Abstract
Posttraumatic stress disorder (PTSD) is a highly heterogeneous psychiatric disorder, complicating efforts to identify consistent biological markers and develop targeted treatments for individuals exposed to trauma. Recent research has identified a distinct intrusive-hypervigilant (IH) phenotype, which is characterized by heightened intrusive reexperiencing and hypervigilance symptoms along with elevated levels of pituitary adenylate cyclase-activating polypeptide (PACAP), a neuropeptide involved in stress response via amygdala signaling. In an independent sample of 172 symptomatic trauma-exposed adults, we replicated this IH phenotype using latent profile analysis of Clinician-Administered PTSD Scale for DSM-5 symptom severity ratings and expanded its biological characterization using resting-state functional magnetic resonance imaging (rs-fMRI). Consistent with prior work, the identified IH group demonstrated more severe intrusive reexperiencing (Cohen’s d’s = 0.61-6.93) and hypervigilance symptoms (d’s = 0.57-0.88) and higher PACAP levels compared to groups with generally High (d = 0.35) or Low (d = 0.44) symptom severity. Additionally, the IH phenotype exhibited stronger functional connectivity of the centromedial, but not basolateral, amygdala with regions in the occipital cortex (d’s = 0.78-0.95), precuneus (d’s = 1.20-1.21), and medial prefrontal cortex (d’s = 0.81-1.18)—areas primarily within the Default Mode and Visual Networks. Meta-analytic decoding linked these regions to mental imagery, memory processing, fear, and threat perception. These findings support the existence of an IH phenotype of posttraumatic stress that may exhibit a distinct biological profile, characterized by exaggerated interactions between memory, threat, and arousal systems that may be mediated by PACAP and its effects on amygdala connectivity. This phenotype may serve as a promising target for precision psychiatry approaches, including pharmacological and neurotherapeutic interventions that modulate PACAP signaling and amygdala connectivity.
Similar content being viewed by others
Introduction
Posttraumatic stress disorder (PTSD) is a markedly heterogeneous psychiatric disorder, with potentially 636,129 different combinations of 20 symptoms across four distinct symptom clusters that fulfill DSM-5 diagnostic criteria for PTSD [1, 2]. Beyond the diagnosis of PTSD, a significant number of trauma-exposed individuals report clinically significant distress and impairment through the endorsement of PTSD symptoms that do not meet full DSM-5 diagnostic criteria (i.e., subthreshold PTSD) [3]. Combined, this vast clinical heterogeneity hinders efforts to identify reliable biological substrates that are needed to advance mechanism-based therapeutics of posttraumatic stress.
The heterogeneity of posttraumatic stress has motivated the identification of “subtypes”, or distinct clinical phenotypes that may have more homogeneous biological mechanisms that can be reliably targeted under a precision psychiatry framework. However, to date, the emergence of reliable subtypes remains elusive. A dissociative subtype of PTSD is the only recognized diagnostic subtype based on DSM-5 criteria and is characterized by the additional endorsement of dissociative symptoms beyond the “classic” 20 PTSD symptoms. Additionally, the ICD-11 recognizes Complex PTSD as a distinct diagnosis that adds disturbances in self-organization to core PTSD symptoms [4]. Beyond these recognized subtypes, additional work has offered evidence for other subtypes based on a predominance of internalizing versus externalizing or fear versus dysphoria symptoms [5,6,7,8,9]. However, other work suggests that the most common subgroups across latent profile and class analyses of PTSD symptoms are “low”, “medium”, and “high” symptoms [10, 11].
Importantly, much of this work has utilized samples of participants with shared or similar traumatic experiences. This approach may fail to capture the diverse etiology of PTSD, as different types of traumatic experiences (e.g., single versus recurrent, interpersonal versus non-interpersonal) may result in different symptoms and neurobiological alterations [12,13,14,15]. Moreover, prior latent class and profile analyses focus on samples meeting full diagnostic criteria for PTSD. This overlooks the significant number of trauma-exposed individuals who experience clinically-significant distress and impairment but do not meet diagnostic thresholds for all four symptom clusters [3].
Addressing these concerns in a diverse, trauma-exposed sample with subthreshold and threshold PTSD, Adams et al. [16] recently identified a distinct clinical phenotype of posttraumatic stress that was marked by specific elevations in intrusive reexperiencing and hypervigilance symptoms [16]. Moreover, they demonstrated that the intrusive-hypervigilant (IH) clinical profile had impaired fear extinction retention. This clinical phenotype aligns with prior work demonstrating a link between arousal processes and intrusive trauma memories: hyperarousal symptoms predict later reexperiencing symptoms [17,18,19], experimental manipulation of physiological arousal triggers the reactivation of aversive memories [20, 21], and aversive memories are coupled with elevations in sympathetic arousal [22,23,24,25,26]. At the neurobiological level, arousal facilitates reinstatement of salient memory information via increased activity of locus coeruleus (LC) and amygdala pathways that enhance the expression of prioritized memory representations in cortical regions [27]. Conceptual models further propose that LC and amygdala circuit activation leads to rapid reactivation in sensory-perceptual cortices [28,29,30], such that retrieval under high arousal leads to rapid and involuntary reactivation of sensory memory traces [31].
Consistent with the neurobiological link between arousal and memory processes, Adams et al. [16] further demonstrated that the IH clinical phenotype had robust elevations in circulating levels of pituitary adenylate cyclase-activating polypeptide (PACAP). PACAP is a highly-conserved neuropeptide that serves as a critical regulator of stress response, including the activation of physiological arousal and defensive responding through signaling within the amygdala complex, particularly the central amygdala [32,33,34,35,36,37,38,39]. It has been linked to the pathophysiology of PTSD through alterations in amygdala activity and subsequent effects on arousal, threat processing, and fear memory [40, 41]. Recent evidence further links PACAP to exaggerated functional connectivity (FC) of the amygdala with regions of the default mode network (DMN), a network frequently implicated in PTSD through its role in autobiographical memory, arousal, and internally (versus externally) oriented attention [42]. Additional evidence suggests that PACAP’s effects are stronger in female than male patients, indicating that it may reveal mechanistic insights into sex differences in PTSD risk and prevalence [16, 40]. Correspondingly, Adams et al. demonstrated that the IH subtype with elevated PACAP was predominantly female, which is consistent with prior work highlighting interactions between estrogen and PACAP systems in conferring risk for PTSD [40]. This suggests a biological phenotype of posttraumatic stress that is tied to biological sex. Overall, findings from Adams et al. reveal a potentially more homogenous clinical-biological subgroup that pairs elevated intrusive reexperiencing and hypervigilance symptoms with biological and psychophysiological markers of fear memory and arousal. This may represent an important step towards a biologically-grounded clinical subtype to target through mechanism-based interventions in a precision psychiatry approach.
The current study sought to replicate findings of an IH clinical phenotype with elevations in PACAP and build upon their biological profile by providing novel evidence for distinct patterns of amygdala FC. Using resting-state functional magnetic resonance imaging (rs-fMRI) in an independent sample of symptomatic trauma-exposed adults, we tested the hypothesis that an IH phenotype would be associated with exaggerated FC of the amygdala with networks implicated in trauma memory and arousal. Consistent with prior work, we hypothesized these effects would be specific to the right central amygdala, where PACAP-resting state associations are strongest [42].
Methods
Participants
One-hundred and seventy-two trauma-exposed adults were recruited and enrolled via advertisements in the local community. This sample is inclusive of the sample reported in previously published work examining the association between PACAP and amygdala FC in trauma-exposed adults, along with additional participants recruited after the initial publication [42]. Study procedures were approved by the Mass General Brigham Human Research Committee, and all participants provided written informed consent. Participants completed a fasting blood draw, clinical interview, self-report questionnaires, and a 13-min eyes-open resting-state fMRI scan. Participants were included if they met DSM-5 diagnostic criteria for at least two PTSD symptom clusters based on the Clinician Administered PTSD Scale for DSM-5 (CAPS-5), constituting subthreshold (n = 57; 33%) and threshold (n = 115; 67%) PTSD [3]. Given a prior interest in biological sex differences motivated by prior work [43], participants were required to be the same sex as assigned at birth, female participants were to be premenopausal, and participants with a history of receiving hormonal replacement therapy or undergoing surgery to change biological sex were excluded. Other exclusion criteria are detailed in Clancy et al. [42].
Of the 172 enrolled participants with complete clinical interview data, 158 (106 female) had detectable serum PACAP levels (excluded: undetectable values = 13, levels below reliable detection threshold = 1). A total of 134 (85 female) had usable fMRI data (excluded: excessive motion = 14, incomplete scanning = 7, poor structural-functional alignment = 4, significant artifact = 6, no scanning performed = 7). Demographic and clinical characteristics of the sample are provided in Table 1.
Clinician-administered PTSD scale for DSM-5 (CAPS-5)
The CAPS-5 [44], the gold-standard diagnostic interview for PTSD, was administered by doctoral-level clinicians. This interview consists of 30 items designed to assess the onset, duration, and impact of PTSD symptoms, yielding a determination of PTSD diagnosis and symptom severity. Specifically, each of the 20 DSM-5 PTSD symptoms is given an overall severity rating, which reflects the intensity and frequency of the symptom within the past month. Severity ratings for each symptom are made using a 0-4 Likert Scale (0 = Absent, 4 = Extreme/incapacitating).
PACAP
Details about PACAP collection and analysis are provided in the Supplementary Methods and Clancy et al. [42]. Fourteen samples were excluded due to undetectable (n = 13) or unreliably low (n = 1) PACAP levels, resulting in a final sample of n = 158 for PACAP analyses. Consistent with Adams et al. [16], PACAP values were square-root-transformed to reduce data compression and correct a left-skewed distribution.
PACAP levels were analyzed in two batches. There was a significant effect of Batch on PACAP levels (Batch 1 mean = 29.53, Batch 2 mean = 20.98; t = 3.08, p = 0.003), with unequal variances across the two batches (Levene’s Test F = 6.22, p = 0.014). These Batch differences were not explained by clinical or demographic variables; therefore, Batch (binary 1 or 2) was included as a covariate in all PACAP analyses.
Latent symptom profile analysis
Gaussian mixture modeling was performed using the Mclust package in R [45] to identify latent symptom profiles using symptom severity ratings from the 20-item CAPS-5 interview. Specifically, we aimed to replicate the IH symptom profile identified in Adams et al. [16] through a complementary yet distinct approach to demonstrate the external reliability of this symptom profile, choosing to utilize latent profile over latent class analysis to retain information gained from the continuous nature of CAPS-5 symptom severity data. Four models with different covariance structures examined a range of 1 to 4 profiles. Optimal model fit was determined using the Bayesian Information Criterion (BIC). Additional details are reported in the Supplementary Methods.
MRI data acquisition and preprocessing
Imaging was conducted at the McLean Hospital Imaging Center on a 3T Siemens Prisma scanner. MRI data were preprocessed using fMRIPrep version 20.2.7 [46]. Fourteen participants were excluded for excessive motion, 7 had incomplete scanning, 4 had inadequate alignment of structural and functional scans, 6 had significant imaging artifacts, and 7 had no scanning performed, resulting in a final sample of n = 136 for fMRI analyses. Notably, participants excluded from fMRI analyses did not differ from those included on any demographic, clinical, or biological variables of interest (Supplementary Table 1). Protocol details and additional denoising steps are reported in the supplement and Clancy et al. [42]. Cleaned timeseries from these preprocessing and denoising steps were subsequently submitted to resting-state functional connectivity analyses.
Resting-state functional connectivity analyses
Seed-based resting state connectivity analyses were conducted in CONN using cleaned timeseries of BOLD signal extracted from left and right (l/r) centromedial (CMA) and basolateral (BLA) amygdala regions of interest (ROIs) generated from the JuBrain Atlas (Supplementary Fig. S1) [47, 48]. Whole-brain seed-based correlations were performed for l/rCMA and l/rBLA seeds to compute whole-brain FC maps. Seed-based FC values were Fisher’s z-transformed prior to further statistical analyses.
Analytic plan
Given our specific a priori interest in an IH symptom profile, an identified group matching this profile was compared to other symptom groups to determine the clinical and biological correlates of this IH phenotype. First, the identified IH group was compared to all other symptom profiles combined into a single non-IH group, as prior findings showed no PACAP differences between non-IH clinical phenotypes [16]. This approach was contingent on the absence of any statistically significant difference between the non-IH clinical phenotypes across biological markers of interest in our sample. Comparisons of the IH group versus the single non-IH group were followed by separate comparisons between the IH group and other groups individually.
Categorical characteristics were compared between IH and non-IH phenotypes using Chi-squared tests of proportions for sex assigned at birth (male vs. female) and PTSD diagnosis (yes vs. no) using the proportions test function in R [49], testing for higher female representation and PTSD diagnosis rates in the IH group. Model-based estimated marginal means in PACAP levels by phenotypic group, and test statistics and p-values for associated significance tests, were calculated from results of linear regression models controlling for Batch using the emmeans package for R [50]. Robust standard errors were used for all analyses examining PACAP with Batch as a covariate due to unequal variances between batches. Complementary mixed-effects models with Batch as a random effect are presented in the Supplementary Results; primary analyses treated batch as a fixed effect to avoid unstable model fit associated with very few levels of random effects (ex., 2 batches) [51]. T-test contrasts examined Group differences in whole-brain seed-based amygdala FC maps. Whole-brain FC results were thresholded at p < 0.005 (uncorrected height threshold), p < 0.0125 FDR-corrected cluster threshold, to correct for multiple comparisons across the 4 amygdala ROIs (0.05/4 ROIs = 0.0125). The SPM Anatomy Toolbox was used to assign estimated anatomical labels for significant clusters [52]. Meta-analytic decoding of identified clusters was performed using the NiMARE v0.0.11 package, which ascribes functional labels to images using the NeuroSynth meta-analytic database [53,54,55,56].
Finally, we tested associations between clinical symptoms and biological markers. Individual linear regressions controlling for Batch examined associations between PACAP levels and symptoms that differentiated the IH group from the non-IH group. Pearson correlations examined associations between symptom severity and amygdala FC. Symptom associations were corrected for multiple comparisons using family-wise error (FWE) correction across all examined symptoms.
One-tailed tests were used for replication analyses (e.g., elevated PACAP in IH), and two-tailed tests for novel analyses (e.g., FC differences between groups, symptom associations with identified biological markers). One-tailed p-values are explicitly labeled in the results and are accompanied by two-tailed p-values; all other p-values are two-tailed. Given a priori findings that implicate biological sex as an important factor of the IH phenotype and its underlying neurobiology [16, 40], sex assigned at birth was not included as a covariate in models; however, models incorporating sex assigned at birth are presented in the Supplementary Results.
Results
Latent profile analysis
Consistent with Adams et al. [16], a three-profile model of CAPS-5 symptoms provided the best fit based on the BIC. The resulting profiles included: a High group (n = 73) characterized by elevated severity across most PTSD symptoms; a Low group (n = 61) with generally low symptom severity; and an Intrusive-Hypervigilant (IH) group (n = 38) marked by differential elevations in intrusion and hypervigilance symptoms. Compared to the High and Low groups, the IH Group demonstrated significantly greater severity of trauma-related intrusive memories (B1; IH > High/Low: t = 3.04/6.38, p = 0.008/ < 0.0001, Cohen’s d = 0.61/1.32), flashbacks (B3; IH > High/Low: t = 34.17/33.55, p’s < 0.0001, d = 6.83/6.93), and hypervigilance (E3; IH > High/Low: t = 2.87/4.27, p = 0.013/0.0001, d = 0.57/0.88; Fig. 1). Complete descriptive statistics for all CAPS-5 symptoms are reported in Supplementary Table 2.
* indicates where the Intrusive-Hypervigilant (IH) group differed from both the High and Low groups. B = Intrusive Reexperiencing symptoms (Cluster B); C = Avoidance symptoms (Cluster C); D = Negative Alterations in Cognitions and Mood symptoms (Cluster D); E = Hyperarousal symptoms (Cluster E).
Also in line with prior findings, the IH group had a greater proportion of females (79%) compared to the High (63%; IH > High χ2 = 2.94, p = 0.043) and Low groups (52%; IH > Low χ2 = 7.02, p = 0.004). The High and Low groups did not differ in sex composition (High ≠ Low χ2 = 2.94, p = 0.217). We also observed differences in the proportion of threshold (vs. subthreshold) PTSD diagnoses, with the IH group having a greater proportion (79%) compared to the Low group (36%; IH > Low χ2 = 17.27, p < 0.001) but not the High group (86%; IH > High χ2 = 0.99, p = 0.841). The High group also had significantly more full PTSD cases than the Low group (High > Low χ2 = 36.16, p < 0.001).
PACAP38
There were no significant differences in PACAP levels between the High (M = 24.02, SE = 1.63) and Low groups (M = 22.86, SE = 1.77; t(120) = 0.52, p = 0.607), justifying their combination into a single non-IH comparison group. The IH group (M = 29.08, SE = 2.53) had significantly higher PACAP levels compared to the combined non-IH group (B = 0.38, SE = 0.18, t(155) = 1.97, p = 0.025 one-tailed, p = 0.050 two-tailed, d = 0.39). Follow-up contrasts confirmed elevated PACAP levels in the IH group compared to both the High (t(154) = 1.65, p = 0.050 one-tailed, p = 0.101 two-tailed, d = 0.35) and Low groups (t(154) = 2.04, p = 0.025 one-tailed, p = 0.050 two-tailed, d = 0.44; Fig. 2).
Participants with selectively elevated Intrusive-Hypervigilant (IH) PTSD symptom profiles demonstrated greater PACAP levels than participants with generally high (High) and generally low (Low) PTSD symptom profiles. Bars represent estimated marginal means from linear regression models controlling for batch effects; error bars reflect standard errors from the mean. Individual points reflect raw (untransformed) PACAP values. Data. PACAP values were square-root transformed for statistical analysis. † p = 0.05 one-tailed; * p < 0.05 one-tailed.
Functional connectivity
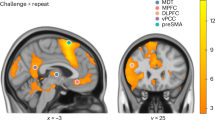
Whole-brain rCMA-seed connectivity maps confirmed no differences between the High and Low groups, again supporting their combination into a single, non-IH group. Relative to this combined non-IH group, the IH group exhibited stronger rCMA connectivity with three midline clusters (Fig. 3A): a cluster spanning the parietooccipital sulcus and intracalcarine cortex (k = 302 voxels, cluster FDR q = 0.001, peak MNI = [–4, –72, 16], T = 4.15), a precuneus cortex cluster (k = 287, cluster FDR q = 0.001, peak = [–4, –56, 68], T = 4.73), and a medial prefrontal cortex (mPFC) cluster comprising the paracingulate gyrus and anterior cingulate cortex (ACC; k = 210, cluster FDR q = 0.005, peak = [–2, 46, 14], T = 4.63). Mapping these clusters onto Yeo’s 7-network atlas (Fig. 3B) situated them predominantly within the Default Mode Network (k = 212) and Visual Network (k = 208), and to a lesser extent the Dorsal Attention (k = 99), Ventral Attention (k = 81), Somatomotor (k = 76), and Frontoparietal (k = 46) networks. Follow-up group contrasts revealed the IH group demonstrated greater rCMA FC across all three clusters compared to both the High and Low groups (Fig. 3C and Supplementary Table 3). No significant group differences emerged for seed connectivity from the rBLA, lCMA, or lBLA.
A Whole-brain contrasts (IH > non-IH) revealed three significant clusters showing greater rCMA FC in the Intrusive-Hypervigilant (IH) group within the parietooccipital sulcus (POS), precuneus (Precun), and medial prefrontal cortex (mPFC). B Radar plot depicting the number of voxels within the significant clusters that overlap with the 7 networks of the Yeo atlas, demonstrating the clusters were predominantly within the default mode network (DMN) and visual network. C Post hoc individual group contrasts revealed that the IH group showed significantly greater rCMA FC than both the High and Low symptom groups across all three clusters. FPN frontoparietal network, SM somatomotor network, VAN ventral attention network, DAN dorsal attention network, **** < 0.001, ***** < 0.0005, ****** < 0.0001.
Associations with clinical symptoms
Across all participants, PACAP levels were modestly associated with CAPS-5 item severity scores for flashbacks (β = 0.17, SE = 0.08, p = 0.025; Fig. 4A), which did not survive FWE correction for multiple comparisons across the three examined symptoms. PACAP levels were not associated with trauma-related intrusive memories (β = 0.08, SE = 0.08, p = 0.336) or hypervigilance (β = 0.02, SE = 0.08, p = 0.787). Flashback severity was additionally associated with rCMA-POS (r = .35, p < 0.0001; Fig. 4B), rCMA-Precun (r = 0.43, p < 0.001; Fig. 4C), and rCMA-mPFC FC (r = 0.37, p < 0.0001; Fig. 4D). Additional weak and trending associations were seen between rCMA-mPFC FC and hypervigilance (r = 0.17, p = 0.048), as well as intrusive memories (r = 0.15, p = 0.079; Fig. 4D), which did not survive FWE correction for multiple comparisons.
A Pituitary adenylate cyclase-activating (PACAP) levels (square-root-transformed) were significantly associated with flashback severity (CAPS-5, item B3) but not with trauma-related intrusive memories (B1) or hypervigilance (E3). B–D Right centromedial amygdala (rCMA) FC with the parieto-occipital sulcus (POS), precuneus (Precun), and medial prefrontal cortex (mPFC) was positively associated with flashback severity. Weaker, trending associations were also observed between rCMA–mPFC connectivity and both intrusive memories and hypervigilance. CAPS-5: Clinician-Administered PTSD Scale for DSM-5.
Discussion
In an independent sample of trauma-exposed adults with symptoms spanning subthreshold to threshold DSM-5 PTSD, we replicated the findings of Adams et al. [16], identifying an IH clinical phenotype characterized by more severe intrusive memories, flashbacks, and hypervigilance, as well as elevated levels of circulating PACAP. Extending prior work, we provide novel evidence that this IH phenotype is associated with stronger rCMA FC with midline cortical regions, particularly within the default mode and visual networks. Importantly, both PACAP levels and rCMA FC were significantly associated with more severe flashbacks, supporting their potential as biomarkers for this clinical phenotype.
Our identification of an IH clinical phenotype aligns with existing neurocognitive models of PTSD symptoms that highlight the intersection of arousal, memory, and attention. The “warning signal” hypothesis of trauma reexperiencing symptoms posits that neutral sensory cues can elicit exaggerated arousal and trigger intrusive memories or flashbacks through acquired temporal associations with the traumatic event [57]. This conditioned threat to sensory cues one encounters in everyday life may lead to exaggerated externally-oriented attention to detect and respond to such threat cues—the hallmark presentation of hypervigilance. Translational evidence suggests that “hypervigilance” and its proposed neurochemical underpinnings increase sensory sensitivity, reduce sensory gating, and facilitate rapid sensory processing, potentially through alterations in attention [58,59,60,61,62,63,64,65,66,67]. Therefore, hypervigilance may contribute to the detection and rapid processing of sensory cues that trigger intrusive memories and flashbacks through the activation of sensory-bound representations of the traumatic event. Moreover, physiological arousal may further exacerbate this cycle, as inductions of physiological arousal can elicit flashback-like memories [68,69,70]. Taken together, these findings suggest that the IH phenotype represents a clinical presentation of posttraumatic stress associated with interactions between arousal, memory, and attention systems that amplify specific reexperiencing symptoms.
In keeping with this notion, the observed elevations in circulating PACAP levels within the IH phenotype align with the demonstrated role of PACAP in stress physiology and fear memory. PACAP is a critical regulator of arousal through its signaling in autonomic stress pathways [33, 34, 71]. PACAP and its predominant type 1 receptor (PAC1R) are densely expressed within the central extended amygdala [32,33,34,35], encompassing both the central amygdala (CeA) and bed nucleus of the stria terminalis (BNST), which activate fear and panic reflexes in response to threat through downstream projections to physiological arousal systems [72]. PACAP is also linked to the acquisition, consolidation, and extinction of contextual or cue-based fear memory through alterations in synaptic plasticity within fear memory circuits [73,74,75,76,77]. Notably, PACAP signaling facilitates trace fear memory, a form of fear learning that is dependent on sustained attention and episodic memory systems [78] and is highly aligned with the “warning signal” model of intrusive reexperiencing symptoms. To this end, PACAP may facilitate attentional states of hypervigilance for threat as a consequence of sustained conditioned fear. Moreover, PACAP may enhance consolidation and impair extinction of contextual fear memories that are linked to episodic memory systems, akin to trauma-related intrusive memories and flashbacks. Aligning with the neurocognitive models of PTSD, this cycle may be maintained and exacerbated by the elevated states of physiological arousal driven by PACAP signaling. Therefore, PACAP is optimally positioned at the intersection of arousal, memory, and attention systems to give rise to the IH phenotype.
Strengthened amygdala FC with the identified posterior and prefrontal regions further supports a role of arousal-memory-attention interactions. Stronger functional coupling between the amygdala and precuneus is associated with greater threat reactivity [79], hyperarousal [42], and sustained attention to aversive stimuli [80]. Additionally, both the amygdala and our identified precuneus cluster are linked to the retrieval of emotional autobiographical memory [81,82,83,84]. Our identified mPFC cluster overlaps with the pregenual ACC (Brodmann’s Area 32), whose rodent analogue, the prelimbic cortex, is consistently implicated in the expression of conditioned fear through connectivity with the amygdala [85,86,87]. Specifically, the retrieval of long-term conditioned fear memory may be supported by strengthened connections between the prelimbic cortex and central amygdala through the paraventricular thalamus [88, 89]. In humans, the pregenual ACC is engaged during the retrieval of remote contextual fear memories that involve the amygdala and hippocampus [90,91,92,93]. Similarly, the POS and adjacent visual cortex have been tied to the retrieval of vivid sensory details of episodic memories [31, 94,95,96] through their role in visual imagery and visuospatial processing [97, 98]. More broadly, these posterior cortices are involved in emotional arousal [99], vigilance [100], and directed attention [101]. In sum, while these regions support numerous functions, they converge on a pattern of processes that are tied to the detection, processing, and responding to threat and the consolidation and retrieval of emotional episodic or fear memories. Our meta-analytic decoding of this collective pattern of posterior and prefrontal clusters provides evidence for this convergence, as it revealed a spatial overlap with studies ascribing functions such as mental imagery, memory retrieval and encoding, negative emotions, fear and threat, and attention (Supplementary Table 4).
Collectively, these identified regions fall predominantly within the DMN and Visual Network (Fig. 3B). Both networks are consistently implicated in posttraumatic stress given their respective functions in episodic memory, attention, and arousal [36, 102,103,104,105,106,107]. Typically, these networks demonstrate robust anticorrelation and are positioned at opposite ends of the hierarchical organization of cortical networks, reflecting a maximal segregation in their patterns of activity and connectivity [53]. However, there is accumulating evidence for a breakdown in the functional segregation of these intrinsic networks in posttraumatic stress [108, 109], particularly in relation to symptoms of hypervigilance [104] and trauma reexperiencing [110]. Additionally, an imbalance in their anticorrelation, or co-deactivation, has been linked to the sensory-perceptual features of trauma memories, including their emotional intensity and sense of reliving, which are key features of flashbacks [95]. The current findings of strengthened connectivity between the amygdala and central brain regions of both the DMN and Visual Network may similarly reflect increased functional integration of these normally divergent networks. This suggests that the IH clinical phenotype is marked by abnormal coupling between these intrinsic resting state networks and the amygdala, reflecting a potential biomarker for this subtype. Future work incorporating dynamic FC of these networks is needed to elucidate the network-wide versus region-specific alterations and which patterns maximally differentiate the IH phenotype. Moreover, task-based neuroimaging may be able to determine whether these changes in organization are in fact tied to behavioral measures of arousal, memory, and attention.
Interestingly, our amygdala FC findings were specific to the rCMA, as no effects emerged with the lCMA or r/lBLA. The BLA generally demonstrates more robust FC with cortical structures compared to the CMA, which preferentially connects with subcortical structures like the midbrain and striatum [111,112,113,114]. Our findings replicate this pattern, as the non-IH groups demonstrated near-zero average connectivity between the CMA and cortical regions (Fig. 3C). Prior work has demonstrated increased BLA-cortical connectivity in PTSD compared to trauma-exposed controls, but no differences in CMA connectivity due potentially to the minimal intrinsic CMA-cortical connectivity [111]. However, we previously demonstrated that elevated levels of circulating PACAP are associated with strengthened cortical connectivity of the rCMA, and not the BLA, with midline regions of the DMN, including the precuneus [42]. Our present findings qualify this unusual strengthening of CMA-cortical connectivity by demonstrating a specificity of this effect to the IH clinical phenotype, which was the only clinical phenotype with elevations in PACAP levels. Therefore, this abnormal rCMA connectivity pattern may reflect a PACAP-mediated shift in the functional organization of the amygdala and its cortical interactions that is specific to the IH phenotype. These converging findings position the combination of high PACAP and rCMA FC as a candidate biomarker for the IH clinical phenotype, reflecting a distinct neurobiological profile not observed in other posttraumatic stress presentations.
Critically, the High and Low symptom groups did not differ in PACAP or rCMA FC despite notable differences in nearly every symptom, including intrusive memories and hypervigilance (Fig. 1). This is consistent with Adams et al. [16], who likewise reported nearly identical PACAP levels between these two groups despite notable differences in nearly every PTSD symptom. This specificity reinforces the hypothesis that the IH phenotype may reflect a biologically distinct subtype of posttraumatic stress, characterized clinically by differentially elevated intrusive memories, flashbacks, and hypervigilance. Among these, flashbacks emerged as the only individual symptom significantly associated with PACAP and rCMA connectivity. This may be attributable to the greater physiological arousal and cognitive alterations often associated with flashbacks compared to other trauma-related intrusive memories, reflecting an exaggerated interaction among memory, attention, and arousal systems [115]. Neuroimaging studies examining the reliving or re-enacting of traumatic events further emphasize a coupling between the DMN and sensory cortex in flashbacks, reflecting a reorganization of intrinsic connectivity patterns that is marked by the integration of sensory processing and autobiographical memory retrieval [108]. Notably, flashbacks were also the most robust differentiator between the IH and non-IH groups. These findings suggest that flashbacks may serve as a core clinical marker of the distinct biological profile underlying the IH phenotype. Because flashbacks are characterized by vivid, sensory-based reliving of the traumatic event, this profile may be particularly amenable to precision therapeutics that target the sensory elements of posttraumatic reexperiencing [61, 110, 116], either through neurofeedback [117], brain stimulation [118,119,120], or more integrative approaches [121]. Although speculative, these possibilities highlight how delineating biologically-informed PTSD subtypes may ultimately guide personalized treatment development. External replications with independent trauma-exposed samples are needed to confirm this specificity and validate these biomarkers of an IH phenotype, including the potential centrality of flashback symptoms.
The present findings are not without limitations. First, although we replicated prior findings of elevated PACAP in the IH-phenotype, the effect sizes were markedly smaller than those reported by Adams et al.[16]. Several factors may have contributed to these discrepancies. Notably, the present study used a single measurement of PACAP, whereas Adams et al. acquired three consecutive measurements, which may yield more reliable estimates of circulating PACAP levels. Additionally, Adams et al. had a more even distribution of male and female participants as well as a more diverse sample with regard to ethnoracial identity. They further demonstrated that the IH phenotype was characterized by a higher proportion of women from marginalized ethnoracial groups; therefore, our predominantly white female sample may have lacked the sociocultural and stress-related diversity to capture these nuanced effects. More broadly, variability in PACAP levels may reflect heterogeneity in trauma exposures, including cumulative stressors faced by women from marginalized ethnoracial groups. Thus, future studies incorporating repeated PACAP assessments and more diverse samples, as well as comprehensively characterizing trauma history and chronic stress burden, will be critical for clarifying the conditions under which PACAP most robustly indexes the IH phenotype. As this study represents the first independent replication of the IH phenotype, any proposed guidelines for its derivation are necessarily preliminary. Nonetheless, the convergence of findings across latent class and latent profile approaches suggests that the IH clinical presentation phenotype represents a reproducible pattern of elevated intrusive reexperiencing and hyperarousal symptoms that is not dependent on the specific clustering method. Future studies should examine the stability of this phenotype across and within subjects, and determine whether empirically derived symptom thresholds can support clinically meaningful identification.
Additional limitations include the cross-sectional design of our study, which precludes inference of directionality in associations between PACAP, rCMA connectivity, and clinical phenotypes. Longitudinal studies are needed to gain insights into the temporal relationship and interdependency between these biological and clinical markers. Relatedly, all imaging data were collected at rest; as such, we cannot directly support claims linking PACAP and rCMA-cortical connectivity to the active processes of memory, attention, and arousal that are implicated in intrusive reexperiencing and hypervigilance symptoms. Future studies incorporating cognitive testing or behavioral measures of these constructs in conjunction with task-based neuroimaging are needed to clarify the mechanistic processes giving rise to the IH phenotype. Additionally, prior work has demonstrated interactions between PACAP and estrogen [40, 122]. While our current sample did not differ with regard to menstrual status, hormonal contraceptive use, or history of hysterectomy across the identified clinical phenotypes (Table 1), more nuanced investigations into the interaction between estrogen and PACAP are encouraged in order to provide critical insights into the role of biological sex in the IH phenotype. Finally, our FC analyses were constrained to amygdala subregions, specifically the BLA and CMA, given prior work demonstrating robust PACAP signaling within these regions. However, PACAP is expressed in numerous other subcortical and midbrain regions that are tied to behavioral arousal, attention, and memory, such as locus coeruleus (LC) and the bed nucleus of the stria terminalis (BNST) [33]. Many of these structures require greater anatomical precision or individual-level, hand-drawn anatomical masks to delineate them from adjacent structures and sources of physiological noise. Future studies utilizing ultra-high-field MRI (i.e., 7 T) or specialized sequences (e.g., neuromelanin-sensitive imaging) would afford deeper insights into other subcortical structures that may be mechanistically linked to the IH phenotype.
In conclusion, this study replicates the identification of an intrusive-hypervigilant clinical phenotype of posttraumatic stress that is characterized by elevated circulating PACAP levels and provides novel evidence for altered patterns of FC of the rCMA. This IH phenotype may reflect a distinct clinical presentation that is characterized by exaggerated interactions between memory and arousal processes, thus driving specific symptoms that are situated at this intersection, like flashbacks. Our findings further add to the accumulating evidence linking both PACAP and altered organization of subcortical-cortical networks to memory, arousal, and PTSD. More specifically, the IH phenotype exhibits these biomarkers previously implicated in the PTSD literature, which have been stymied by inconsistent replication across different, diverse samples. Therefore, the IH phenotype may be a candidate clinical presentation for a precision psychiatry framework that targets these biomarkers through pharmacotherapy or neurotherapeutics.
Data availability
Data are available through the NIMH National Data Archive (NDA; https://nda.nih.gov/edit_collection.html?id=3166) and are available upon reasonable request to the senior author, IMR.
References
Bryant RA, Galatzer-Levy I, Hadzi-Pavlovic D. The heterogeneity of posttraumatic stress disorder in DSM-5. JAMA Psychiatry. 2023;80:189–91.
Galatzer-Levy IR, Bryant RA. 636,120 ways to have posttraumatic stress disorder. Perspect Psychol Sci. 2013;8:651–62.
McLaughlin KA, Koenen KC, Friedman MJ, Ruscio AM, Karam EG, Shahly V, et al. Sub-threshold post traumatic stress disorder in the WHO World Mental Health Surveys. Biol Psychiatry. 2015;77:375–84.
Cloitre M, Garvert DW, Brewin CR, Bryant RA, Maercker A. Evidence for proposed ICD-11 PTSD and complex PTSD: a latent profile analysis. Eur J Psychotraumatology. 2013;4:20706.
Campbell SarahB, Trachik B, Goldberg S, Simpson TL. Identifying PTSD symptom typologies: a latent class analysis. Psychiatry Res. 2020;285:112779.
Forbes D, Elhai JD, Miller MW, Creamer M. Internalizing and externalizing classes in posttraumatic stress disorder: a latent class analysis. J Trauma Stress. 2010;23:340–9.
Miller MW, Kaloupek DG, Dillon AL, Keane TM. Externalizing and internalizing subtypes of combat-related PTSD: a replication and extension using the PSY-5 scales. J Abnorm Psychol. 2004;113:636–45.
Miller MW, Resick PA. Internalizing and externalizing subtypes in female sexual assault survivors: implications for the understanding of complex PTSD. Behav Ther. 2007;38:58–71.
Zoellner LA, Pruitt LD, Farach FJ, Jun JJ. Understanding heterogeneity in Ptsd: fear, dysphoria, and distress. Depress Anxiety. 2014;31:97–106.
Sanghvi DE, Rackoff GN, Newman MG. Latent class analysis of post-traumatic stress disorder symptoms following exposure to Hurricane Ike. Soc Sci Med. 2023;327:115942.
Whiteman SE, Lee DJ, Kramer LB, Petri JM, Weathers FW. Subgroup differences in PTSD symptom presentations: a latent class analysis. Eur J Trauma Dissociation. 2024;8:100430.
Kelley LP, Weathers FW, McDevitt-Murphy ME, Eakin DE, Flood AM. A comparison of PTSD symptom patterns in three types of civilian trauma. J Trauma Stress. 2009;22:227–35.
Lewis MW, Jones RT, Davis MT. Exploring the impact of trauma type and extent of exposure on posttraumatic alterations in 5-HT1A expression. Transl Psychiatry. 2020;10:237.
Lewis SJ, Arseneault L, Caspi A, Fisher HL, Matthews T, Moffitt TE, et al. The epidemiology of trauma and post-traumatic stress disorder in a representative cohort of young people in England and Wales. Lancet Psychiatry. 2019;6:247–56.
Smith HL, Summers BJ, Dillon KH, Cougle JR. Is the worst-event trauma type related to PTSD symptom presentation and associated features? J Anxiety Disord. 2016;38:55–61.
Adams SW, Neylan TC, May V, Hammack SE, Ressler K, Inslicht SS. PACAP associated with precise PTSD and fear extinction response in women. Psychoneuroendocrinology. 2025;173:107375.
Marshall GN, Schell TL, Glynn SM, Shetty V. The role of hyperarousal in the manifestation of posttraumatic psychological distress following injury. J Abnorm Psychol. 2006;115:624–8.
Schell TL, Marshall GN, Jaycox LH. All symptoms are not created equal: the prominent role of hyperarousal in the natural course of posttraumatic psychological distress. J Abnorm Psychol. 2004;113:189–97.
Weston CSE. Posttraumatic stress disorder: a theoretical model of the hyperarousal subtype. Front Psychiatry. 2014;5:37.
Gieselmann A, Ait Aoudia M, Carr M, Germain A, Gorzka R, Holzinger B, et al. Aetiology and treatment of nightmare disorder: state of the art and future perspectives. J Sleep Res. 2019;28:e12820.
Nielsen T. The stress acceleration hypothesis of nightmares. Front Neurol. 2017;8:201.
Zoladz PR, Diamond DM. Current status on behavioral and biological markers of PTSD: a search for clarity in a conflicting literature. Neurosci Biobehav Rev. 2013;37:860–95.
Campbell AA, Wisco BE, Silvia PJ, Gay NG. Resting respiratory sinus arrhythmia and posttraumatic stress disorder: a meta-analysis. Biol Psychol. 2019;144:125–35.
Germain A, Nielsen TA. Sleep pathophysiology in posttraumatic stress disorder and idiopathic nightmare sufferers. Biol Psychiatry. 2003;54:1092–8.
Simor P, Bódizs R, Horváth K, Ferri R. Disturbed dreaming and the instability of sleep: altered nonrapid eye movement sleep microstructure in individuals with frequent nightmares as revealed by the cyclic alternating pattern. Sleep. 2013;36:413–9.
Mäder T, Oliver KI, Daffre C, Kim S, Orr SP, Lasko NB, et al. Autonomic activity, posttraumatic and nontraumatic nightmares, and PTSD after trauma exposure. Psychol Med. 2023;53:731–40.
Polack P-O, Friedman J, Golshani P. Cellular mechanisms of brain state–dependent gain modulation in visual cortex. Nat Neurosci. 2013;16:1331–9.
Clewett D, Murty VP. Echoes of emotions past: how neuromodulators determine what we recollect. eNeuro. 2019;6:ENEURO.0108-18.2019.
Roozendaal B, McGaugh JL. Memory modulation. Behav Neurosci. 2011;125:797–824.
Mather M, Clewett D, Sakaki M, Harley CW. GANEing traction: the broad applicability of NE hotspots to diverse cognitive and arousal phenomena. Behav Brain Sci. 2016;39:e228.
Waldhauser GT, Braun V, Hanslmayr S. Episodic memory retrieval functionally relies on very rapid reactivation of sensory information. J Neurosci. 2016;36:251–60.
Boucher MN, May V, Braas KM, Hammack SE. PACAP orchestration of stress-related responses in neural circuits. Peptides. 2021;142:170554.
Hammack SE, May V. Pituitary adenylate cyclase activating polypeptide in stress-related disorders: data convergence from animal and human studies. Biol Psychiatry. 2015;78:167–77.
Hashimoto H, Shintani N, Tanida M, Hayata A, Hashimoto R, Baba A. PACAP is implicated in the stress axes. Curr Pharmacol Des. 2011;17:985–9.
Missig G, Roman CW, Vizzard MA, Braas KM, Hammack SE, May V. Parabrachial nucleus (PBn) pituitary adenylate cyclase activating polypeptide (PACAP) signaling in the amygdala: implication for the sensory and behavioral effects of pain. Neuropharmacology. 2014;86:38–48.
Patel R, Spreng RN, Shin LM, Girard TA. Neurocircuitry models of posttraumatic stress disorder and beyond: a meta-analysis of functional neuroimaging studies. Neurosci Biobehav Rev. 2012;36:2130–42.
Rauch SL, Shin LM, Phelps EA. Neurocircuitry models of posttraumatic stress disorder and extinction: human neuroimaging research-past, present, and future. Biol Psychiatry. 2006;60:376–82.
Ressler KJ, Berretta S, Bolshakov VY, Rosso IM, Meloni EG, Rauch SL, et al. Post-traumatic stress disorder: clinical and translational neuroscience from cells to circuits. Nat Rev Neurol. 2022;18:273–88.
Shin LM, Rauch SL, Pitman RK. Amygdala, medial prefrontal cortex, and hippocampal function in PTSD. Ann N Y Acad Sci. 2006;1071:67–79.
Ressler KJ, Mercer KB, Bradley B, Jovanovic T, Mahan A, Kerley K, et al. Post-traumatic stress disorder is associated with PACAP and the PAC1 receptor. Nature. 2011;470:492–7.
Stevens JS, Almli LM, Fani N, Gutman DA, Bradley B, Norrholm SD, et al. PACAP receptor gene polymorphism impacts fear responses in the amygdala and hippocampus. Proc Natl Acad Sci USA. 2014;111:3158–63.
Clancy KJ, Devignes Q, Kumar P, May V, Hammack SE, Akman E, et al. Circulating PACAP levels are associated with increased amygdala-default mode network resting-state connectivity in posttraumatic stress disorder. Neuropsychopharmacol. 2023;48:1245–54.
King SB, Toufexis DJ, Hammack SE. Pituitary adenylate cyclase activating polypeptide (PACAP), stress, and sex hormones. Stress. 2017;20:465–75.
Weathers FW, Bovin MJ, Lee DJ, Sloan DM, Schnurr PP, Kaloupek DG, et al. The clinician-administered PTSD scale for DSM-5 (CAPS-5): development and initial psychometric evaluation in military veterans. Psychol Assess. 2018;30:383–95.
Scrucca L, Fraley C, Murphy TB, Raftery AE. Model-based clustering, classification, and density estimation using mclust in R. New York: Chapman and Hall/CRC; 2023.
Esteban O, Markiewicz CJ, Blair RW, Moodie CA, Isik AI, Erramuzpe A, et al. fMRIPrep: a robust preprocessing pipeline for functional MRI. Nat Methods. 2019;16:111–6.
Eickhoff S, Walters NB, Schleicher A, Kril J, Egan GF, Zilles K, et al. High-resolution MRI reflects myeloarchitecture and cytoarchitecture of the human cerebral cortex. Hum Brain Mapp. 2005;24:206–15.
Amunts K, Kedo O, Kindler M, Pieperhoff P, Mohlberg H, Shah NJ, et al. Cytoarchitectonic mapping of the human amygdala, hippocampal region and entorhinal cortex: intersubject variability and probability maps. Anat Embryol. 2005;210:343–52.
Newcombe RG. Interval estimation for the difference between independent proportions: comparison of eleven methods. Stat Med. 1998;17:873–90.
Lenth R, Singmann H, Love J, Buerkner P, Herve M. Package ‘emmeans’. R package version. 2019;1(3.2).
Oberpriller J, de Souza Leite M, Pichler M. Fixed or random? On the reliability of mixed-effects models for a small number of levels in grouping variables. Ecol Evolution. 2022;12:e9062.
Eickhoff SB, Stephan KE, Mohlberg H, Grefkes C, Fink GR, Amunts K, et al. A new SPM toolbox for combining probabilistic cytoarchitectonic maps and functional imaging data. Neuroimage. 2005;25:1325–35.
Margulies DS, Ghosh SS, Goulas A, Falkiewicz M, Huntenburg JM, Langs G, et al. Situating the default-mode network along a principal gradient of macroscale cortical organization. Proc Natl Acad Sci. 2016;113:12574–9.
Salo T, Yarkoni T, Nichols TE, Poline J-B, Kent JD, Gorgolewski KJ, et al. NiMARE: neuroimaging meta-analysis research environment. NeuroLibre. 2022;1:7.
Salo T, Yarkoni T, Nichols T, Poline J-B, Bilgel M, Bottenhorn K, et al. NiMARE: neuroimaging meta-analysis research environment. Aperture Neuro. 2023. https://doi.org/10.52294/001c.87480.
Yarkoni T, Poldrack RA, Nichols TE, Van Essen DC, Wager TD. Large-scale automated synthesis of human functional neuroimaging data. Nat Methods. 2011;8:665–70.
Ehlers A, Hackmann A, Steil R, Clohessy S, Wenninger K, Winter H. The nature of intrusive memories after trauma: the warning signal hypothesis. Behav Res Ther. 2002;40:995–1002.
Adler LE, Pang K, Gerhardt G, Rose GM. Modulation of the gating of auditory evoked potentials by norepinephrine: pharmacological evidence obtained using a selective neurotoxin. Biol Psychiatry. 1988;24:179–90.
Aston-Jones G, Rajkowski J, Kubiak P, Alexinsky T. Locus coeruleus neurons in monkey are selectively activated by attended cues in a vigilance task. J Neurosci. 1994;14:4467–80.
Berridge CW, Waterhouse BD. The locus coeruleus–noradrenergic system: modulation of behavioral state and state-dependent cognitive processes. Brain Res Rev. 2003;42:33–84.
Clancy K, Ding M, Bernat E, Schmidt NB, Li W. Restless “rest”: intrinsic sensory hyperactivity and disinhibition in post-traumatic stress disorder. Brain. 2017;140:2041–50.
Gelbard-Sagiv H, Magidov E, Sharon H, Hendler T, Nir Y. Noradrenaline modulates visual perception and late visually evoked activity. Curr Biol. 2018;28:2239–2249.e6.
McBurney-Lin J, Lu J, Zuo Y, Yang H. Locus coeruleus-norepinephrine modulation of sensory processing and perception: a focused review. Neurosci Biobehav Rev. 2019;105:190–9.
Mehrpour V, Martinez-Trujillo JC, Treue S. Attention amplifies neural representations of changes in sensory input at the expense of perceptual accuracy. Nat Commun. 2020;11:2128.
Pessoa L, Kastner S, Ungerleider LG. Neuroimaging studies of attention: from modulation of sensory processing to top-down control. J Neurosci. 2003;23:3990–8.
Southwick SM, Krystal JH, Bremner JD, Morgan CA III, Nicolaou AL, Nagy LM, et al. Noradrenergic and serotonergic function in posttraumatic stress disorder. Arch Gen Psychiatry. 1997;54:749–58.
Southwick SM, Bremner JD, Rasmusson A, Morgan CA, Arnsten A, Charney DS. Role of norepinephrine in the pathophysiology and treatment of posttraumatic stress disorder. Biol Psychiatry. 1999;46:1192–204.
Bremner JD, Innis RB, Ng CK, Staib LH, Salomon RM, Bronen RA, et al. Positron emission tomography measurement of cerebral metabolic correlates of yohimbine administration in combat-related posttraumatic stress disorder. Arch Gen Psychiatry. 1997;54:246–54.
Kellner M, Levengood R, Yehuda R, Wiedemann K. Provocation of a posttraumatic flashback by cholecystokinin tetrapeptide? Am J Psychiatry. 1998;155:1299.
Nixon RDV, Bryant RA. Induced arousal and reexperiencing in acute stress disorder. J Anxiety Disord. 2005;19:587–94.
Stroth N, Holighaus Y, Ait-Ali D, Eiden LE. PACAP: a master regulator of neuroendocrine stress circuits and the cellular stress response. Ann N Y Acad Sci. 2011;1220:49–59.
Davis M. The role of the amygdala in fear and anxiety. Annu Rev Neurosci. 1992;15:353–75.
Ago Y, Hayata-Takano A, Kawanai T, Yamauchi R, Takeuchi S, Cushman JD, et al. Impaired extinction of cued fear memory and abnormal dendritic morphology in the prelimbic and infralimbic cortices in VPAC2 receptor (VIPR2)-deficient mice. Neurobiol Learn Mem. 2017;145:222–31.
Cho J-H, Zushida K, Shumyatsky GP, Carlezon WA, Meloni EG, Bolshakov VY. Pituitary adenylate cyclase-activating polypeptide induces postsynaptically expressed potentiation in the intra-amygdala circuit. J Neurosci. 2012;32:14165–77.
Meloni EG, Venkataraman A, Donahue RJ, Carlezon WA. Bi-directional effects of pituitary adenylate cyclase-activating polypeptide (PACAP) on fear-related behavior and c-Fos expression after fear conditioning in rats. Psychoneuroendocrinology. 2016;64:12–21.
Schmidt SD, Myskiw JC, Furini CRG, Schmidt BE, Cavalcante LE, Izquierdo I. PACAP modulates the consolidation and extinction of the contextual fear conditioning through NMDA receptors. Neurobiol Learn Mem. 2015;118:120–4.
Velasco ER, Florido A, Flores Á, Senabre E, Gomez-Gomez A, Torres A, et al. PACAP-PAC1R modulates fear extinction via the ventromedial hypothalamus. Nat Commun. 2022;13:4374.
Kirry AJ, Herbst MR, Poirier SE, Maskeri MM, Rothwell AC, Twining RC, et al. Pituitary adenylate cyclase-activating polypeptide (PACAP) signaling in the prefrontal cortex modulates cued fear learning, but not spatial working memory, in female rats. Neuropharmacology. 2018;133:145–54.
Rabellino D, Tursich M, Frewen PA, Daniels JK, Densmore M, Théberge J, et al. Intrinsic connectivity networks in post-traumatic stress disorder during sub- and supraliminal processing of threat-related stimuli. Acta Psychiatr Scand. 2015;132:365–78.
Ferri J, Schmidt J, Hajcak G, Canli T. Emotion regulation and amygdala-precuneus connectivity: Focusing on attentional deployment. Cogn Affect Behav Neurosci. 2016;16:991–1002.
Daselaar SM, Rice HJ, Greenberg DL, Cabeza R, LaBar KS, Rubin DC. The spatiotemporal dynamics of autobiographical memory: neural correlates of recall, emotional intensity, and reliving. Cereb Cortex. 2008;18:217–29.
Mazzoni G, Clark A, De Bartolo A, Guerrini C, Nahouli Z, Duzzi D, et al. Brain activation in highly superior autobiographical memory: The role of the precuneus in the autobiographical memory retrieval network. Cortex. 2019;120:588–602.
Richter FR, Cooper RA, Bays PM, Simons JS. Distinct neural mechanisms underlie the success, precision, and vividness of episodic memory. eLife. 2016;5:e18260.
Young KD, Siegle GJ, Misaki M, Zotev V, Phillips R, Drevets WC, et al. Altered task-based and resting-state amygdala functional connectivity following real-time fMRI amygdala neurofeedback training in major depressive disorder. NeuroImage Clin. 2018;17:691–703.
Burgos-Robles A, Vidal-González I, Quirk GJ. Sustained conditioned responses in prelimbic prefrontal neurons are correlated with fear expression and extinction failure. J Neurosci. 2009;29:8474–82.
Etkin A, Egner T, Kalisch R. Emotional processing in anterior cingulate and medial prefrontal cortex. Trends Cogn Sci. 2011;15:85–93.
Tovote P, Fadok JP, Lüthi A. Neuronal circuits for fear and anxiety. Nat Rev Neurosci. 2015;16:317–31.
Arruda-Carvalho M, Clem RL. Prefrontal-amygdala fear networks come into focus. Front Syst Neurosci. 2015;9:145.
Do-Monte FH, Quiñones-Laracuente K, Quirk GJ. A temporal shift in the circuits mediating retrieval of fear memory. Nature. 2015;519:460–3.
Cullen PK, Gilman TL, Winiecki P, Riccio DC, Jasnow AM. Activity of the anterior cingulate cortex and ventral hippocampus underlie increases in contextual fear generalization. Neurobiol Learn Mem. 2015;124:19–27.
de Lima MAX, Baldo MVC, Oliveira FA, Canteras NS. The anterior cingulate cortex and its role in controlling contextual fear memory to predatory threats. eLife. 2022;11:e67007.
Frankland PW, Bontempi B, Talton LE, Kaczmarek L, Silva AJ. The involvement of the anterior cingulate cortex in remote contextual fear memory. Science. 2004;304:881–3.
Vetere G, Restivo L, Cole CJ, Ross PJ, Ammassari-Teule M, Josselyn SA, et al. Spine growth in the anterior cingulate cortex is necessary for the consolidation of contextual fear memory. Proc Natl Acad Sci USA. 2011;108:8456–60.
Bone MB, Buchsbaum BR. Detailed episodic memory depends on concurrent reactivation of basic visual features within the posterior hippocampus and early visual cortex. Cereb Cortex Commun. 2021;2:tgab045.
Clancy KJ, Devignes Q, Ren B, Pollmann Y, Nielsen SR, Howell K, et al. Spatiotemporal dynamics of hippocampal-cortical networks underlying the unique phenomenological properties of trauma-related intrusive memories. Mol Psychiatry. 2024;29:2161–9.
Wheeler ME, Petersen SE, Buckner RL. Memory’s echo: Vivid remembering reactivates sensory-specific cortex. Proc Natl Acad Sci. 2000;97:11125–9.
Kravitz DJ, Saleem KS, Baker CI, Mishkin M. A new neural framework for visuospatial processing. Nat Rev Neurosci. 2011;12:217–30.
Pearson J. The human imagination: the cognitive neuroscience of visual mental imagery. Nat Rev Neurosci. 2019;20:624–34.
Schubring D, Schupp HT. Emotion and brain oscillations: high arousal is associated with decreases in alpha- and lower beta-band power. Cereb Cortex. 2021;31:1597–608.
Breckel TPK, Giessing C, Thiel CM. Impact of brain networks involved in vigilance on processing irrelevant visual motion. NeuroImage. 2011;55:1754–62.
Kastner S, De Weerd P, Desimone R, Ungerleider LG. Mechanisms of directed attention in the human extrastriate cortex as revealed by functional MRI. Science. 1998;282:108–11.
Akiki TJ, Averill CL, Abdallah CG. A network-based neurobiological model of PTSD: evidence from structural and functional neuroimaging studies. Curr Psychiatry Rep. 2017;19:81.
Akiki TJ, Averill CL, Wrocklage KM, Scott JC, Averill LA, Schweinsburg B, et al. Default mode network abnormalities in posttraumatic stress disorder: a novel network-restricted topology approach. Neuroimage. 2018;176:489–98.
Clancy KJ, Andrzejewski JA, Simon J, Ding M, Schmidt NB, Li W. Posttraumatic stress disorder is associated with α dysrhythmia across the visual cortex and the default mode network. eNeuro. 2020;7.
Miller DR, Hayes SM, Hayes JP, Spielberg JM, Lafleche G, Verfaellie M. Default mode network subsystems are differentially disrupted in posttraumatic stress disorder. Biol Psychiatry Cogn Neurosci Neuroimaging. 2017;2:363–71.
Pitman RK, Rasmusson AM, Koenen KC, Shin LM, Orr SP, Gilbertson MW, et al. Biological studies of post-traumatic stress disorder. Nat Rev Neurosci. 2012;13:769–87.
Sripada RK, King AP, Welsh RC, Garfinkel SN, Wang X, Sripada CS, et al. Neural dysregulation in posttraumatic stress disorder: evidence for disrupted equilibrium between salience and default mode brain networks. Psychosom Med. 2012;74:904–11.
Kearney BE, Terpou BA, Densmore M, Shaw SB, Théberge J, Jetly R, et al. How the body remembers: examining the default mode and sensorimotor networks during moral injury autobiographical memory retrieval in PTSD. Neuroimage Clin. 2023;38:103426.
Kearney BE, Lanius RA. Why reliving is not remembering and the unique neurobiological representation of traumatic memory. Nat Ment Health. 2024;2:1142–51.
Clancy KJ, Albizu A, Schmidt NB, Li W. Intrinsic sensory disinhibition contributes to intrusive re-experiencing in combat veterans. Sci Rep. 2020;10:936.
Brown VM, LaBar KS, Haswell CC, Gold AL, McCarthy G, Morey RA. Altered resting-state functional connectivity of basolateral and centromedial amygdala complexes in posttraumatic stress disorder. Neuropsychopharmacol. 2014;39:351–9.
Etkin A, Prater KE, Schatzberg AF, Menon V, Greicius MD. Disrupted amygdalar subregion functional connectivity and evidence of a compensatory network in generalized anxiety disorder. Arch Gen Psychiatry. 2009;66:1361–72.
Roy AK, Shehzad Z, Margulies DS, Kelly AMC, Uddin LQ, Gotimer K, et al. Functional connectivity of the human amygdala using resting state fMRI. NeuroImage. 2009;45:614–26.
Roy AK, Fudge JL, Kelly C, Perry JSA, Daniele T, Carlisi C, et al. Intrinsic functional connectivity of amygdala-based networks in adolescent generalized anxiety disorder. J Am Acad Child Adolesc Psychiatry. 2013;52:290–299.e2.
Brewin CR. Re-experiencing traumatic events in PTSD: new avenues in research on intrusive memories and flashbacks. Eur J Psychotraumatol. 2015;6:https://doi.org/10.3402/ejpt.v6.27180.
Kearney BE, Lanius RA. The brain-body disconnect: a somatic sensory basis for trauma-related disorders. Front Neurosci. 2022;16:1015749.
Nicholson AA, Ros T, Densmore M, Frewen PA, Neufeld RWJ, Théberge J, et al. A randomized, controlled trial of alpha-rhythm EEG neurofeedback in posttraumatic stress disorder: a preliminary investigation showing evidence of decreased PTSD symptoms and restored default mode and salience network connectivity using fMRI. Neuroimage Clin. 2020;28:102490.
Clancy KJ, Baisley SK, Albizu A, Kartvelishvili N, Ding M, Li W. Lasting connectivity increase and anxiety reduction via transcranial alternating current stimulation. Soc Cogn Affect Neurosci. 2018;13:1305–16.
Herz N, Bar-Haim Y, Tavor I, Tik N, Sharon H, Holmes EA, et al. Neuromodulation of the visual cortex reduces the intensity of intrusive memories. Cereb Cortex. 2021;32:408–17.
Clancy KJ, Andrzejewski JA, You Y, Rosenberg JT, Ding M, Li W. Transcranial stimulation of alpha oscillations up-regulates the default mode network. Proc Natl Acad Sci USA. 2022;119:e2110868119.
Lanius R. Sensory Motor Arousal Regulation Treatment (SMART) Study. ClinicalTrials.gov identifier: NCT06035809. Available at: https://clinicaltrials.gov/study/NCT06035809.
Mercer KB, Dias B, Shafer D, Maddox SA, Mulle JG, Hu P, et al. Functional evaluation of a PTSD-associated genetic variant: estradiol regulation and ADCYAP1R1. Transl Psychiatry. 2016;6:e978–e978.
Kubany ES, Leisen MB, Kaplan AS, Watson SB, Haynes SN, Owens JA, et al. Development and preliminary validation of a brief broad-spectrum measure of trauma exposure: the traumatic life events questionnaire. Psychol Assess. 2000;12:210–24.
Acknowledgements
The authors would like to thank all participants for dedicating their time and energy to completing the study. We also thank the MRI technologists of the McLean Imaging Center.
Funding
This work was supported by NIH awards P50-MH115874 (to WAC, KJR; Project 4: IMR, SLR). Additionally, IMR was supported by NIH awards R01-MH120400 and R01-MH125852, KJC was supported by NIH award K23-MH137459, and SEH and VM were supported by NIH award R01-MH97988.
Author information
Authors and Affiliations
Contributions
KJC performed data processing, statistical analyses, and wrote the initial draft of the manuscript. CR provided consultation on all statistical analyses and provided critical feedback on the analytic plan and reporting of the statistical methods and results. VM and SEH performed analysis of PACAP data. SAJ contributed to data collection, management, and processing. WAC, KJR, SLR, and IMR obtained funding and contributed to study design and conceptualization. SLR and IMR conceived the study. All authors provided critical input to the manuscript, contributed to revised drafts, and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
WAC and KJR are members of the NPP Editorial Board. For completeness of disclosure: SLR has been employed by Mass General Brigham/McLean Hospital; paid as secretary of Society of Biological Psychiatry, and for Board service to Mindpath Health/Community Psychiatry and National Association of Behavioral Healthcare; served as volunteer member of the Board for Anxiety & Depression Association of America, and The National Network of Depression Centers; received royalties from Oxford University Press, American Psychiatric Publishing Inc, and Springer Publishing; received research funding from NIMH. Within the past 3 years, WAC has served as a consultant for Psy Therapeutics and has had sponsored research agreements with Cerevel Therapeutics and Delix Therapeutics. KJR has performed scientific consultation for Bioxcel, Bionomics, Acer, and Jazz Pharma; serves on Scientific Advisory Boards for Sage, Boehringer Ingelheim, Senseye, and the Brain Research Foundation. He has received sponsored research support from Alto Neuroscience. None of these relationships is related to the current manuscript. The remaining authors have no disclosures.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Clancy, K.J., Ravichandran, C., Jobson, S.A. et al. Identification of an intrusive-hypervigilant phenotype of posttraumatic stress symptoms with unique stress peptide and amygdala functional connectivity profiles. Neuropsychopharmacol. (2026). https://doi.org/10.1038/s41386-026-02396-0
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41386-026-02396-0