Abstract
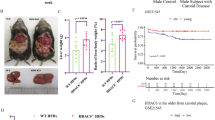
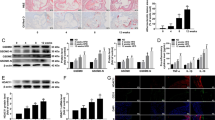
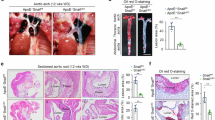
Histone deacetylase 3 (HDAC3) is an epigenetic modifying enzyme closely linked to the development of atherosclerosis. Endothelial inflammation is a critical factor in atherosclerosis. However, the role of HDAC3 in mediating epigenetic modifications and regulating endothelial inflammation in atherosclerosis remains unclear. This study aims to investigate the impact of HDAC3 on endothelial inflammation and its contribution to atherosclerosis. Firstly, single-cell transcriptomic analysis identified elevated expression of HDAC3 and nucleotide-binding oligomerization domain-like receptor protein 3 (NLRP3) in inflammatory endothelial cells of atherosclerotic plaques in symptomatic patients. Endothelial-specific knockout HDAC3 in an apolipoprotein E knockout (ApoE−/−) mice decreased atherosclerotic lesion by reducing lipid deposition and endothelial NLRP3 inflammasome activation compared with control mice. Consistently, experiments using HDAC3 inhibitor and overexpression in human umbilical vein endothelial cells (HUVECs) demonstrated that HDAC3 enhanced the transcriptional upregulation of NLRP3 inflammasome by promoting nuclear factor kappa-B pathway, thereby contributing to the activation of the NLRP3 inflammasome and cellular injury. Further studies revealed that HDAC3 reduced specificity protein 1 (SP1) Lys-703 acetylation, thereby enhancing SP1 binding to the NLRP3 promoter and promoting NLRP3 transcription. Additionally, pharmacological inhibition of HDAC3 effectively ameliorated atherosclerosis by reducing endothelial inflammation and increasing SP1 acetylation in ApoE−/− mice. Thus, these findings demonstrate a crucial role of HDAC3 in endothelial inflammation and shed light on potential therapeutic strategy for atherosclerosis via inhibition of HDAC3.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The datasets generated or analyzed during the current study are available in the human carotid atherosclerotic plaques GEO database with the link: https://www.ncbi.nlm.nih.gov/geo/. The computer code, proteomics data and other data employed to generate the results, is available from the corresponding author upon reasonable request. Uncropped immunoblots are provided as Supplemental Material-Original Blots.
References
Shvedunova M, Akhtar A. Modulation of cellular processes by histone and non-histone protein acetylation. Nat Rev Mol Cell Biol. 2022;23:329–49.
Emmett MJ, Lazar MA. Integrative regulation of physiology by histone deacetylase 3. Nat Rev Mol Cell Biol. 2019;20:102–15.
Hoeksema MA, Gijbels MJ, Van den Bossche J, van der Velden S, Sijm A, Neele AE, et al. Targeting macrophage Histone deacetylase 3 stabilizes atherosclerotic lesions. EMBO Mol Med. 2014;6:1124–32.
Li X, Chen M, Chen X, He X, Li X, Wei H, et al. TRAP1 drives smooth muscle cell senescence and promotes atherosclerosis via HDAC3-primed histone H4 lysine 12 lactylation. Eur Heart J. 2024;45:4219–35.
Augustin HG, Koh GY. A systems view of the vascular endothelium in health and disease. Cell. 2024;187:4833–58.
Inoue K, Kobayashi M, Yano K, Miura M, Izumi A, Mataki C, et al. Histone deacetylase inhibitor reduces monocyte adhesion to endothelium through the suppression of vascular cell adhesion molecule-1 expression. Arterioscler Thromb Vasc Biol. 2006;26:2652–9.
Martin D, Li Y, Yang J, Wang G, Margariti A, Jiang Z, et al. Unspliced X-box-binding protein 1 (XBP1) protects endothelial cells from oxidative stress through interaction with histone deacetylase 3. J Biol Chem. 2014;289:30625–34.
Bai Y, Du S, Li F, Huang F, Deng R, Zhou J, et al. Histone deacetylase-high mobility group box-1 pathway targeted by hypaconitine suppresses the apoptosis of endothelial cells. Exp Biol Med. 2017;242:527–35.
Huang S, Chen G, Sun J, Chen Y, Wang N, Dong Y, et al. Histone deacetylase 3 inhibition alleviates type 2 diabetes mellitus-induced endothelial dysfunction via Nrf2. Cell Commun Signal. 2021;19:35.
Chen L, Shang C, Wang B, Wang G, Jin Z, Yao F, et al. HDAC3 inhibitor suppresses endothelial-to-mesenchymal transition via modulating inflammatory response in atherosclerosis. Biochem Pharm. 2021;192:114716.
Song J, Nabeel-Shah S, Pu S, Lee H, Braunschweig U, Ni Z, et al. Regulation of alternative polyadenylation by the C2H2-zinc-finger protein Sp1. Mol Cell. 2022;82:3135–50.e9.
Sugawara A, Uruno A, Kudo M, Ikeda Y, Sato K, Taniyama Y, et al. Transcription suppression of thromboxane receptor gene by peroxisome proliferator-activated receptor-gamma via an interaction with Sp1 in vascular smooth muscle cells. J Biol Chem. 2002;277:9676–83.
Lee SJ, Choi EK, Seo KW, Bae JU, Kim YH, Park SY, et al. 5-Lipoxygenase plays a pivotal role in endothelial adhesion of monocytes via an increased expression of Mac-1. Cardiovasc Res. 2013;99:724–33.
Zhao J, Liu Z, Chang Z. Lipopolysaccharide induces vascular endothelial cell pyroptosis via the SP1/RCN2/ROS signaling pathway. Eur J Cell Biol. 2021;100:151164.
Tang Y, Yan JH, Ge ZW, Fei AH, Zhang YC. LncRNA Gaplinc promotes the pyroptosis of vascular endothelial cells through SP1 binding to enhance NLRP3 transcription in atherosclerosis. Cell Signal. 2022;99:110420.
Tan NY, Khachigian LM. Sp1 phosphorylation and its regulation of gene transcription. Mol Cell Biol. 2009;29:2483–8.
Goldberg HJ, Whiteside CI, Hart GW, Fantus IG. Posttranslational, reversible O-glycosylation is stimulated by high glucose and mediates plasminogen activator inhibitor-1 gene expression and Sp1 transcriptional activity in glomerular mesangial cells. Endocrinology. 2006;147:222–31.
Yun S, Dardik A, Haga M, Yamashita A, Yamaguchi S, Koh Y, et al. Transcription factor Sp1 phosphorylation induced by shear stress inhibits membrane type 1-matrix metalloproteinase expression in endothelium. J Biol Chem. 2002;277:34808–14.
Lu H, Jiang X, He L, Ji X, Li X, Liu S, et al. Endothelial Sp1/Sp3 are essential to the effect of captopril on blood pressure in male mice. Nat Commun. 2023;14:5891.
Lu H, Yuan P, Ma X, Jiang X, Liu S, Ma C, et al. Angiotensin-converting enzyme inhibitor promotes angiogenesis through Sp1/Sp3-mediated inhibition of notch signaling in male mice. Nat Commun. 2023;14:731.
Bashore AC, Yan H, Xue C, Zhu LY, Kim E, Mawson T, et al. High-dimensional single-cell multimodal landscape of human carotid atherosclerosis. Arterioscler Thromb Vasc Biol. 2024;44:930–45.
Jiang LP, Yu XH, Chen JZ, Hu M, Zhang YK, Lin HL, et al. Histone deacetylase 3: a potential therapeutic target for atherosclerosis. Aging Dis. 2022;13:773–86.
Zhang L, Chen Y, Jiang Q, Song W, Zhang L. Therapeutic potential of selective histone deacetylase 3 inhibition. Eur J Med Chem. 2019;162:534–42.
Zampetaki A, Zeng L, Margariti A, Xiao Q, Li H, Zhang Z, et al. Histone deacetylase 3 is critical in endothelial survival and atherosclerosis development in response to disturbed flow. Circulation. 2010;121:132–42.
Xiao Q, Zeng L, Zhang Z, Margariti A, Ali ZA, Channon KM, et al. Sca-1+ progenitors derived from embryonic stem cells differentiate into endothelial cells capable of vascular repair after arterial injury. Arterioscler Thromb Vasc Biol. 2006;26:2244–51.
Zeng L, Xiao Q, Margariti A, Zhang Z, Zampetaki A, Patel S, et al. HDAC3 is crucial in shear- and VEGF-induced stem cell differentiation toward endothelial cells. J Cell Biol. 2006;174:1059–69.
Bhaskara S, Chyla BJ, Amann JM, Knutson SK, Cortez D, Sun ZW, et al. Deletion of histone deacetylase 3 reveals critical roles in S phase progression and DNA damage control. Mol Cell. 2008;30:61–72.
Jung SB, Kim CS, Naqvi A, Yamamori T, Mattagajasingh I, Hoffman TA, et al. Histone deacetylase 3 antagonizes aspirin-stimulated endothelial nitric oxide production by reversing aspirin-induced lysine acetylation of endothelial nitric oxide synthase. Circ Res. 2010;107:877–87.
Zhuang T, Liu J, Chen X, Zhang L, Pi J, Sun H, et al. Endothelial Foxp1 suppresses atherosclerosis via modulation of Nlrp3 inflammasome activation. Circ Res. 2019;125:590–605.
McKee CM, Coll RC. NLRP3 inflammasome priming: a riddle wrapped in a mystery inside an enigma. J Leukoc Biol. 2020;108:937–52.
Swanson KV, Deng M, Ting JP. The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat Rev Immunol. 2019;19:477–89.
Guo H, Callaway JB, Ting JP. Inflammasomes: mechanism of action, role in disease, and therapeutics. Nat Med. 2015;21:677–87.
Lee SJ, Seo KW, Kim CD. LPS increases 5-LO expression on monocytes via an activation of Akt-Sp1/NF-κB Pathways. Korean J Physiol Pharm. 2015;19:263–8.
Pazdrak K, Shi XZ, Sarna SK. TNFalpha suppresses human colonic circular smooth muscle cell contractility by SP1- and NF-kappaB-mediated induction of ICAM-1. Gastroenterology. 2004;127:1096–109.
Ishibashi H, Nakagawa K, Onimaru M, Castellanous EJ, Kaneda Y, Nakashima Y, et al. Sp1 decoy transfected to carcinoma cells suppresses the expression of vascular endothelial growth factor, transforming growth factor beta1, and tissue factor and also cell growth and invasion activities. Cancer Res. 2000;60:6531–6.
Ye Y, Jin L, Wilmott JS, Hu WL, Yosufi B, Thorne RF, et al. PI(4,5)P2 5-phosphatase A regulates PI3K/Akt signalling and has a tumour suppressive role in human melanoma. Nat Commun. 2013;4:1508.
Luan Y, Liu H, Luan Y, Yang Y, Yang J, Ren KD. New insight in HDACs: potential therapeutic targets for the treatment of atherosclerosis. Front Pharm. 2022;13:863677.
Gao Q, Wei A, Chen F, Chen X, Ding W, Ding Z, et al. Enhancing PPARγ by HDAC inhibition reduces foam cell formation and atherosclerosis in ApoE deficient mice. Pharmacol Res. 2020;160:105059.
Chi Z, Chen S, Xu T, Zhen W, Yu W, Jiang D, et al. Histone deacetylase 3 couples mitochondria to drive IL-1β-dependent inflammation by configuring fatty acid oxidation. Mol Cell. 2020;80:43–58.e7.
Acknowledgements
This study was supported by National Natural Science Foundation of China (No.81873520, China), Innovation Capability Support Program of Shaanxi (No.2023-CX-PT-07, China). We thank Laboratory Animal Center of Xi’an Jiaotong University for their assistance with detection of ultrasonography on mice. We thank Instrumental Analysis Center of Xi’an Jiaotong University for their help in the detection of super-resolution confocal microscope. We thank Professor Shengpeng Wang (Xi’an Jiaotong University, Xi’an, China) provides us Tie2-CreERT2 mice. We acknowledge the National Local Joint Engineering Research Center for Precision Surgery & Regenerative Medicine, Shaanxi Province Center for Regenerative Medicine and Surgery Engineering Research for the laboratory and experimental platform. The graphical abstract was created with medpeer and Fig. 7A was created with FigDraw.
Author information
Authors and Affiliations
Contributions
LC and WZ: Investigation, Methodology, Project administration, Writing-original draft; HC: Single-cell analysis; YZ and LR: Software; BG and LY: Formal analysis. CY and QZ: Methodology; LB, RW and SZ: Data curation; EL: Supervision; WW: Conceptualization, Funding acquisition, Writing-review & editing; All authors have read the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All animal experiments were approved by the Experimental Animal Ethics Committee of Xi’an Jiaotong University (2023-1534) and performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, L., Zhang, W., Chen, H. et al. HDAC3 activates endothelial NLRP3 inflammasome and promotes atherosclerosis via inhibiting the acetylation of specificity protein 1. Cell Death Differ 33, 930–944 (2026). https://doi.org/10.1038/s41418-025-01620-6
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41418-025-01620-6