 2-hour burrowing test, the filled burrowing tube was placed in the cage around 2:00-3:00 pm and the mouse was allowed to behave freely for two hours. Following the 2-hour period, the amount of food left in the tube was weighed and used to calculate the amount burrowed. For the overnight burrowing test, the burrow tube was emptied and refilled with 200 g of new food pellets and placed back in the cage overnight. On the following morning, the amount of food left in the tube was weighed and used to calculate the amount burrowed [154, 155]. Smaller amount burrowed will indicate greater apathy-like behavior.
Open Field (OF)
Mice were placed in a white plastic chamber (40 cm x 40 cm x 40 cm) and allowed to explore for 15 min. Time spent in the center and periphery of the chamber, as well as distance moved, and average velocity was measured using EthoVision XT Tracking Software (Noldus Information Technology). The percentage of total time spent in the periphery of the chamber was calculated.
Novelty-Induced Hypophagia (NIH)
NIH was performed as previously cited with minor adjustments [48]. Mice were trained to drink vanilla Ensure® in their home cage over the course of three days for 30 min each day. The Ensure® was presented in a 50 mL conical tube with a rubber stopper through the wire rack of the cage lid. The amount consumed was weighed in grams and the latency to drink was recorded for each training day. Following training, mice underwent home cage testing following the same procedure as during training. Training and home cage testing were performed in low light. The day after home cage testing, mice underwent novel cage testing where the mice were presented the ensure in a novel cage without a wire rack or bedding and in bright light conditions. The amount consumed was weighed in grams and the latency to drink was recorded for both testing days.
Novelty-Suppressed Feeding Test (NSFT)
NSFT was performed as previously described with minor adjustments [40]. Mice were food deprived for 48 h prior to behavioral testing with a 2-hour free feeding period after the first 24 h. Following food deprivation, mice were placed in a white plastic chamber (40 cm x 40 cm x 40 cm) that had 2 cm of bedding in the bottom and a food pellet position in the middle of the arena and secured to a platform at the level of the bedding. The latency for the mouse to begin eating the food pellet was recorded. The mouse was then immediately returned to its home cage with a pre-weighed food pellet for 5 min. the weight of food consumed after 5 min was recorded.
Sucrose Preference Test (SPT)
SPT was performed with minor adaptations as previously described [40]. Mice were first habituated to two bottles of water in their home cage overnight. Water was presented in a 50 mL conical tube fitted with a rubber stopper on the left and right side of the cage. The amount of water consumed was weighed after 24-hour access. One bottle was replaced with a 1% (w/v) sucrose solution. The position of the sucrose solution was randomly assigned for each mouse. The amount of sucrose consumed was weighed after 24-hour access. This was repeated for 2 more days, and the position of the sucrose solution was alternated each consecutive day.
Forced Swim Test (FST)
Mice were placed in a 4-L glass beaker (Corning Pyrex 1000-4 L) containing 3 L of 24 °C water (75% full) [31]. They were recorded at the level of the animal horizontally for 6 minutes. The final 4 minutes were used for scoring immobility as described in literature. The videos from FST were scored using DBscorerV2 which is an automatic mobility based quantification software [39]. We used the recommended threshold of 1.6. Results were compared to blinded hand scored results and found to be accurate.
Tail Suspension Test (TST)
Mice were suspended by their tail in a custom-designed white chamber [31]. Seventeen cm of tape was attached at the end of their tail with 1 cm of tail exposed. The tape was then wrapped around the chamber to suspend the mouse with 12 cm of slack. Mice were recorded for 6 minutes and the entire video was used for scoring using DBscorerV2 with the recommended threshold of 0.6 [39]. Results were compared to blinded hand scored results and found to be accurate.
3-Chamber Sociability Test (3-CST)
Protocol for 3-chamber sociability test was adapted from Yang, et al. [45]. Stranger mice were habituated in wire cups prior to behavioral testing. Multiple stranger mice were used and alternated between experimental mice so that each stranger mouse did not spend significant amounts of time in the wire cup. The experiment mouse was first habituated for 5 min in the middle compartment of the 3-chamber apparatus with the doors closed. Immediately following, empty wire cups were placed in the left and right-side chambers, doors were opened, and experiment mouse was allowed to explore all three chambers and cups for another 5 min. After the 5 min, the experiment mouse was placed back in the middle chamber with the doors closed while the stranger 1 mouse was placed in a cup. The side for stranger mouse was determined to be the opposite side of the chamber the experiment mouse was last in. The doors were opened and the experiment mouse was allowed to explore for 10 min. The experiment mouse was then placed back in the middle compartment with the doors closed while the stranger 2 mouse was added to the empty wire cup on the opposite side of the stranger 1 mouse. The doors were opened and the experiment mouse was allowed to explore for another 10 min. The time the experiment mouse spent interacting with each cup during each 10 min session was measured.
Composite z-score Analysis
Z-scores were calculated as previously described for each behavior test independently for each mouse relative to the mean and standard deviation (SD) of Cre controls so that changes of ± 1 z-score unit represented ± 1 SD of the control sample’s behavior [37]. To reduce the variance and enhance the reliability of our data, we further created composite scores (i.e., a combined z-score for each mouse across all tests) for 4 different behavioral domains: anxiety-like behavior, apathy-like behavior, despair-like behavior, and anhedonia-like behaviors. The raw z-scores were corrected so that a higher value was indicative of greater anxiety, apathy, despair, or anhedonia, respectively. The composite score for anxiety-like behaviors was calculated using the following equation: (zi(OF) + zi(NIH Latency) + zi(NIH Consumption) + zi(NSFT))/4, which will indicate overall anxiety-like behavior severity. The composite score for apathy-like behaviors was calculated using the following equation: (zi(NS) + zi(MB) + zi(NB % unshredded) + zi(NB score) + zi(2 hr burrowing) + zi(overnight burrowing))/6, which will indicate overall apathy-like behavior severity [42, 81]. The composite score for despair-like behaviors was calculated using the following equation: (zi(FST Immobility) + zi(FST Latency) + zi(TST Immobility) + zi(TST Latency))/4, which will indicate overall despair-like behavior severity. The score for anhedonia-like behavior was comprised of the individual z-score for sucrose preference.
Proteomics
Mouse hippocampi were dissected and flash frozen on dry ice. Tissue was lysed in PBS with 2x protease inhibitor using a bead mill homogenizer. Lysed tissue was incubated in 2x RIPA buffer on ice for 30 min. Samples were centrifuged at 18,000 x g for 10 min and supernatant collected and protein quantification assay performed. 10 mM dithiothreitol (DTT) was added to 50 μg of protein and incubated at 55 °C for 30 min. 30 mM iodoacetamide (IAM) was added and samples incubated in the dark at room temperature for 30 min. 4x sample volume of cold acetone was added and samples incubated overnight at -20 °C. Samples were centrifuged at 14,000 x g for 10 min at 4 °C and supernatant discarded. Pellet was suspended in 100 μL of 50 mM ammonium bicarbonate. Samples were incubated with 1.0 μg of trypsin/lys-C mixture overnight at 37 °C. Digestion was quenched by adding formic acid (1.0% w/w final) and the mixture was purified using C18 tip. Peptides were prepared in 100 μL of 5% acetonitrile with 1% formic acid for LC-MS/MS analysis.
Samples were analyzed on a LC-MS/MS system consisting of an Orbitrap Eclipse Mass Spectrometer (Thermo Scientific, Waltham, MA) and a Vanquish Neo nano-UPLC system (Thermo Scientific, Waltham, MA). Peptides were separated on a DNV PepMap Neo (1500 bar, 75 μm x 500 mm) column for 120 min employing linear gradient elution consisted of water (A) and 80% acetonitrile (B) both of which contained 0.1% formic acid. Data were acquired by top speed data dependent mode where maximum MS/MS scans were acquired per cycle time of 3 seconds between adjacent survey spectra. Dynamic exclusion option was enabled which duration was set to 120 seconds. To identify proteins, spectra were searched against the UniProt mouse protein FASTA database (17,082 annotated entries, Oct 2021) using the Sequest HT search engine with the Proteome Discoverer v2.5 (Thermo Scientific, Waltham, MA). Search parameters were as follows: FT-trap instrument; parent mass error tolerance, 10 ppm; fragment mass error tolerance, 0.6 Da (monoisotopic); enzyme, trypsin (full); # maximum missed cleavages, 2; variable modification, +15.995 Da (oxidation) on methionine; static modification, +57.021 Da (carbamidomethyl) on cysteine.
In 2 batches, 16 age and sex matched samples (n = 8 per genotype) underwent LC-MS/MS analysis. Two samples, one from each batch, were determined to be outliers via hierarchical clustering and were excluded from further analysis. The resulting 14 samples (n = 7 for each genotype) were corrected for batch effects using the sva package [80]. Principle component analysis was performed using PCAtools package [156]. Normalized abundances were analyzed for all samples using R (version 4.5.0), excluding proteins with missing values. A 2-tailed t-test was performed for each protein reported and FDR-corrected p-values were used to determine significantly changed proteins. Proteins that had a q-value < 0.05 and a fold change greater than 20% were reported. Gene ontology enrichment analysis was performed on all proteins that met the above criteria.
Immunofluorescence
Mice were anesthetized using Avertin (Sigma-Aldrich #T4, 840-2) intraperitoneal injection at a dose of 0.80 g per gram of body weight. Mice were transcardially perfused with 15 mL of cold PBS then perfused with 50 mL of cold 4% paraformaldehyde (PFA) at a 5 mL/min flow rate. The brain was dissected out and fixed overnight in cold 4% PFA in PBS. Half brain was subsectioned to 60 μm using a Leica VT1200S vibratome and washed twice in 0.02% Triton X-100 in PBS for 10 min each. Free-floating sections were then permeabilized in 300 mM Glycine and 10% DMSO, in 0.02% Triton X-100 in PBS for 1.5 h. Slices were washed twice in 0.02% Triton X-100 in PBS for 5 min each. Slices were blocked for 2 h in 6% normal goat serum in 0.02% Triton X-100 in PBS. Slices were washed twice in 0.01% Triton X-100 in PBS for 5 min each. Slices were then additionally blocked against endogenous IgG with Fab Fragment Goat Anti-Mouse IgG (1:50, Jackson ImmunoResearch, 115-007-003) in 0.01% Triton X-100 in PBS at 4 °C overnight on a shaker. Slices were washed 3 times for 15 min in 0.2% Tween-20 in PBS with 0.001 mg/mL Heparin (PTwH). Slices were then incubated for 2 days at 4 °C in primary antibodies: GFAP (1:500, Cell Signaling Technology, catalog 12389) and Iba1 (1:500, Abcam, ab178846) in 0.5x PTwH with 3% normal goat serum. Slices were washed with 0.5xPTwH 4 times for 20 min. Slices were then incubated for 36-48 h at 4 °C in secondary antiobdies: Goat anti-Mouse IgG Alexa Fluor® 488 conjugate (1:500, Invitrogen, A11001) and Goat anti-Rabbit IgG Alexa Fluor® 568 conjugate (1:500, Invitrogen, A-11011) in 0.5x PTwH with 3% normal goat serum. Sections were washed 3 times for 20 min in 0.5x PTwH then another 3 times for 20 min in PBS. Sections were incubated in Dapi (Sigma-Aldrich, MBD0015) for 4 h. Sections were washed 3 times with PBS for 30 min then mounted with Vectashield anti-fade mounting medium (Vector Laboratories, H-1000-10). The sections were then imaged using an Olympus FV1200 confocal microscope. All parameters (pinhole, contrast, gain, and offset) were held constant for all sections across the same experiment. Images were analyzed using ImageJ software (NIH).
Statistical analysis
Data are presented as mean ± SEM. For comparisons between two groups, a two-tailed unpaired Student’s t-test was used. For significant differences in variance, Welch’s correction was used. When applicable, two-way ANOVA was used followed by individual post hoc tests. Error probabilities of p < 0.05 were considered statistically significant. Sample size was chosen following previous publications. Variance was similar between the groups that were being statistically compared based on our observation. Data were analyzed using GraphPad Prism software.
Ethics approval
All methods and protocols involving animals were approved by the Institutional Animal Care and Use Committee (IACUC) of Wake Forest University School of Medicine (A24-094). Mice were kept in compliance with the NIH Guide for the Care and Use of Laboratory Animals (National Academies Press, 2011).
Data availability
All data are available upon request.
References
Laguesse S, Ron D. Protein translation and psychiatric disorders. The Neuroscientist. 2020;26:21–42.
Abelaira HM, Réus GZ, Neotti MV, Quevedo J. The role of mTOR in depression and antidepressant responses. Life Sci. 2014;101:10–14.
Oliveira MM, Klann E. eIF2-dependent translation initiation: memory consolidation and disruption in Alzheimer’s disease. Seminars in cell & developmental biology. 2022;125:101–9.
Cozachenco D, Ribeiro FC, Ferreira ST. Defective proteostasis in Alzheimer’s disease. Ageing research reviews. 2023;85:101862.
Jan A, Jansonius B, Delaidelli A, Bhanshali F, An YA, Ferreira N, et al. Activity of translation regulator eukaryotic elongation factor-2 kinase is increased in Parkinson disease brain and its inhibition reduces alpha synuclein toxicity. Acta Neuropathol Commun. 2018;6:54.
Monteggia LM, Gideons E, Kavalali ET. The role of eukaryotic elongation factor 2 kinase in rapid antidepressant action of ketamine. Biological Psychiatry. 2013;73:1199–203.
Alberini CM. The role of protein synthesis during the labile phases of memory: revisiting the skepticism. Neurobiology of learning and memory. 2008;89:234–46.
Klann E, Dever TE. Biochemical mechanisms for translational regulation in synaptic plasticity. Nat Rev Neurosci. 2004;5:931–42.
Gebauer F, Hentze MW. Molecular mechanisms of translational control. Nature Reviews Molecular Cell Biology. 2004;5:827–35.
James CC, Smyth JW. Alternative mechanisms of translation initiation: An emerging dynamic regulator of the proteome in health and disease. Life Sci. 2018;212:138–44.
Gismondi A, Caldarola S, Lisi G, Juli G, Chellini L, Iadevaia V, et al. Ribosomal stress activates eEF2K-eEF2 pathway causing translation elongation inhibition and recruitment of terminal oligopyrimidine (TOP) mRNAs on polysomes. Nucleic Acids Res. 2014;42:12668–80.
Smith PR, Garcia G, Meyer AR, Ryazanov AG, Ma T, Loerch S, et al. eEF2K regulates pain through translational control of BDNF. Molecular Cell. 2025;85:756–69.e755.
Christopher. Regulation and roles of elongation factor 2 kinase. Biochemical Society Transactions. 2015;43:328–32.
Browne GJ, Proud CG. Regulation of peptide-chain elongation in mammalian cells. Eur J Biochem. 2002;269:5360–8.
Kenney JW, Moore CE, Wang X, Proud CG. Eukaryotic elongation factor 2 kinase, an unusual enzyme with multiple roles. Advances in Biological Regulation. 2014;55:15–27.
Ma T. Roles of eukaryotic elongation factor 2 kinase (eEF2K) in neuronal plasticity, cognition, and Alzheimer disease. J Neurochem. 2023;166:47–57.
Szwed A, Kim E, Jacinto E. Regulation and metabolic functions of mTORC1 and mTORC2. Physiol Rev. 2021;101:1371–426.
Hardie DG. Sensing of energy and nutrients by AMP-activated protein kinase. The American Journal of Clinical Nutrition. 2011;93:891S–6S.
Hardie DG. AMP-activated protein kinase: maintaining energy homeostasis at the cellular and whole-body levels. Annual Review of Nutrition. 2014;34:31–55.
Inoki K, Li Y, Xu T, Guan KL. Rheb GTPase is a direct target of TSC2 GAP activity and regulates mTOR signaling. Genes Dev. 2003;17:1829–34.
Smiles WJ, Ovens AJ, Kemp BE, Galic S, Petersen J, Oakhill JS. New developments in AMPK and mTORC1 cross-talk. Essays in biochemistry. 2024;68:321–36.
Horman S, Browne G, Krause U, Patel J, Vertommen D, Bertrand L, et al. Activation of AMP-activated protein kinase leads to the phosphorylation of elongation factor 2 and an inhibition of protein synthesis. Curr Biol. 2002;12:1419–23.
Johanns M, Pyr dit Ruys S, Houddane A, Vertommen D, Herinckx G, Hue L, et al. Direct and indirect activation of eukaryotic elongation factor 2 kinase by AMP-activated protein kinase. Cellular Signalling. 2017;36:212–21.
Browne GJ, Finn SG, Proud CG. Stimulation of the AMP-activated protein kinase leads to activation of eukaryotic elongation factor 2 kinase and to its phosphorylation at a novel site, serine 398. J Biol Chem. 2004;279:12220–31.
Py BF, Boyce M, Yuan J. A critical role of eEF-2K in mediating autophagy in response to multiple cellular stresses. Autophagy. 2009;5:393–6.
Kruiswijk F, Yuniati L, Magliozzi R, Low TY, Lim R, Bolder R, et al. Coupled activation and degradation of eEF2K regulates protein synthesis in response to genotoxic stress. Science signaling. 2012;5:ra40.
Huber-Keener KJ, Evans BR, Ren X, Cheng Y, Zhang Y, Hait WN, et al. Phosphorylation of elongation factor-2 kinase differentially regulates the enzyme’s stability under stress conditions. Biochem Biophys Res Commun. 2012;424:308–14.
Leprivier G, Remke M, Rotblat B, Dubuc A, Abigail R, Kool M, et al. The eEF2 kinase confers resistance to nutrient deprivation by blocking translation elongation. Cell. 2013;153:1064–79.
Isaac AR, Chauvet MG, Lima-Filho R, Wagner BdA, Caroli BG, Leite REP, et al. Defective regulation of the eIF2-eIF2B translational axis underlies depressive-like behavior in mice and correlates with major depressive disorder in humans. Translational Psychiatry. 2024;14:397.
Blaze J, Chen S, Heissel S, Alwaseem H, Landinez Macias MP, Peter C, et al. Altered tRNA expression profile associated with codon-specific proteomic changes in the suicide brain. Molecular Psychiatry. 2025;30:2871–9.
Adaikkan C, Taha E, Barrera I, David O, Rosenblum K. Calcium/calmodulin-dependent protein kinase ii and eukaryotic elongation factor 2 kinase pathways mediate the antidepressant action of ketamine. Biological Psychiatry. 2018;84:65–75.
Feng J, Zhou Q, Gao W, Wu Y, Mu R. Seeking for potential pathogenic genes of major depressive disorder in the gene expression omnibus database. Asia-Pacific Psychiatry. 2020;12:e12379.
Chu VT, Weber T, Graf R, Sommermann T, Petsch K, Sack U, et al. Efficient generation of Rosa26 knock-in mice using CRISPR/Cas9 in C57BL/6 zygotes. BMC biotechnology. 2016;16:4.
Yang W, Zhou X, Zimmermann HR, Ma T. Brain-specific suppression of AMPKα2 isoform impairs cognition and hippocampal LTP by PERK-mediated eIF2α phosphorylation. Molecular Psychiatry. 2021;26:1880–97.
Wiebe S, Nagpal A, Sonenberg N. Dysregulated translational control in brain disorders: from genes to behavior. Current Opinion in Genetics & Development. 2020;65:34–41.
Santini E, Klann E. Dysregulated mTORC1-dependent translational control: from brain disorders to psychoactive drugs. Frontiers in Behavioral Neuroscience. 2011;5:76.
Zhang T, Wang X, Jester HM, Zhou X, Ma T. Characterization of apathy-like behaviors in mouse models of down syndrome. J Alzheimers Dis. 2024;101:1217–26.
Acikgoz B, Dalkiran B, Dayi A. An overview of the currency and usefulness of behavioral tests used from past to present to assess anxiety, social behavior and depression in rats and mice. Behavioural processes. 2022;200:104670.
Nandi A, Virmani G, Barve A, Marathe S. DBscorer: an open-source software for automated accurate analysis of rodent behavior in forced swim test and tail suspension test. eneuro. 2021;8:ENEURO.0305-0321.2021.
Pang TY, Renoir T, Du X, Lawrence AJ, Hannan AJ. Depression-related behaviours displayed by female C57BL/6J mice during abstinence from chronic ethanol consumption are rescued by wheel-running. The European journal of neuroscience. 2013;37:1803–10.
Pless A, Ware D, Saggu S, Rehman H, Morgan J, Wang Q. Understanding neuropsychiatric symptoms in Alzheimer’s disease: challenges and advances in diagnosis and treatment. Front Neurosci. 2023;17:1263771.
Keszycki R, Rodriguez G, Dunn JT, Locci A, Orellana H, Haupfear I, et al. Characterization of apathy-like behaviors in the 5xFAD mouse model of Alzheimer’s disease. Neurobiol Aging. 2023;126:113–22.
Deacon RM. Assessing nest building in mice. Nat Protoc. 2006;1:1117–9.
Lozupone M, Dibello V, Sardone R, Altamura M, Bellomo A, Daniele A, et al. Social dysfunction and apathy: transdiagnostic domains in late-life cognitive disorders. J Alzheimers Dis. 2024;100:S57–61.
Yang M, Silverman JL, Crawley JN. Automated three-chambered social approach task for mice. Current protocols in neuroscience 2011;Chapter 8:Unit 8.26.
Martinetz S, Meinung CP, Jurek B, von Schack D, van den Burg EH, Slattery DA, et al. De novo protein synthesis mediated by the eukaryotic elongation factor 2 is required for the anxiolytic effect of oxytocin. Biol Psychiatry. 2019;85:802–11.
Ma X, Li L, Li Z, Huang Z, Yang Y, Liu P, et al. eEF2 in the prefrontal cortex promotes excitatory synaptic transmission and social novelty behavior. EMBO Rep. 2022;23:e54543.
Dulawa SC, Hen R. Recent advances in animal models of chronic antidepressant effects: the novelty-induced hypophagia test. Neuroscience and biobehavioral reviews. 2005;29:771–83.
Kasica NP, Zhou X, Jester HM, Holland CE, Ryazanov AG, Forshaw TE, et al. Homozygous knockout of eEF2K alleviates cognitive deficits in APP/PS1 Alzheimer’s disease model mice independent of brain amyloid β pathology. Front Aging Neurosci. 2022;14:959326.
Kasica NP, Zhou X, Yang Q, Wang X, Yang W, Zimmermann HR, et al. Antagonists targeting eEF2 kinase rescue multiple aspects of pathophysiology in Alzheimer’s disease model mice. J Neurochem. 2022;160:524–39.
Wang X, Yang Q, Zhou X, Keene CD, Ryazanov AG, Ma T. Suppression of eEF2 phosphorylation alleviates synaptic failure and cognitive deficits in mouse models of Down syndrome. Alzheimers Dement. 2024;20:5357–74.
Antunes M, Biala G. The novel object recognition memory: Neurobiology, test procedure, and its modifications. Cognitive Processing. 2012;13:93–110.
Beckelman BC, Yang W, Kasica NP, Zimmermann HR, Zhou X, Keene CD, et al. Genetic reduction of eEF2 kinase alleviates pathophysiology in Alzheimer’s disease model mice. J Clin Invest. 2019;129:820–33.
Bromley-Brits K, Deng Y, Song W. Morris water maze test for learning and memory deficits in Alzheimer’s disease model mice. Journal of visualized experiments : JoVE. 2011.
Bliss TV, Collingridge GL. A synaptic model of memory: long-term potentiation in the hippocampus. Nature. 1993;361:31–9.
Heise C, Gardoni F, Culotta L, di Luca M, Verpelli C, Sala C. Elongation factor-2 phosphorylation in dendrites and the regulation of dendritic mRNA translation in neurons. Frontiers in Cellular Neuroscience. 2014;8:35.
Park S, Park JM, Kim S, Kim JA, Shepherd JD, Smith-Hicks CL, et al. Elongation factor 2 and fragile X mental retardation protein control the dynamic translation of Arc/Arg3.1 essential for mGluR-LTD. Neuron. 2008;59:70–83.
Scheetz AJ, Nairn AC, Constantine-Paton M. NMDA receptor-mediated control of protein synthesis at developing synapses. Nat Neurosci. 2000;3:211–6.
Taha E, Gildish I, Gal-Ben-Ari S, Rosenblum K. The role of eEF2 pathway in learning and synaptic plasticity. Neurobiology of learning and memory. 2013;105:100–6.
Mäki-Marttunen T, Blackwell KT, Akkouh I, Shadrin A, Valstad M, Elvsåshagen T, et al. Genetic mechanisms for impaired synaptic plasticity in schizophrenia revealed by computational modeling. Proc Natl Acad Sci USA. 2024;121:e2312511121.
Fries GR, Saldana VA, Finnstein J, Rein T. Molecular pathways of major depressive disorder converge on the synapse. Molecular Psychiatry. 2023;28:284–97.
Liu W, Ge T, Leng Y, Pan Z, Fan J, Yang W, et al. The role of neural plasticity in depression: from hippocampus to prefrontal cortex. Neural plasticity. 2017;2017:6871089.
Tartt AN, Mariani MB, Hen R, Mann JJ, Boldrini M. Dysregulation of adult hippocampal neuroplasticity in major depression: pathogenesis and therapeutic implications. Mol Psychiatry. 2022;27:2689–99.
Ma T, Chen Y, Vingtdeux V, Zhao H, Viollet B, Marambaud P, et al. Inhibition of AMP-activated protein kinase signaling alleviates impairments in hippocampal synaptic plasticity induced by amyloid β. Journal of Neuroscience. 2014;34:12230–8.
Ma T, Hoeffer CA, Capetillo-Zarate E, Yu F, Wong H, Lin MT, et al. Dysregulation of the mTOR pathway mediates impairment of synaptic plasticity in a mouse model of alzheimer’s disease. PLoS ONE. 2010;5:e12845.
Zhou Y, Wu H, Li S, Chen Q, Cheng XW, Zheng J, et al. Requirement of TORC1 for late-phase long-term potentiation in the hippocampus. PLoS One. 2006;1:e16.
Creager R, Dunwiddie T, Lynch G. Paired-pulse and frequency facilitation in the CA1 region of the in vitro rat hippocampus. The Journal of physiology. 1980;299:409–24.
Schulz PE, Cook EP, Johnston D. Changes in paired-pulse facilitation suggest presynaptic involvement in long-term potentiation. J Neurosci. 1994;14:5325–37.
Costa-Mattioli M, Sossin WS, Klann E, Sonenberg N. Translational control of long-lasting synaptic plasticity and memory. Neuron. 2009;61:10–26.
Sutton MA, Schuman EM. Dendritic protein synthesis, synaptic plasticity, and memory. Cell. 2006;127:49–58.
Tan AM. Dendritic spine dysgenesis: An emerging concept in neuropsychiatric disease. Neurosci Lett. 2015;601:1–3.
Akhgari A, Michel TM, Vafaee MS. Dendritic spines and their role in the pathogenesis of neurodevelopmental and neurological disorders. Rev Neurosci. 2024;35:489–502.
Glausier JR, Lewis DA. Dendritic spine pathology in schizophrenia. Neuroscience. 2013;251:90–107.
Moyer CE, Shelton MA, Sweet RA. Dendritic spine alterations in schizophrenia. Neurosci Lett. 2015;601:46–53.
Risher WC, Ustunkaya T, Alvarado JS, Eroglu C. Rapid golgi analysis method for efficient and unbiased classification of dendritic spines. PLoS ONE. 2014;9:e107591.
Okabe S. Molecular anatomy of the postsynaptic density. Molecular and cellular neurosciences. 2007;34:503–18.
Sheng M, Kim E. The postsynaptic organization of synapses. Cold Spring Harbor Perspectives in Biology. 2011;3:a005678–a005678.
Schmidt EK, Clavarino G, Ceppi M, Pierre P. SUnSET, a nonradioactive method to monitor protein synthesis. Nature Methods. 2009;6:275–7.
Ostroff LE, Fiala JC, Allwardt B, Harris KM. Polyribosomes redistribute from dendritic shafts into spines with enlarged synapses during LTP in developing rat hippocampal slices. Neuron. 2002;35:535–45.
Leek JT, Johnson WE, Parker HS, Jaffe AE, Storey JD. The sva package for removing batch effects and other unwanted variation in high-throughput experiments. Bioinformatics. 2012;28:882–3.
Guilloux JP, Seney M, Edgar N, Sibille E. Integrated behavioral z-scoring increases the sensitivity and reliability of behavioral phenotyping in mice: relevance to emotionality and sex. Journal of neuroscience methods. 2011;197:21–31.
Kraeuter A-K. The use of integrated behavioural z-scoring in behavioural neuroscience – A perspective article. Journal of neuroscience methods. 2023;384:109751.
Lanctôt KL, Ismail Z, Bawa KK, Cummings JL, Husain M, Mortby ME, et al. Distinguishing apathy from depression: A review differentiating the behavioral, neuroanatomic, and treatment-related aspects of apathy from depression in neurocognitive disorders. Int J Geriatr Psychiatry. 2023;38:e5882.
Robert PH, Mulin E, Malléa P, David R. REVIEW: Apathy diagnosis, assessment, and treatment in Alzheimer’s disease. CNS Neurosci Ther. 2010;16:263–71.
Nobis L, Husain M. Apathy in Alzheimer’s disease. Current opinion in behavioral sciences. 2018;22:7–13.
Zhao QF, Tan L, Wang HF, Jiang T, Tan MS, Tan L, et al. The prevalence of neuropsychiatric symptoms in alzheimer’s disease: systematic review and meta-analysis. Journal of affective disorders. 2016;190:264–71.
Siafarikas N, Selbaek G, Fladby T, Šaltytė Benth J, Auning E, Aarsland D. Frequency and subgroups of neuropsychiatric symptoms in mild cognitive impairment and different stages of dementia in Alzheimer’s disease. Int Psychogeriatr. 2018;30:103–13.
Dauphinot V, Delphin-Combe F, Mouchoux C, Dorey A, Bathsavanis A, Makaroff Z, et al. Risk factors of caregiver burden among patients with Alzheimer’s disease or related disorders: a cross-sectional study. J Alzheimers Dis. 2015;44:907–16.
Riedijk SR, De Vugt ME, Duivenvoorden HJ, Niermeijer MF, Van Swieten JC, Verhey FR, et al. Caregiver burden, health-related quality of life and coping in dementia caregivers: a comparison of frontotemporal dementia and Alzheimer’s disease. Dement Geriatr Cogn Disord. 2006;22:405–12.
Chen CT, Chang CC, Chang WN, Tsai NW, Huang CC, Chang YT, et al. Neuropsychiatric symptoms in Alzheimer’s disease: associations with caregiver burden and treatment outcomes. Qjm. 2017;110:565–70.
Hongisto K, Hallikainen I, Selander T, Törmälehto S, Väätäinen S, Martikainen J, et al. Quality of Life in relation to neuropsychiatric symptoms in Alzheimer’s disease: 5-year prospective ALSOVA cohort study. Int J Geriatr Psychiatry. 2018;33:47–57.
Spalletta G, Long JD, Robinson RG, Trequattrini A, Pizzoli S, Caltagirone C, et al. Longitudinal neuropsychiatric predictors of death in alzheimer’s disease. J Alzheimers Dis. 2015;48:627–36.
Sousa M, Moreira F, Jesus-Ribeiro J, Marques I, Cunha F, Canário N, et al. Apathy profile in parkinson’s and huntington’s disease: a comparative cross-sectional study. European neurology. 2018;79:13–20.
Caga J, Turner MR, Hsieh S, Ahmed RM, Devenney E, Ramsey E, et al. Apathy is associated with poor prognosis in amyotrophic lateral sclerosis. European journal of neurology. 2016;23:891–7.
Funes CM, Lavretsky H, Ercoli L, St Cyr N, Siddarth P. Apathy mediates cognitive difficulties in geriatric depression. The American journal of geriatric psychiatry : official journal of the American Association for Geriatric Psychiatry. 2018;26:100–6.
Sultzer DL. Why apathy in alzheimer’s matters. The American journal of psychiatry. 2018;175:99–100.
Raimo S, Spitaleri D, Trojano L, Santangelo G. Apathy as a herald of cognitive changes in multiple sclerosis: A 2-year follow-up study. Multiple sclerosis (Houndmills, Basingstoke, England). 2020;26:363–71.
Andrews SC, Langbehn DR, Craufurd D, Durr A, Leavitt BR, Roos RA, et al. Apathy predicts rate of cognitive decline over 24 months in premanifest Huntington’s disease. Psychological medicine. 2021;51:1338–44.
Breitve MH, Brønnick K, Chwiszczuk LJ, Hynninen MJ, Aarsland D, Rongve A. Apathy is associated with faster global cognitive decline and early nursing home admission in dementia with lewy bodies. Alzheimer’s research & therapy. 2018;10:83.
D’Iorio A, Maggi G, Vitale C, Trojano L, Santangelo G. Pure apathy and cognitive dysfunctions in Parkinson’s disease: A meta-analytic study. Neuroscience and biobehavioral reviews. 2018;94:1–10.
Li H, Cui L, Wang M, Liao M, Li JB, Ouyang F, et al. Apathy is associated with striatal atrophy and cognitive impairment in cerebral small vessel disease. Journal of affective disorders. 2023;328:39–46.
Malpetti M, Jones PS, Tsvetanov KA, Rittman T, van Swieten JC, Borroni B, et al. Apathy in presymptomatic genetic frontotemporal dementia predicts cognitive decline and is driven by structural brain changes. Alzheimers Dement. 2021;17:969–83.
Seligman SC, Kamath V, Giovannetti T, Arnold SE, Moberg PJ. Olfaction and apathy in Alzheimer’s disease, mild cognitive impairment, and healthy older adults. Aging & mental health. 2013;17:564–70.
Cramer CK, Friedman JH, Amick MM. Olfaction and apathy in Parkinson’s disease. Parkinsonism & related disorders. 2010;16:124–6.
Hizli AA, Chi Y, Swanger J, Carter JH, Liao Y, Welcker M, et al. Phosphorylation of eukaryotic elongation factor 2 (eEF2) by cyclin A-cyclin-dependent kinase 2 regulates its inhibition by eEF2 kinase. Mol Cell Biol. 2013;33:596–604.
Henis M, Rücker T, Scharrenberg R, Richter M, Baltussen L, Hong S, et al. The autism susceptibility kinase, TAOK2, phosphorylates eEF2 and modulates translation. Science advances. 2024;10:eadf7001.
Meyuhas O, Kahan T. The race to decipher the top secrets of TOP mRNAs. Biochimica et Biophysica Acta (BBA) - Gene Regulatory Mechanisms. 2015;1849:801–11.
Verpelli C, Piccoli G, Zibetti C, Zanchi A, Gardoni F, Huang K, et al. Synaptic activity controls dendritic spine morphology by modulating eEF2-dependent BDNF synthesis. Journal of Neuroscience. 2010;30:5830–42.
Carlisle HJ, Kennedy MB. Spine architecture and synaptic plasticity. Trends Neurosci. 2005;28:182–7.
Tada T, Sheng M. Molecular mechanisms of dendritic spine morphogenesis. Curr Opin Neurobiol. 2006;16:95–101.
Nägerl UV, Eberhorn N, Cambridge SB, Bonhoeffer T. Bidirectional activity-dependent morphological plasticity in hippocampal neurons. Neuron. 2004;44:759–67.
De Roo M, Klauser P, Garcia PM, Poglia L, Muller D. Spine dynamics and synapse remodeling during LTP and memory processes. Prog Brain Res. 2008;169:199–207.
Heck N, Santos MD. Dendritic spines in learning and memory: from first discoveries to current insights. Advances in neurobiology. 2023;34:311–48.
Kasai H, Fukuda M, Watanabe S, Hayashi-Takagi A, Noguchi J. Structural dynamics of dendritic spines in memory and cognition. Trends Neurosci. 2010;33:121–9.
van der Zee EA. Synapses, spines and kinases in mammalian learning and memory, and the impact of aging. Neuroscience and biobehavioral reviews. 2015;50:77–85.
Reyes-Lizaola S, Luna-Zarate U, Tendilla-Beltrán H, Morales-Medina JC, Flores G. Structural and biochemical alterations in dendritic spines as key mechanisms for severe mental illnesses. Progress in neuro-psychopharmacology & biological psychiatry. 2024;129:110876.
Li Y, Abdourahman A, Tamm JA, Pehrson AL, Sánchez C, Gulinello M. Reversal of age-associated cognitive deficits is accompanied by increased plasticity-related gene expression after chronic antidepressant administration in middle-aged mice. Pharmacology, biochemistry, and behavior. 2015;135:70–82.
Sarowar T, Grabrucker AM. Actin-dependent alterations of dendritic spine morphology in shankopathies. Neural plasticity. 2016;2016:8051861.
McKinney BC, MacDonald ML, Newman JT, Shelton MA, DeGiosio RA, Kelly RM, et al. Density of small dendritic spines and microtubule-associated-protein-2 immunoreactivity in the primary auditory cortex of subjects with schizophrenia. Neuropsychopharmacology. 2019;44:1055–61.
Feng J, Yan Z, Ferreira A, Tomizawa K, Liauw JA, Zhuo M, et al. Spinophilin regulates the formation and function of dendritic spines. Proc Natl Acad Sci USA. 2000;97:9287–92.
Muhammad K, Reddy-Alla S, Driller JH, Schreiner D, Rey U, Böhme MA, et al. Presynaptic spinophilin tunes neurexin signalling to control active zone architecture and function. Nat Commun. 2015;6:8362.
Dityatev A, Bukalo O, Schachner M. Modulation of synaptic transmission and plasticity by cell adhesion and repulsion molecules. Neuron glia biology. 2008;4:197–209.
Guilmatre A, Huguet G, Delorme R, Bourgeron T. The emerging role of SHANK genes in neuropsychiatric disorders. Developmental neurobiology. 2014;74:113–22.
Chow F, Gong Y, Lippa CF. The potential role of insulin on the shank-postsynaptic platform in neurodegenerative diseases involving cognition. American journal of Alzheimer’s disease and other dementias. 2014;29:303–10.
Sun Y, Lin Y, Liang N, Xue Z, Xu J, Lin L, et al. Methylome-wide association study of adolescent depressive episode with psychotic symptoms and childhood trauma. Journal of affective disorders. 2025;370:439–48.
Grubisha MJ, Sun X, MacDonald ML, Garver M, Sun Z, Paris KA, et al. MAP2 is differentially phosphorylated in schizophrenia, altering its function. Mol Psychiatry. 2021;26:5371–88.
Marchisella F, Coffey ET, Hollos P. Microtubule and microtubule associated protein anomalies in psychiatric disease. Cytoskeleton (Hoboken, NJ). 2016;73:596–611.
Ren SY, Wang ZZ, Zhang Y, Chen NH. Potential application of endocannabinoid system agents in neuropsychiatric and neurodegenerative diseases-focusing on FAAH/MAGL inhibitors. Acta pharmacologica Sinica. 2020;41:1263–71.
Rafiei D, Kolla NJ. Elevated brain fatty acid amide hydrolase induces depressive-like phenotypes in rodent models: a review. Int J Mol Sci. 2021;22:1047.
Morris G, Sominsky L, Walder KR, Berk M, Marx W, Carvalho AF, et al. Inflammation and nitro-oxidative stress as drivers of endocannabinoid system aberrations in mood disorders and schizophrenia. Mol Neurobiol. 2022;59:3485–503.
Brocos-Mosquera I, Gabilondo AM, Meana JJ, Callado LF, Erdozain AM. Spinophilin expression in postmortem prefrontal cortex of schizophrenic subjects: Effects of antipsychotic treatment. European neuropsychopharmacology : the journal of the European College of Neuropsychopharmacology. 2021;42:12–21.
Najera K, Fagan BM, Thompson PM. SNAP-25 in major psychiatric disorders: a review. Neuroscience. 2019;420:79–85.
Leung E, Lau EW, Liang A, de Dios C, Suchting R, Östlundh L, et al. Alterations in brain synaptic proteins and mRNAs in mood disorders: a systematic review and meta-analysis of postmortem brain studies. Mol Psychiatry. 2022;27:1362–72.
Kim KY, Shin KY, Chang KA. GFAP as a potential biomarker for alzheimer’s disease: a systematic review and meta-analysis. Cells. 2023;12:1309.
Garcia-Escobar G, Manero RM, Fernández-Lebrero A, Ois A, Navalpotro-Gómez I, Puente-Periz V, et al. Blood biomarkers of alzheimer’s disease and cognition: a literature review. Biomolecules. 2024;14:93.
Sanchez E, Wilkinson T, Coughlan G, Mirza S, Baril AA, Ramirez J, et al. Association of plasma biomarkers with cognition, cognitive decline, and daily function across and within neurodegenerative diseases: results from the ontario neurodegenerative disease research initiative. Alzheimers Dement. 2024;20:1753–70.
Wei YC, Kung YC, Lin C, Yeh CH, Chen PY, Huang WY, et al. Differential neuropsychiatric associations of plasma biomarkers in older adults with major depression and subjective cognitive decline. Transl Psychiatry. 2024;14:333.
Kim R, Healey KL, Sepulveda-Orengo MT, Reissner KJ. Astroglial correlates of neuropsychiatric disease: From astrocytopathy to astrogliosis. Progress in neuro-psychopharmacology & biological psychiatry. 2018;87:126–46.
Michel M, Fiebich BL, Kuzior H, Meixensberger S, Berger B, Maier S, et al. Increased GFAP concentrations in the cerebrospinal fluid of patients with unipolar depression. Transl Psychiatry. 2021;11:308.
Ellis RJ, Fan Y, Grelotti D, Tang B, Letendre S, He JJ. Astrocyte activation is a potential mechanism underlying depressed mood and apathy in people with HIV. Journal of neurology and psychology. 2022;9:05.
Cheng P, Huang W, Yang M, Chen Z, Geng Y, Zhang X, et al. Autoimmune GFAP astrocytopathy after viral encephalitis: a case report of bimodal overlapping encephalitis. Frontiers in immunology. 2023;14:1258048.
Yin P, Niu X, Guan C, Zhang Z, Liu Y, Li J, et al. Relationship between increased serum neurofilament light chain and glial fibrillary acidic protein levels with non-motor symptoms in patients with Parkinson’s disease. Psychogeriatrics. 2024;24:415–25.
Acevedo JM, Hoermann B, Schlimbach T, Teleman AA. Changes in global translation elongation or initiation rates shape the proteome via the Kozak sequence. Sci Rep. 2018;8:4018.
Chu D, Kazana E, Bellanger N, Singh T, Tuite MF, von der Haar T. Translation elongation can control translation initiation on eukaryotic mRNAs. Embo j. 2014;33:21–34.
Campbell S, Macqueen G. The role of the hippocampus in the pathophysiology of major depression. J Psychiatry Neurosci. 2004;29:417–26.
Cao B, Passos IC, Mwangi B, Amaral-Silva H, Tannous J, Wu MJ, et al. Hippocampal subfield volumes in mood disorders. Mol Psychiatry. 2017;22:1352–8.
Femenía T, Gómez-Galán M, Lindskog M, Magara S. Dysfunctional hippocampal activity affects emotion and cognition in mood disorders. Brain Research. 2012;1476:58–70.
Matuskova V, Ismail Z, Nikolai T, Markova H, Cechova K, Nedelska Z, et al. Corrigendum: mild behavioral impairment is associated with atrophy of entorhinal cortex and hippocampus in a memory clinic cohort. Front Aging Neurosci. 2022;14:884077.
Steffens DC, Fahed M, Manning KJ, Wang L. The neurobiology of apathy in depression and neurocognitive impairment in older adults: a review of epidemiological, clinical, neuropsychological and biological research. Transl Psychiatry. 2022;12:525.
Ma, Trinh MA T, Wexler AJ, Bourbon C, Gatti E, Pierre P, et al. Suppression of eIF2α kinases alleviates Alzheimer’s disease-related plasticity and memory deficits. Nature Neuroscience. 2013;16:1299–305.
Zimmermann HR, Yang W, Beckelman BC, Kasica NP, Zhou X, Galli LD, et al. Genetic removal of eIF2α kinase PERK in mice enables hippocampal L-LTP independent of mTORC1 activity. J Neurochem. 2018;146:133–44.
Kraeuter A-K, Guest PC, Sarnyai Z The Y-maze for assessment of spatial working and reference memory in mice. Springer New York 2019, pp 105-11.
Angoa-Pérez M, Kane MJ, Briggs DI, Francescutti DM, Kuhn DM Marble burying and nestlet shredding as tests of repetitive, compulsive-like behaviors in mice. Journal of visualized experiments : JoVE 2013;82:50978.
Jirkof P. Burrowing and nest building behavior as indicators of well-being in mice. Journal of neuroscience methods. 2014;234:139–46.
Deacon RM. Burrowing in rodents: a sensitive method for detecting behavioral dysfunction. Nat Protoc. 2006;1:118–21.
PCAtools: Everything Principle Component Analysis. https://github.com/kevinblighe/PCAtools, 2019, Accessed Date Accessed 2019 Accessed.
Acknowledgements
We would like to acknowledge the support provided by the Proteomics and Metabolomics Shared Resource of the Wake Forest School of Medicine and Wake Forest Baptist Comprehensive Cancer Center (NIH/NCI P30 CA12197). We would like to acknowledge the support provided by the Cellular Imaging Shared Resource of the Wake Forest School of Medicine. We would also like to acknowledge advice and training received for behavior paradigms from Sara Jones, Katherine Holleran, Kim Raab-Graham, and This work was supported by National Institutes of Health Grants R01 AG073823 and RF1 AG082388 (T.M.).
Funding
Open access funding provided by the Carolinas Consortium.
Author information
Authors and Affiliations
Contributions
HMJ conceptualized experiments, collected and analyzed data, and wrote the manuscript. NN collected and analyzed data and assisted with proteomics analysis. QY performed electrophysiology experiments, collected and analyzed data. YZ performed immunofluorescence experiments. AS performed Western blot experiments. XY maintained mouse colony and genotyping experiments. DC performed the RT-PCR experiment. EU, HP, JL, and CMF advised, and collected data for proteomics experiments. PS, CGP and KR advised and edited the manuscript. TM conceptualized experiments, advised and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jester, H.M., Nicol, N., Yang, Q. et al. Overactive Neuronal eEF2K/eEF2 signaling is associated with cognitive impairment and apathy-like behavior. Mol Psychiatry (2025). https://doi.org/10.1038/s41380-025-03408-z
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41380-025-03408-z