Abstract
The safety of multi-dose mesenchymal stem cell (MSC) regimens has seldom been systematically investigated. The PRIME-HFrEF (Prospective Randomized Controlled Study of Multiple Intravenous Infusions of Umbilical Cord–derived MSCs in Patients with Heart Failure and Reduced Ejection Fraction) trial was a single-center, randomized, placebo-controlled, investigator-initiated study (ClinicalTrials.gov identifier: NCT04992832) that enrolled 40 patients. The trial aimed to evaluate the safety of three intravenous infusions of Umbilical Cord–derived MSCs (UC-MSCs) administered at six-week intervals in patients with heart failure and reduced ejection fraction (HFrEF), while also collecting exploratory efficacy data. The primary safety endpoint was the incidence of serious adverse events (SAEs), and the primary efficacy endpoint was the change (Δ) in left ventricular ejection fraction (LVEF). Secondary efficacy endpoints included changes in right ventricular (RV) end-systolic and end-diastolic volumes (ESV and EDV). Thirty-nine patients completed 12 study visits over a 360-day follow-up period or until death. The incidence of SAEs did not differ significantly between treatment groups. However, UC-MSC–treated patients exhibited elevated D-dimer levels, suggesting a treatment-associated increase in coagulability. D-dimer levels were negatively correlated with LVEF, and no significant difference in ΔLVEF was observed between groups. In contrast, the improvement in ΔRVESV was significantly greater in the UC-MSC group than in placebo-treated patients (P = 0.033). In summary, multi-dose UC-MSC infusions were safely administered to patients with HFrEF and were associated with improvements in RV volumes. However, these benefits were accompanied by transient increases in coagulability, which may have attenuated potential improvements in left ventricular contractile function.
Similar content being viewed by others
Introduction
Angiotensin receptor-neprilysin inhibitors (ARNIs) and sodium-glucose cotransporter 2 inhibitors (SGLT2i) have recently been designated as Class I therapies for the treatment of heart failure with reduced ejection fraction (HFrEF).1 Although the implementation of these updated guideline-directed medical therapies (GDMT) is expected to substantially reduce heart failure-related mortality and hospitalization, the overall prognosis for patients with HFrEF remains poor.2 Consequently, there is continued interest in developing novel therapeutic strategies to further improve clinical outcomes, including cell-based therapies, which have demonstrated encouraging yet heterogeneous results in clinical trials.3 In the recently completed DREAM-HF (Double-Blind Randomized Assessment of Clinical Events With Allogeneic Mesenchymal Precursor Cells in Heart Failure) trial, 537 patients with HFrEF received a single transendocardial injection of mesenchymal precursor cells or control treatment. Although the change (Δ) in left ventricular (LV) ejection fraction (EF) from baseline to Month 12 was significantly greater in the cell-treated group, no significant differences were observed in the primary efficacy endpoint—time to recurrent events—or in terminal cardiac outcomes.4
The therapeutic effects of cardiac cell therapy are generally attributed to two principal mechanisms: (1) direct replacement of damaged cardiac cells, particularly cardiomyocytes, and (2) paracrine signaling that supports cardiac repair and regeneration.3,5 Mesenchymal stem cells (MSCs) primarily exert their effects through paracrine pathways, secreting a broad range of bioactive factors that promote angiogenesis and confer protection against apoptosis, inflammation, and fibrosis.6 In addition, MSCs are relatively easy to obtain and, due to their low expression of major histocompatibility complex molecules in culture, are considered minimally immunogenic, enabling allogeneic administration. Despite these advantages, clinical benefits derived from allogeneic MSC therapy have generally been modest and transient,7 potentially because MSCs persist in vivo for only a few weeks.8,9 Whether repeated dosing of MSCs can enhance and sustain their therapeutic efficacy has rarely been systematically examined.
The transendocardial delivery approach employed in DREAM-HF is technically complex and poorly suited for repeated administration. In contrast, the phase 1/2 RIMECARD trial (Randomized Clinical Trial of Intravenous Infusion of Umbilical Cord Mesenchymal Stem Cells on Cardiopathy) utilized an intravenous delivery route and demonstrated that a single infusion of umbilical cord–derived MSCs (UC-MSCs) was safe and associated with improvements in cardiac function and quality of life in patients with HFrEF.10 However, participants in both DREAM-HF and RIMECARD were not treated with the currently recommended combination of ARNIs and SGLT2i, which constitutes contemporary GDMT and has been shown to yield meaningful improvements in patient outcomes. Moreover, although numerous studies have established the safety of a single intravenous MSC infusion, the safety profile of multiple MSC administrations remains insufficiently characterized and must be evaluated in smaller exploratory studies prior to the initiation of large-scale efficacy trials.
Here, we report the findings of the PRIME-HFrEF (Prospective Randomized Controlled Study of Multiple Intravenous Infusions of UC-MSCs in Patients with Heart Failure and Reduced Ejection Fraction) trial, an investigator-initiated, randomized, double-blind, placebo-controlled study conducted in patients with HFrEF receiving combined ARNIs and SGLT2i therapy. Modeled as a phase 1/2 trial, PRIME-HFrEF was designed to evaluate the safety of UC-MSCs administered intravenously on three occasions at six-week intervals, while concurrently collecting exploratory efficacy data. The incidence of serious adverse events (SAEs) served as the primary safety endpoint, and ΔLVEF was the primary efficacy endpoint. Given that intravenously infused cells transit through the right ventricle (RV) and pulmonary circulation before reaching the LV, we hypothesized that therapeutic effects might be more pronounced in the RV. Accordingly, RV chamber dimensions were also assessed to more comprehensively characterize the cardiovascular effects of repeated intravenous UC-MSC administration.
Results
Patient demographics
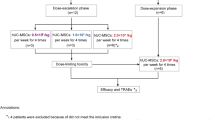
A total of 40 patients with HFrEF were enrolled in the study. One participant in the placebo group withdrew after enrollment for personal reasons (Fig. 1a); therefore, 39 patients were included in the full analysis set (FAS). All participants were classified as New York Heart Association (NYHA) functional class II–III. Baseline characteristics were generally well balanced between the two groups, with the exception of total cholesterol (CHOL), which was significantly higher in the placebo group (P = 0.011) (Table 1). All patients received GDMT: 100% of participants in both groups were treated with ARNIs, and SGLT2i were prescribed to 90% of UC-MSC-treated patients and 84% of placebo-treated patients. The schedule of study procedures and follow-up visits is shown in Fig. 1b.
Patient disposition and study schedule. a Flow diagram illustrating patient enrollment, randomization, treatment allocation, and follow-up. b Schematic overview of treatment administration and scheduled follow-up visits. UC-MSCs umbilical cord-derived mesenchymal stem cells, ARNI angiotensin receptor-neprilysin inhibitor, SGLT2i sodium-glucose cotransporter-2 inhibitor, MRA mineralocorticoid receptor antagonist. Red arrows denote UC-MSC or placebo infusion time points; black arrows denote clinical laboratory measurement time points; yellow dots denote imaging examination time points
Safety
Serious adverse events and adverse events
Four patients in the UC-MSC group and two patients in the placebo group experienced SAEs (Table 2), with no significant difference in SAE incidence between groups. Two deaths occurred in the UC-MSC group. The first was a 67-year-old male whose warfarin therapy was discontinued for 81 days following a retroperitoneal hemorrhage; he subsequently died of cerebral infarction on Day 131. The second was a 66-year-old female who experienced three hospitalizations for worsening heart failure and died suddenly on Day 341. In both cases, the causes of death were more plausibly attributable to anticoagulation interruption, arrhythmia, or progression of heart failure rather than to UC-MSC treatment. No deaths occurred in the placebo group.
The incidence of other adverse events-including inflammatory, cardiovascular, gastrointestinal, neurological, and endocrine/metabolic disorders-was comparable between groups (Supplementary Table 1). Hepatic and renal function remained within normal ranges throughout the study (Table 3). Serial computed tomography (CT) and ultrasonography revealed no evidence of tumorigenesis or ectopic tissue formation, and the incidence of arrhythmia did not differ between groups (Supplementary Tables 2-3).
Coagulation and blood lipids
Principal component analysis (PCA) of baseline-adjusted laboratory parameters revealed a clear separation between UC-MSC- and placebo-treated patients (Fig. 2a). When these parameters were further grouped into ten functional modules, PCA indicated that the “coagulation” and “lipid” modules contributed most prominently to intergroup separation (Supplementary Fig 1). In contrast, the “immune factors” module, which is typically associated with the anti-inflammatory activity of MSCs, showed little separation between groups. Within the coagulation module, D-dimer and international normalized ratio (INR) were the principal contributors to between-group differences, whereas CHOL and small Low-Density Lipoprotein (sLDL) were the dominant contributors within the lipid module (Fig. 2b). Across all post-baseline visits, procoagulant markers and lipid parameters tended to increase in the UC-MSC group, while anticoagulation indicators showed a relative decline (Fig. 2c; Supplementary Table 4). Notably, D-dimer levels in UC-MSC-treated patients rose to pathological levels after the second infusion (V5) and remained elevated through the third infusion (V8), before returning to the normal range by Day 180 (V11) and Day 360 (V12) (Fig. 2d). INR values in the UC-MSC group remained consistently lower than baseline from one week after the first infusion through the end of follow-up. Collectively, these findings indicate that a multi-dose UC-MSC infusion regimen is associated with transient alterations in lipid metabolism and a shift toward a hypercoagulable state.
Blood lipids and coagulation. Principal component analysis (PCA) of laboratory parameters shown in two dimensions based on (a) patient group and visit and (b) key contributing indicators; in panel (b), cos² values indicate the relative contribution of each indicator to the principal components. c Heat map depicting baseline-relative changes in blood lipid and coagulation indicators across all visits; representative indicators are highlighted in red. d Raw serum D-dimer and international normalized ratio (INR) values at each visit. V0 denotes the 1st MSC infusion; V5 denotes the 2nd MSC infusion; and V8 denotes the 3rd MSC infusion. e Serum total cholesterol (CHOL) and small dense LDL (sLDL) levels at each visit. Dashed lines denote the upper limit of normal. Statistical significance in (d) and (e) was assessed using baseline-adjusted repeated-measures analysis of variance (ANOVA). Data are presented as mean ± SEM. Red P values indicate P < α (0.05)
Elevation of D-dimer occurred almost exclusively in UC-MSC-treated patients who were not receiving anticoagulation therapy (Fig. 3a, b). Moreover, baseline-relative changes in D-dimer were negatively correlated with changes in LVEF (Fig. 3c, d; Supplementary Tables 5-6). In parallel, UC-MSC-treated patients exhibited transient, nonpathological increases in CHOL and sLDL (Fig. 2e), which were positively correlated with changes in LVEF (Fig.3c, d; Supplementary Tables 5–6). To integrate cardiac functional and laboratory measures, raw data for cardiac parameters-including LVEF, left ventricular fractional shortening (LVFS), left ventricular end-systolic and end-diastolic volumes (LVESV and LVEDV), right ventricular end-systolic and end-diastolic volumes (RVESV and RVEDV), fractional area change (FAC), and tricuspid annular plane systolic excursion (TAPSE), together with circulating biomarkers of N-terminal pro-B-type natriuretic peptide (NT-proBNP) and growth stimulation-expressed gene 2 protein (ST2) at V0, V11, and V12 were used to construct a composite Heart Function Index (HFI) matrix. Mantel testing demonstrated that inter-individual dissimilarities in baseline-relative D-dimer changes from V0 to the mean of V9 and V10 were significantly correlated with the HFI matrix at both V0 and V12, but only in the UC-MSC treatment group (Fig. 3e, f; Supplementary Table 7).
Correlation between coagulation or blood-lipid measurements and cardiac function indicators. a Summary of anticoagulant use in each group (1 = users; 0 = non-users); P values were calculated using the chi-square test. b D-dimer levels stratified by anticoagulant status; P values were obtained using Spearman correlation. Red P values indicate P < α (0.05). c, d Pearson correlation coefficients (r) between baseline-relative fractional changes (BRF; averaged over V9-V10) in coagulation/blood-lipid metrics and cardiac function measures (averaged over V11-V12), displayed as heat maps (positive correlations in coral orange; negative correlations in mint green; color intensity reflects |r | ); significant correlations (P < 0.05) are marked with an asterisk. e, f Pearson and Mantel correlation analyses between the unadjusted Heart Function Index (HFI; composite of LVEF, LVFS, LVESV, LVEDV, FAC, RVESV, RVEDV, TAPSE, NT-proBNP, and ST2) at baseline (V0), Day 180 (V11), and Day 360 (V12), and coagulation or lipid metrics. Pearson correlations are shown as heat maps (positive in pink; negative in light blue; color intensity reflects |r | ). Mantel coefficients are represented by line thickness, and Mantel P values by line color (gray, P > 0.05; red, 0.01 < P ≤ 0.05; deep red, 0.001 < P ≤ 0.01). BRF baseline-relative fractional change, defined as (Xt − X0) / X0, where X0 is the baseline value at V0 and Xt is the mean of V9 and V10
Immunogenicity
Despite the allogeneic origin of UC-MSCs, PCA of immune cell-related indicators revealed no significant differences between the UC-MSC and placebo groups (Supplementary Fig 1). Consistently, most autoantibodies remained undetectable in UC-MSC-treated patients throughout follow-up (Supplementary Table 8). However, positive antinuclear antibody (ANA) titers (1:300 or 1:1000) were observed in 6 of 20 UC-MSC recipients (30.0%), compared to 1 of 19 patients (5.3%) in the placebo group, a difference that did not reach statistical significance (P = 0.091). Among UC-MSC-treated patients who died or required rehospitalization, two (33.3%) exhibited elevated ANA titers, whereas four (66.7%) maintained normal ANA levels.
To explore whether an inflammatory host microenvironment (Supplementary Fig 2a) might prime UC-MSCs and enhance their immunogenic potential, we analyzed publicly available transcriptomic data (GSE117387) derived from cytokine-stimulated UC-MSCs. Cytokine exposure was associated with marked upregulation of human leukocyte antigen (HLA) gene expression (Supplementary Fig 2b), including HLA-DRA and β2-microglobulin (B2M) (Supplementary Fig 2c). These findings suggest that exposure to proinflammatory conditions may increase MSC immunogenicity, providing a potential mechanistic context for the ANA responses observed in a subset of UC-MSC-treated patients.
Exploratory efficacy
Left ventricular measures
LV function was evaluated using cardiac magnetic resonance imaging (MRI) and echocardiography. Cardiac MRI demonstrated significant improvements in LVEF from baseline to V12 in both the UC-MSC group (ΔLVEF = 8.73 ± 8.81%, P = 0.003) and the placebo group (ΔLVEF = 12.63 ± 6.99%, P < 0.001); however, the magnitude of improvement did not differ significantly between groups (Fig. 4a). Both groups also exhibited significant reductions in LVESV and LVEDV over the same period (P = 0.001 and P = 0.010, respectively), with no significant between-group differences. Echocardiographic assessments yielded concordant results (Fig. 4b). Positron emission tomography (PET)-MRI analyses of LV fibrosis and myocardial 18F-fluorodeoxyglucose uptake revealed no significant changes from baseline to V11 in either group, and the extent of change did not differ between groups (Supplementary Fig 3a). Additional analyses stratified by disease severity and age similarly showed no significant differences in LV functional outcomes between treatment groups (Supplementary Fig 4).
Left ventricular function and dimensions. Changes (Δ) in left ventricular ejection fraction (LVEF), left ventricular end-systolic volume (LVESV), and left ventricular end-diastolic volume (LVEDV) from baseline (V0) to the 360-day follow-up (V12), assessed by (a) cardiac magnetic resonance imaging (MRI) (n = 13 in placebo group, n = 14 in UC-MSCs group) and (b) echocardiography (n = 19 in placebo group, n = 18 in UC-MSCs group). P values were determined using independent t tests. Data are presented as mean ± SEM
Right ventricular measures
In contrast to LV findings, cardiac MRI analyses indicated significant improvements in (RV) parameters in the UC-MSC group. From baseline to V11, UC-MSC-treated patients demonstrated a significant reduction in RVESV and a trend toward reduced RVEDV (ΔRVESV = –39.46 ± 59.85 ml, P = 0.027; ΔRVEDV = –24.44 ± 44.75 ml, P = 0.062). By comparison, placebo-treated patients exhibited small, non-significant increases in RV volumes (ΔRVEDV = 20.00 ± 34.13 ml, P = 0.067; ΔRVESV = 3.46 ± 16.26 ml, P = 0.477). Between-group differences in ΔRVEDV and ΔRVESV were statistically significant (P = 0.010 and P = 0.033, respectively; Fig. 5a, b). The reduction in RVESV in the UC-MSC group persisted at V12 (ΔRVESV = -50.31 ± 72.60 ml), remaining significant both relative to baseline (P = 0.022) and compared with placebo (P = 0.041; Fig. 5c). Consistent results were observed when data were analyzed using ANCOVA with baseline values as covariates, which revealed significant between-group differences in RVESV at V11-V12 and in RVEDV at V11 (Supplementary Tables 9, 10).
Right ventricular dimensions and function. a Representative cardiac MRI images of the right ventricle (RV) at baseline (V0) and at Day 180 (V11) in the placebo and UC-MSC groups. Changes (Δ) in RV end-systolic volume (RVESV) and RV end-diastolic volume (RVEDV) from baseline (V0) to (b) Day 180 (V11) (n = 12 in placebo group, n = 14 in UC-MSCs group) and (c) Day 360 (V12) (n = 11 in placebo group, n = 14 in UC-MSCs group), measured by cardiac MRI. d Changes (Δ) in tricuspid annular plane systolic excursion (TAPSE) from baseline (V0) to Day 180 (V11) (n = 18 in placebo group, n = 19 in UC-MSCs group) and Day 360 (V12) (n = 19 in placebo group, n = 18 in UC-MSCs group), measured by echocardiography. P values were calculated using independent t-tests. Data are presented as mean ± SEM. Red P values indicate P < α (0.05)
TAPSE also improved significantly in the UC-MSC group at both V11 and V12 (P = 0.043 and P = 0.023, respectively), whereas no significant changes were observed in the placebo group (P = 0.498 and P = 0.595). Between-group differences in TAPSE improvement approached statistical significance at both time points (V11: P = 0.071; V12: P = 0.068; Fig. 5d). Post hoc analyses suggested that the most pronounced improvements in ΔRVESV and ΔTAPSE occurred in patients with more severe baseline dysfunction (LVEF <26% or TAPSE < 17 mm) or in younger patients (≤59 years) (Supplementary Fig 4).
Other efficacy assessments
During the 12-month follow-up period (V0-V12), the composite clinical endpoint of death or heart failure hospitalization occurred in 30.0% of UC-MSC-treated patients (6/20) and 26.3% of placebo-treated patients (5/19), with no significant difference between groups (P = 0.764; Fig. 6). Patients in the UC-MSC group demonstrated a significant improvement in the 6-minute walk test (6MWT) distance from baseline to V12 (Δ6MWT = 41.86 ± 57.09 m, P = 0.017), whereas no significant change was observed in the placebo group (P = 0.414); however, the between-group difference did not reach statistical significance. Minnesota Living with Heart Failure Questionnaire (MLHFQ) scores were comparable between groups throughout follow-up (Supplementary Fig 3b).
Kaplan-Meier analysis of composite clinical outcomes. Kaplan–Meier curves comparing freedom from heart failure–related rehospitalization or death over time between the UC-MSC and placebo groups
Baseline NT-proBNP and ST2 levels were non-normally distributed and exhibited substantial inter-individual variability, particularly in the UC-MSC group (Supplementary Tables 11-12). Significant between-group differences in ΔNT-proBNP (P = 0.035) and ΔST2 (P = 0.015) were observed using standard non-parametric analyses, but these differences were no longer significant after adjustment for baseline values using non-parametric rank-based ANCOVA (Supplementary Table 12).
Discussion
MSCs are attractive candidates for cardiac regenerative therapy because they can be readily isolated from multiple tissue sources and secrete a broad array of paracrine factors with pro-angiogenic, anti-inflammatory, and antifibrotic properties.3 However, exogenously administered MSCs typically persist in vivo for only a few weeks, suggesting that the therapeutic effects of a single administration may be transient. Early clinical observations support this notion: a randomized trial conducted in 2006 demonstrated that intracoronary infusion of bone marrow–derived cells (BMCs) improved LVEF at 6 months, but this benefit was no longer evident at 18 months.7 Despite these findings, systematic investigation of multi-dose cell therapy strategies has only begun in recent years (Supplementary Table 13).
UC-MSCs are particularly well suited for clinical application because they can be harvested noninvasively, exhibit robust proliferative capacity, and may exert more potent therapeutic effects than MSCs derived from adult tissues (Supplementary Table 14). The PRIME-HFrEF trial represents the first randomized, double-blind, placebo-controlled study to evaluate a multi-dose regimen of intravenously administered allogeneic UC-MSCs in patients with HFrEF. In terms of safety, this therapeutic strategy demonstrated a favorable profile overall. UC-MSC treatment was not associated with an increased incidence of SAEs or adverse events (AEs), including acute infusion reactions, ventricular arrhythmias, stroke, myocardial infarction, malignancy, or death. These observations are consistent with prior studies evaluating single-dose MSC infusion,10 as well as a double-blind trial in which patients with chronic obstructive pulmonary disease received monthly intravenous MSC infusions for four months.11
Hypercoagulability is a recognized safety concern associated with intravenous MSC administration.12 In the present study, patients in the UC-MSC group exhibited evidence of coagulation perturbations, most notably pathologically elevated D-dimer levels that persisted for several weeks following infusion. Importantly, these elevations occurred predominantly in patients who were not receiving antithrombotic therapy, and changes in D-dimer levels were negatively correlated with indices of LV function. MSCs are known to express high levels of tissue factor,13 a potent initiator of the instant blood-mediated inflammatory reaction (IBMIR), which can activate the coagulation cascade.14 Preclinical studies further suggest that heparin can attenuate IBMIR and improve MSC engraftment and therapeutic efficacy.15 Although no patients in the PRIME-HFrEF trial experienced overt thrombotic events, the observed subclinical procoagulant changes may have attenuated the therapeutic effects of UC-MSCs. These findings suggest that future trials employing repeated intravenous UC-MSC infusions should consider systematic evaluation of concomitant anticoagulation strategies.
In exploratory analyses, the lipid module-driven primarily by CHOL and sLDL-contributed to intergroup separation. However, these associations do not establish a direct causal effect of UC-MSC infusions on lipid metabolism. Given the baseline imbalance in CHOL between treatment groups, and potential confounding by concomitant lipid-lowering therapy and other systemic factors, correlations between lipid parameters and cardiac function should be interpreted with caution and regarded as hypothesis-generating. Future studies incorporating prespecified adjustment for lipid-lowering medications, targeted metabolomic profiling, and broader immunometabolic characterization will be required to distinguish direct MSC-mediated effects from secondary changes related to cardiac remodeling or systemic hemodynamic improvement.
MSCs are generally considered hypoimmunogenic due to their low expression of HLA molecules and their secretion of immunomodulatory factors, such as indoleamine 2,3-dioxygenase.16 This characterization is consistent with the absence of significant between-group differences in immune cell counts observed in the present study. However, exposure to proinflammatory conditions has been shown to shift MSCs toward a more immunogenic phenotype,17 and the immunological behavior of UC-MSCs administered in a multi-dose regimen has not been fully characterized. Our transcriptomic analyses provide supportive evidence that inflammatory cytokines can markedly upregulate HLA expression in UC-MSCs, including HLA-DRA and B2M, suggesting that host inflammatory milieus may influence MSC immunogenicity. Accordingly, further studies are warranted to determine whether circulating factors in patient serum can modulate the functional phenotype and immunological properties of MSCs following repeated administration. MSC-based therapies have been associated with reductions in autoantibody levels in autoimmune disease settings,18,19 although these studies did not employ multi-dose protocols, and few trials in nonautoimmune conditions have systematically monitored ANA over time. A systematic review of 356 patients treated with allogeneic MSCs reported donor-specific antibody development in 11.5% of cases.20 In the present study, while no inflammatory cytokines differed significantly between groups, several approached statistical significance (e.g., IL-1: F = 3.626, P = 0.065; IL-6: F = 3.489, P = 0.070), and the modest sample size may have limited the detection of subtle treatment-related effects. Notably, ANA positivity was observed in 30.0% of UC-MSC-treated patients, compared with 5.3% of placebo-treated patients; however, none of the ANA-positive individuals developed clinical manifestations of autoimmune disease. Taken together, these findings suggest that the observed increase in ANA positivity should be regarded as exploratory and does not constitute evidence of a definitive safety signal. Nonetheless, they underscore the importance of longitudinal immunological monitoring in future trials evaluating multi-dose administration of allogeneic MSCs.
Although the PRIME-HFrEF trial was primarily designed to evaluate the safety of a multi-dose UC-MSC regimen in patients with HFrEF, exploratory analyses were also performed to assess potential efficacy signals. The primary efficacy endpoint-ΔLVEF from baseline to V12 (Day 360)—did not differ significantly between the UC-MSC and placebo groups. Likewise, no between-group differences were observed in ΔLVEDV, ΔLVESV, heart failure hospitalizations, 6MWT performance, or MLHFQ scores. These findings contrast with results from several earlier trials in which MSC administration was associated with modest improvements in LVEF, often when cells were delivered via intramyocardial routes,21 or in patient populations receiving less optimized background medical therapy.10 In addition, recent meta-analyses have reported small but significant increases in LVEF (approximately 3–6%), reductions in LV volumes, and fewer heart failure-related hospitalizations among MSC-treated patients.22,23,24 However, it is notable that many of these studies were conducted before the widespread adoption of contemporary “gold standard” GDMT, which now consists of quadruple therapy with an ARNI or angiotensin-converting enzyme inhibitor, a β-blocker, a mineralocorticoid receptor antagonist, and a SGLT2i.25,26 In PRIME-HFrEF, nearly all participants were receiving concomitant ARNI and SGLT2i therapy, in contrast to earlier MSC trials (e.g., 0% ARNI/SGLT2i use in RIMECARD, and 25.1% ARNI and 1.1% SGLT2i use in DREAM-HF).4,10 The substantial benefits conferred by contemporary GDMT may therefore have masked any incremental improvements attributable to MSC therapy, particularly given that PRIME-HFrEF was not powered to detect small-to-moderate changes in LVEF. Additional factors that may have influenced LV outcomes include the relatively short GDMT stabilization period prior to the first infusion, lower baseline LVEF values, and the use of methylprednisolone to mitigate infusion-related reactions, which may have dampened inflammatory signaling pathways relevant to MSC activation and engagement. Moreover, the effects of MSC therapy on LV remodeling appear to be highly context dependent, varying with disease etiology, cell dose, and delivery route. For example, transendocardial MSC administration has been associated with LVEF improvement in nonischemic dilated cardiomyopathy,27 but not in ischemic cardiomyopathy,28 and no significant LVEF benefit has been reported when adipose-derived or hypoxia-conditioned MSCs were delivered via intramyocardial or intravenous routes.29,30 Collectively, these considerations underscore the complexity of interpreting LV functional outcomes in the setting of advanced background therapy and highlight the need for carefully tailored trial designs to detect incremental benefits of cell-based interventions.
With respect to right ventricular remodeling, at V11 (Day 180), patients treated with UC-MSCs demonstrated significantly greater improvements in ΔRVEDV and ΔRVESV compared with placebo-treated patients, and the beneficial effect on ΔRVESV persisted at V12 (Day 360). TAPSE also improved at both V11 and V12 in UC-MSC recipients but not in the placebo group. Given the exploratory nature of this investigator-initiated trial, the modest sample size, and baseline variability, these improvements in RV dimensions should be interpreted as preliminary efficacy signals rather than definitive evidence. Nevertheless, in light of the absence of approved pharmacological therapies that specifically target RV dysfunction, these findings could be clinically meaningful if confirmed in larger, multicenter studies. The right ventricle is uniquely vulnerable to pathological stress, in part because its angiogenic response is impaired and its antioxidant defenses are weaker than those of the left ventricle.31 MSCs secrete multiple pro-angiogenic cytokines as well as superoxide dismutase 3, thereby promoting neovascularization, reducing reactive oxygen species, and protecting cardiomyocytes from ischemia-reperfusion injury.3,6,31 In preclinical models of RV pressure overload, MSC therapy more effectively reversed disease-associated proteomic alterations than other progenitor cell types, resulting in greater improvements in RV structure and function.32 In addition, intravenously administered UC-MSCs traverse the right ventricle and pulmonary circulation before reaching the left ventricle, and animal studies have demonstrated substantial pulmonary retention of infused MSCs.33 This “pulmonary filter” effect likely limits the number of MSCs reaching the left ventricle relative to the right ventricle and may, at least in part, explain the absence of significant LV functional improvement observed in the UC-MSC group.
NT-proBNP and ST2 are well-established biomarkers for the diagnosis and clinical assessment of heart failure.34,35 In PRIME-HFrEF, baseline distributions of both biomarkers were highly skewed, a pattern commonly observed in larger heart failure cohorts, and baseline values tended to be higher, although not significantly, in the UC-MSC group than in placebo-treated patients. While initial nonparametric analyses identified significant between-group differences in ΔNT-proBNP and ΔST2, these differences were no longer evident after adjustment for baseline levels using ANCOVA, indicating that factors such as regression to the mean cannot be excluded as potential contributors to the observed changes. Accordingly, whether repeated UC-MSC infusions can induce sustained and clinically meaningful improvements in these biomarkers remains unproven, but also not disproven, and merits further investigation in adequately powered studies.
In the PRIME-HFrEF trial, patients received three intravenous infusions of UC-MSCs or placebo at 6-week intervals, with the final infusion administered near the end of the third month within the 12-month follow-up period. This dosing strategy was informed by preclinical evidence indicating that intravenously delivered MSCs are largely cleared within days to weeks and exert their therapeutic effects predominantly through transient paracrine signaling rather than long-term engraftment, with biological activity peaking several weeks after administration.36 Consistent with this concept, multiple preclinical studies across diverse animal models of cardiac disease have demonstrated that repeated cell dosing over several weeks to months produces greater and more durable improvements in ventricular function and remodeling than a single administration of the same cells.37,38 Moreover, emerging clinical evidence in patients with heart failure suggests that the benefits of bone marrow-derived or hematopoietic stem cell therapies may be incrementally enhanced and/or prolonged when cells are delivered repeatedly rather than as a one-time intervention.39,40
At present, available preclinical and translational evidence suggests a biologically plausible re-dosing window of approximately 2–8 weeks, during which MSC-mediated paracrine effects and downstream cellular responses evolve and then attenuate. For example, MSC-induced c-kit⁺ cardiac progenitor cell responses peak at approximately 2 weeks and return toward baseline by around 8 weeks,37 and biodistribution studies indicate that systemically delivered MSCs are largely cleared within weeks and are typically undetectable by 8 weeks.38 In addition, clinical experience supports the feasibility of repeated dosing at roughly monthly intervals (e.g., monthly infusions in chronic obstructive pulmonary disease without infusion-related adverse events during follow-up).11 Based on this window, we selected a 6-week interval to (i) avoid re-dosing at the putative peak of the preceding dose-thereby reducing the risk of masking the effect of the first infusion-while (ii) re-dosing before the biological activity is expected to fully wane, aiming to sustain or augment potential benefit. We also note in the manuscript that the optimal interval remains to be determined and should be formally evaluated in future trials.
Study limitations
Several limitations should be acknowledged when interpreting the findings of this study. First, the sample size was modest (n = 20 per treatment arm), and the duration of follow-up was limited to one year, which restricts statistical power and the ability to assess long-term safety and efficacy. Second, all UC-MSCs were derived from only three healthy donors, raising the possibility that donor-specific differences in immunogenicity or therapeutic potency may have influenced the observed outcomes. Third, although this was a single-center trial, participants were recruited from multiple regions across China, including areas with limited access to advanced medical resources (e.g., Heilongjiang Province). This recruitment pattern likely contributed to the low prevalence of implantable cardioverter defibrillator (ICD) use–three patients in the placebo group and none in the UC-MSC group–which is lower than expected based on current guideline recommendations for the prevention of sudden cardiac death in patients with HFrEF.41 However, this observation reflects broader systemic constraints within the Chinese healthcare system: despite an estimated 540,000 individuals at risk of sudden cardiac death annually, only approximately 8000 ICDs are implanted in China each year.42 Accordingly, the low rate of ICD utilization in this study is consistent with real-world practice in resource-limited settings and underscores the relevance of these findings to patient populations that are often underrepresented in clinical trials. Finally, despite rigorous randomization procedures, notable-though not always statistically significant-baseline imbalances were observed, particularly in cholesterol levels, NT-proBNP, ST2, and indices of RV function. Several potentially meaningful trends in cytokine and biomarker levels did not reach statistical significance, likely due, at least in part, to the limited sample size and insufficient statistical power. These observations suggest that future studies may benefit from the use of stratified randomization based on key prognostic variables, implemented in an unblinded manner at enrollment but followed by fully blinded treatment administration and outcome assessment. Such an approach could improve baseline comparability and enhance the ability to detect treatment-related effects.
Conclusion
In summary, the PRIME-HFrEF trial demonstrates that administration of three intravenous doses of allogeneic UC-MSCs in patients with HFrEF was not associated with clinically significant immune activation or inflammation and did not increase the incidence of SAEs or AEs compared with placebo. However, UC-MSC treatment was associated with evidence of transient hypercoagulability, which may have attenuated cardiac functional responses, indicating that the potential role of concomitant anticoagulation strategies warrants further investigation. Notably, this study is the first to systematically evaluate RV responses to UC-MSC therapy and provides preliminary evidence that multi-dose intravenous UC-MSC infusions may reduce RV dilatation. Although exploratory in nature, these findings highlight a potentially novel therapeutic avenue and underscore the need for validation in larger, multicenter clinical trials.
Materials and methods
Comprehensive methodological details of the PRIME-HFrEF trial (ClinicalTrials.gov identifier: NCT04992832) have been published previously in the study protocol43 and are further detailed in the Online Appendix. The study was approved by the Ethics Committee and Scientific Research Committee of Shanghai East Hospital (approval number EC.D (BG).016.03.1) and was conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent was obtained from all participants prior to enrollment.
Randomization and blinding
Blocks of 10 enrolled patients were randomly assigned in a 1:1 ratio to either the UC-MSC group or the Placebo group. The randomization list was generated with SAS 9.1.3 software by an independent facility (the Department of Preventive Medicine, Medical College, Tongji University). All patients, study sponsors, nurses, data collectors, and statisticians were blinded to the treatment allocation. Treatments were manufactured by the laboratory of the National Stem Cell Translational Resource Center, and all manufacturing information was withheld from the clinical team. The packaging and appearance of the experimental (UC-MSC) and control (placebo) products were indistinguishable.
Patient population and treatment
HFrEF, defined as a LVEF ≤ 40%, who had been receiving stable GDMT for at least three months were eligible for enrollment. Full inclusion and exclusion criteria are provided in Supplementary Table 16. Participants were randomized to receive three intravenous infusions at 6-week intervals of either placebo (1% human serum albumin alone) or UC-MSCs suspended in 1% human serum albumin. UC-MSCs were isolated from Wharton’s jelly of human umbilical cords. The administered dose (1 × 10⁶ cells per kilogram of body weight) was selected based on prior clinical experience from the RIMECARD trial.10 To minimize inter-donor variability, all three infusions administered to an individual participant were derived from the same donor.
Study endpoints
The primary safety endpoint was the occurrence of any SAE within 12 months following treatment. SAEs included, but were not limited to, death, nonfatal myocardial infarction, stroke, and sustained ventricular arrhythmias lasting more than 15 seconds or associated with hemodynamic compromise. Cardiac arrhythmias were assessed using serial Holter monitoring during both the intervention and follow-up periods. Additional SAEs included persistent or significant disability, tumor formation, and any clinical condition requiring inpatient hospitalization or prolongation of an existing hospital stay.
The primary efficacy endpoint was ΔLVEF from baseline to the 12-month follow-up visit, assessed by contrast-enhanced echocardiography or cardiac MRI. Secondary efficacy endpoints included all-cause mortality and heart failure–related rehospitalization; circulating levels of NT-proBNP and ST2; performance on the 6MWT; LVESV/LVEDV; RVESV/RVEDV; and TAPSE measured by echocardiography. Additional imaging-based endpoints included myocardial fibrosis, quantified as cardiac enhancement volume by MRI, and myocardial glucose uptake, assessed as standardized uptake value by PET/MRI.
Contrast echocardiography, cardiac MRI, and PET
Contrast-enhanced echocardiography, cardiac MRI, and PET were performed to assess cardiac structure, function, and myocardial metabolism. Contrast echocardiography was acquired after intravenous administration of SonoVue and was digitally stored for quantitative analysis. Cardiac MRI was performed on a 3.0-T scanner to quantify LVESV, LVEDV, RVESV, RVEDV, and LVEF, and to obtain late gadolinium enhancement (LGE) images; LGE was defined as myocardial post-contrast signal intensity ≥6 standard deviations above that of a reference region of non-enhanced myocardium within the same slice,44 and the LGE volume ratio was calculated as the percentage of left ventricular volume exhibiting LGE. PET imaging was conducted on an integrated 3.0-T PET-MR system using 18F-fluorodeoxyglucose under glucose-loading conditions, with standardized uptake values (SUVs) used as a relative measure of myocardial 18F-FDG uptake.45 The detailed operating procedures can be found in the supplementary materials.
UC-MSC isolation and expansion
Clinical-grade allogeneic UC-MSC were produced in the Good Manufacturing Practice (GMP) laboratory of Stem Cell East Hospital (affiliated with Tongji University and the National Stem Cell Translational Resource Center). Cells were isolated from the Wharton’s Jelly of umbilical cords obtained from healthy, fully screened donors and expanded under standardized GMP conditions. The cells displayed the defining pattern of surface marker for MSCs—positive for CD73, CD90, and CD105 expression, and negative or low expression for CD11b, CD19, CD31, CD34, CD45, and HLA-DR. In addition, the trilineage differentiation potential of the UC-MSCs (adipogenic, osteogenic, and chondrogenic) was verified as part of cell characterization (Supplementary Fig 5). The cells met predefined release criteria for identity, viability, sterility, mycoplasma, endotoxin, and a full panel of human viral tests (including HIV). Qualified seed stocks could be resuscitated and cultured for up to five passages, and the final product was prepared at a dose of 1×10⁶ cells/kg per infusion, with all three infusions for each patient derived from the same batch of donor cells.
Statistical analysis
Primary statistical analyses were conducted in the full analysis set (FAS; defined as all randomized participants who received at least one infusion and had ≥1 post-baseline assessment), using SPSS version 22.0 (IBM Corp., Armonk, NY, USA) and R version 4.2.3 software. Two-sided P values < 0.05 were considered statistically significant. Death and hospitalization related to heart failure were summarized by Kaplan–Meier curves and compared using the log-rank test. For efficacy endpoints, between-group differences were assessed using Student’s t-tests or Wilcoxon rank-sum tests, as appropriate for the data distribution. Because the principal goal of the trial was to evaluate the safety of three intravenous UC-MSC injections, no a priori power analysis was performed to determine the sample size required to establish efficacy. A post hoc power analysis at a two-sided significance level of 0.05 suggested limited power overall, with the highest estimated power observed for RVESV at Day 180 (83.92%) and Day 360 (72.96%).
Multivariate patterns in safety- and efficacy-related clinical indicators were explored using PCA. Dimensionality was reduced via orthogonal transformation to yield two principal components for visualization. In addition, repeated-measures analysis of variance was performed for key indicators with adjustment for baseline values. Pearson correlation analysis was applied to assess linear associations among continuous variables, while Spearman rank correlation was used for analyses involving binary variables. The Mantel test was used to evaluate correlations between distance matrices or dissimilarity structures derived from sets of variables. To evaluate potential heterogeneity and reduce confounding, stratified analyses were conducted according to LVEF, TAPSE, age, or ANA; within each stratum, between-group differences in efficacy endpoints were assessed using Student’s t-tests or Wilcoxon rank-sum tests, depending on the distribution.
Data availability
The study protocol, including the statistical analysis plan, and other supporting materials are provided within this article and its supplementary information files. Due to national regulatory requirements in China, research involving UC-MSCs is subject to oversight, and the raw data from this study have been transferred to Jiangsu Kanion Pharmaceutical Co., Ltd. under a data transfer agreement. Therefore, the full dataset cannot be made publicly available due to ethical, regulatory, and confidentiality restrictions. Anonymized data supporting the findings of this study may be made available for non-commercial academic research upon reasonable request to the corresponding authors and Jiangsu Kanion Pharmaceutical Co., Ltd. Requests should include a detailed research proposal and analysis plan and will be reviewed within two weeks. Access to approved data will require a signed data access agreement.
References
McDonagh, T. A. et al. 2023 Focused Update of the 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 44, 3627–3639 (2023).
Burger, P. M. et al. Personalized lifetime prediction of survival and treatment benefit in patients with heart failure with reduced ejection fraction: The LIFE-HF model. Eur. J. Heart Fail 25, 1962–1975 (2023).
Bolli, R., Solankhi, M., Tang, X. L. & Kahlon, A. Cell therapy in patients with heart failure: a comprehensive review and emerging concepts. Cardiovasc Res. 118, 951–976 (2022).
Perin, E. C. et al. Randomized trial of targeted transendocardial mesenchymal precursor cell therapy in patients with heart failure. J. Am. Coll. Cardiol. 81, 849–863 (2023).
Sareen, N., Srivastava, A., Alagarsamy, K. N., Lionetti, V. & Dhingra, S. Stem cells derived exosomes and biomaterials to modulate autophagy and mend broken hearts. Biochim Biophys. Acta Mol. Basis Dis. 1869, 166806 (2023).
Ulpiano, C., da Silva, C. L. & Monteiro, G. A. Mesenchymal stromal cells (MSCs): A promising tool for cell-based angiogenic therapy. Curr. Gene Ther. 21, 382–405 (2021).
Meyer, G. P. et al. Intracoronary bone marrow cell transfer after myocardial infarction: eighteen months’ follow-up data from the randomized, controlled BOOST (BOne marrOw transfer to enhance ST-elevation infarct regeneration) trial. Circulation 113, 1287–1294 (2006).
Tokita, Y. et al. Repeated administrations of cardiac progenitor cells are markedly more effective than a single administration: a new paradigm in cell therapy. Circ. Res. 119, 635–651 (2016).
Hou, D. et al. Radiolabeled cell distribution after intramyocardial, intracoronary, and interstitial retrograde coronary venous delivery: Implications for current clinical trials. Circulation 112, I150–I156 (2005).
Bartolucci, J. et al. Safety and efficacy of the intravenous infusion of umbilical cord mesenchymal stem cells in patients with heart failure: A phase 1/2 randomized controlled trial (RIMECARD trial [randomized clinical trial of intravenous infusion umbilical cord mesenchymal stem cells on cardiopathy]). Circ. Res 121, 1192–1204 (2017).
Weiss, D. J., Casaburi, R., Flannery, R., LeRoux-Williams, M. & Tashkin, D. P. A placebo-controlled, randomized trial of mesenchymal stem cells in COPD. Chest 143, 1590–1598 (2013).
Yang, B. et al. Procoagulant properties of mesenchymal stem cells and extracellular vesicles: a novel aspect of thrombosis pathogenesis. Stem Cells 42, 98–106 (2024).
Hoang, V. T. et al. Impact of tissue factor expression and administration routes on thrombosis development induced by mesenchymal stem/stromal cell infusions: Re-evaluating the dogma. Stem Cell Res. Ther. 15, 56 (2024).
Moll, G. et al. Intravascular mesenchymal stromal/stem cell therapy product diversification: Time for new clinical guidelines. Trends Mol. Med. 25, 149–163 (2019).
Liao, L. et al. Heparin improves BMSC cell therapy: Anticoagulant treatment by heparin improves the safety and therapeutic effect of bone marrow-derived mesenchymal stem cell cytotherapy. Theranostics 7, 106–116 (2017).
Zhang, C. et al. Eradication of specific donor-dependent variations of mesenchymal stem cells in immunomodulation to enhance therapeutic values. Cell Death Dis. 12, 357 (2021).
Sivanathan, K. N. & Coates, P. T. IL-17A-induced mesenchymal stem cells have promising therapeutic value for clinical translation. Kidney Int. 93, 771–773 (2018).
Wang, D. et al. Umbilical cord mesenchymal stem cell transplantation in active and refractory systemic lupus erythematosus: A multicenter clinical study. Arthritis Res. Ther. 16, R79 (2014).
Liang, J. et al. Allogenic mesenchymal stem cells transplantation in refractory systemic lupus erythematosus: a pilot clinical study. Ann. Rheum. Dis. 69, 1423–1429 (2010).
Guan, A. et al. Clinical translation of mesenchymal stem cells in ischemic heart failure: Challenges and future perspectives. Vasc. Pharm. 159, 107491 (2025).
Mathiasen, A. B. et al. Bone marrow-derived mesenchymal stromal cell treatment in patients with severe ischaemic heart failure: A randomized placebo-controlled trial (MSC-HF trial). Eur. Heart J. 36, 1744–1753 (2015).
Shen, T. et al. A systematic review and meta-analysis: Safety and efficacy of mesenchymal stem cells therapy for heart failure. Curr. Stem Cell Res Ther. 16, 354–365 (2021).
Tran, T. T. et al. A comprehensive review of clinical trials and Progress in stem cell therapies for advanced heart failure. Regen. Ther. 30, 812–837 (2025).
Abouzid, M. R. et al. The safety and efficacy of human umbilical cord-derived mesenchymal stem cells in patients with heart failure and myocardial infarction: A meta-analysis of clinical trials. Cureus 15, e49645 (2023).
D’Amato, A. et al. Early SGLT2i therapy facilitates in-hospital ARNI Introduction improving 6-month systolic function in patients with HFrEF. Am. J. Cardiovasc Drugs 26, 205–214 (2026).
Januzzi, J. L. Jr et al. Association of change in N-terminal pro-B-type natriuretic peptide following initiation of sacubitril-valsartan treatment with cardiac structure and function in patients with heart failure with reduced ejection fraction. JAMA 322, 1085–1095 (2019).
Hare, J. M. et al. Randomized comparison of allogeneic versus autologous mesenchymal stem cells for nonischemic dilated cardiomyopathy: POSEIDON-DCM trial. J. Am. Coll. Cardiol. 69, 526–537 (2017).
Heldman, A. W. et al. Transendocardial mesenchymal stem cells and mononuclear bone marrow cells for ischemic cardiomyopathy: The TAC-HFT randomized trial. Jama 311, 62–73 (2014).
Butler, J. et al. Intravenous allogeneic mesenchymal stem cells for nonischemic cardiomyopathy: Safety and efficacy results of a phase II-A randomized trial. Circ. Res. 120, 332–340 (2017).
Qayyum, A. A. et al. Effect of allogeneic adipose tissue-derived mesenchymal stromal cell treatment in chronic ischaemic heart failure with reduced ejection fraction - the SCIENCE trial. Eur. J. Heart Fail 25, 576–587 (2023).
Si, M. S. & Ohye, R. G. Stem cell therapy for the systemic right ventricle. Expert Rev. Cardiovasc Ther. 15, 813–823 (2017).
Makkaoui, N. et al. Cell-based therapies reverse the heart failure-altered right ventricular proteome towards a pre-disease state. Stem Cell Res. Ther. 15, 420 (2024).
Li, X. et al. Intravenous injection of human umbilical cord-derived mesenchymal stem cells ameliorates not only blood glucose but also nephrotic complication of diabetic rats through autophagy-mediated anti-senescent mechanism. Stem Cell Res. Ther. 14, 146 (2023).
Armstrong, P. W. et al. Sequential evaluation of NT-proBNP in heart failure: insights into clinical outcomes and efficacy of vericiguat. JACC Heart Fail 10, 677–688 (2022).
Savarimuthu, S., Goel, P. & Harky, A. Soluble ST2: A valuable prognostic marker in heart failure. Heart Fail Rev. 27, 2155–2164 (2022).
Shan, Y. et al. Pharmacokinetic characteristics of mesenchymal stem cells in translational challenges. Signal Transduct. Target Ther. 9, 242 (2024).
Tang, X. L. et al. Intravenous infusions of mesenchymal stromal cells have cumulative beneficial effects in a porcine model of chronic ischaemic cardiomyopathy. Cardiovasc Res. 120, 1939–1952 (2024).
Gong, C. et al. Infusion of two-dose mesenchymal stem cells is more effective than a single dose in a dilated cardiomyopathy rat model by upregulating indoleamine 2,3-dioxygenase expression. Stem Cell Res. Ther. 13, 409 (2022).
Yao, K. et al. Repeated autologous bone marrow mononuclear cell therapy in patients with large myocardial infarction. Eur. J. Heart Fail 11, 691–698 (2009).
Gu, X. et al. Repeated intracoronary infusion of peripheral blood stem cells with G-CSF in patients with refractory ischemic heart failure-a pilot study. Circ. J. 75, 955–963 (2011).
Heidenreich, P. A. et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 79, e263–e421 (2022).
Jiang, J. & Zhang, S. Current status and challenges of interventional therapy for diagnosis and treatment of arrhythmia in China. Int. J. Heart Rhythm 9, 1–6 (2024).
Gong, X. et al. A prospective randomized controlled study of multi-intravenous infusion of umbilical cord mesenchymal stem cells in patients with heart failure and reduced ejection fraction (PRIME-HFrEF) trial: Rationale and design. Contemp. Clin. Trials Commun. 41, 101350 (2024).
Harrigan, C. J. et al. Hypertrophic cardiomyopathy: Quantification of late gadolinium enhancement with contrast-enhanced cardiovascular MR imaging. Radiology 258, 128–133 (2011).
Thie, J. A. Understanding the standardized uptake value, its methods, and implications for usage. J. Nucl. Med. 45, 1431–1434 (2004).
Acknowledgements
We gratefully acknowledge the support of Shenghu He, MD (SuBei People’s Hospital affiliated with Yangzhou University), and Shouming Zhao, MD (Department of Cardiology, Taicang First People’s Hospital, China), for their assistance with patient selection. We also thank Bo Zhang and Lianghua Xia at Shanghai East Hospital for their contributions to cardiac ultrasound examinations, as well as Bin He for oversight of stem cell regulatory compliance and verification. This work was supported by the National Key Research and Development Program of China (Grant No. 2020YFA0112600), the Peak Disciplines (Type IV) of Institutions of Higher Learning in Shanghai, and the Key Discipline Construction Project of the Shanghai Pudong New Area Health Commission (Grant No. PWZxk2022-20).
Author information
Authors and Affiliations
Contributions
W.H. designed and implemented the study and drafted the manuscript. Y.J. was responsible for clinical data organization and verification and contributed to manuscript writing. W.C. and L.Z. contributed to statistical analyses. X.G. led the clinical implementation and performed quality monitoring. H.H., Y.X., S.G., Q.Z., B.Y., and J.P. were responsible for patient selection and clinical management. J.Z. and H.W. performed and analyzed cardiac PET-MRI scans. Q.L. and S.C. conducted and analyzed cardiac MRI examinations. D.C. performed clinical data verification. W.J. and Z.H. prepared the stem cell products and performed quality control. Y.E.S. and H.Y. analyzed and interpreted the experimental results. Z.L. conceived the overall project and supervised the research. All authors have read and approved the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Han, W., Jiao, Y., Chen, W. et al. PRIME-HFrEF Trial: a randomized, double-blind, multi-dose umbilical cord-derived mesenchymal stem cell regimen for heart failure. Sig Transduct Target Ther 11, 112 (2026). https://doi.org/10.1038/s41392-026-02678-5
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41392-026-02678-5