Abstract
We report the results of a Phase I radiation dose escalation study using an yttrium-90 (90Y) labelled anti-CD66 monoclonal antibody given with standard conditioning regimen for patients receiving haematopoietic stem cell transplants for myeloid leukaemia or myeloma. The 90Y-labelled anti-CD66 was infused prior to standard conditioning. In total, 30 patients entered the trial and 29 received 90Y-labelled mAb, at infused radiation activity levels of 5, 10, 25, or 37.5 megaBequerel (MBq)/kg lean body weight. A prerequisite for receiving the 90Y-labelled mAb was favourable dosimetry determined by single-photon emission computerised tomography (SPECT) dosimetry following administration of indium-111 (111In) anti-CD66. Estimated absorbed radiation doses delivered to the red marrow demonstrated a linear relationship with the infused activity of 90Y-labelled mAb. At the highest activity level of 37.5 MBq/kg, mean estimated radiation doses for red marrow, liver, spleen, kidneys and lungs were 24.6 ± 5.6 Gy, 5.8 ± 2.7 Gy, 19.1 ± 8.0 Gy, 2.1 ± 1.1 and 2.2 ± 0.9, respectively. All patients engrafted, treatment-related mortality 1-year post-transplant was zero. Toxicities were no greater than those anticipated for similar conditioning regimens without targeted radiation. The ability to substantially intensify conditioning prior to haematopoietic stem cell transplantation without increasing toxicity warrants further testing to determine efficacy. clinicaltrials.gov identifier: NCT01521611.
Similar content being viewed by others
Introduction
Autologous or allogeneic haematopoietic stem cell transplantation (HSCT) can improve outcomes for a wide range of haematological malignancies, however, the risks of treatment toxicity must be balanced against the risk of disease recurrence. In the allogeneic setting, total body irradiation (TBI) has been shown to reduce disease recurrence in acute myeloid leukaemia (AML) and chronic myeloid leukaemia (CML) in a dose-dependent manner [1,2,3]. The reduced relapse rate was, however, countered by corresponding increases in transplant-related mortality (TRM) at higher doses of radiation. Additionally, the doses of TBI used in full-intensity allogeneic transplantation protocols have excessive toxicity for older patients. Reduced-intensity conditioning protocols using lower radiation doses or avoiding TBI allow the extension of allogeneic transplantation to older patients and those with significant co-morbid conditions. The reduction of conditioning intensity results in lower TRMs, allowing engraftment and stable high donor chimerism but has an increased risk of disease recurrence, shown by several retrospective studies [4,5,6,7,8]. Essentially, the risks from the toxicity of the conditioning regimen are exchanged for increased risk of relapse resulting in similar overall survivals (OS) [9]. This has been confirmed in a large retrospective analysis of transplant outcomes [10] although randomised prospective trials have shown conflicting results, possibly due to differences in the details of conditioning used and the age limits applied [11,12,13,14]. A long-term follow-up study of a randomised trial showed a lower relapse risk in patients that received myeloablative conditioning while another long-term analysis showed no differences in TRM, relapse or OS [12, 13]. However, in patients aged 41–60 years, there was a significantly higher TRM with the higher TBI dose impacting on OS in these patients [12].
In the treatment of myeloma, the dose–response relationship led to the development of high-dose therapy and autologous stem cell transplantation, shown by several randomised trials to be of more benefit than chemotherapy alone and remains the recommended standard treatment [15,16,17,18,19,20]. Further dose escalation is limited by toxicity to non-haematopoietic organs. The use of tandem autologous transplantation allows dose intensification by performing two procedures temporally close, shown in some trials to result in improved outcomes [21,22,23] but at the expense of increased toxicity [24]. The addition of TBI to high-dose melphalan has been tested but was associated with increased TRM but no improvement in response rates or OS [25, 26].
An alternative to TBI that may reduce the incidence of complications while maintaining therapeutic intensity is targeted molecular radiotherapy (MRT) where radiation is targeted to antigens present on haematopoietic cells such as CD45 [27,28,29,30,31], CD33 [32, 33] and CD66 [34,35,36], principally using monoclonal antibodies as vector [37]. Several radioisotopes have been used clinically, mainly the beta particle emitting isotopes iodine-131 (131I), rhenium-188 (188Re) and 90Y [32] but also alpha-emitting isotopes such as bismuth-213 (213Bi) or astatine-211 (211As) [38], each radionuclide having advantages and disadvantages in the setting of HSCT. Although therapeutic doses of radiation can be delivered to haematopoietic tissues, a potential problem with the use of MRT has been the variable uptake and retention of the radiolabelled agent by non-target organs, particularly liver and kidneys, causing unwanted toxicities such as renal impairment and hepatic toxicity necessitating adjustment of the infused activity [39, 40], impacting on the potential benefit of the targeted radiation. The reasons for uptake by non-haematopoietic tissues are complex involving specific and non-specific binding or instability of the antibody-radioisotope construct in vivo. The variation of absorbed radiation in red marrow also complicates the assessment of disease response. Identifying optimal combinations of vectors and radioisotopes would allow the maximum potential benefit of targeted therapy to be achieved in all patients.
The cell surface antigen CD66 is an ideal target for MRT with isoforms of CD66 a, b, c and e present on cells of myeloid origin from promyelocytes through to mature neutrophils [39] and are also expressed by leukaemic blasts in 40% of patients with AML and in 80–100% of patients with B cell acute lymphoblastic leukaemia [40]. The isoform CD66a is expressed on plasma cells in the majority of cases of myeloma [41].
We report the results of a Phase I radiation dose escalation study using an 90Y-anti-CD66 in patients undergoing autologous or allogeneic HSCT for myeloma or high-risk myeloid leukaemia.
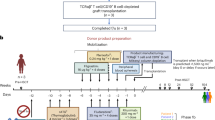
Study design, patients, materials and methods
This was an open label, non-randomised Phase I study with four levels of infused 90Y-anti-CD66 radioactivity. Patients initially received 111In-anti-CD66 to determine the biodistribution and to derive organ dosimetry from whole body and SPECT gamma-camera images. Five patients were treated at each activity level with a planned expansion of the highest activity level to 15 patients if no dose-limiting toxicity (DLT) was demonstrated. The study was approved by the UK National Research Ethics Committee, the Administration of Radioactive Substances Advisory Committee and the Medicines and Healthcare Products and Regulatory Authority in accordance with the Declaration of Helsinki. Patients were recruited from referrals to the Wessex Blood and Marrow Transplantation Program for autologous or allogeneic HSCT as treatment for myeloma, AML or CML. Recruitment was dependent on the availability of nuclear medicine scanning time. Patients with AML had a high risk of relapse based on the presence of adverse cytogenetics, CR > 1 or secondary AML and were ineligible for full intensity conditioning due to age or co-morbid conditions. Patients with CML either had poor response to tyrosine kinase inhibitors or were in an accelerated phase. Exclusion criteria included hypocellular bone marrow with <10% red marrow content. AML patients in relapse were permitted with marrow blasts <20% of nucleated cells and peripheral blood leucocytes <30 × 109/L.
Patients were informed of the investigational nature of the study and received verbal and written information before giving signed consent. The primary objective was to determine the safety profile of 90Y-anti-CD66 when included in standard transplant conditioning. Secondary objectives included determination of the maximum tolerated infused activity of 90Y-anti-CD66 as MBq/kg lean body weight, the radiation-related DLT in the context of standard autologous and allogeneic transplant conditioning schedules and development of a dosimetry model based on whole body and SPECT gamma imaging, blood clearance and thereby determine the pharmacokinetics of the radiolabelled antibody in vivo.
Antibody description and radiolabelling
The anti-CD66 mAb is a murine IgG1, specific for a common epitope on CD66 isoforms a, b, c, e [42], International Non-proprietary Name, Besilesomab. The antibody was produced according to Good Manufacturing Practice and provided in a purified form by TheraPharm GmbH (Zug, Switzerland). Conjugation to isothiocyanato-2-aminobenzyl-3-methyl-diethylenetriamine-pentaacetic acid (SCN-2B3M-DTPA) [43] was performed on bulk batches and stored frozen in single use vials. For dosimetry the anti-CD66 was radiolabelled with ∼185 MBq of 111In and for therapy 90Y, the activity required based on treatment cohort and lean body weight (details in Supplementary Information).
Dosimetry and pharmacokinetic studies
Patient-specific internal dosimetry was estimated using a methodology based on the Medical Internal Radiation Dose Committee and the EANM Dosimetry Committee [44, 45]. Cumulative activity in the blood was calculated from samples taken immediately at the end of infusion of 185 MBq 111In-anti-CD66 (T = 0) and at intervals up to day 7 post-infusion. Serial whole-body (WB) gamma-camera images were taken within 1 h of completion of infusion and at least three additional whole-body acquisitions between days 2 and 7 (details in Supplementary Information).
Therapy with 90Y-labelled anti-CD66
Patients with a minimum twofold ratio between the calculated radiation dose to red marrow compared with the next highest non-haematopoietic organ, in all cases the liver, received therapy with 90Y-anti-CD66, administered on D-14 of the transplant schedule. Patients were discharged 2 h post-infusion. Patients receiving autologous HSCT were admitted on D-2 for melphalan 200 mg/m2. Patients undergoing allogeneic HSCT were admitted on day 5 post-infusion to start standard conditioning with fludarabine, melphalan and CAMPATH 1H, transplant conditioning index 2.5 [46] (detailed in Supplementary Information). Imaging and therapy were delivered as planned day case episodes.
Patient characteristics
Thirty patients with myeloma, poor risk AML or CML were recruited into the study over a 5-year time period, mean age 54.4 years, range 20–68, 5 females and 25 males. Details of disease characteristics and number of prior therapies are summarised in Table 1. In patients with myeloma, disease stage was determined using the Durie–Salmon staging system which was in use at the time of this study [47].
Results
111In activity in whole blood
Blood 111In activity displayed similar biphasic retention in all patients, consistent with the sum of two exponential functions. In each patient, an initial rapid fall in blood 111In activity occurred within 2 h, followed by a slower decrease over 5–10 h (Fig. 1). After 24 h, the decrease in activity was markedly slower, approximating the physical half-life of the isotope and most of the radiation was located in the red marrow as indicated by whole-body planar gamma images (Fig. 2a) and SPECT(-CT) (Fig. 2b). From the early time-activity plots, the mean derived T½ alpha 2.06 ± 0.96 h (range 0.9–3.4), T½ beta 6.0 ± 3.2 h (range 4.0–9.0).
Mean blood 111In activity for each infused activity cohort in blood samples taken immediately at the end of infusion (T0) of 111In-anti-CD66 infusion and at timed intervals post-infusion. Initial blood 111In activity from T0 blood sample set as 100%; for subsequent samples, radioactivity expressed as a % of T0 activity, time in minutes post end of infusion.
a Whole-body gamma-camera image showing distribution of 111In-anti-CD66 monoclonal antibody. b Superimposed SPECT gamma radiation (coloured) and CT images (grey scale). (i) Cross-section showing strong gamma radiation signal in L3 vertebral body (yellow), low in liver and spleen (blue) and undetectable in muscle, bowel. (ii) Coronal section image showing strong gamma radiation signal in vertebrae and pelvis (yellow), low in liver and spleen (blue) and very low in kidneys (grey scale image of CT). (iii) Sagittal image showing high gamma signal (yellow) in vertebral bodies, sternum and virtually none in other organs.
Dosimetry
Thirty patients received 111In-anti-CD66. In the first cohort, one patient failed dosimetry with red marrow:liver ratio < 2:1 and was excluded from the study. Within the trial, 29 patients received 90Y-anti-CD66 at infused activity levels of 5, 10, 25 and 37.5 MBq 90Y per kilogram body weight, activity ranged from 229 to 2758 MBq, determined by patient weight and treatment cohort. In Table 2, the mean absorbed radiation dose to red marrow, spleen, liver, lungs and kidneys are expressed as milliGray per megaBequerel (mGy/MBq) of infused 90Y-anti-CD66 and the mean total organ doses are in Gy. The mean (±1 SD, range) estimated radiation dose delivered per unit infused radiation activity expressed as mGy/MBq were marrow 10.2 (±3.4, 6.5–16.2), liver 2.4 (±1.1, 1.4–5.6), spleen 9.0 (±3.9, 3.3–20.3), renal 0.6 (±0.4, 0.4–1.2), lungs 0.9 (±0.5, 0.5–1.8) and whole body 0.4 (±0.1, 0.2–0.6). The uptake of radiolabelled antibody by red marrow showed a high degree of consistency between patients. The mean red marrow to liver ratio was 4.3:1, estimated radiation doses to lung, kidneys, muscle and gut were considerably lower than to red marrow, shown graphically in Fig. 3. In most patients, the distribution of radiation was similar with marked uptake in the axial skeleton, ribs, sternum, pelvis, base of skull and proximal ends of the femur and humerus (Fig. 2a), consistent with red marrow distribution in adults.
The mean organ radiation dose determined from gamma-images for each of the four infused activity cohorts. Bars represent 1 standard deviation from the mean.
There was a linear relationship between the infused activity of 90Y in MBq and the absorbed dose to the bone marrow in Gy over all dose levels (Fig. 4) indicating that the uptake of labelled anti-CD66 mAb was consistent between patients for any given infused activity cohort. The targeted radiation remained localised within red marrow over the period of imaging. Additionally, red marrow time/activity curves for each patient showed a similar effective half-life (Teff) of 48.14 ± 12.1 h that was slightly shorter than the physical t½ for 111In (67 h) indicating good retention in the target organ (Fig. 5).
The graph shows the calculated radiation dose in Gray (Gy) to the bone marrow plotted against the infused 90Y-anti-CD66 activity for each individual patient. For the 90Y-anti-CD66 activity levels used, the coefficient of determination, R2, was 0.76 indicating a close fit to a linear relationship between infused activity and bone marrow radiation dose. R2 is calculated using the Microsoft (MS) Excel software.
Curve fitting achieved with the MS Excel software with a single exponential fit using gamma-camera-derived data. Lumbar vertebral body red marrow: Teff = 48.14 ± 12.1 h. Liver: Teff = 59.7 ± 12.9 h.
Engraftment
Neutrophil recovery reached >0.5 × 109/L at a median of 15 days (range 9–55) and platelet recovery >50 × 109/L median 15 days (range 10–119) post-transplant. All patients had marrow aspirate and trephine biopsy performed at approximately day 100 post-transplantation, tri-lineage engraftment was seen in all cases. Importantly, no late graft failures occurred. Chimerism analysis in recipients of allogeneic transplants showed >97% donors in unselected peripheral blood cells by day 30 post-transplant in all patients.
Safety profile
No immediate adverse effects were seen during the infusion of either 111In or 90Y-anti-CD66. Following 111In-anti-CD66 infusion, there were no changes in peripheral blood counts. Post 90Y-anti-CD66 infusion, all patients experienced grades 1–4 haematological toxicity, as indicated by a fall in peripheral blood counts, from days 8 to 12 post infusion. There was an increasing rate and depth of decline in counts with increasing total activity of 90Y-anti-CD66 infused. One patient experienced transient grade 1 gut toxicity with diarrhoea lasting 24 h post 90Y-anti-CD66 infusion, and another experienced transient grade 2 infection due to bacteraemia related to routine flushing of the Hickman line. The remaining patients experienced no toxicities, other than haematological, from the time of infusion up to the start of the conventional transplant conditioning. All episodes of grades 1–4 (WHO Toxicity Criteria) [48] nausea and vomiting were related to the infusion of high-dose melphalan, the severity recorded was no different to that seen in patients who had not received radiolabelled antibody (Table 3).
Organ toxicities recorded following completion of standard conditioning therapy and post-stem cell transplantation are summarised in Table 3 using WHO Toxicity Criteria. As predicted for patients receiving conditioning therapy for HSCT, all patients experienced grade ≥ 3 haematological toxicity. Gastrointestinal toxicity was comparable to that caused by conventional transplant conditioning, particularly that caused by high-dose melphalan. There was a trend for more patients to experience grade ≥ 3 oral mucositis at the higher radiation dose levels. The severity was within that expected for high-dose therapy alone, but because of the trend, this was considered to represent the DLT when used in conjunction with the dose of melphalan of 200 mg/m2. In patients undergoing allogeneic HSCT, the lower dose of melphalan, 140 mg/m2, caused less generalised mucositis and no clear DLT was seen. The use of enteral or parenteral nutrition was similar to that required in patients undergoing standard transplant conditioning without MRT. Two patients developed HAMA post autologous HSCT.
Transplant-related mortality was 0 at days 30, 100, 180 and 1-year post-transplant for autologous and allogeneic transplant recipients, an important indicator of regimen-related toxicity. For patients with myeloma undergoing autologous HSCT, 13 of 15 patients relapsed or experienced disease progression, mean time to progression 41 months (range 1.5– 245 months); one patient remains in long-term complete remissions (CR) 20 years post-transplant and another proceeded to receive a sibling allogeneic transplant. All 14 patients who received allogeneic HSCT achieved CR with donor chimerism of >97% at D + 30. There were no cases of grade ≥ 3 acute graft versus host disease (GvHD) and no cases of chronic GvHD. Outcomes are detailed in Table 4.
Discussion
In this study, we demonstrated that 90Y-anti-CD66 consistently delivered radiation to red marrow and spleen with minimal uptake by non-haematopoietic organs. The excellent biodistribution of the radiolabelled antibody allowed the use of a simple method based on lean body weight to determine the total activity of 90Y for infusion. It should be noted that patients were in remission or very good partial remission at the time of imaging and therapy which would have contributed to the consistent BM targeting as the target antigen CD66 is predominantly expressed on normal myeloid cells. Radiolabelling episodes were reproducible and of high efficiency, both 111In and 90Y routinely achieving >95% radiochemical purity (RCP) contributing to the observed excellent inter-patient biodistribution and red marrow dosimetry. Similar excellent RCP results have been obtained with besilesomab conjugated to other backbone-substituted derivatives of DTPA such as MX-DTPA [35] and CHXA”-DTPA [49]. At the highest infused activity level of 37.5 MBq/kg, the mean estimated radiation dose to red marrow was 24.5 ± 5.6 Gy, 19.1 ± 8.0 Gy to spleen, 5.8 ± 2.7 Gy to liver, 2.1 ± 0.8 Gy to kidneys, and 2.2 ± 0.9 Gy to lungs with a whole-body dose of 0.9 ± 0.2 Gy. The use of 90Y, which emits a high energy (2.3 MeV) beta particle without gamma-emission, allowed patients to receive therapy as outpatients, hospital admission was required only for the completion of standard conditioning. In the period between 90Y-anti-CD66 therapy and the start of standard conditioning, there were no serious adverse events attributed to the radiolabelled antibody. Overall toxicities for the transplant period were within the limits anticipated for allogeneic transplantation using fludarabine and melphalan 140 mg/m2 or for autologous transplantation with melphalan 200 mg/m2. Mucosal toxicity was within anticipated limits with no severe or life-threatening events. However, in patients receiving 200 mg/m2 melphalan prior to autologous HSCT for myeloma, there was a trend to grade \(\ge\)3 dysphagia at the higher activity level as indicated by higher pain scores. Therefore, 37.5 MBq/kg LBW was selected as the activity for a subsequent randomised Phase IIb trial in myeloma autologous HSCT (manuscript in preparation). Although 13 of 15 patients that received autologous HSCT for myeloma experienced disease progression there was a trend for longer time to disease progression between the lowest activity level to the highest with a mean of 14.4 months in cohort 1 and 43.2 months in cohort 4.
Of the 14 patients receiving allogeneic transplants, all engrafted and achieved >97% donor chimerism at day 30 that was maintained. All achieved sustained CRs with ten patients remaining alive in CR, median follow-up of 220 months.
While not encountered in the study, it is possible that the marrow will be the dose-limiting organ because of potential damage to stromal cellular components, compromising stem cell survival. In a canine model where bone-seeking 166Ho-EDTMP was used to indirectly target the marrow, the DLT was reversible marrow fibrosis [50]. External beam radiation of 20 Gy or more was shown to cause marrow fibrosis in a rat femur model with >40 Gy causing permanent aplasia [51, 52]. In a clinical trial using an 131I-labelled anti-CD45, graft failure occurred in one patient who received a total estimated radiation dose of 42.7 Gy consisting of 12 Gy TBI and 30.7 Gy from MRT [28]. Based on this limited experience, it would seem prudent in future clinical studies to use an upper limit of radiation delivered by targeted radiotherapy to the marrow in the order of 45 Gy.
We were able to demonstrate a dose–response relationship between the infused radiation activity and the effect of this red marrow-targeted radiation on the peripheral blood counts over a 12-day period. In the first activity cohort (5 MBq/kg), two patients showed a modest fall in leucocytes. In contrast, at the highest activity level, all patients had significant falls in leucocytes with the majority of patients having zero neutrophils at the time of admission for standard chemotherapy, demonstrating that the targeted molecular radiotherapy was functioning as a form of ‘pre-conditioning’ rendering the patients aplastic prior to the start of standard conditioning.
The favourable biodistribution and delivery of high absorbed radiation doses targeted to red marrow and spleen with the radiolabelled anti-CD66 mAb added substantial therapy to standard conditioning without additional toxicity and warrants further investigation in Phase II trials, particularly in patients with a high risk of disease relapse post-transplantation.
Data availability
The data that support the findings of this study are available from Telix Pharmaceuticals, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are, however, available from the authors upon reasonable request and with permission of Telix Pharmaceuticals.
Change history
05 March 2026
The original online version of this article was revised: During the production process tab. 2 has been typeset wrongly.
04 March 2026
A Correction to this paper has been published: https://doi.org/10.1038/s41409-026-02810-7
References
Clift RA, Buckner CD, Appelbaum FR, Bearman SI, Petersen FB, Fisher LD, et al. Allogeneic marrow transplantatig1on in patients with acute myeloid leukemia in first remission: a randomized trial of two irradiation regimens. Blood. 1990;76:1867–71.
Clift RA, Buckner CD, Appelbaum FR, Bryant E, Bearman SI, Petersen FB, et al. Allogeneic marrow transplantation in patients with chronic myeloid leukemia in the chronic phase: a randomized trial of two irradiation regimens. Blood. 1991;77:1660–5.
Clift RA, Buckner CD, Appelbaum FR, Sullivan KM, Storb R, Thomas ED. Long-term follow-up of a randomized trial of two irradiation regimens for patients receiving allogeneic marrow transplants during first remission of acute myeloid leukemia. Blood. 1998;92:1455–6.
Alyea EP, Kim HT, Ho V, Cutler C, DeAngelo DJ, Stone R, et al. Impact of conditioning regimen intensity on outcome of allogeneic hematopoietic cell transplantation for advanced acute myelogenous leukemia and myelodysplastic syndrome. Biol Blood Marrow Transplant. 2006;12:1047–55.
Aoudjhane M, Labopin M, Gorin NC, Shimoni A, Ruutu T, Kolb HJ, et al. Comparative outcome of reduced intensity and myeloablative conditioning regimen in HLA identical sibling allogeneic haematopoietic stem cell transplantation for patients older than 50 years of age with acute myeloblastic leukaemia: a retrospective survey from the Acute Leukemia Working Party (ALWP) of the European group for Blood and Marrow Transplantation (EBMT). Leukemia. 2005;19:2304–12.
de Lima M, Anagnostopoulos A, Munsell M, Shahjahan M, Ueno N, Ippoliti C, et al. Nonablative versus reduced-intensity conditioning regimens in the treatment of acute myeloid leukemia and high-risk myelodysplastic syndrome: dose is relevant for long-term disease control after allogeneic hematopoietic stem cell transplantation. Blood. 2004;104:865–72.
Martino R, Iacobelli S, Brand R, Jansen T, van Biezen A, Finke J, et al. Retrospective comparison of reduced-intensity conditioning and conventional high-dose conditioning for allogeneic hematopoietic stem cell transplantation using HLA-identical sibling donors in myelodysplastic syndromes. Blood. 2006;108:836–46.
Abdul Wahid SF, Ismail NA, Mohd-Idris MR, Jamaluddin FW, Tumian N, Sze-Wei EY, et al. Comparison of reduced-intensity and myeloablative conditioning regimens for allogeneic hematopoietic stem cell transplantation in patients with acute myeloid leukemia and acute lymphoblastic leukemia: a meta-analysis. Stem Cells Dev. 2014;23:2535–52.
Pingali SR, Champlin RE. Pushing the envelope-nonmyeloablative and reduced intensity preparative regimens for allogeneic hematopoietic transplantation. Bone Marrow Transplant. 2015;50:1157–67.
Luger SM, Ringden O, Zhang MJ, Perez WS, Bishop MR, Bornhauser M, et al. Similar outcomes using myeloablative vs reduced-intensity allogeneic transplant preparative regimens for AML or MDS. Bone Marrow Transplant. 2012;47:203–11.
Bornhauser M, Kienast J, Trenschel R, Burchert A, Hegenbart U, Stadler M, et al. Reduced-intensity conditioning versus standard conditioning before allogeneic haemopoietic cell transplantation in patients with acute myeloid leukaemia in first complete remission: a prospective, open-label randomised phase 3 trial. Lancet Oncol. 2012;13:1035–44.
Fasslrinner F, Schetelig J, Burchert A, Kramer M, Trenschel R, Hegenbart U, et al. Long-term efficacy of reduced-intensity versus myeloablative conditioning before allogeneic haemopoietic cell transplantation in patients with acute myeloid leukaemia in first complete remission: retrospective follow-up of an open-label, randomised phase 3 trial. Lancet Haematol. 2018;5:e161–9.
Scott BL, Pasquini MC, Logan BR, Wu J, Devine SM, Porter DL, et al. Myeloablative versus reduced-intensity hematopoietic cell transplantation for acute myeloid leukemia and myelodysplastic syndromes. J Clin Oncol. 2017;35:1154–61.
Kröger N, Iacobelli S, Franke GN, Platzbecker U, Uddin R, Hübel K, et al. Dose-reduced versus standard conditioning followed by allogeneic stem-cell transplantation for patients with myelodysplastic syndrome: a prospective randomized phase III study of the EBMT (RICMAC Trial). J Clin Oncol. 2017;35:2157–64.
Child JA, Morgan GJ, Davies FE, Owen RG, Bell SE, Hawkins K, et al. High-dose chemotherapy with hematopoietic stem-cell rescue for multiple myeloma. N Engl J Med. 2003;348:1875–83.
Attal M, Harousseau JL, Stoppa AM, Sotto JJ, Fuzibet JG, Rossi JF, Intergroupe Francais du Myelome et al. A prospective, randomized trial of autologous bone marrow transplantation and chemotherapy in multiple myeloma. N Engl J Med. 1996;335:91–7.
Lenhoff S, Hjorth M, Holmberg E, Turesson I, Westin J, Nielsen JL, Nordic Myeloma Study Group et al. Impact on survival of high-dose therapy with autologous stem cell support in patients younger than 60 years with newly diagnosed multiple myeloma: a population-based study. Blood. 2000;95:7–11.
Bird JM, Owen RG, D’Sa S, Snowden JA, Pratt G, Ashcroft J, et al. Guidelines for the diagnosis and management of multiple myeloma 2011. Br J Haematol. 2011;154:32–75.
Shah N, Callander N, Ganguly S, Gul Z, Hamadani M, Costa L, et al. Hematopoietic stem cell transplantation for multiple myeloma: guidelines from the American Society for Blood and Marrow Transplantation. Biol Blood Marrow Transplant. 2015;21:1155–66.
Dimopoulos MA, Moreau P, Terpos E, Mateos MV, Zweegman S, Cook G, et al. Multiple myeloma: EHA-ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up†. Ann Oncol. 2021;32:309–22.
Barlogie B, Jagannath S, Desikan KR, Mattox S, Vesole D, Siegel D, et al. Total therapy with tandem transplants for newly diagnosed multiple myeloma. Blood. 1999;93:55–65.
Lahuerta JJ, Grande C, Martinez-Lopez J, De La Serna J, Toscano R, Ortiz MC, et al. Tandem transplants with different high-dose regimens improve the complete remission rates in multiple myeloma. Results of a Grupo Espanol de Sindromes Linfoproliferativos/Trasplante Autologo de Medula Osea phase II trial. Br J Haematol. 2003;120:296–303.
Attal M, Harousseau JL, Facon T, Guilhot F, Doyen C, Fuzibet JG, et al. Single versus double autologous stem-cell transplantation for multiple myeloma. N Engl J Med. 2003;349:2495–502.
Kumar A, Kharfan-Dabaja MA, Glasmacher A, Djulbegovic B. Tandem versus single autologous hematopoietic cell transplantation for the treatment of multiple myeloma: a systematic review and meta-analysis. J Natl Cancer Inst. 2009;101:100–6.
Moreau P, Facon T, Attal M, Hulin C, Michallet M, Maloisel F, et al. Comparison of 200 mg/m(2) melphalan and 8 Gy total body irradiation plus 140 mg/m(2) melphalan as conditioning regimens for peripheral blood stem cell transplantation in patients with newly diagnosed multiple myeloma: final analysis of the Intergroupe Francophone du Myélome 9502 randomized trial. Blood. 2002;99:731–5.
Abraham R, Chen C, Tsang R, Simpson D, Murray C, Davidson M, et al. Intensification of the stem cell transplant induction regimen results in increased treatment-related mortality without improved outcome in multiple myeloma. Bone Marrow Transplant. 1999;24:1291–7.
Matthews DC, Martin PJ, Nourigat C, Appelbaum FR, Fisher DR, Bernstein ID. Marrow ablative and immunosuppressive effects of 131I-anti-CD45 antibody in congenic and H2-mismatched murine transplant models. Blood. 1999;93:737–45.
Matthews DC, Appelbaum FR, Eary JF, Fisher DR, Durack LD, Bush SA, et al. Development of a marrow transplant regimen for acute leukemia using targeted hematopoietic irradiation delivered by 131I-labeled anti-CD45 antibody, combined with cyclophosphamide and total body irradiation. Blood. 1995;85:1122–31.
Matthews DC, Appelbaum FR, Eary JF, Fisher DR, Durack LD, Hui TE, et al. Phase I study of (131)I-anti-CD45 antibody plus cyclophosphamide and total body irradiation for advanced acute leukemia and myelodysplastic syndrome. Blood. 1999;94:1237–47.
Orozco JJ, Zeller J, Pagel JM. Radiolabeled antibodies directed at CD45 for conditioning prior to allogeneic transplantation in acute myeloid leukemia and myelodysplastic syndrome. Ther Adv Hematol. 2012;3:5–16.
Mawad R, Gooley TA, Rajendran JG, Fisher DR, Gopal AK, Shields AT, et al. Radiolabeled anti-CD45 antibody with reduced-intensity conditioning and allogeneic transplantation for younger patients with advanced acute myeloid leukemia or myelodysplastic syndrome. Biol Blood Marrow Transplant. 2014;20:1363–8.
Jurcic JG. Radioimmunotherapy for hematopoietic cell transplantation. Immunotherapy. 2013;5:383–94.
Burke JM, Caron PC, Papadopoulos EB, Divgi CR, Sgouros G, Panageas KS, et al. Cytoreduction with iodine-131-anti-CD33 antibodies before bone marrow transplantation for advanced myeloid leukemias. Bone Marrow Transplant. 2003;32:549–56.
Buchmann I, Bunjes D, Kotzerke J, Martin H, Glatting G, Seitz U, et al. Myeloablative radioimmunotherapy with Re-188-anti-CD66-antibody for conditioning of high-risk leukemia patients prior to stem cell transplantation: biodistribution, biokinetics and immediate toxicities. Cancer Biother Radiopharm. 2002;17:151–63.
Schulz AS, Glatting G, Hoenig M, Schuetz C, Gatz SA, Grewendorf S, et al. Radioimmunotherapy-based conditioning for hematopoietic cell transplantation in children with malignant and nonmalignant diseases. Blood. 2011;117:4642–50.
Zenz T, Glatting G, Schlenk RF, Buchmann I, Dohner H, Reske SN, et al. Targeted marrow irradiation with radioactively labeled anti-CD66 monoclonal antibody prior to allogeneic stem cell transplantation for patients with leukemia: results of a phase I-II study. Haematologica. 2006;91:285–6.
Ali AM, Dehdashti F, DiPersio JF, Cashen AF. Radioimmunotherapy-based conditioning for hematopoietic stem cell transplantation: another step forward. Blood Rev. 2016;30:389–99.
Chen Y, Kornblit B, Hamlin DK, Sale GE, Santos EB, Wilbur DS, et al. Durable donor engraftment after radioimmunotherapy using alpha-emitter astatine-211-labeled anti-CD45 antibody for conditioning in allogeneic hematopoietic cell transplantation. Blood. 2012;119:1130–8.
Stocks SC, Ruchaud-Sparagano MH, Kerr MA, Grunert F, Haslett C, Dransfield I. CD66: role in the regulation of neutrophil effector function. Eur J Immunol. 1996;26:2924–32.
Ratei R, Karawajew L, Schabath R, Ehrfeldt A, Grunert F, Ludwig WD. Differential expression of the carcinoembryonic antigen-related cell adhesion molecules panCD66, CD66a, CD66c and of sialyl-Lewis x (CD15s) on blast cells of acute leukemias. Int J Hematol. 2008;87:137–43.
Lee C, Guinn BA, Brooks SE, Richardson D, Orchard K. CD66a (CEACAM1) is the only CD66 variant expressed on the surface of plasma cells in multiple myeloma: a refined target for radiotherapy trials? Br J Haematol. 2010;149:795–6.
Bosslet K, Luben G, Schwarz A, Hundt E, Harthus HP, Seiler FR, et al. Immunohistochemical localization and molecular characteristics of three monoclonal antibody-defined epitopes detectable on carcinoembryonic antigen (CEA). Int J Cancer. 1985;36:75–84.
Quadri SMMH. A convenient synthesis of 2-p-aminobenzyl-3-methyl and 2-p-aminobenzyl-3-benzyl derivatives of diethylenetriaminepentaacetic acid (DTPA): carbon backbone modified chelating agents. Bio Med Chem Lett. 1992;2:1661–4.
Hindorf C, Glatting G, Chiesa C, Linden O, Flux G, Committee ED. EANM Dosimetry Committee guidelines for bone marrow and whole-body dosimetry. Eur J Nucl Med Mol Imaging. 2010;37:1238–50.
Loevinger R, Berman M. A formalism for calculation of absorbed dose from radionuclides. Phys Med Biol. 1968;13:205–17.
Spyridonidis A, Labopin M, Savani BN, Niittyvuopio R, Blaise D, Craddock C, et al. Redefining and measuring transplant conditioning intensity in current era: a study in acute myeloid leukemia patients. Bone Marrow Transplant. 2020;55:1114–25.
Durie BG, Salmon SE. A clinical staging system for multiple myeloma. Correlation of measured myeloma cell mass with presenting clinical features, response to treatment, and survival. Cancer. 1975;36:842–54.
WHO. WHO handbook for reporting results of cancer treatment. Neoplasma. 1980;20:37–46.
Winter G, Hamp-Goldstein C, Fischer G, Kletting P, Glatting G, Solbach C, et al. Optimization of radiolabeling of a [(90)Y]Y-anti-CD66-antibody for radioimmunotherapy before allogeneic hematopoietic cell transplantation. Cancers. 2023;15:3660.
Appelbaum FR, Brown PA, Sandmaier BM, Storb R, Fisher DR, Shulman HM, et al. Specific marrow ablation before marrow transplantation using an aminophosphonic acid conjugate 166Ho-EDTMP. Blood. 1992;80:1608–13.
Knospe WH, Blom J, Crosby WH. Regeneration of locally irradiated bone marrow: I. Dose dependent, long-term changes in the rat, with particular emphasis upon vascular and stromal reaction. Blood. 1966;28:398–415.
Knospe WH, Blom J, Crosby WH. Regeneration of locally irradiated bone marrow. II. Induction of regeneration in permanently aplastic medullary cavities. Blood. 1968;31:400–5.
Acknowledgements
Therapharm GmbH for providing the native anti-CD66 monoclonal antibody. Dr. Syed Quadri for providing the bifunctional chelator. Dr. Paul Borchardt for advice on conjugation chemistry and radiolabelling. Dr. Maria Tristram for contributions to the development of the dosimetry model. Mr. James Thom for preparing the radiolabelled products in the initial stages of the study. Mrs. Maureen Zivanovic for quality control and patient management in the early stages of the study. KO greatly appreciated the encouragement and support in the initial stages of the project from Professor John Goldman, Professor Richard Begent and Professor Grant Prentice. Grateful appreciation to the patients who participated in the trial and to the staff on the Leukaemia and Transplant Unit in University Hospital Southampton.
Funding
Wessex Cancer Trust; National Health Service Research and Development Grant; Experiment Cancer Medicine Centre (ECMC) core funding.
Author information
Authors and Affiliations
Contributions
KO led the development of the antibody radiolabelling procedures, produced the study protocol and applications for ethical and regulatory approvals; patient management during treatment and post-transplantation and preparation of paper. JL was responsible for the technical management of the radiolabelling procedure, prepared documents and had quality control oversight of the radiolabelled products during the trial; contributed to dosimetry analysis. MG refined the dosimetry model and had oversight of medical physics aspects of the study. GL performed dosimetry analysis. SM contributed to the dosimetry analysis. MC helped in the pre-clinical preparation and evaluation of the conjugated anti-CD66 and in the bulk manufacture of the conjugated Mab according to Good Manufacturing Practice. CZ performed the radiolabelling. DR was involved with patient care during their transplants and post-transplant periods. VL helped to design the study and contributed to the preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
KO has contributed to advisory boards for Gilead, Janssen, Takeda and on consultancy basis for Telix Pharmaceuticals. KO has IP in the targeted radiotherapy technology. JL has been employed on a consultancy basis by Telix Pharmaceuticals. VL has contributed to advisory boards for Immedica Pharma AG. The other authors have declared no conflicts of interest. The radiolabelled anti-CD66 technology was acquired by Telix Pharmaceuticals in 2020. All the work described in this report was completed before acquisition by Telix.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: During the production process tab. 2 has been typeset wrongly.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Orchard, K., Langford, J., Guy, M. et al. Efficient bone marrow irradiation and low uptake by non-haematological organs with an yttrium-90-anti-CD66 antibody prior to haematopoietic stem cell transplantation. Bone Marrow Transplant 59, 1247–1257 (2024). https://doi.org/10.1038/s41409-024-02317-z
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-024-02317-z
This article is cited by
-
The molecular blueprint of targeted radionuclide therapy
Nature Reviews Clinical Oncology (2025)