Abstract
Addictive drugs engage molecular pathways of associative learning and memory. Drug craving triggered by cues that were once associated with drug intoxication contributes to continued drug-seeking behaviors. Reactivated memories are vulnerable to disruption by interference with the process of reconsolidation, hence targeting reconsolidation could be beneficial in reducing cue-induced drug craving and relapse. Here, we sought to identify the neuronal pathways and neuroplasticity involved in cocaine contextual memory reconsolidation. Mice expressing inhibitory DREADDs in either nucleus accumbens (NAc) neurons or neurons in the ventral hippocampus (vHPC) projecting to NAc underwent cocaine place conditioning to establish cocaine contextual memories. Clozapine-N-oxide (CNO) administered after memory reactivation was used to inhibit NAc neurons or vHPC neurons projecting to the NAc during the reconsolidation period. Inhibition of either neuronal population abolished the previously established preference for the cocaine context. FosTRAP2-Ai14 mice were used to identify and characterize neurons in the NAc and vHPC activated during reconsolidation of cocaine memory. Results demonstrate NAc medium spiny neurons activated by cocaine contextual memory recall had greater dendritic spine density, length and complexity than without memory recall. vHPC pyramidal neurons also showed higher dendritic spine density after cocaine contextual memory recall. The neuroplastic changes induced by cocaine memory reactivation suggest synaptic strengthening and eventual maturation of synapses. The results of this work reveal a critical role for a circuit involving glutamatergic projections from the vHPC onto NAc neurons in the reconsolidation of cocaine memories; disruption of the activity of this circuit abolishes previously established cocaine contextual memories.
Similar content being viewed by others
Introduction
Addictive drugs, including cocaine, engage molecular signaling pathways that are involved in associative learning processes. Cocaine-associated memories are highly resistant to extinction [1] and exposure to cues previously associated with cocaine availability can elicit a conditioned physiological response, intense drug craving, and return to cocaine use [1, 2]. Once drug reward memories are reactivated, they undergo a process of reconsolidation through synaptic plasticity that strengthens the memory [3, 4]. During reactivation, memory traces are labile and can be manipulated behaviorally or pharmacologically [5, 6]. Since drug-associated cues can trigger craving and return to drug use, pharmacological disruption of reconsolidation-related plasticity that serves to maintain intrusive cocaine-related memories may be a useful approach to prevent relapse [7]. Indeed, interference with reconsolidation after cue re-exposure can reduce craving for cocaine and other drugs, at least transiently [7, 8]. In a study of persons with cocaine use disorder, propranolol administered following cued memory reactivation temporarily reduced craving and cardiovascular reactivity during a subsequent cocaine cue re-exposure [7]. Since exposure to drug cues can trigger relapse, a treatment objective is to diminish previously learned associations between the positive effects of cocaine and environmental cues signaling cocaine availability [7, 9,10,11,12,13]. The development of successful therapeutic strategies for dampening cue-induced craving depends on a greater understanding of the circuitry and molecular processes involved in the maintenance of cocaine memories.
The goal of this study was to identify the neural circuitry engaged in cocaine memory maintenance and elucidate specific cell populations required for reconsolidation of cocaine contextual memories. The literature supports the involvement of the ventral hippocampus (vHPC) in circuits of fear memory reconsolidation [14,15,16]. Evidence also supports the importance of the projections from the vHPC to the nucleus accumbens (NAc) in the retrieval of recent cocaine memory [17] and the NAc as a prominent site for maintaining drug memories [18,19,20]. Our previous work identified a signaling pathway consisting of NMDA receptors→GSK3β→mTORC1 in the HPC and NAc whose activity is required for reconsolidation of cocaine contextual memories [21,22,23]. Given these findings, we hypothesized that the maintenance of cocaine reward memories relies on activation of neurons in the NAc and their afferent connections from the vHPC. As such, we anticipated that inhibiting the vHPC→NAc circuit after cocaine memory recall would abolish cocaine contextual memories. To test this, we utilized mice expressing pathway-targeted inhibitory DREADDs to silence specific neurons during reconsolidation of cocaine contextual memories using a conditioned place preference procedure. Cells activated in the NAc and vHPC during reconsolidation of cocaine contextual memories were identified and examined for dendritic spine plasticity.
Materials and methods
Animals
Animal procedures were performed in compliance with the National Institutes of Health guidelines for the Care and Use of Laboratory Animals, and animal use was reviewed and approved by Temple University Institutional Animal Care and Use Committee. Male C57BL/6 (8 weeks old on delivery, Charles River Laboratories, Wilmington, MA) were single housed under a 12-h light/dark cycle (7:00 AM lights on/7:00 PM lights off) without enrichment objects with ad libitum standard chow and water. Animals were housed for five days before experiments began and weighed daily. Mice were arbitrarily assigned to experimental groups; no randomization was performed. Behavioral procedures were conducted between 9:00-11:00 AM. Some studies utilized male and female FosTRAP2-Ai14 mice, 8-12 weeks old. FosTRAP2 mice were crossed with Ai14 reporter mice (expressing TdTomato (TdT)), both purchased from Jackson Labs (Stock 030323 & 007914) and bred in our facility. TRAP2 mice utilize a Fos promoter to drive transcription of CreER permitting temporally controlled recombination in active neurons when 4-hydroxytamoxifen (4-OHT) is administered. With the TRAP2-Ai14 cross, 4-OHT leads to the permanent expression of TdT in Fos-expressing neurons.
Drugs
Cocaine hydrochloride, generously supplied by the National Institute on Drug Abuse Drug Supply Program, and clozapine-N-oxide hydrochloride (CNO; Cayman Chemicals, Ann Arbor, MI, #25780) were dissolved in sterile saline. 4-Hydroxytamoxifen (4-OHT; Sigma-Aldrich, Burlington, MA, #H6278) was dissolved in corn oil according to a modification of published methods [24]. All drugs were injected intraperitoneally (ip) in a volume of 5 ml/kg body weight and equal volumes of saline or corn oil served as vehicle controls.
Viral vectors
A chemogenetic approach allowed for selective expression of iDREADDs and neuronal inhibition. In experiment 1, iDREADDs were expressed in the nucleus accumbens (NAc) neuronal cell bodies using AAV2-hSyn-hM4Di-mCherry. In experiment 3, glutamatergic neurons in the ventral hippocampus (vHPC) projecting to the NAc were targeted using a two-vector system approach. In this case, AAV5-calcium/calmodulin dependent protein kinase II-cre was delivered bilaterally into the vHPC, and a Cre-dependent retrograde iDREADD (retroAAV2-DIO-hM4D(Gi)-mCherry) was delivered into the bilateral NAc. The AAV vectors were obtained from the Neural Repair Viral Core (Shriners grant #84051-PHI-21, GM Smith), where they were fully characterized and tested for specificity. See Supplementary Information for details on generation of recombinant adeno-associated viruses.
Intracranial vector injections
Surgical procedures were carried out to inject viral vectors into the NAc and vHPC as described previously by us [22, 25] and detailed in the Supplementary Information. The following coordinates were used to target the NAc and vHPC: NAc (AP: +0.85 mm; ML: ±0.70 mm; DV: -4.2 mm); vHPC (AP: -3.3, ML: ±3.0; DV: -3.0 and -4.0).
Electrophysiology of hippocampal neurons expressing iDREADD
The functionality of the hM4Di synthetic receptor in the presence of CNO was verified ex vivo using whole-cell patch-clamp slice electrophysiology. Methods and data are provided in the Supplementary Information. Results demonstrate that CNO was effective in inhibiting the firing of hippocampal neurons of mice expressing the hM4Di construct (Supplementary Figure 1).
Cocaine-induced conditioned place preference (CPP)
An unbiased procedure was used following our published methods [21]. Place conditioning occurred in rectangular plastic chambers (45 ×20 x 20 cm) consisting of two unique compartments, one with white and black vertical striped walls and smooth flooring and the other with white walls with black circles and rough flooring. The two compartments were separated by a removable wall during conditioning and a door during testing. Illumination in both compartments was equal, and mice showed no initial preference to either compartment. An unbiased procedure was used. Mice were injected with 10 mg/kg ip of cocaine or saline and were immediately confined to one compartment of the conditioning chamber, where they remained for 30 min. Conditioning occurred once per day for 8 consecutive days, resulting in four conditioning sessions with saline in one compartment and four sessions with cocaine in the opposite compartment of the conditioning chamber, in a counterbalanced design. The test for place preference occurred on day 9, when mice had access to both compartments for 15 min in a drug-free state. The time spent in each compartment was recorded. Preference scores were calculated as: (time spent in the cocaine-paired compartment) minus (time spent in the saline-paired compartment) and reported in seconds. Mice that showed less than a 60 second preference for the cocaine-paired compartment on day 9 did not continue in the study. On day 10, 24 h following assessment of cocaine place preference, half the mice were re-exposed to the previously cocaine-paired compartment for 10 min to reactivate and destabilize cocaine-associated memory, while the others remained in the home cage in the testing room.
Chemogenetic targeted neuronal inhibition
The experimental timeline is shown in Fig. 1A. Mice expressing iDREADDs underwent cocaine CPP. On day 10, mice received CNO (5 mg/kg ip) or saline immediately after the 10 min re-exposure to the cocaine context [26], and again 2 and 4 h post re-exposure to inhibit neuronal activity for the entire duration of reconsolidation ( ~4-6 h) [27,28,29]. Mice were re-tested for cocaine place preference in a drug-free state 3 days post re-exposure. As a control for memory re-activation, some mice received an injection of CNO, same dose and schedule, without memory reactivation. The control mice were brought into the CPP testing room on day 10 and received CNO or vehicle injections in their home cage. Following behavioral testing, mice underwent transcardiac perfusions and brain extractions. The brains were frozen and cryosectioned for immunostaining to verify iDREADD location, using primary antibody anti-RFP (1:1000, #600-401-379, Rockland). Sections were imaged at 10x using the Nikon AR2 confocal microscope.
A For Experiments 1 and 3 utilizing iDREADDs, mice received intracranial infusions of viral vectors 10-14 days prior to beginning cocaine CPP to allow stable expression of iDREADDs by the memory reactivation on Day 10 of CPP (ie, 20-24 days post viral injection). B Timeline for studies using FosTRAP2xAi14 mice to identify neurons activated during the recall of cocaine-associated contextual memories.
Identification of activated neurons
The experimental timeline is shown in Fig. 1B. Male and female FosTRAP2-Ai14 mice underwent an unbiased cocaine CPP procedure as described above. On day 10, mice were re-exposed to the cocaine compartment for 10 min to reactivate the cocaine memory; control mice were brought into the testing room but remained in their home cages. 4-OHT (50 mg/kg, i.p.) or vehicle (corn oil) was injected immediately after memory reactivation to “trap” cells that were activated during cocaine contextual memory recall [30]. After 7 days (maximal TdT expression [24, 31]), mice underwent transcardiac perfusions and brains were extracted. Brains were frozen and cryosectioned at 50 µm into free floating wells. IHC was conducted using rabbit primary antibody dsRed (1:500, #632496, Takara) to label TdT+ cells. For quantification of TdT+ cells, sections were imaged on fluorescent microscope (Keynce BZ-X) at 10x for localization of TdT+ labeled cells. Using FIJI (ImageJ), regions of interest were traced in accordance with the mouse brain atlas [32] and cell counts were completed manually by an investigator blind to experimental group. For each mouse, the number of TdT+ cells per region were counted on 6 coronal sections spanning the nucleus accumbens or ventral hippocampus. Data are reported as the mean cell count/region/mouse.
Dendritic spine and morphology analysis
Sections labeled for TdT+ neurons from the TRAP2 mice were imaged first at 10x using Nikon AR2 confocal microscope for localization of TdT-labeled accumbens medium spiny neurons (MSNs) and hippocampal pyramidal neurons, then individual neurons were imaged at 60x oil lens for analysis of dendritic spines. After confocal imaging, cells were reconstructed using Neurolucida 360 software (Microbrightfield Bioscience). The cell soma was detected, then all dendritic branches were traced using the directional kernels method. The tracing was exported to Neurolucida explorer. Cells were selected based on thorough expression of TdT, and dendrites free of obstructions from other cells or blood vessels that could be traced without interruption. Morphometrics were obtained using Neurolucida Explorer software [33]. Spines were automatically detected on the reconstructed dendrites; corrections were manually made for accuracy. Parameters included total number of dendritic spines, head diameter, density per cell, morphology (mushroom, stubby, thin), and density per morphology, completed for 6-10 neurons per region per animal. Dendritic spines were classified into canonical types by Neurolucida 360 using a combination of morphological measurements and ratios derived from 3D reconstructions [34]. While stubby, mushroom and thin-type spines were abundant, few filopodia were detected. As such, they were not included in the analysis. Randomly selected reconstructed cells from dendritic spine analysis were used to assess dendritic tree plasticity using Sholl analysis. Using the Neurolucida Explorer software, total dendritic length and number of dendritic intersections were counted in concentric circles of increasing diameter (10 μm) from the nucleus of the cell. The investigators performing these analyses were blind to the treatment groups.
Statistical analyses
GraphPad Prism was used for performing statistical tests. CPP data were analyzed by two-way RM ANOVA (repeated across test days) with Sidak post-tests. Dendritic morphology was analyzed by either two-way ANOVA or Students two-tailed t-test. Estimates of variation within each data set were performed to justify use of parametric statistical tests. Sample size was chosen by power analyses to be sufficient to detect a difference of 30% between group means based on the variability previously found using these methods, assuming alpha level of 0.05 and power of 80%. Data are presented as individual data points, means, and SEMs.
Results
Inhibition of nucleus accumbens neuronal activity prevents memory reconsolidation and reduces expression of a previously established cocaine place preference
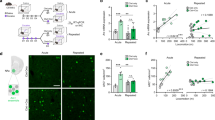
Experiment 1 determined the involvement of NAc neurons in cocaine contextual memory reconsolidation. Figure 1 summarizes the experimental design of the chemogenetic studies (Exp 1 and 3). AAV2-hSyn-hM4D(Gi)-mCherry was injected bilaterally into the accumbens prior to cocaine place conditioning and testing. Sixteen mice underwent 8 days of alternating cocaine and saline conditioning followed by a post-conditioning test for place preference on day 9; 14/16 mice showed a cocaine place preference and comprised the two groups that continued in the study. Figure 2A shows similar CPP scores on day 9 for the two groups of mice. On day 10, 14 mice were re-exposed to the cocaine context for 10 min and CNO or vehicle was administered immediately thereafter. Three days later, mice were re-tested for place preference. Preference scores from days 9 and 13 were analyzed by a two-way ANOVA with day and treatment factors (Fig. 2A). Results indicate a significant interaction (F(1,12) = 4.847, p = 0.048) and main effect of day (F(1,12) = 13.37, p = 0.0033; treatment F(1,12) = 2.865, p = 0.1163). Post hoc tests shows significantly lower CPP scores in mice expressing the iDREADD and injected with CNO following recall of the cocaine context, compared to mice injected with vehicle on Day 13 (iDREADD CNO vs vehicle; p < 0.05) and also significantly lower preference scores on day 13 vs day 9 for the iDREADD+CNO group (day 9 vs day 13, p < 0.005). Figure 2B shows an example of bilateral expression of the iDREADD (AAV2-hSyn-hM4D(Gi)-mCherry) in the accumbens, corroborating site specific expression of iDREADDs. A diagram of a coronal section of the nucleus accumbens is shown in Fig. 2B right for orientation [32].
AAV2-hSyn-hM4D(Gi)-mCherry was injected bilaterally into the nucleus accumbens of mice to express iDREADDs, prior to undergoing cocaine conditioned place preference (CPP). A Two groups of mice (N = 7/group) expressing iDREADDs in the NAc show similar cocaine (10 mg/kg) place preference on Day 9. On Day 10, mice were re-exposed to the cocaine context to activate memory and injected with vehicle or CNO (5 mg/kg) to inhibit NAc neuronal firing. CPP was re-tested on Day 13. CPP scores (time in cocaine side – time in saline side, sec) were significantly lower in iDREADD-expressing mice injected with CNO compared to vehicle-injected mice on day 13 (*p < 0.05) and significantly lower on day 13 than on day 9 (***p < 0.005). B Representative images of NAc showing expression of mCherry-tagged iDREADDs [32]. C Two groups of mice (N = 7-8) expressing NAc iDREADDs showed similar cocaine CPP scores on Day 9. CNO (5 mg/kg) or vehicle was administered in the home cage, without cocaine context re-exposure, and preference retested 3 days later. No significant differences in CPP scores were found on Day 13.
In a control experiment, 18 mice expressing iDREADDs underwent cocaine CPP with 15 demonstrating preference for cocaine context on CPP test day 9 (Fig. 2C). On day 10, all mice remained in their home cage (no reactivation of cocaine contextual memory), 8 received CNO to inhibit NAc neurons and 7 received vehicle. Preference was retested three days later. Data shown in Fig. 2C demonstrate that inhibition of NAc neurons via administration of CNO did not alter place preference when CNO was administered to mice in their home cage environment without re-exposure to the cocaine-paired context; two-way ANOVA day factor: F(1,26) = 0.44, p = 0.51; treatment factor: F(1,26) = 0.11, p = 0.73). These data demonstrate that recall of cocaine memories was necessary for interferance of an established place preference. Taken together, the results in Fig. 2 suggest that inhibiting NAc neurons after reactivation of a cocaine contextual memory prevents reconsolidation as demonstrated by the loss in preference for the cocaine context 72 h post CNO administration.
Nucleus accumbens neurons are activated by recall of cocaine contextual memories
Experiment 2 utilized male and female FosTRAP2-Ai14 mice to visualize and characterize neurons activated by recall of cocaine contextual memories (experimental timeline shown in Fig. 1B). Mice underwent an unbiased cocaine CPP procedure and were separated into 2 groups. CPP scores are presented in Fig. 3A; both groups had comparable preference for the cocaine-paired context on day 9. On day 10, one group of mice was re-exposed to the cocaine compartment (10 min, drug free) for reactivation of cocaine contextual memory and immediately thereafter given 4-OHT (50 mg/kg ip) to label (ie “trap”) Fos+ cells with TdT. The other group of mice remained in their home cages in the testing room and were injected with 4-OHT (50 mg/kg). Brains were prepared for analysis of TdT labeled cells in the nucleus accumbens. Figure 3B presents representative images of TdT+ cells in the accumbens after re-exposure to the cocaine context and for home cage controls. Reactivation of cocaine contextual memory led to an increase in the number of TdT+ cells compared to home cage controls in both the accumbens core and shell (Fig. 3C). Two-way ANOVA of TdT+ cell counts reveal significant differences between treatment groups in the NAc with Sidak’s post-hoc tests revealing significantly more TdT+ cells in the core (p = 0.0014) and shell region (p = 0.001) of 4-OHT injected mice re-exposed to the cocaine context compared to those remaining in home cage.
A Cocaine place preference scores of two groups of TRAP2xAi14 mice showing both groups developed a similar preference for the cocaine-paired context. B Representative images of TdT+ neurons following 4-OHT (50 mg/kg) administration in the home cage (left) or following re-exposure to the cocaine context (middle) indicating neuronal activation. High magnification (60X) image of an activated medium spiny neuron. C Quantification of TdT+ neurons showed a significantly greater number of activated TdT+ cells in the nucleus accumbens core (**p < 0.01) and shell (**p < 0.01) following recall of cocaine contextual memory compared to home cage controls. Coronal brain section at the level of the NAc [32] showing NAc core and shell regions where activated cells were quantified. N = 6/group, 2 male/4 female home cage; 4 male/2 female re-exposure.
Dendritic spine plasticity of nucleus accumbens neurons following recall of cocaine contextual memories
Brains from the FosTRAP2-Ai14 mice used in Experiment 2 were further examined for synaptic plasticity following recall of cocaine memories. Medium spiny neurons (MSNs) were identified by morphological examination of TdT+ cells and dendrites were analyzed for spine density and characteristics. Figure 4A is a representative image of a TdT+ MSNs at high magnification (left) along with representative images of dendrite reconstruction and spine capture using Neurolucida 360 imaging software (right). Recall of cocaine contextual memory significantly increased total spine density compared to home cage controls (t = 3.137, df=12, p = 0.0086; Fig. 4B) and this was driven by an increase in the density of thin spines (treatment factor F(1,35) = 15.61, p = 0.0004; morphology F(2,35) = 10.33, p < 0.0001; interaction F(2,35) = 11.12, p = 0.0002; post-hoc thin spines, p < 0.0001). Total spine surface area was significantly higher following the recall of cocaine context as compared to no recall (Fig. 4C; t = 2.432, df=12; p = 0.031). Spine neck diameter (left) and spine head diameter (right) were significantly smaller in dendrites of MSNs from mice re-exposed to the cocaine context compared to home-cage controls (Fig. 4D; neck diameter t = 4.549, df=12, p = 0.0007; head diameter t = 4.884, df=11, p = 0.0005). Lower spine neck and head diameter likely reflect the higher density of thin spines which have smaller head diameter than the other spine types. Sholl analysis indicated significant differences in dendritic length and complexity of MSNs of mice re-exposed to the cocaine context compared to home-cage controls (Fig. 4E; two-way ANOVA length: treatment F(1157) = 14.78, p = 0.0002; intersections: treatment F(1148) = 13.22, p = 0.0004).
A Representative 60x image of a TdT+ medium spiny neuron from a mouse re-exposed to the cocaine context (left). Representative segments of dendrites from a home cage and a re-exposed mouse, along with the dendrite reconstruction and spine analysis performed by Neurolucida 360 software [33]. Mushroom spines are shown as blue, stubby spines as green, and thin spines as red (right). B Quantification of total spine density (left) and spine density by morphology (right) following recall of cocaine contextual memory compared to home cage controls (**p < 0.01, ****p < 0.0001). Recall of cocaine contextual memory resulted in higher total spine density and higher density specifically of thin spines in accumbens medium spiny neurons. C Quantification of spine surface area following recall of cocaine contextual memory compared to home cage controls (*p < 0.05). D Spine neck diameter (left) and head diameter (right) following recall of cocaine contextual memory were significantly smaller compared to home cage controls (***p < 0.001). E Sholl analysis of dendritic length (left) and number of intersections (right) showed that MSNs dendrites were significantly longer (***p = 0.0002) and more complex (***p = 0.0004) following cocaine memory recall than in the absence of reactivated memories. 8-10 neurons/mouse were analyzed for morphometric characteristics; mean values/mouse are shown. N = 7/group, 4 males + 3 females.
Inhibition of glutamatergic projections from the ventral hippocampus to the nucleus accumbens prevents cocaine contextual memory reconsolidation
Cocaine memory reactivation caused significant neuroplasticity in the nucleus accumbens as demonstrated by changes in dendritic spines on the medium spiny neurons. Experiment 3 sought to identify the circuit that innervates the accumbens (NAc) and contributes to the reconsolidation process. This study targeted ventral hippocampal (vHPC) neurons that project selectively to the NAc and assessed their role in the reconsolidation of cocaine contextual memories. Figure 1A summarizes the experimental design. AAVretro-hSyn-DIO-hM4D(Gi)-mCherry or control AAVretro-hSyn-mCherry vector was injected bilaterally into the nucleus accumbens and AAV-CamKII-Cre into the vHPC of mice, in order to express iDREADDs specifically in the vHPC→NAc pathway. Mice expressing iDREADDs underwent cocaine conditioning followed a test for place preference. Day 9 CPP scores are shown in Fig. 5A for the 25 mice that were conditioned. On day 10, cocaine contextual memory was reactivated via a 10 min re-exposure to the cocaine context followed immediately by CNO to activate the iDREADD or vehicle control injection. Results demonstrated that inhibition of vHPC→NAc neurons by administration of CNO during reconsolidation abolished a previously established place preference when retested three days later on day 13 (Fig. 5A). Mice expressing the control vector and injected with CNO maintained their cocaine place preference, as did mice expressing iDREADDs and injected with vehicle. CPP scores were analyzed by two-way ANOVA (treatment F(2,22) = 5.440, p = 0.012; day F(1,22) = 31.44, p < 0.0001; interaction F(2,22) = 3.529, p = 0.0468). Post-hoc analysis shows a significant difference of the rAAV-iDREADD+CNO group between day 9 and 13 (p = 0.0001), and a significant difference between groups on day 13 (p = 0.0111 rAAV-iDREADD+CNO vs rAAV-control+CNO; p = 0.001 iDREADD+CNO vs iDREADD+vehicle). Thus the established cocaine preference was abolished by CNO-induced chemogenetic inhibition of the vHPC to NAc glutamatergic projection. Location of vectors was verified and representative images of the NAc injection site of the AAVretro-hSyn-DIO-hM4D(Gi)-mCherry and the expression of mCherry by vHPC neurons are provided in Fig. 5B. These data demonstrate that activation of neurons in the vHPC projecting to the NAc was important in the maintenance of cocaine contextual memories.
AAVretro-hSyn-DIO-hM4D(Gi)-mCherry or control AAVretro-hSyn-mCherry vector was injected bilaterally into the nucleus accumbens and AAV-CamKII-Cre into the vHPC of mice, to express iDREADDs specifically in the vHPC→NAc pathway. A Mice expressing vHPC to NAc iDREADDs or vector controls show similar cocaine CPP on Day 9. On Day 10, mice were re-exposed to the cocaine context and injected with CNO (5 mg/kg) or vehicle. CPP was retested on Day 13. CPP scores on Day 13 were significantly lower in mice expressing iDREADDs and injected with CNO compared to both vehicle and vector control mice (*p < 0.05; ***p < 0.001). Day 13 scores were also significantly lower for the iDREADD + CNO group on Day 13 versus Day 9 (**** p < 0.0001). B Representative image of expression of mCherry in the NAc (left) and in the vHPC (right).
Ventral hippocampal neurons are activated by recall of cocaine contextual memories and show associated dendritic plasticity
FosTRAP2-Ai14 mice were used to examine the activation and neuroplasticity of pyramidal cells in the vHPC following recall of cocaine contextual memories. Male and female mice underwent cocaine CPP (see Fig. 1B for experimental timeline). One group was re-exposed to the cocaine context and administered 4-OHT while another group received 4-OHT in home-cage. 4-OHT leads to TdT expression following Fos promoter activation. The number of activated (TdT + ) neurons in the vHPC was significantly higher in mice that were re-exposed to the cocaine context compared to those remaining in home cage (t = 5.305, df=10, p = 0.003 (Fig. 6A). A representative TdT+ pyramidal neuron from a mouse re-exposed to the cocaine context is shown in Fig. 6B along with representative segments of dendrites (top) and their reconstruction by Neurolucida software (bottom). Analysis of TdT+ dendrites of ventral hippocampal pyramidal neurons demonstrated significantly greater spine density following recall of cocaine memories compared with home-cage controls as shown in Fig. 6C, left (t = 4.903, df=9, p = 0.0008). Analysis of spine density by morphology (Fig. 6C, right) showed a significant effect of re-exposure (re-exposure F(1,27) = 30.76, p < 0.0001; morphology F(2,27) = 1021, p < 0.0001; interaction F(2,27) = 0.5059, p = 0.6086). Sidak’s post-hoc test showed significant differences in the density of each spine type, thin spines (*p = 0.0388), stubby spines (**p = 0.0013), and mushroom spines (*p = 0.0196), between home cage controls and cocaine context re-exposure. Total spine surface area in the vHPC was significantly higher following the recall of cocaine context as compared to no recall (Fig. 6D; t = 9.373, df=9; p < 0.0001). Spine neck diameter (Fig. 6E left; t = 2.397, df=9, p = 0.0401) and head diameter (Fig. 6E right; t = 2.783, df=9, p = 0.0213) were significantly larger in dendrites of pyramidal neurons from mice re-exposed to the cocaine context as compared to home cage controls. Total spine volume was significantly greater in mice re-exposed to the cocaine context when compared to the home cage controls (Fig. 6E right; t = 9.754, df=9, p < 0.0001). Sholl analyses of dendritic length and complexity of pyramidal cells are shown in Fig. 6F; no significant differences in dendritic length or intersections were found. Thus, recall of cocaine contextual memories activated vHPC neurons and induced significant neuroplastic changes.
A Quantification of TdT+ neurons in the vHPC following 4-OHT (50 mg/kg) administration in the home cage or following re-exposure to the cocaine context indicating neuronal activation. There was a significantly greater number of activated TdT+ cells in the vHPC (***p = 0.0005) following recall of cocaine contextual memory compared to home cage controls. B Representative 60x image of a TdT+ pyramidal neuron in the vHPC of a mouse re-exposed to the cocaine context (left). Representative segments of dendrites from a home cage and a cocaine context re-exposed mouse (top), along with the dendrite reconstruction and spine analysis performed by Neurolucida 360 software (bottom). Mushroom spines are shown as blue, stubby spines as green, and thin spines as red (right). C Quantification of total spine density and spine density by morphology following recall of cocaine contextual memory compared to home cage controls. Recall of cocaine contextual memory resulted in higher total spine density (***p = 0.0003), due to greater density of all spine types (*p < 0.05, **p < 0.005). D Spine surface area was significantly greater after cocaine memory recall as compared with home cage controls (****p < 0.0001). E Overall spine head diameter (*p < 0.05), neck diameter (*p < 0.05), and spine volume (****<0.0001) were significantly greater in mice re-exposed to the cocaine context as compared to the home cage controls. F Sholl analysis of dendritic length and complexity are shown and were not significantly different between groups. 5-8 neurons/mouse were analyzed for morphometric characteristics; mean values per mouse are shown. N = 5 (3 male + 2 female) re-exposed and 6 (2 male + 4 female) home cage mice.
Discussion
Environmental cues can form strong and long-lasting associations with cocaine intoxication. This can occur when a context is paired with the rewarding effects of cocaine over multiple occasions. Repetition of such salience with a specific context leads to the context acquiring conditioned reinforcing properties [35,36,37,38,39]. Reactivation of drug memories induced by contextual cues can trigger a physiological response and intense drug craving which can perpetuate drug-seeking behaviors. Disruption of cocaine contextual memories may be a valuable strategy to prevent cue-induced drug seeking behavior.
The present study sought to identify the circuit engram involved in the reconsolidation of cocaine memories. Our results demonstrate that cocaine memory reconsolidation can be disrupted by chemogenetically inhibiting NAc intrinsic neurons or the glutamatergic projections from the vHPC to the NAc. Inhibition of NAc neuronal firing immediately after reactivation of cocaine-associated contextual memory abolished a previously established cocaine place preference. NAc neuronal silencing in the absence of memory recall (i.e., CNO to home cage controls) had no effect on the maintenance of the place preference, supporting the specific targeting of reconsolidation and the requirement of NAc neuronal activity therein. The reconsolidation process begins shortly after memory recall and is completed within 6 h [40, 41]. In the present study, CNO was administered immediately after memory recall and again 2 and 4 h later to maintain neuronal inhibition throughout the duration of the reconsolidation process. Because of this study design, it is unknown if selective inhibition of early- or late-stage reconsolidation would be sufficient to reduce memory strength as was examined in other studies [42]. Disruption of reconsolidation decreases the retention of that specific memory and has been studied as a potential therapeutic strategy to ameliorate trauma-associated memories in patients with post-traumatic stress disorder [43, 44].
Using fosTRAP2XAi14 mice that had an established cocaine place preference, neurons activated during the recall of cocaine contextual memory were ‘trapped’ for further study. Results demonstrate that NAc MSNs in both the core and shell regions were activated by recall of cocaine contextual memories. We further analyzed activated MSNs for structural changes. Analysis of MSNs ‘trapped’ following cocaine memory reactivation showed changes indicative of synaptic strengthening (increased total spine density) and eventual maturation such as the increase in density of thin spines. Thin spines are highly dynamic (compared to mushrooms) and believed to represent “learning spines”, responsible for forming new memories [45]. These changes were also evident after examining spine surface area, neck diameter and head diameter. Greater surface area following reactivation of cocaine memory indicates the rapid expansion of spines following recall versus no memory reactivation. Reductions in spine neck and head diameter lend further support that the increase in spine density was driven by thin spines, whose characteristics include smaller head and neck diameter compared to other spine types. Prior literature on dendritic spines and cocaine in rodent models describes cocaine-induced changes in thin spines. For example, an increase in the number of thin spines, without changes in other spine subtypes, was found in the medial prefrontal cortex of rats following 7 days of forced abstinence from cocaine vs saline iv self-administration [46]. Interestingly, in a study assessing dendritic spine density of D1R- and D2R-expressing MSNs, increased spine density was observed in both MSNs populations when measured 2 days after chronic cocaine administration, while the increased spine density was maintained only in D1R-expressing MSNs 30 days after the last cocaine injection [46, 47]. In contrast to prior reports [46,47,48], our study measured dendritic plasticity induced by reactivation of cocaine memories, not by cocaine itself. Both experimental groups of mice had identical cocaine exposure and differed only by cocaine memory reactivation achieved by re-exposure to the cocaine-paired context or no memory reactivation. Thus, reactivation of cocaine memories induced changes in spine morphology. In the current study, spine density was not evaluated in phenotypically defined MSNs, so it remains unknown if recall of cocaine memories produced dendritic changes in D1R- and/or D2R-expressing MSNs.
Results presented here demonstrate that cocaine memory reconsolidation depends on NAc neuronal activation. The coupling of NAc neuronal activity and MSNs dendritic plasticity suggests increases in afferent connections to these neurons. To identify other components of the cocaine memory engram that may synapse on NAc neurons, the role of vHPC projections onto NAc neurons was investigated. This is the first study to demonstrate that activation of the vHPC to NAc projections is necessary for the reconsolidation of cocaine contextual memories. The vHPC sends excitatory glutamatergic projections to the NAc [49]. This excitatory projection has been implicated in encoding contextual information of addictive behaviors [50]. Cue-induced recall of cocaine memory depends on specific projections from the vHPC to the NAc (i.e., ventral CA1→NAcore) [17], and inhibition of vHPC neurons with a GABA receptor agonist attenuates reinstatement to cocaine seeking following a presentation of either cues or a cocaine priming injection in a rat cocaine self-administration procedure [51]. The vHPC also mediates associative processing of cues during classical fear conditioning [52].
Prior literature described the engram that facilitates initial consolidation of cocaine CPP as being comprised of the ventral CA1 region of the hippocampus and the NAc core in mice [53]. In that study, chemogenetic silencing of either NAc core or vCA1 engram cells prior to the test for cocaine place preference impaired the expression of a cocaine CPP, suggesting these cells are involved in either the storage and/or retrieval of the CPP memory [53]. Their study differed from ours in several ways. Most importantly for our focus on reconsolidation, we administered CNO after, not before, memory reactivation, and CPP expression was tested 3 days later without further CNO administration. Chemogenetic inhibition of vHPC neurons projecting specifically to the NAc after cocaine memory recall interfered with reconsolidation as evidenced by abolishment of the established cocaine place preference. Dendrites of activated pyramidal neurons in the vHPC underwent neuroplastic adaptations following reactivation of cocaine contextual memories. Changes in the density and morphology of dendritic spines reflect altered synaptic strength, particularly of excitatory synapses. Increases in dendritic spine density were noted and, in contrast to the nucleus accumbens where thin spines were most upregulated, the densities of all spine types were increased in vHPC pyramidal neurons after cocaine memory recall, resulting in overall increases in spine surface area and volume.
Our prior studies using a similar mouse CPP model of cocaine contextual memory processes identified a cellular signaling pathway that is critical for the reconsolidation of cocaine memories. Recall of cocaine contextual memories resulted in activation of glycogen synthase kinase 3β (GSK3β) and its downstream effector, mTORC1, and inhibition of either was sufficient to block cocaine memory reconsolidation [21, 23]. Further investigation demonstrated that NMDA receptor activation, specifically GluN2A/B subtypes, is upstream of the activation of GSK3β in the NAc and hippocampus, and activation of these NMDA receptors is required for cocaine memory reconsolidation [22]. Thus, the current findings and our previous work support the hypothesis that cocaine memory reconsolidation involves activation of vHPC glutamatergic projections to the NAc, the resulting stimulation of MSNs NMDA receptors, and GSK3β - mTORC1 signaling to produce changes in protein synthesis underlying long-term memory [54]. The literature supports glutamatergic synapses as key cellular sites where cocaine experiences create memory traces that promote cocaine craving and seeking, and these synapses can be generated in the NAc following cocaine experience [55].
The current investigation did not identify the target(s) of NAc MSNs involved in cocaine memory reconsolidation. The MSNs of the NAc project to several brain areas including the ventral pallidum, the mesencephalon including the ventral tegmental area, and the hypothalamus [56]. Other work demonstrated that D1 receptor-expressing MSNs collateralize to both the ventral pallidum and the ventral mesencephalon. It was further demonstrated that activation of the MSNs specifically innervating the ventral pallidum was necessary for cue-induced cocaine reinstatement in rats [57]. Thus, the ventral pallidum may be a target of the MSNs that mediate reconsolidation of cocaine memories. Further studies are needed to confirm this proposed circuit.
Conclusions
Cue-induced relapse is a major barrier for successful remission in patients battling cocaine use disorder, and there are no FDA approved medications to treat cocaine use disorder [58,59,60,61]. Targeting the reconsolidation of cocaine contextual memories may be an important strategy to help prevent relapse and reduce drug seeking behaviors. Future directions include identifying the output pathway from the NAc involved in the reconsolidation of cocaine contextual memories, as well characterizing possible sex differences in the circuitry responsible for the reconsolidation of cocaine contextual memories.
Data availability
Data and protocols are available on request from the corresponding author (ellen.unterwald@temple.edu).
References
Miller CA, Marshall JF. Molecular substrates for retrieval and reconsolidation of cocaine-associated contextual memory. Neuron. 2005;47:873–84.
Ehrman RN, Robbins SJ, Childress AR, O’Brien CP. Conditioned responses to cocaine-related stimuli in cocaine abuse patients. Psychopharmacology (Berl). 1992;107:523–9.
Mactutus CF, Riccio DC, Ferek JM. Retrograde amnesia for old (reactivated) memory: some anomalous characteristics. Science. 1979;204:1319–20.
Przybyslawski J, Sara SJ. Reconsolidation of memory after its reactivation. Behav Brain Res. 1997;84:241–6.
Sorg BA. Reconsolidation of drug memories. Neurosci Biobehav Rev. 2012;36:1400–17.
Reichelt AC, Lee JL. Memory reconsolidation in aversive and appetitive settings. Front Behav Neurosci. 2013;7:118.
Saladin ME, Gray KM, McRae-Clark AL, Larowe SD, Yeatts SD, Baker NL, et al. A double blind, placebo-controlled study of the effects of post-retrieval propranolol on reconsolidation of memory for craving and cue reactivity in cocaine dependent humans. Psychopharmacology (Berl). 2013;226:721–37.
Lonergan M, Saumier D, Tremblay J, Kieffer B, Brown TG, Brunet A. Reactivating addiction-related memories under propranolol to reduce craving: A pilot randomized controlled trial. J Behav Ther Exp Psychiatry. 2016;50:245–9.
Rich MT, Torregrossa MM. Molecular and synaptic mechanisms regulating drug-associated memories: towards a bidirectional treatment strategy. Brain Res Bull. 2018;141:58–71.
Germeroth LJ, Carpenter MJ, Baker NL, Froeliger B, LaRowe SD, Saladin ME. Effect of a brief memory updating intervention on smoking behavior: a randomized clinical trial. JAMA psychiatry. 2017;74:214–23.
Milton AL, Everitt BJ. The persistence of maladaptive memory: addiction, drug memories and anti-relapse treatments. Neurosci Biobehav Rev. 2012;36:1119–39.
Pachas GN, Gilman J, Orr SP, Hoeppner B, Carlini SV, Grasser EB, et al. Single dose propranolol does not affect physiologic or emotional reactivity to smoking cues. Psychopharmacology (Berl). 2015;232:1619–28.
Xue YX, Luo YX, Wu P, Shi HS, Xue LF, Chen C, et al. A memory retrieval-extinction procedure to prevent drug craving and relapse. Science. 2012;336:241–5.
Chia C, Otto T. Hippocampal Arc (Arg3.1) expression is induced by memory recall and required for memory reconsolidation in trace fear conditioning. Neurobiol Learn Mem. 2013;106:48–55.
Pitkänen A, Pikkarainen M, Nurminen N, Ylinen A. Reciprocal connections between the amygdala and the hippocampal formation, perirhinal cortex, and postrhinal cortex in rat. A review. Ann N. Y Acad Sci. 2000;911:369–91.
Tao S, Wang Y, Peng J, Zhao Y, He X, Yu X, et al. Whole-brain mapping the direct inputs of dorsal and ventral CA1 projection neurons. Front Neural Circuits. 2021;15:643230.
Zhou Y, Yan E, Cheng D, Zhu H, Liu Z, Chen X, et al. The projection from ventral CA1, not prefrontal cortex, to nucleus accumbens core mediates recent memory retrieval of cocaine-conditioned place preference. Front Behav Neurosci. 2020;14:558074.
Shen Q, Xie B, Galaj E, Yu H, Li X, Lu Y, et al. CircTmeff-1 in the nucleus accumbens regulates the reconsolidation of cocaine-associated memory. Brain Res Bull. 2022;185:64–73.
Wright WJ, Graziane NM, Neumann PA, Hamilton PJ, Cates HM, Fuerst L, et al. Silent synapses dictate cocaine memory destabilization and reconsolidation. Nat Neurosci. 2020;23:32–46.
Liang J, Li JL, Han Y, Luo YX, Xue YX, Zhang Y, et al. Calpain-GRIP signaling in nucleus accumbens core mediates the reconsolidation of drug reward memory. J Neurosci. 2017;37:8938–51.
Shi X, von Weltin E, Fitzsimmons E, Do C, Caban Rivera C, Chen C, et al. Reactivation of cocaine contextual memory engages mechanistic target of rapamycin/S6 kinase 1 signaling. Front Pharm. 2022;13:976932.
Shi X, von Weltin E, Barr JL, Unterwald EM. Activation of GSK3β induced by recall of cocaine reward memories is dependent on GluN2A/B NMDA receptor signaling. J Neurochem. 2019;151:91–102.
Shi X, Miller JS, Harper LJ, Poole RL, Gould TJ, Unterwald EM. Reactivation of cocaine reward memory engages the Akt/GSK3/mTOR signaling pathway and can be disrupted by GSK3 inhibition. Psychopharmacology (Berl). 2014;231:3109–18.
Guenthner CJ, Miyamichi K, Yang HH, Heller HC, Luo L. Permanent genetic access to transiently active neurons via TRAP: targeted recombination in active populations. Neuron. 2013;78:773–84.
Barr JL, Brailoiu GC, Abood ME, Rawls SM, Unterwald EM, Brailoiu E. Acute cocaine administration alters permeability of blood-brain barrier in freely-moving rats- Evidence using miniaturized fluorescence microscopy. Drug Alcohol Depend. 2020;206:107637.
Werner CT, Mitra S, Auerbach BD, Wang ZJ, Martin JA, Stewart AF, et al. Neuroadaptations in the dorsal hippocampus underlie cocaine seeking during prolonged abstinence. Proc Natl Acad Sci USA. 2020;117:26460–9.
Pedreira ME, Pérez-Cuesta LM, Maldonado H. Reactivation and reconsolidation of long-term memory in the crab Chasmagnathus: protein synthesis requirement and mediation by NMDA-type glutamatergic receptors. J Neurosci. 2002;22:8305–11.
Nader K, Hardt O. A single standard for memory: the case for reconsolidation. Nat Rev Neurosci. 2009;10:224–34.
Lee JLC, Nader K, Schiller D. An update on memory reconsolidation updating. Trends Cogn Sci. 2017;21:531–45.
Cazzulino AS, Martinez R, Tomm NK, Denny CA. Improved specificity of hippocampal memory trace labeling. Hippocampus. 2016;26:752–62.
Festa LK, Irollo E, Platt BJ, Tian Y, Floresco S, Meucci O. CXCL12-induced rescue of cortical dendritic spines and cognitive flexibility. eLife. 2020;9:e49717.
Paxinos G, and Keith BJ Franklin. The mouse brain in stereotaxic coordinates: hard cover edition2001.
Dickstein DL, Dickstein DR, Janssen WGM, Hof PR, Glaser JR, Rodriguez A, et al. Automatic dendritic spine quantification from confocal data with neurolucida 360. Curr Protoc Neurosci. 2016;77:1 27 1–1.27.21.
Rodriguez A, Ehlenberger D, Dickstein D, Hof P, Wearne S. Automated three-dimensional detection and shape classification of dendritic spines from fluorescence microscopy images. PLoS ONE. 2008;3:e1997.
Shi H-S, Luo Y-X, Yin X, Wu H-H, Xue G, Geng X-H, et al. Reconsolidation of a cocaine associated memory requires DNA methyltransferase activity in the basolateral amygdala. Sci Rep. 2015;5:13327.
Stewart J. Reinstatement of heroin and cocaine self-administration behavior in the rat by intracerebral application of morphine in the ventral tegmental area. Pharmacol Biochem Behav. 1984;20:917–23.
Childress AR, Mozley PD, McElgin W, Fitzgerald J, Reivich M, O’Brien CP. Limbic activation during cue-induced cocaine craving. American J Psychiatry. 1999;156:11–8.
Arroyo M, Markou A, Robbins TW, Everitt BJ. Acquisition, maintenance and reinstatement of intravenous cocaine self-administration under a second-order schedule of reinforcement in rats: effects of conditioned cues and continuous access to cocaine. Psychopharmacology (Berl). 1998;140:331–44.
Davis WM, Smith SG, Werner TE. Intragastric alcohol: effects of unit dosage on self-administration and on conditioned reinforcement. Proc West Pharm Soc. 1976;19:346–50.
Nader K, Schafe GE, Le Doux JE. Fear memories require protein synthesis in the amygdala for reconsolidation after retrieval. Nature. 2000;406:722–6.
Duvarci S, Nader K. Characterization of fear memory reconsolidation. J Neurosci. 2004;24:9269–75.
Qi S, Tan SM, Wang R, Higginbotham JA, Ritchie JL, Ibarra CK, et al. Optogenetic inhibition of the dorsal hippocampus CA3 region during early-stage cocaine-memory reconsolidation disrupts subsequent context-induced cocaine seeking in rats. Neuropsychopharmacology. 2022;47:1473–83.
Alberini CM, LeDoux JE. Memory reconsolidation. Curr Biol. 2013;23:R746–R50.
Nader K. Reconsolidation and the dynamic nature of memory. Cold Spring Harb Perspect Biol. 2015;7:a021782.
Pchitskaya E, Bezprozvanny I. Dendritic spines shape analysis—classification or clusterization? perspective. Front Synaptic Neurosci. 2020;12:31.
Rasakham K, Schmidt HD, Kay K, Huizenga MN, Calcagno N, Pierce RC, et al. Synapse density and dendritic complexity are reduced in the prefrontal cortex following seven days of forced abstinence from cocaine self-administration. PLoS ONE. 2014;9:e102524.
Lee K-W, Kim Y, Kim AM, Helmin K, Nairn AC, Greengard P. Cocaine-induced dendritic spine formation in D1 and D2 dopamine receptor-containing medium spiny neurons in nucleus accumbens. Proceedings Natl Acad Sci. 2006;103:3399–404.
Anderson EM, Wissman AM, Chemplanikal J, Buzin N, Guzman D, Larson EB, et al. BDNF-TrkB controls cocaine-induced dendritic spines in rodent nucleus accumbens dissociated from increases in addictive behaviors. Proceedings Natl Acad Sci. 2017;114:9469–74.
Floresco SB, Todd CL, Grace AA. Glutamatergic afferents from the hippocampus to the nucleus accumbens regulate activity of ventral tegmental area dopamine neurons. J Neurosci. 2001;21:4915–22.
Barrientos C, Knowland D, Wu MMJ, Lilascharoen V, Huang KW, Malenka RC, et al. Cocaine-induced structural plasticity in input regions to distinct cell types in nucleus accumbens. Biol Psychiatry. 2018;84:893–904.
Rogers JL, See RE. Selective inactivation of the ventral hippocampus attenuates cue-induced and cocaine-primed reinstatement of drug-seeking in rats. Neurobiol Learn Mem. 2007;87:688–92.
Zhang WN, Bast T, Feldon J. The ventral hippocampus and fear conditioning in rats: different anterograde amnesias of fear after infusion of N-methyl-D-aspartate or its noncompetitive antagonist MK-801 into the ventral hippocampus. Behav Brain Res. 2001;126:159–74.
Zhou Y, Zhu H, Liu Z, Chen X, Su X, Ma C, et al. A ventral CA1 to nucleus accumbens core engram circuit mediates conditioned place preference for cocaine. Nat Neurosci. 2019;22:1986–99.
Santini E, Huynh TN, Klann E. Mechanisms of translation control underlying long-lasting synaptic plasticity and the consolidation of long-term memory. Prog Mol Biol Transl Sci. 2014;122:131–67.
Wright WJ, Dong Y. Silent synapses in cocaine-associated memory and beyond. J Neurosci. 2021;41:9275–85.
Scofield MD, Heinsbroek JA, Gipson CD, Kupchik YM, Spencer S, Smith AC, et al. The nucleus accumbens: mechanisms of addiction across drug classes reflect the importance of glutamate homeostasis. Pharmacol Rev. 2016;68:816–71.
Pardo-Garcia TR, Garcia-Keller C, Penaloza T, Richie CT, Pickel J, Hope BT, et al. Ventral pallidum is the primary target for accumbens D1 projections driving cocaine seeking. J Neurosci. 2019;39:2041–51.
Johnson BA, Ait-Daoud N, Wang X-Q, Penberthy JK, Javors MA, Seneviratne C, et al. Topiramate for the treatment of cocaine addiction: a randomized clinical trial. JAMA Psychiatry. 2013;70:1338–46.
Rohsenow DJ, Niaura RS, Childress AR, Abrams DB, Monti PM. Cue reactivity in addictive behaviors: theoretical and treatment implications. Int J Addict. 1990;25:957–93.
Sinha R. The clinical neurobiology of drug craving. Curr Opin Neurobiol. 2013;23:649–54.
Yahyavi-Firouz-Abadi N, See RE. Anti-relapse medications: preclinical models for drug addiction treatment. Pharmacol Ther. 2009;124:235–47.
Acknowledgements
We thank Joseph Meissler for managing the mouse breeding colony that generated the TRAP2XAi14 mice for this study, Chongguang Chen for his assistance in setting up imaging protocols, and Thomas Campion for viral vector preparation. We also thank Danielle Stern and Daniel Lopez for their help with figure preparation.
Funding
This work was supported by the National Institutes of Health grants P30DA013429 (to EMU), R01DA043988 (to EMU), and T32DA007237 (to EMU/CCR). The Shriners Neural Repair Viral Core is supported by Shriners grant #84051-PHI-21 to GM Smith.
Author information
Authors and Affiliations
Contributions
Ellen Unterwald (EU) was the principal investigator, obtained funding, designed experiments, supervised the studies, reviewed data and statistical analyses, and contributed to the writing and editing of the manuscript. CCR, RP, RPF, CL, CD, JS, MG, and XS performed experiments and data analysis. LK supervised electrophysiology studies. GS designed the vectors and supervised their synthesis. CCR created the first draft of the manuscript. The figures were generated by CCR, RP, RPF, CL, and EU. All authors discussed the results and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have nothing to disclose. There are no competing financial interests in relation to the work described.
Regulations & approvals
Animal procedures were performed in compliance with the National Institutes of Health guidelines for the Care and Use of Laboratory Animals. Animal use was reviewed and approved by Temple University Institutional Animal Care and Use Committee (Animal Care and Use Protocol #4915).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Caban Rivera, C., Price, R., Petrilli Fortuna, R. et al. The ventral hippocampus and nucleus accumbens as neural substrates for cocaine contextual memory reconsolidation. Transl Psychiatry 16, 2 (2026). https://doi.org/10.1038/s41398-025-03734-4
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-025-03734-4