Abstract
Background
Prostate cancer(PCa) patients treated with androgen deprivation therapy(ADT) may experience major adverse cardiovascular events(MACE) [1]. Racial disparities in PCa incidence and outcomes have been noted. In contrast to older studies, three recent studies found significantly longer overall survival in Black vs. White patients: 2019 meta-analysis of nine phase III trials in men with metastatic castration-resistant PCa(CRPC) (n = 8820) [2]; 2020 registry study in men with metastatic CRPC (n = 1902) [3]; and 2023 study in men with non-metastatic CRPC (n = 12,992) [4]. Our “real-world” data study compared MACE and all-cause mortality risk for Black vs. White PCa patients. Compared to prior studies [1,2,3,4], our study encompassed a broader scope and was not exclusive to CRPC patients.
Methods
Historical, longitudinal patient-level were collected from the Decision Resources Group (DRG, now Clarivate) Real World Evidence repository. The analysis included PCa patients receiving ≥1 ADT 1991–2020. Multivariable regression model accounted for baseline metastasis, BMI (<18.5 vs. ≥18.5 kg/m2), oncology vs. urology setting, antagonist vs. agonist, personal MACE history, tobacco history, baseline prostate-specific antigen (>4 vs. ≤4 ng/mL), race (White vs. Black), statin use, increasing age per year, ethnicity (non-Hispanic vs. Hispanic), increasing ADT exposure per year, diabetes, hypertension, and family MACE history.
Results
MACE risk was higher for White patients than Black (4.0% vs. 2.4% at one year after ADT initiation; 21.0% vs. 13.3% at four years). Mortality risk after ADT initiation was 1.6% and 2.6% at 1 year and 11.7% and 18.1% at 4 years for Black and White patients, respectively.
Conclusions
Our analysis reveals a unique finding that MACE and all-cause mortality incidence were higher in White vs. Black patients. Black race is associated with lower MACE rates and improved survival for men undergoing ADT treatment. Whether selection bias, underlying biology or other factors are responsible for these differences remains unknown.
Similar content being viewed by others
Introduction
Both prostate cancer (PCa) [5] and cardiovascular disease (CVD) [6] are common in the United States. Prostate cancer is the second most common cancer among men [5], and CVD is the leading cause of death [6]. Much research has been devoted to studying how prostate cancer and cardiovascular disease intertwine. Within the population of men with PCa and CVD, outcomes may vary by race. First, disparities in PCa outcomes between racial groups have been noted, with studies showing ~67% higher incidence and a greater than 2-fold increased risk of mortality in Black vs. White men [7]. Additionally, age-adjusted cardiovascular mortality rates are higher in Black vs. White men (rate ratio = 1.33, 95% CI 1.32–1.34) in the general population [8]. African American patients also demonstrate higher prostate-specific antigen (PSA) values [9] and potentially higher tumor cell burden compared to White patients at presentation.
Counter to SEER data reporting increased mortality risk in Black vs. White men with PCa [10], several studies have recently reported improved survival among Black men with PCa, albeit only in the subgroup of patients with metastatic castration-resistant PCa (mCRPC). For example, a 2023 systematic review [11] found that several studies for specific treatments (sipuleucel-T [3, 12, 13], radium [14], abiraterone alone [15], and abiraterone or enzalutamide [16, 17]) reported better survival for Black vs. White patients. As Black men have higher CVD risk than White men in the general population [8], and cardiovascular risk and mortality by race in men with PCa on androgen deprivation therapy (ADT) has not been well examined, our real-world data study explored major adverse cardiovascular events (MACE) and all-cause mortality risk for Black vs. White patients with PCa treated with ADT. Compared to prior studies [1,2,3,4], our study encompassed a broader scope and was not exclusive to men with CRPC. Based on previous data from men with mCRPC, we hypothesized that Black men may have lower MACE risk and longer/better survival after initiation of ADT, adding to the body of evidence for Black men with PCa on ADT.
Methods
Study design
Data were collected from the Decision Resources Group (DRG, now Clarivate) Real World Evidence repository, which links medical claims, prescription claims, and US Electronic Healthcare Records to provide historical, longitudinal patient-level data. The analysis set included PCa patients who received ≥1 ADT injection between 1991 and 2020 (99% of patients started ADT between 2010 and 2020).
Definitions and queries
In our analysis set, only PCa patients who have taken ADT medication are considered. Prostate cancer is defined as the patient being diagnosed with PCa. The DRG extracted PCa patients from their database using keywords documented in Supplementary Table 1. Androgen deprivation therapy medication includes subcutaneous and intramuscular leuprolide, triptorelin, goserelin, histrelin, and degarelix. Similar to PCa, ADT data are extracted from the database using keywords (documented in Supplementary Table 2). Patients without any data after their earliest ADT in the DRG database are excluded.
Androgen deprivation therapy start date is the primary reference time point, and data are classified as before or after ADT start. Androgen deprivation therapy start date is defined as the date of the earliest ADT documented in the DRG database for each patient.
For MACE-related analysis, MACE is defined as the first event since ADT start. Since only the first event is considered, each patient contributes only one event maximum to the analysis i.e., MACE, which is not the first event after ADT start or happened before ADT start, is not considered in the risk analysis. Baseline values are defined as the average value within 30 days prior to ADT start, and if no value exists, the latest data point available before ADT start is used.
Major adverse CV event and all-cause mortality are the clinical outcomes we considered within our analysis. Components for MACE are extracted using keywords and ICD codes documented in Supplementary Table 3. Patients are excluded if they experienced a MACE within the 6-month (180-day) window prior to ADT start (inclusive), consistent with HERO trial methodology. Comorbidities (diabetes and hypertension) are defined as having taken medication to treat comorbidities or being diagnosed with the comorbidities prior to the first events after ADT start. Keywords and ICD codes to extract comorbidities data are documented in Supplementary Table 4. All-cause mortality is defined as a recorded “deceased” status for each patient in the database. The date of all-cause mortality is defined as the most recent date of data entry in the database for all of those who are recorded as “deceased”.
Analysis methodology
An analysis of retrospective data from patients with PCa treated with ADT (n = 44,439) was performed. Kaplan–Meier curves were generated to compare risk of MACE and all-cause mortality following ADT initiation for Black and White PCa patients. MACE was defined as all-cause mortality, stroke, and myocardial infarction based on 2 recent studies in PCa patients: HERO (randomized trial comparing relugolix and leuprolide over 48 weeks) [18] and PRONOUNCE (randomized trial comparing degarelix and leuprolide in patients with PCa and concomitant athero-sclerotic cardiovascular (CV) disease over 12 months) [19]. Univariable Cox regressions to calculate the unadjusted hazard ratio (HR) and 95% confidence interval (CI) were performed by analyzing all data after first dose of ADT for each variable.
All available confounding variables were evaluated in a multivariable Cox regression analysis to calculate the adjusted HR and 95% CI. The multivariable regression model adjusted for the following variables: baseline metastasis (with vs. without), BMI (<18.5 vs. ≥18.5 kg/m2), treatment setting (oncology vs. urology), drug type (GnRH antagonist vs. LHRH agonist), personal MACE history, tobacco history, baseline PSA (>4 vs. ≤4 ng/mL), race (White vs. Black), statin use, increasing age per year, ethnicity (non-Hispanic vs. Hispanic), increasing ADT exposure per year, diabetes, hypertension, and family MACE history. Another multivariable regression model adjusted for 2 variables: BMI (<18.5 vs. ≥18.5 kg/m2) and race (White vs. Black) to identify whether and how much the 2 variables accounted for the other factor’s risk of MACE and/or mortality.
Body mass index was analyzed using two approaches: as a continuous variable and as a categorical variable. While both methods were employed, the group with a BMI < 18.5 kg/m2 demonstrated the largest absolute difference relative to other BMI groups. This difference was also statistically significant compared to all other BMI groups in the univariable analysis, and therefore BMI < 18.5 kg/m2 vs ≥18.5 kg/m2 was selected as the categorical variable for univariable and multivariable analysis.
Results
Demographics
34,762 patients were included in the Black vs. White analyses (Fig. 1) and 5817 were Black (Table 1). The proportion of Black patients included in our analysis (13%) is representative of the US population (14%) [20].
1MACE (recent urology studies HERO and PRONOUNCE definition) defined as myocardial infarction, stroke, and mortality from any cause. 34,762 and 33,891 patients were included in the mortality and MACE analyses, respectively. MACE Major adverse cardiovascular events, ADT Androgen deprivation therapy, MI Myocardial infarction.
MACE
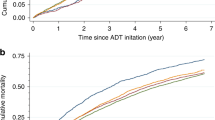
MACE risk was higher for White patients than Black (4.0% and 2.4%, for White and Black patients, respectively at 1 year after ADT initiation; 21.0% and 13.3%, respectively, at 4 years) (Fig. 2a). The unadjusted and adjusted HRs for MACE risk in White vs. Black patients were 1.68 (95% CI 1.56–1.82, p < 0.001) and 1.30 (95% CI 1.09–1.56, p < 0.05), respectively.
a MACE risk was higher for White patients than Black (4.0% and 2.4%, for White and Black patients, respectively at one year after ADT initiation; 21.0% and 13.3%, respectively, at four years). MACE Major adverse cardiovascular events, ADT Androgen deprivation therapy, LHRH Luteinizing hormone-releasing hormone. b The mortality risk after ADT initiation was 1.6% and 2.6% at 1 year and 11.7% and 18.1% at 4 years for Black and White patients, respectively. ADT Androgen deprivation therapy, LHRH Luteinizing hormone-releasing hormone. a Kaplan–Meier Curve for MACE Since ADT Start by Race. 1 Major Adverse Cardiovascular Events (recent urology studies HERO and PRONOUNCE definition) defined as MI, stroke, and mortality from any cause. 2 Date of earliest LHRH injection recorded for patient 3 Excluded patients who had a MACE < 6 months prior to ADT Start. b Kaplan–Meier Curve for All-Cause Mortality 4-Years Since ADT Start by Race. 1 Date of earliest LHRH injection recorded for patient. 2 Excluded patients with no race data.
When evaluating only the impact of BMI and race on MACE, unadjusted MACE risk was higher for patients with BMI < 18.5 vs. ≥18.5 kg/m2 (HR = 2.29, 95% CI 1.82–2.90, p < 0.001) and for White vs. Black patients (HR = 1.69, 95% CI 1.56–1.82, p < 0.001) (Table 2). In the bivariate analysis evaluating BMI and race, adjusted MACE risk was higher for patients with BMI < 18.5 vs. BMI ≥ 18.5 kg/m2 (HR = 2.45, 95% CI 1.87–3.22, p < 0.001) and for White vs. Black patients (HR = 1.72, 95% CI 1.56–1.89, p < 0.001).
Mortality
Mortality risk was higher for White patients than Black (2.6% and 1.6% for White and Black patients, respectively, at 1 year after ADT initiation; 18.1% and 11.7%, respectively, at 4 years) (Fig. 2b). The unadjusted and adjusted HRs for all-cause mortality risk in White vs. Black patients were 1.66 (95% CI 1.53–1.80, p < 0.001) and 1.24 (95% CI 1.01–1.52, p < 0.05), respectively.
When evaluating only the impact of BMI and race on all-cause mortality, unadjusted mortality risk was higher for patients with BMI < 18.5 vs. ≥18.5 kg/m2 (HR = 2.56, 95% CI 2.03–3.24, p < 0.001) and for White vs. Black patients (HR = 1.66, 95% CI 1.53–1.80, p < 0.001) (Table 3). In the bivariate analysis evaluating BMI and race, adjusted mortality risk was higher for patients with BMI < 18.5 vs. BMI ≥ 18.5 kg/m2 (HR = 2.91, 95% CI 2.23–3.80, p < 0.001) and White vs. Black patients (HR = 1.67, 95% CI 1.51–1.84, p < 0.001).
Discussion
Previously published studies have evaluated outcomes by race in patients with PCa [2,3,4, 21,22,23,24,25,26,27,28,29,30]. A 2023 review of PCa, race, and healthy disparity [31] included nine studies that demonstrated a racial disparity in the screening, early detection, and treatment of PCa [32,33,34,35,36,37,38,39,40], with findings such as Black men are less likely to receive aggressive PCa treatment [32] and definitive treatment by radiation or surgery [33, 39], and Black men have less knowledge of PCa and early detection [35]. This review also reported that, of 12 phase 3 randomized clinical trials reporting outcomes by race, only one suggested a worse outcome for African American men [24]. Additionally, three studies support longer/better overall survival in Black vs. White patients with CRPC: a 2019 meta-analysis of nine phase III trials in men with metastatic CRPC (n = 8820) [2]; a 2020 registry study in men with metastatic CRPC (n = 1902) [3]; and a 2023 study in patients with non-metastatic CRPC (n = 12,992) [4]. Another study analyzing data from five phase III randomized radiotherapy PCa trials (n = 5624) found similar 10-year overall survival rates in Black vs. White patients (58% vs. 60%) [41]. Expanding on these previously published studies [2,3,4], our real-world data study evaluated the two separate endpoints of all-cause mortality risk by race and MACE risk by race in patients with PCa on ADT.
Compared to prior studies, our study encompassed a broader scope and was not exclusive to men with CRPC. Our analysis of data over the most recent decade from ~45,000 PCa patients is likely an accurate reflection of the real world.
Contrary to expectations based on SEER data reporting increased mortality risk in Black vs. White patients with PCa [10], our analysis found that both MACE and all-cause mortality were lower in Black vs. White patients with PCa on ADT. This is consistent with other literature suggesting improved outcomes among Black patients with CRPC compared to non-Hispanic White patients [15, 42]. Adding to this body of evidence for Black men with PCa, our research found that Black race is associated with lower MACE rates and improved overall survival for men undergoing treatment with ADT. One potential explanation for our findings is that White men had higher baseline rates of MACE compared to Black men, which could have led to increased MACE risk. Another potential hypothesis for higher mortality risk in White patients is survival bias of Black PCa patients. As Black adults have a significantly younger age of cardiovascular disease diagnosis compared to White adults (50 vs. 56 years) [43], it is possible that Black patients with higher MACE risk passed away before developing PCa and were not captured in the database used for this study. A third potential hypothesis for higher mortality in White vs. Black patients is that Black patients have denser lean body mass than White patients [44], which can protect them from cancer cachexia and allow for longer/better overall survival. Indeed, consistent with previous literature reporting better prognosis in patients with cardiovascular disease who were classified as overweight or obese [45], our analysis results suggest that BMI contributes to improved survival (and lower MACE) independent of race. Of course, the categorical variable of BMI < 18.5 kg/m2 is “underweight” [46] and may be proxy for cancer cachexia rather than a more direct lean/obesity related mechanism. Finally, a fourth potential hypothesis for higher mortality in White vs. Black patients is the HSD3B1 (1245C) allele inheritance, which can cause ADT resistance in men with PCa [47] and, thereby, lower overall survival [47, 48]. This germline is more prevalent in White (10%) than Black (1%) populations [48]. Therefore, White race could be considered a predisposing risk factor for MACE and mortality in PCa patients undergoing ADT.
A multi-disciplinary care team (e.g., primary care physician, urologist, oncologist, cardiologist/cardio-oncologist) should collaborate in patient care, with the goal of providing optimal CV treatment in all PCa patients. Additionally, physicians should actively manage lifestyle habits, including dietary habits, to further mitigate CV risks and improve patient outcomes. Indeed, within the context of phase 3 trials [19, 49], providing optimal cardiac care can minimize the MACE inducing effects of ADT. As such, ensuring all patients have equal access will go a long way to improving outcomes among all patients, especially those with decreased access to care (i.e., Black patients).
Our study has limitations. First, retrospective database studies are hypothesis-generating rather than confirmatory. However, the large size (~45,000 patients from a database containing >300 million patients), long follow-up (10 years for some patients), recent clinical experience (99% from 2010–2020), and diversity of the dataset give weight to the results being an accurate representation of current clinical experience.
Second, our findings are based on a database that used ICD codes, thus the reliability is limited by the accuracy of coding practices. However, any misclassification bias would likely occur uniformly across treatment groups. Third, several other PCa treatments, such as ARI and the co-administration of glucocorticoid [50], have been found to be associated with cardiovascular risk in patients. This may have been a confounding factor and future studies are needed. Additionally, the MACE and mortality analyses are not truly independent since mortality is part of MACE and takes up a large proportion of MACE.
Lastly, socioeconomic factors were not assessed and may be potential confounders. Socioeconomic factors impact patients’ ability to afford and access healthcare, and it has been established that low socioeconomic status is associated with higher risk of morbidity and mortality due to CV disease [51]. It should also be noted that “race” is a social construct. Thus, any differences by “race” are likely due to social factors e.g., systemic racism, lower socioeconomic status, food deserts, lack of health insurance, and lack of preventative care. However, given the real-world knowledge of the distribution of these factors, we might expect Black men to experience higher MACE and mortality risks. That said, equal access to care would likely attenuate differences in survival by “race” and is an important determinant of racial equity. Indeed, a study of over 60,000 men with PCa treated in the equal-access Veterans Affairs medical system found that the 10-year PCa-specific mortality rate was slightly lower for African American men compared to Non-Hispanic White men (4.4 vs. 5.1%; p = 0.005) [52]. These data, along with our study, suggest that, in contrast to national trends, African American men diagnosed with PCa do not appear to present with more advanced disease or experience worse outcomes in comparison to Non-Hispanic White Men. Future studies should analyze factors such as social determinants of health, regional differences, types of insurance, and annual income, to reduce potential bias to due socioeconomic factors.
Conclusion
Our analysis reveals a unique finding that both MACE and all-cause mortality incidence were higher in White vs. Black patients. BMI and race are largely independent and do not account for the other factor’s increased MACE and mortality risk. Adding to the body of evidence for Black men with PCa, our research reveals that the Black race is associated with lower MACE rates and improved survival in men on ADT. Potential explanations for these findings may include genetic factors or other hypotheses warranting further investigation. Future studies should evaluate the role of co-morbidities on MACE risk for PCa patients during ADT to identify other CV predictors, confirm our findings that White PCa patients have higher MACE and mortality risk compared to Black patients, and investigate the above hypotheses. Finally, despite the common belief dating back more than 20 years, Black men with PCa do not appear to have inherently higher risks of MACE and mortality.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request. Restrictions apply to the availability of data generated or analyzed during this study to preserve patient confidentiality or because they were used under license. The corresponding author [judd.moul@duke.edu] will on request detail the restrictions and any conditions under which access to some data may be provided.
References
Zhao J, Zhu S, Sun L, Meng F, Zhao L, Zhao Y, et al. Androgen deprivation therapy for prostate cancer is associated with cardiovascular morbidity and mortality: a meta-analysis of population-based observational studies. PLoS One. 2014;9:e107516.
Halabi S, Dutta S, Tangen CM, Rosenthal M, Petrylak DP, Thompson IM Jr, et al. Overall Survival of Black and White Men With Metastatic Castration-Resistant Prostate Cancer Treated With Docetaxel. J Clin Oncol. 2019;37:403–10.
Sartor O, Armstrong AJ, Ahaghotu C, McLeod DG, Cooperberg MR, Penson DF, et al. Survival of African-American and Caucasian men after sipuleucel-T immunotherapy: outcomes from the PROCEED registry. Prostate Cancer Prostatic Dis. 2020;23:517–26.
Rasmussen KM, Patil V, Li C, Yong C, Appukkuttan S, Grossman JP, et al. Survival Outcomes by Race and Ethnicity in Veterans With Nonmetastatic Castration-Resistant Prostate Cancer. JAMA Netw Open. 2023;6:e2337272.
Prostate Cancer Statistics: Centers for Disease Control and Prevention. 2024. Available from: https://www.cdc.gov/prostate-cancer/statistics/index.html.
Heart Disease Facts: Centers for Disease Control and Prevention. 2024. Available from: https://www.cdc.gov/heart-disease/data-research/facts-stats/index.html.
Saka AH, Giaquinto AN, McCullough LE, Tossas KY, Star J, Jemal A, et al. Cancer statistics for African American and Black people, 2025. CA Cancer J Clin. 2025;75:111‒40.
Kyalwazi AN, Loccoh EC, Brewer LC, Ofili EO, Xu J, Song Y, et al. Disparities in Cardiovascular Mortality Between Black and White Adults in the United States, 1999 to 2019. Circulation. 2022;146:211–28.
Barlow M, Down L, Mounce LTA, Merriel SWD, Watson J, Martins T, et al. Ethnic differences in prostate-specific antigen levels in men without prostate cancer: a systematic review. Prostate Cancer Prostatic Dis. 2023;26:249–56.
Cancer Stat Facts: Prostate Cancer: National Cancer Institute Surveillance, Epidemiology, and End Results Program; Accessed January 2025. [Available from: https://seer.cancer.gov/statfacts/html/prost.html.
Freedland SJ, Samjoo IA, Rosta E, Lansing A, Worthington E, Niyazov A, et al. The impact of race on survival in metastatic prostate cancer: a systematic literature review. Prostate Cancer Prostatic Dis. 2023;26:461–74.
Muralidhar V, Dee EC, Mahal BA, Wei XX, Sartor O, Mouw KW, et al. Association Between Black Race And Improved Survival Following Sipuleucel-T Immunotherapy In Metastatic Castrate-Resistant Prostate Cancer: Implications For Immune Biology And Integration Of Radiation Therapy With Immunotherapy. Int J Radiat Oncol Biol Phys. 2020;108:e901.
Tutrone R, Pieczonka C, Nordquist L, Concepcion R, Flanders S, Armstrong A. Survival Outcomes for Metastatic Castration-Resistant Prostate Cancer with PSA less than 5.0 ng/mL treated with Sipuleucel-T, Overall and by Race: Data from the PROCEED Registry. 20th Annual Meeting of the Society of Urologic Oncology; Washington, DC. 2019.
Zhao H, Howard LE, De Hoedt A, Terris MK, Amling CL, Kane CJ, et al. Racial Discrepancies in Overall Survival among Men Treated with (223)Radium. J Urol. 2020;203:331–7.
Marar M, Long Q, Mamtani R, Narayan V, Vapiwala N, Parikh RB. Outcomes Among African American and Non-Hispanic White Men With Metastatic Castration-Resistant Prostate Cancer With First-Line Abiraterone. JAMA Netw Open. 2022;5:e2142093.
George DJ, Ramaswamy K, Huang A, Russell D, Mardekian J, Schultz NM, et al. Survival by race in men with chemotherapy-naive enzalutamide- or abiraterone-treated metastatic castration-resistant prostate cancer. Prostate Cancer Prostatic Dis. 2022;25:524–30.
Leuva H, Sigel K, Zhou M, Wilkerson J, Aggen DH, Park YA, et al. A novel approach to assess real-world efficacy of cancer therapy in metastatic prostate cancer. Analysis of national data on Veterans treated with abiraterone and enzalutamide. Semin Oncol. 2019;46:351–61.
Shore ND, Saad F, Cookson MS, George DJ, Saltzstein DR, Tutrone R, et al. Oral Relugolix for Androgen-Deprivation Therapy in Advanced Prostate Cancer. N Engl J Med. 2020;382:2187–96.
Lopes RD, Higano CS, Slovin SF, Nelson AJ, Bigelow R, Sørensen PS, et al. Cardiovascular Safety of Degarelix versus Leuprolide in Patients with Prostate Cancer: The Primary Results of the PRONOUNCE Randomized Trial. Circulation. 2021:144:1295–1307.
QuickFacts United States: United States Census Bureau; 2023. Available from: https://www.census.gov/quickfacts/fact/table/US/PST045222.
Crawford ED, Blumenstein BA, Goodman PJ, Davis MA, Eisenberger MA, McLeod DG, et al. Leuprolide with and without flutamide in advanced prostate cancer. Cancer. 1990;66:1039–44.
Vogelzang NJ, Chodak GW, Soloway MS, Block NL, Schellhammer PF, Smith JA Jr., et al. Goserelin versus orchiectomy in the treatment of advanced prostate cancer: final results of a randomized trial. Zoladex Prostate Study Group. Urology. 1995;46:220–6.
McLeod DG, Schellhammer PF, Vogelzang NJ, Soloway MS, Sharifi R, Block NL, et al. Exploratory analysis on the effect of race on clinical outcome in patients with advanced prostate cancer receiving bicalutamide or flutamide, each in combination with LHRH analogues. The Casodex Combination Study Group. Prostate. 1999;40:218–24.
Thompson I, Tangen C, Tolcher A, Crawford E, Eisenberger M, Moinpour C. Association of African-American ethnic background with survival in men with metastatic prostate cancer. J Natl Cancer Inst. 2001;93:219–25.
Roach M 3rd, Lu J, Pilepich MV, Asbell SO, Mohiuddin M, Grignon D. Race and survival of men treated for prostate cancer on radiation therapy oncology group phase III randomized trials. J Urol. 2003;169:245–50.
Halabi S, Small EJ, Vogelzang NJ, Barrier RC Jr, George SL, Gilligan TD. Impact of race on survival in men with metastatic hormone-refractory prostate cancer. Urology. 2004;64:212–7.
Roach M 3rd, De Silvio M, Rebbick T, Grignon D, Rotman M, Wolkov H, et al. Racial differences in CYP3A4 genotype and survival among men treated on Radiation Therapy Oncology Group (RTOG) 9202: a phase III randomized trial. Int J Radiat Oncol Biol Phys. 2007;69:79–87.
Spratt DE, Chen YW, Mahal BA, Osborne JR, Zhao SG, Morgan TM, et al. Individual Patient Data Analysis of Randomized Clinical Trials: Impact of Black Race on Castration-resistant Prostate Cancer Outcomes. Eur Urol Focus. 2016;2:532–9.
Dess RT, Hartman HE, Mahal BA, Soni PD, Jackson WC, Cooperberg MR, et al. Association of Black Race With Prostate Cancer-Specific and Other-Cause Mortality. JAMA Oncol. 2019;5:975–83.
Ma TM, Romero T, Nickols NG, Rettig MB, Garraway IP, Roach M 3rd, et al. Comparison of Response to Definitive Radiotherapy for Localized Prostate Cancer in Black and White Men: A Meta-analysis. JAMA Netw Open. 2021;4:e2139769.
Roach M 3rd, Coleman PW, Kittles R. Prostate Cancer, Race, and Health Disparity: What We Know. Cancer J. 2023;29:328–37.
Barocas DA, Penson DF. Racial variation in the pattern and quality of care for prostate cancer in the USA: mind the gap. BJU Int. 2010;106:322–8.
Pollack CE, Bekelman JE, Liao KJ, Armstrong K. Hospital racial composition and the treatment of localized prostate cancer. Cancer. 2011;117:5569–78.
Barocas DA, Grubb R 3rd, Black A, Penson DF, Fowke JH, Andriole G, et al. Association between race and follow-up diagnostic care after a positive prostate cancer screening test in the prostate, lung, colorectal, and ovarian cancer screening trial. Cancer. 2013;119:2223–9.
Coughlin SS, Vernon M, Klaassen Z, Tingen MS, Cortes JE. Knowledge of prostate cancer among African American men: A systematic review. Prostate. 2021;81:202–13.
Hoge C, Verma S, Lama DJ, Bergelson I, Haj-Hamed M, Maynor S, et al. Racial disparity in the utilization of multiparametric MRI-ultrasound fusion biopsy for the detection of prostate cancer. Prostate Cancer Prostatic Dis. 2020;23:567–72.
Abashidze N, Stecher C, Rosenkrantz AB, Duszak R Jr, Hughes DR. Racial and Ethnic Disparities in the Use of Prostate Magnetic Resonance Imaging Following an Elevated Prostate-Specific Antigen Test. JAMA Netw Open. 2021;4:e2132388.
Kensler KH, Pernar CH, Mahal BA, Nguyen PL, Trinh QD, Kibel AS, et al. Racial and Ethnic Variation in PSA Testing and Prostate Cancer Incidence Following the 2012 USPSTF Recommendation. J Natl Cancer Inst. 2021;113:719–26.
Agochukwu-Mmonu N, Qin Y, Kaufman S, Oerline M, Vince R, Makarov D, et al. Understanding the Role of Urology Practice Organization and Racial Composition in Prostate Cancer Treatment Disparities. JCO Oncol Pr. 2023;19:e763–e72.
Nyame YA, Holt SK, Etzioni RD, Gore JL. Racial inequities in the quality of surgical care among Medicare beneficiaries with localized prostate cancer. Cancer. 2023;129:1402–10.
Roach M, Zhang J, Esteva A, Mohamad O, Wal DVD, Simko J, et al. Prostate cancer risk in African American men evaluated via digital histopathology multi-modal deep learning models developed on NRG Oncology phase III clinical trials. J Clin Oncol. 2022;40:108.
George DJ, Halabi S, Heath EI, Sartor AO, Sonpavde GP, Das D, et al. A prospective trial of abiraterone acetate plus prednisone in Black and White men with metastatic castrate-resistant prostate cancer. Cancer. 2021;127:2954–65.
Lee K, Huang X, Wang MC, Shah NS, Khan SS. Age at Diagnosis of CVDs by Race and Ethnicity in the U.S., 2011 to 2020. JACC Adv. 2022;1:100053.
Schutte JE, Townsend EJ, Hugg J, Shoup RF, Malina RM, Blomqvist CG. Density of lean body mass is greater in blacks than in whites. J Appl Physiol Respir Environ Exerc Physiol. 1984;56:1647–9.
Elagizi A, Kachur S, Lavie CJ, Carbone S, Pandey A, Ortega FB, et al. An Overview and Update on Obesity and the Obesity Paradox in Cardiovascular Diseases. Prog Cardiovasc Dis. 2018;61:142–50.
Assessing Your Weight: Centers for Disease Control and Prevention. 2022. Available from: https://www.cdc.gov/healthyweight/assessing/index.html#:~:text=If%20your%20BMI%20is%20less,falls%20within%20the%20obese%20range.
Sharifi N. Homozygous HSD3B1(1245C) inheritance and poor outcomes in metastatic castration-resistant prostate cancer with abiraterone or enzalutamide: what does it mean? Ann Oncol. 2020;31:1103–5.
McKay RR, Nelson TJ, Pagadala MS, Teerlink CC, Gao A, Bryant AK, et al. Adrenal-Permissive Germline HSD3B1 Allele and Prostate Cancer Outcomes. JAMA Netw Open. 2024;7:e242976.
Nguyen PL, Je Y, Schutz FA, Hoffman KE, Hu JC, Parekh A, et al. Association of androgen deprivation therapy with cardiovascular death in patients with prostate cancer: a meta-analysis of randomized trials. JAMA. 2011;306:2359–66.
Chan JSK, Lee YHA, Leung CH, Leung DKW, Dee EC, Ng K, et al. Associations between glucocorticoid use and major adverse cardiovascular events in patients with prostate cancer receiving antiandrogen: a retrospective cohort study. Prostate Cancer Prostatic Dis. 2024.
Schultz WM, Kelli HM, Lisko JC, Varghese T, Shen J, Sandesara P, et al. Socioeconomic Status and Cardiovascular Outcomes: Challenges and Interventions. Circulation. 2018;137:2166–78.
Riviere P, Luterstein E, Kumar A, Vitzthum LK, Deka R, Sarkar RR, et al. Survival of African American and non-Hispanic white men with prostate cancer in an equal-access health care system. Cancer. 2020;126:1683–90.
Acknowledgements
Editorial support was provided by Xelay Acumen Group, Inc. (funded by Tolmar, Inc.). All the authors have authorized the submission of their manuscript via Xelay Acumen Group, Inc. and have approved all statements and declarations, including conflicting interests and funding.
Funding
This work was supported by Tolmar, Inc.
Author information
Authors and Affiliations
Contributions
Judd W. Moul, Mack Roach III, Deborah M. Boldt-Houle participated in research design, data analysis, and writing and editing the manuscript. All authors have reviewed the manuscript, believe it represents valid work, and approved it for submission.
Corresponding author
Ethics declarations
Competing interests
DMB-H is an employee of Tolmar, Inc. JWM and MR III have no conflicts of interest to declare. Editorial support was funded by Tolmar, Inc.
Ethics approval and consent to participate
All methods were performed in accordance with the relevant guidelines and regulations. This study was conducted in accordance with the Declaration of Helsinki. Because this is a retrospective study, there is no approval number for each study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Moul, J.W., Boldt-Houle, D.M. & Roach, M. Real-world analyses of major adverse cardiovascular events and mortality risk after androgen deprivation therapy initiation in black vs. white prostate cancer patients. Prostate Cancer Prostatic Dis 28, 946–952 (2025). https://doi.org/10.1038/s41391-025-00963-y
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41391-025-00963-y