Abstract
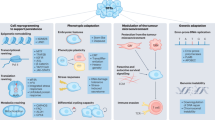
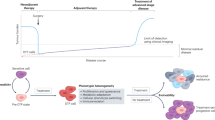
Persisters are a rare sub-population of tumor cells that survive anti-cancer therapy and are thought to be a major cause of recurrence. These cells have been identified following both drug- and immune-therapy but are generally considered to be distinct entities. Since both pharmacological agents and immune cells often kill via apoptosis, we tested a hypothesis that both types of cells survive based on reduced mitochondrial apoptotic sensitivity, which in turn would yield a similar and reciprocal multi-agent resistant phenotype. Supporting this hypothesis, we indeed observed that IPCs acquired a reduced sensitivity to multiple drug classes and radiotherapy, suggesting non-immune mechanisms are important in the survival of cancer cells after immunotherapy. Likewise, DTPs developed not only a reduced sensitivity to multiple drug classes and radiotherapy, but also acquired a reduced sensitivity to T cell killing. Both IPCs and DTPs developed decreased sensitivity to mitochondrial apoptosis. A sub-population of IPCs downregulated antigen and upregulated PD-L1. Intriguingly, in the IPCs that didn’t employ these mechanisms of resistance, a greater decrease in sensitivity to mitochondrial apoptosis was observed, suggesting that the presence or absence of a resistance mechanism can exert selective pressures over the emergence of others. Targeting anti-apoptotic dependencies in persisters increased sensitivity to chemotherapy or CAR T therapy. These results suggest that common biological mechanisms underly survival of persisters, whether derived from immune or drug therapy, and offer an explanation for the acquired cross-resistance to these two types of therapies often observed in the clinic.

Highlights
-
Immunotherapy persister cells (IPCs) are less sensitive to drugs and radiation.
-
Drug tolerant persisters (DTPs) are less sensitive to radiation and CAR T cell attack.
-
IPCs and DTPs are less sensitive to mitochondrial apoptosis.
-
Targeting anti-apoptotic dependencies helps eliminate IPCs/DTPs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Data availability
Available upon request.
References
Hangauer MJ, Viswanathan VS, Ryan MJ, Bole D, Eaton JK, Matov A, et al. Drug-tolerant persister cancer cells are vulnerable to GPX4 inhibition. Nature. 2017;551:247–50.
Oren Y, Tsabar M, Cuoco MS, Amir-Zilberstein L, Cabanos HF, Hütter JC, et al. Cycling cancer persister cells arise from lineages with distinct programs. Nature. 2021;596:576–82.
Shen S, Vagner S, Robert C. Persistent cancer cells: the deadly survivors. Cell. 2020;183:860–74.
Park SY, Nam JS. The force awakens: metastatic dormant cancer cells. Exp Mol Med. 2020;52:569–81.
Dhanyamraju PK, Schell TD, Amin S, Robertson GP. Drug-tolerant persister cells in cancer therapy resistance. Cancer Res. 2022;82:2503–14.
Bai R, Chen N, Li L, Du N, Bai L, Lv Z, et al. Mechanisms of cancer resistance to immunotherapy. Front Oncol. 2020;10:1290.
O’Donnell JS, Teng MWL, Smyth MJ. Cancer immunoediting and resistance to T cell-based immunotherapy. Nat Rev Clin Oncol. 2018;16:151–67.
Sehgal K, Portell A, Ivanova EV, Lizotte PH, Mahadevan NR, Greene JR, et al. Dynamic single-cell RNA sequencing identifies immunotherapy persister cells following PD-1 blockade. J Clin Invest. 2021;131. Available from: https://pubmed.ncbi.nlm.nih.gov/33151910/.
Joung J, Kirchgatterer PC, Singh A, Cho JH, Nety SP, Larson RC, et al. CRISPR activation screen identifies BCL-2 proteins and B3GNT2 as drivers of cancer resistance to T cell-mediated cytotoxicity. Nat Commun. 2022;13:1606.
Catalán E, Jaime-Sánchez P, Aguilo N, Simon MM, Froelich CJ, Pardo J. Mouse cytotoxic T cell-derived granzyme B activates the mitochondrial cell death pathway in a Bim-dependent fashion. J Biol Chem. 2015;290:6868–77.
Vasan N, Baselga J, Hyman DM. A view on drug resistance in cancer. Nature. 2019;575:299–309.
Adhikari S, Bhattacharya A, Adhikary S, Singh V, Gadad SS, Roy S, et al. The paradigm of drug resistance in cancer: an epigenetic perspective. Biosci Rep. 2022;42:BSR20211812.
Hannun YA. Apoptosis and the dilemma of cancer chemotherapy. Blood. 1997;89:1845–53. https://doi.org/10.1182/blood.V89.6.1845.
Pourzia AL, Olson ML, Bailey SR, Boroughs AC, Aryal A, Ryan J, et al. Quantifying requirements for mitochondrial apoptosis in CAR T killing of cancer cells. Cell Death Dis. 2023;14. Available from: https://pubmed.ncbi.nlm.nih.gov/37055388/
Potter DS, Du R, Bhola P, Bueno R, Letai A. Dynamic BH3 profiling identifies active BH3 mimetic combinations in non-small cell lung cancer. Cell Death Dis. 2021;12. Available from: https://pubmed.ncbi.nlm.nih.gov/34315868/
Touzeau C, Ryan J, Guerriero J, Moreau P, Chonghaile TN, Le Gouill S, et al. BH3-profiling identifies heterogeneous dependency on Bcl-2 family members in multiple myeloma and predicts sensitivity to BH3 mimetics. Leukemia. 2016;30:761.
Bhatt S, Pioso MS, Olesinski EA, Yilma B, Ryan JA, Mashaka T, et al. Reduced mitochondrial apoptotic priming drives resistance to BH3 mimetics in acute myeloid leukemia. Cancer Cell. 2020;38:872–890.e6.
Bhola PD, Ahmed E, Guerriero JL, Sicinska E, Su E, Lavrova E, et al. High-throughput dynamic BH3 profiling may quickly and accurately predict effective therapies in solid tumors. Sci Signal. 2020;13. Available from: https://pubmed.ncbi.nlm.nih.gov/32546544/
Pan R, Ryan J, Pan D, Wucherpfennig KW, Letai A. Augmenting NK cell-based immunotherapy by targeting mitochondrial apoptosis. Cell. 2022;185:1521–1538.e18.
Song EZ, Wang X, Philipson BI, Zhang Q, Thokala R, Zhang L, et al. The IAP antagonist birinapant enhances chimeric antigen receptor T cell therapy for glioblastoma by overcoming antigen heterogeneity. Mol Ther Oncolytics. 2022;27:288–304.
Johnson LA, Scholler J, Ohkuri T, Kosaka A, Patel PR, McGettigan SE, et al. Rational development and characterization of humanized anti–EGFR variant III chimeric antigen receptor T cells for glioblastoma. Sci Transl Med. 2015;7:275ra22.
Jiao Y, Cao F, Liu H. Radiation-induced cell death and its mechanisms. Health Phys. 2022;123:376.
Debatin KM. Apoptosis pathways in cancer and cancer therapy. Cancer Immunol Immunother. 2004;53:153–9.
Chen M, Mainardi S, Lieftink C, Velds A, de Rink I, Yang C, et al. Targeting of vulnerabilities of drug-tolerant persisters identified through functional genetics delays tumor relapse. Cell Rep Med. 2024;5:101471.
Russo M, Chen M, Mariella E, Peng H, Rehman SK, Sancho E, et al. Cancer drug-tolerant persister cells: from biological questions to clinical opportunities. Nat Rev Cancer. 2024;24:694–717.
Pu Y, Li L, Peng H, Liu L, Heymann D, Robert C, et al. Drug-tolerant persister cells in cancer: the cutting edges and future directions. Nat Rev Clin Oncol. 2023;20:799–813.
Sharma SV, Lee DY, Li B, Quinlan MP, Takahashi F, Maheswaran S, et al. A chromatin-mediated reversible drug-tolerant state in cancer cell subpopulations. Cell. 2010;141:69–80.
Mikubo M, Inoue Y, Liu G, Tsao MS. Mechanism of drug tolerant persister cancer cells: the landscape and clinical implication for therapy. J Thoracic Oncol. 2021;16:1798–809.
Kohlhapp FJ, Haribhai D, Mathew R, Duggan R, Ellis PA, Wang R, et al. Venetoclax increases intratumoral effector t cells and antitumor efficacy in combination with immune checkpoint blockade. Cancer Discov. 2021;11:68–79.
Assaraf YG, Brozovic A, Gonçalves AC, Jurkovicova D, Linē A, Machuqueiro M, et al. The multi-factorial nature of clinical multidrug resistance in cancer. Drug Resist Updat. 2019;46. Available from: https://pubmed.ncbi.nlm.nih.gov/31585396/
Mesner PW, Budihardjo II, Kaufmann SH. Chemotherapy-induced apoptosis. Adv Pharmacol. 1997;41:461–99.
Waterhouse NJ, Sedelies KA, Browne KA, Wowk ME, Newbold A, Sutton VR, et al. A central role for Bid in granzyme B-induced apoptosis. J Biol Chem. 2005;280:4476–82.
Goping IS, Barry M, Liston P, Sawchuk T, Constantinescu G, Michalak KM, et al. Granzyme B-induced apoptosis requires both direct caspase activation and relief of caspase inhibition. Immunity. 2003;18:355–65.
Mehta AK, Gracias DT, Croft M. TNF activity and T cells. Cytokine. 2018;101:14–8.
Chun N, Ang RL, Chan M, Fairchild RL, Baldwin WM, Horwitz JK, et al. T cell–derived tumor necrosis factor induces cytotoxicity by activating RIPK1-dependent target cell death. JCI Insight. 2021;6:e148643.
Sedger LM, McDermott MF. TNF and TNF-receptors: From mediators of cell death and inflammation to therapeutic giants – past, present and future. Cytokine Growth Factor Rev. 2014;25:453–72.
Chonghaile TN, Sarosiek KA, Vo TT, Ryan JA, Tammareddi A, Moore VDG, et al. Pretreatment mitochondrial priming correlates with clinical response to cytotoxic chemotherapy. Science. 2011;334:1129.
Potter DS, Du R, Bohl SR, Chow KH, Ligon KL, Bueno R, et al. Dynamic BH3 profiling identifies pro-apoptotic drug combinations for the treatment of malignant pleural mesothelioma. Nat Commun. 2023;14. Available from: https://pubmed.ncbi.nlm.nih.gov/37210412/
Chen DS, Mellman I. Elements of cancer immunity and the cancer-immune set point. Nature. 2017;541:321–30.
Kalbasi A, Ribas A. Antigen presentation keeps trending in immunotherapy resistance. Clin Cancer Res. 2018;24:3239–41.
Murciano-Goroff YR, Warner AB, Wolchok JD. The future of cancer immunotherapy: microenvironment-targeting combinations. Cell Res. 2020;30:507–19.
Zhang W, Yuan S, Zhang Z, Fu S, Liu S, Liu J, et al. Regulating tumor cells to awaken T cell antitumor function and enhance melanoma immunotherapy. Biomaterials. 2025;316:123034.
Charles A, Janeway J, Travers P, Walport M, Shlomchik MJ. T cell-mediated cytotoxicity. 2001. https://www.ncbi.nlm.nih.gov/books/NBK27101/.
Halle S, Halle O, Förster R. Mechanisms and dynamics of T cell-mediated cytotoxicity in vivo. Trends Immunol. 2017;38:432–43.
Morad G, Helmink BA, Sharma P, Wargo JA. Hallmarks of response, resistance, and toxicity to immune checkpoint blockade. Cell. 2021;184:5309–37.
Leonard GD, Fojo T, Bates SE. The role of ABC transporters in clinical practice. Oncologist. 2003;8:411–24.
Ozben T. Mechanisms and strategies to overcome multiple drug resistance in cancer. FEBS Lett. 2006;580:2903–9.
Oliver TG, Mercer KL, Sayles LC, Burke JR, Mendus D, Lovejoy KS, et al. Chronic cisplatin treatment promotes enhanced damage repair and tumor progression in a mouse model of lung cancer. Genes Dev. 2010;24:837–52.
Atilla PA, Atilla E. Resistance against anti-CD19 and anti-BCMA CAR T cells: recent advances and coping strategies. Transl Oncol. 2022;22:101459.
Kallingal A, Olszewski M, Maciejewska N, Brankiewicz W, Baginski M. Cancer immune escape: the role of antigen presentation machinery. J Cancer Res Clin Oncol. 2023;149:8131.
Davern M, Donlon NE, Sheppard A, Connell FO, Hayes C, Bhardwaj A, et al. Chemotherapy regimens induce inhibitory immune checkpoint protein expression on stem-like and senescent-like oesophageal adenocarcinoma cells. Transl Oncol. 2021;14:101062.
Lacour M, Hiltbrunner S, Lee SY, Soltermann A, Rushing EJ, Soldini D, et al. Adjuvant chemotherapy increases programmed death-ligand 1 (PD-L1) expression in non-small cell lung cancer recurrence. Clin Lung Cancer. 2019;20:391–6.
Cui JW, Li Y, Yang Y, Yang HK, Dong JM, Xiao ZH, et al. Tumor immunotherapy resistance: revealing the mechanism of PD-1 / PD-L1-mediated tumor immune escape. Biomed Pharmacotherapy. 2024;171:116203.
Stutvoet TS, Kol A, de Vries EGE, de Bruyn M, Fehrmann RSN, Terwisscha van Scheltinga AGT, et al. MAPK pathway activity plays a key role in PD-L1 expression of lung adenocarcinoma cells. J Pathol. 2019;249:52–64.
Shen X, Jin X, Fang S, Chen J. EFEMP2 upregulates PD-L1 expression via EGFR/ERK1/2/c-Jun signaling to promote the invasion of ovarian cancer cells. Cell Mol Biol Lett. 2023;28:53.
Sato H, Niimi A, Yasuhara T, Permata TBM, Hagiwara Y, Isono M, et al. DNA double-strand break repair pathway regulates PD-L1 expression in cancer cells. Nat Commun. 2017;8. https://pubmed.ncbi.nlm.nih.gov/29170499/
Dadsena S, Jenner A, García-Sáez AJ. Mitochondrial outer membrane permeabilization at the single molecule level. Cell Mol Life Sci. 2021;78:3777.
Kalkavan H, Chen MJ, Crawford JC, Quarato G, Fitzgerald P, Tait SWG, et al. Sublethal cytochrome c release generates drug-tolerant persister cells. Cell. 2022;185:3356–74.e22.
Vringer E, Heilig R, Riley JS, Black A, Cloix C, Skalka G, et al. Mitochondrial outer membrane integrity regulates a ubiquitin-dependent and NF-κB-mediated inflammatory response. EMBO J. 2024;43:904–30.
Guo Q, Jin Y, Chen X, Ye X, Shen X, Lin M, et al. NF-κB in biology and targeted therapy: new insights and translational implications. Signal Transduct Target Ther. 2024;9:1–37.
Xia Y, Shen S, Verma IM. NF-κB, an active player in human cancers. Cancer Immunol Res. 2014;2:823.
Li F, Sethi G. Targeting transcription factor NF-κB to overcome chemoresistance and radioresistance in cancer therapy. Biochim Biophys Acta Rev Cancer. 2010;1805:167–80.
Perez R, Crombet T, de Leon J, Moreno E. A view on EGFR-targeted therapies from the oncogene-addiction perspective. Front Pharmacol. 2013;4:53. https://pmc.ncbi.nlm.nih.gov/articles/PMC3636470/
Acknowledgements
This work utilized an Illumina NovaSeq X Plus that was purchased with funding from a National Institutes of Health SIG grant 1S10OD036228-01.
Funding
This work was funded by NCI R01 249062 (AL and MVM). AL also receives funding from Blood Cancer United’s Discovery Grant Program.
Author information
Authors and Affiliations
Contributions
All authors contributed to this study. MD and AL designed the study and wrote the manuscript. MD, CJT, DG, LB, BCC, JY-TK, MLO, CWW, SS, LK, MY, GA, FB and AJA performed experiments. JR, PB, KAS, and MVM provided advice on experimental design. DB, CPP, and PHL provided cell lines.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41418_2025_1656_MOESM6_ESM.jpg (download JPG )
Supplementary Figure 5 (Supplemental for Figure 5): PD-L1-positive DTPs are typically more unprimed for apoptosis compared with PD-L1-negative DTPs.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Davern, M., Turner, C.J., Griffin, D. et al. Drug-tolerant persisters and immunotherapy persister cells exhibit cross-resistance and share common survival mechanisms. Cell Death Differ (2025). https://doi.org/10.1038/s41418-025-01656-8
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41418-025-01656-8