Abstract
To investigate whether αCGRP (Calca) deficiency exacerbates pulmonary fibrosis (PF) by promoting alveolar type 2 (AT2) cell senescence, we retrospectively analyzed clinical data and lung biopsy samples from PF patients (n = 15). In vivo, lung tissues from Calca-knockout (KO) rats and D-galactose (D-gal)-induced senescence models were analyzed using immunohistochemistry, single-cell RNA sequencing (scRNA-seq), and label-free proteomics. PF patient samples showed low αCGRP expression, AT2 subtype differentiation, and high Calca promoter methylation. In Calca-/- rats, AT2 differentiation and oxidative lipid metabolism were enhanced, with increased senescence gene signatures. scRNA-seq revealed upregulation of linoleic, α-linolenic, and arachidonic acid metabolism, alongside suppression of oxidative stress responses in AT2 of the Calca-/- group. D-gal treatment induced alveolitis, fibrotic changes and AT2 subtype differentiation, and the most severe alveolar inflammation was found in Calca-/-+D-gal rats. Proteomics revealed distinct metabolic pathway alterations between WT + D-gal and Calca-/-+D-gal, and Calca-/- and WT + D-gal rats. Differences in metabolic and PPAR pathways were observed between Calca-/- and Calca-/-+D-gal rats. Additionally, both D-gal treatment and Calca-/- affect oxidative phosphorylation. Overall, αCGRP deficiency disrupts AT2 lipid metabolism, and accelerates AT2 inflammatory senescence, ultimately promoting pulmonary fibrosis.
Similar content being viewed by others
Introduction
Interstitial lung disease (ILD) encompasses a group of disorders characterized by diffuse parenchymal damage, alveolar inflammation, and interstitial fibrosis [1]. Pulmonary fibrosis (PF) is a leading driver of disease progression and poor prognosis, largely due to the irreversible fibrotic remodeling that follows chronic alveolitis. Despite ongoing research, the triggers and mechanisms driving PF remain poorly understood, and current treatments are limited, with lung transplantation as the only definitive option [2]. Recent studies have highlighted the impaired repair of alveolar epithelial type II cells (AT2) following injury as an irreversible trigger in PF [1, 3, 4]. However, the molecular mechanisms initiating AT2 dysfunction remain unclear, hindering the development of effective intervention strategies [1].
Senescence is a significant risk factor for many chronic diseases, including PF [5, 6]. Elevated oxidative stress, a hallmark of senescence and the resulting reactive oxygen species (ROS), promote cell apoptosis and necrosis through dysregulation of metabolic pathways, triggering inflammatory senescence [7, 8]. The lungs are particularly vulnerable due to their structure and continuous exposure to oxygen and environmental stressors [9]. Increasing evidence indicates that the senescence of alveolar epithelium and AT2 cell senescence and dysfunction drive PF progression [10,11,12]. Oxidative lipid metabolism disturbances may further exacerbate the inflammatory senescence of alveolar epithelial cells (AECs) [13, 14]. In patients with idiopathic pulmonary fibrosis (IPF), elevated lipid peroxidation products, oxidative protein accumulation, and reduced levels of antioxidant enzymes have been reported [15], although the underlying mechanisms remain unclear.
Calcitonin gene-related peptide (CGRP), a neuroimmune peptide secreted by neurons and immune cells [16], plays roles in vasodilation, immune regulation, pain, and tissue repair [17,18,19,20]. Of its two subtypes, αCGRP, encoded by CT/Calca, and βCGRP, encoded by Calcb, αCGRP is the predominant form, representing approximately 90% of total CGRP [21, 22]. During the physiological process of tissue injury repair, it was found that high expression of CGRP not only promotes the migration of mesenchymal stem cells (MSCs) to damaged tissues, but also promotes stem cell differentiation [23,24,25]. Our previous research demonstrated that Calca+/- rats develop PF-like pathology, with reduced αCGRP expression, AT2 cell injury, and heightened type 2 immune responses [26]. Transcriptomic and proteomic analysis revealed that αCGRP deficiency alters oxidative lipid metabolism and impairs PPARγ nuclear translocation [27].
We therefore hypothesize that αCGRP deficiency promotes PF by inducing AT2 inflammatory senescence through dysregulated oxidative lipid metabolism. To test this, we utilized a Calca-/- rat model and a D-galactose (D-gal)-induced senescence model to evaluate the impact of αCGRP loss on AT2 regeneration, metabolic dysfunction, and fibrosis progression. We further examined the protective role of αCGRP in suppressing AT2 senescence and collagen deposition, providing insight into its therapeutic potential in PF.
Materials and Methods
Patients’ characteristics
A retrospective study was conducted, including patients hospitalized with IPF or progressive pulmonary fibrosis (PPF) between August 2016 and September 2024. A total of 15 patients were included, all diagnosed with ILD through chest CT or pathological examination. IPF and PPF classifications were determined based on established diagnostic criteria [2, 28]. Patients with malignant tumors, bronchiectasis, or pulmonary cystic fibrosis were excluded. Clinical laboratory data from all patients were reviewed.
Patients with pulmonary bullae served as the control group for pathological comparison, while ten healthy individuals were used as controls for peripheral blood testing. Of the 15 patients, three (cases 1–3) underwent CT-guided fine-needle aspiration biopsy of lung lesions. Three patients with pulmonary bullae served as controls for histopathological analysis.
Our study involving human material, and human data have been performed in accordance with the Declaration of Helsinki. This study was approved by the Medical Ethics Committee of the First Affiliated Hospital of Fujian Medical University (Fuzhou, Fujian, China) under number [2023]485. Informed consent for participation and publication was obtained from all patients.
Peripheral blood methylation detection
Bisulfite conversion of DNA samples was performed using the EpiTect Bisulfite Kit (Qiagen, Düsseldorf, Germany), with an output of 1–2 µg of DNA per sample, according to the manufacturer’s protocol. Following single-stranded DNA preparation, target gene sequencing was carried out. Methylation status at the target gene locus was analyzed using supporting software.
The PCR amplification primers: CALCA-F1 (TTATAGTTTYGGGATGTAGTTGTTG); CALCA-R1 (TTTCAACCTCTCCACCATCT (5’-BIOTIN)); Sequencingprimers: CALCA-S (TGTTGAGTTTATYGTATAGGTA; CALCA sequencinggenome segments: GCGCCCGGACCGGCTGCAGCAGATCGCGCGCTGCGCGTT.
Construction of Calca knockout rats
Calca knockout rats were generated by Shanghai Model Organisms Co., LTD., using the CRISPR/Cas9 gene-editing technique. Homozygous (Calca-/-) rats were maintained at 22 °C± 2 °C and 40–60% humidity. Mature rats (>8 weeks) with appropriate genotypes were mated at a 2:1 female-to-male ratio. After a gestation period of approximately 21 days, offspring were genotyped using tail biopsies taken at 7–10 days post-birth and were separated by sex at 4 weeks of age.
All procedures conformed to the ARRIVE guidelines for animal care and use. This study was approved by the Medical Ethics Committee of the First Affiliated Hospital of Fujian Medical University (Fuzhou, Fujian, China) under number (IACUCFJMU 2024-Y-1991). Five Calca-/- rats were successfully generated and designated as the Calca-/- group (Fig. 1).
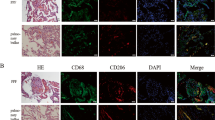
A In PF patients, immunofluorescence revealed increased Lamp3+NPW+AT2 cells and reduced Ager+Rtkn+AT1 cells compared to the pulmonary bulla group (×400). B Similarly, Calca-/- rats showed decreased Ager+Rtkn+AT1 and increased Lamp3+NPW+AT2 expression compared to WT rats (×400).
Experimental senescence models and grouping
Eight-week-old female Sprague-Dawley (SD) rats (weighing 280–320 g) were housed under controlled conditions (22 °C–25 °C) with free access to food and water. All procedures conformed to the ARRIVE guidelines for animal care and use. This study was approved by the Medical Ethics Committee of the First Affiliated Hospital of Fujian Medical University (Fuzhou, Fujian, China) under number (IACUCFJMU 2024-Y-1991).
Construction of the D-gal-induced senescence model
Ten 8-week-old female SD rats were randomly assigned to either the WT group or the WT + D-gal group (n = 5 per group). Similarly, ten 8-week-old Calca-/- female rats were divided into the Calca-/- group and the Calca-/-+D-gal group (n = 5 per group).
Rats in the D-gal intervention groups received daily subcutaneous injections of 10% D-gal (400 mg/kg·d) in the neck and back for eight weeks. Control groups received equal volumes of sterile saline for the same duration. After the treatment period, rats were euthanized by cervical dislocation, and tissue samples were collected for further analysis.
Histological evaluation
Lung tissues from each specimen were fixed in neutral formalin and embedded in paraffin. Some sections were subjected to H&E staining, Masson’s trichrome staining, and apoptosis assay. The remaining sections were reserved for immunohistochemical and immunofluorescence analyzes.
Hematoxylin and eosin staining
Lung sections were stained with H&E following standard pathologic protocols. Slides were deparaffinized in xylene (2 × 5 min) and rehydrated through sequential 1 min washes in 70, 80, 95, and 100% ethanol, successively. Sections were stained with hematoxylin for 2 min, rinsed with distilled water, immersed in 0.1% hydrochloric acid in 50% ethanol, and washed under tap water for 15 min. Eosin staining was then applied for 1 min, followed by a distilled water rinse. Slides were dehydrated in 95 and 100% ethanol, cleared in xylene (2 × 5 min), and mounted with cover slips.
Immunohistochemical studies
Paraffin-embedded lung sections were deparaffinized in xylene and rehydrated through a graded ethanol series. Antigen retrieval was performed by heating sections in the Tris-EDTA buffer for 30 minutes. The following primary antibodies were used: anti-CD68+ (ready-to-use, Maixin, Fuzhou, China), anti-CD3+ (1:1000, Santa Cruz, CA, USA), anti-CD4+ (ready-to-use, Maixin, Fuzhou, China), anti-CD8+ (ready-to-use, Maixin, Fuzhou, China), TGFβ (1:800, Bioworld, Louis Park, MN, USA), αCGRP (1:800, ABclonal, Wuhan, China), and PPARγ (1:800, Bioss, Beijing, China).
Sections were incubated with primary antibodies overnight at 4 °C. Horseradish peroxidase-conjugated secondary antibodies (Dako, Denmark) were used for detection. Negative controls were prepared by replacing the primary antibody with a species-matched isotype control. Samples were stained with diaminobenzidine and lightly counterstained with hematoxylin. Collagen deposition in fibrosis lung lesions was assessed via Masson’s trichrome staining and categorized based on staining intensity compared to background tissue.
Immunofluorescence staining
For immunofluorescence analysis, lung sections were incubated overnight at 4 °C with the following primaries: PPARγ (1:100, Bioss, Beijing, China), LAMP3(1:2000, Proteintech, Wuhan, China), NPW(1:2000,Bioss,Beijing,China), AGER(1:2000, Proteintech, Wuhan, China), and RTKN2 (1:2000, Proteintech, Wuhan, China).
After washing, sections were incubated for 2 h at room temperature with secondary antibodies Goat Anti-Mouse IgG (H + L), FITC-conjugated (1:100, Boster, Wuhan, China) and Goat Anti-Rabbit IgG (H + L), Cy3-conjugated (1:100, Boster, Wuhan, China).
Sections were counterstained with DAPI (4’,6-diamidino-2-phenylindole, Boster, Wuhan, China) for 10 minutes at room temperature and imaged using a fluorescence microscope.
Terminal deoxynucleotidyl transferase dUTP Nick‑End Labeling assay
Apoptotic nerve cells in the cerebral cortex of senescence rats were detected by Terminal deoxynucleotidyl transferase dUTP Nick‑End Labeling reagents and DAPI. Apoptotic cells were visualized and recorded using a fluorescence microscope.
Measurement of malondialdehyde (MDA)
Malondialdehyde (MDA) levels in peripheral blood were measured using a rat MDA ELISA kit, following the protocol.
single-cell RNA sequencing (scRNA-seq) of rat lung tissues
Lung tissues were collected from two rats in the WT group and Calca-/-group and combined for testing. Freshly prepared enzymatic digestion solutions, including trypsin, collagenase types I, II, and IV, BSA, DMEM, Ficoll Plus 1.077, DNase I, and lichen spore hydrolase, were applied. Cell concentration and viability were assessed using a Luna cell counter. Single-cell transcriptome sequencing of two pooled samples was completed. Library preparation, sequencing, and initial data processing were performed by OE Biotech Co, Ltd. (Shanghai, China).
Sequencing data were processed using the official 10x Genomics software, Space Ranger (version 2.0.1), for quality control and gene quantification, in conjunction with bright field microscopy images of tissue sections. Captured regions on the chip were mapped to the reference genome (Rnor 6.0). The total number of captured spots, reads per spot, detected genes, and UMIs were quantified.
Subsequent analysis was conducted using the Seurat R package (version 4.3.0) [29]. Data normalization was performed using the sctransform [30] function, and highly variable genes (HVGs) were identified and stored in the SCT matrix. The FindVariable Genes function was used to select the top 3000 HVGs, which were subjected to Principal Component Analysis for dimensionality reduction. Results were visualized in two dimensions using UMAP (Nonlinear Dimensionality Reduction).
Spatial feature gene identification was performed using the FindAllMarkers function (test. use = “presto”) in Seurat to identify differentially upregulated genes for each spot group, serving as potential marker genes. The VlnPlot and EigenPlot functions were used to visualize marker gene expression. Cell type annotation was carried out using the RCTD [31] (version 1.1.0) method, which applied single-cell RNA-seq-derived cell type profiles to infer spatial cell type composition while correcting for cross-platform differences.
Label-free protein quantitative analysis
Lung tissue was collected and pooled from four rats in each of the following groups: WT, WT + D-gal, Calca-/-, and Calca-/-+D-gal groups. Label-free quantitative proteomics was performed via liquid chromatography-mass spectrometry, without the use of isotopic labeling. Peptide abundance was determined by analyzing the signal intensity of peptide segments across samples. Precursor intensity was used as the primary quantitative metric.
Protein abundance was determined by summing the weighted peak intensities of all peptide fragments corresponding to each protein in the search database. The peak intensities were normalized across samples to yield normalized abundance. Fold changes between groups were calculated by comparing the mean normalized signal intensity within each group. Proteins satisfying two predefined criteria were selected as differentially expressed between groups.
Statistical analysis
All experiments were independently repeated in triplicate. Data are presented as mean ± standard deviation (SD). Statistical analyzes were conducted using SPSS 24.0 statistical software, and figures were generated using GraphPad Prism 8.0. For comparisons between two groups, an independent sample t-test was used when assumptions of normal distribution and equal variance were met. For comparisons involving three or more groups, one-way ANOVA was applied. Non-parametric tests were used when data failed to meet the assumptions of normality. A value of p < 0.05 was considered statistically significant.
Results
Hypermethylation of the Calca promoter and reduced αCGRP expression in PF patients
A retrospective analysis was conducted on clinical data from 15 patients hospitalized with IPF or other forms of PF over a 2-year period ending in September 2024. The most common diagnoses were IPF (3/15), nonspecific interstitial pneumonia (3/15), and IgG4-related ILD (IgG4-RILD; 3/15), followed by polymyositis-related ILD (PM-ILD; 2/15). One patient each was diagnosed with interstitial pneumonia with autoimmune features, sarcoidosis, ANCA-associated vasculitis-related ILD, and cryptogenic organizing pneumonia (Supplemental Fig. 1E).
Phosphoric acid sequencing revealed significant hypermethylation at the second site of the Calca promoter in the peripheral blood of PF patients compared to healthy individuals (Supplemental Fig. 1). In parallel, αCGRP expression in lung tissue was significantly reduced in PF patients. Immunohistochemical staining further demonstrated decreased αCGRP expression in fibrotic lung tissue (Supplemental Fig. 2A).
Differentiation of AT2 subtypes in patients and rats during PF progression
H&E and Masson’s trichrome staining confirmed prominent alveolitis, PF, and collagen deposition in PF patients relative to pulmonary bullae controls (Supplemental Fig. 2A). Immunofluorescence staining indicated a predominance of the Lamp3+NPW+AT2 cell subtype in lung tissue from PF patients, with an associated reduction in Ager+Rtkn+AT1 cells. These findings suggest subtype differentiation and regenerative activity of AT2 cells in response to AT1 injury during PF. In contrast, lung tissue from patients with pulmonary bullae showed a dominance of Ager+Rtkn+AT1 cells, while Lamp3+NPW+AT2 cells were reduced (Fig. 1A).
Similar histological changes were observed in Calca-/- rats, including alveolitis, PF, and collagen accumulation, when compared to WT controls. The Calca-/- lungs exhibited disrupted alveolar architecture, thick collagen bands or lamellar deposits, and marked infiltration of inflammatory cells within alveolar spaces and capillaries (Fig. 5A). Consistent with the human findings, immunofluorescence revealed that the Calca-/- rats had increased differentiation toward the Lamp3+NPW+AT2 subtype and decreased representation of Ager+Rtkn+AT1 cells, supporting the hypothesis of impaired AT1 regeneration and a compensatory AT2 response during fibrosis progression (Fig. 1B).
Disruption of AT2 lipid metabolism in Calca -/- rats during PF
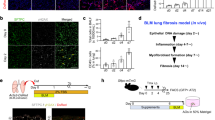
scRNA-seq was performed on lung tissue from WT and Calca-/- rats. After initial quality control by Cell Ranger, the two samples yielded 10,897 and 10,764 high-quality cells, respectively. Following exclusion of doublets, apoptotic, and low-quality cells, final cell counts ranged from 7905 and 8343. The average UMI count per cell was 10,516 and 10,659, the average gene number detected per cell was 2570 and 2667, and the average proportion of mitochondrial UMIs per cell was between 0.0670 and 0.0740.
Dimensionality reduction and clustering analyses were performed. A total of 15 cell subpopulations were identified in lung tissue samples (Fig. 2A, B, D). The proportion of AECs was markedly reduced in the Calca-/-group compared to WT (4.21 vs. 15.08%) (Fig. 2C). Based on marker gene expression and previous literature [32], AECs were further categorized into six subgroups. Subgroups 1–5 were classified as AT2 cells, while only a small portion of subgroup 6 was identified as AT1 cells in the Calca-/- group (Fig. 3A–C, E).
A Dimensionality reduction identified 15 cell subtypes in WT and Calca-/- lung tissues. B Marker genes of 15 cell subtypes. C, D Proportion of 15 cell subtypes were showed in WT and Calca-/- lung tissues. AECs were significantly reduced in Calca-/- rats (4.21%) compared to WT (15.08%).
A AEC was divided into six subgroups; subgroups 1–5 were AT2, and subgroup 6 was AT1. B Subgroup 2 was notably elevated in Calca-/- rats. C, E Marker genes of each subgroup. D KEGG analysis of subgroup 2 showed enrichment in linoleic acid metabolism (rno00591), arachidonic acid metabolism (rno00590), and steroid hormone biosynthesis (rno00140).
Among these, subgroup 2 was significantly expanded in the Calca-/- group (Fig. 3B). KEGG pathway enrichment analysis of subgroup 2 revealed dysregulation in oxidative lipid metabolism, including linoleic acid metabolism (rno00591), arachidonic acid metabolism (rno00590), and steroid hormone biosynthesis (rno00140), indicating metabolic disturbance associated with fibrotic progression (Fig. 3D).
Elevated senescence gene set scores in both lung tissues and the AT2 subgroup of Calca -/- rats
Further analysis of lung tissue and AEC senescence gene set scores revealed that, compared to the WT group, the Calca-/- group exhibited significantly elevated overall senescence scores, particularly within the AT2 cell subpopulation (Fig. 4A, B).
A Senescence gene set scores were elevated in whole lung tissues of Calca-/- rats compared to WT group. B Senescence gene set scores were elevated in AT2 cells of Calca-/- rats compared to WT group. C KEGG analysis indicated upregulation of lipid metabolism (e.g., linoleic acid metabolism (rno00591), α-linoleic acid metabolism (rno00592), and arachidonic acid metabolism (rno00590), fat digestion and absorption (rno04975), and Th1/Th2 cell differentiation (rno04658) in Calca-/- rats; and downregulation of HIF-1 signaling (rno04066), FoxO signaling (rno04068), and cell cycle (rno04110) pathways in Calca-/- rats. D GO analysis showed increased negative regulation of cell fate commitment (GO: 0010454), fatty acid metabolism (GO: 0045923), stem cell proliferation (GO: 0072089), replicative senescence (GO: 0090399), and T-helper 2 cell cytokine production (GO: 0035745) in Calca-/- rats; oxygen transport (GO: 0015671) were downregulated in Calca-/- rats.
KEGG pathway analysis of AEC indicated that the Calca-/- group showed upregulation of linoleic acid metabolism (rno00591), α-linoleic acid metabolism (rno00592), arachidonic acid metabolism (rno00590), and Th1/Th2 cell differentiation (rno04658). Conversely, there was downregulation of HIF-1 signaling (rno04066), FoxO signaling (rno04068), and the cell cycle pathway (rno04110) (Fig. 4C).
GO enrichment analysis further revealed that biological processes in Calca-/- rats such as negative regulation of cell fate commitment (GO: 0010454), positive regulation of fatty acid metabolism (GO: 0045923), stem cell proliferation (GO: 0072089), replicative senescence (GO: 0090399), and T-helper 2 cell cytokine production (GO: 0035745) were upregulated in the Calca-/- group, whereas oxygen transport (GO: 0015671) were downregulated (Fig. 4D).
Since AECs are predominantly AT2 cells, and these cells showed dysregulated oxidative lipid metabolism, we hypothesize that αCGRP deficiency accelerates AT2 cell senescence, thereby promoting fibrosis progression.
αCGRP deficiency promotes AT2 inflammation senescence via oxidative stress and metabolic pathways
To investigate the relationship between αCGRP deficiency and senescence, we constructed a D-gal treatment model. H&E and Masson’s trichrome staining revealed marked pulmonary inflammation and mild fibrosis in WT rats treated with D-gal, confirming its role in driving inflammatory and fibrotic remodeling. In contrast, the Calca-/-+D-gal group exhibited minimal fibrosis but the most pronounced alveolar inflammation (Fig. 5A).
A H&E and Masson’s revealed alveolitis, and PF in Calca-/- rats in the form of alveolar structure destruction and collagen deposition. WT + D-gal rats showed milder pathology, while Calca-/-+D-gal rats exhibited the most severe alveolitis, despite minimal fibrosis (×400). B Compared to the Calca-/- group, immunofluorescence revealed that Calca-/-+D-gal group and WT + D-gal group had a higher degree of Lamp3+Npw+AT2 subtype differentiation and a decreased Ager+Rtkn+AT1 expression (× 400).
Immunohistochemistry showed that αCGRP, PPARγ, CD3+, CD4+, CD8+, CD68+, and TGFβ were primarily localized in the cytoplasm. It was revealed that decreased αCGRP and increased PPARγ in patients vs. controls (Supplemental Fig. 2B). Compared with WT rats, both Calca-/- and WT + D-gal groups exhibited increased infiltration of CD3+CD4+ T lymphocytes, CD68+ macrophages, and elevated expression of TGF β and PPARγ, alongside reduced αCGRP levels. No significant difference was observed in CD8+ T cell levels. Among all groups, the Calca-/-+D-gal group demonstrated the highest levels of CD3+CD4+ cells, the greatest upregulation of PPARγ, and the lowest αCGRP expression (Supplemental Fig. 3A-B). Apoptosis levels were significantly elevated in the Calca-/-, Calca-/-+D-gal, and WT + D-gal groups compared to WT controls (Supplemental Fig. 3A).
Immunofluorescence analysis showed increased differentiation of Lamp3+NPW+AT2 subtypes in both Calca-/-+D-gal and WT + D-gal groups compared to Calca-/- alone, supporting the notion that impaired AT1 regeneration triggers a compensatory AT2 expansion during fibrosis and senescence (Fig. 1B, Fig. 5B).
Measurement of MDA, an oxidative stress marker, revealed that MDA levels were significantly elevated in WT + D-gal, Calca-/-, and Calca-/-+D-gal groups compared to WT controls (p < 0.05). Notably, the Calca-/-+D-gal group showed the highest MDA levels, exceeding those in the WT + D-gal group. These results indicate that both αCGRP deficiency and D-gal treatment induce oxidative stress, while αCGRP may play a protective role by suppressing MDA accumulation (Supplemental Fig. 4).
Label-free proteomics revealed distinct metabolic pathway alterations between WT + D-gal and Calca-/-+D-gal, and Calca-/- and WT + D-gal rats. Differences in metabolic and PPAR signaling pathways were observed between Calca-/- and Calca-/-+D-gal groups. Additionally, changes in oxidative phosphorylation were detected in comparisons between WT and WT + D-gal, Calca-/- and WT, and Calca-/-+D-gal and WT groups. Interestingly, no significant difference in oxidative phosphorylation pathways was found between Calca-/- and WT + D-gal groups (Fig. 6).
A Differences in metabolic pathways were observed between the WT + D-gal group and the Calca-/- + D-gal group; B Differences in metabolic pathways were observed between the Calca-/- group and the WT + D-gal group; C Differences in metabolism and PPAR pathways were observed between the Calca-/- group and the Calca-/-+D-gal group; D Differences in the oxidative phosphorylation pathway were observed between the WT group and the WT + D-gal group; E Differences in the oxidative phosphorylation pathway were observed between the Calca-/- group and the WT group; F Differences in the oxidative phosphorylation pathway were observed between the Calca-/- + D-gal group and the WT group.
These findings suggest that αCGRP deficiency primarily disrupts metabolic and PPAR signaling pathways, while D-gal affects oxidative phosphorylation. Together, both factors contribute to pulmonary inflammation, metabolic dysfunction, AT2 senescence, and fibrosis.
Discussion
The intact alveolar cortex serves as a protective barrier against PF [33]. This study extended prior investigations into the mechanism by which abnormal expression of the PF-associated gene Calca contributes to AT2 cell damage, and further explored the impairment of AT2 regeneration from the perspective of αCGRP deficiency-induced inflammatory senescence. It was established that αCGRP deficiency promotes AT2 inflammatory senescence via activation of oxidative stress and metabolic pathways during PF.
This conclusion was supported by multiple lines of evidence, including characteristic pathological changes such as pulmonary inflammation and alveolar structural damage in PF patients, accompanied by reduced αCGRP expression, elevated PPARγ expression, AT2 subtype differentiation, and hypermethylation of the Calca promoter region in peripheral blood. In Calca-/- rats, pulmonary inflammation, fibrotic remodeling, AT2 subtype differentiation, and increased senescence gene set scores in both lung tissue and AT2 cells were observed. KEGG pathway enrichment analysis of AT2 cells from the Calca-/- group revealed upregulation of linoleic acid metabolism, α-linolenic acid metabolism, arachidonic acid metabolism, fat digestion and absorption. GO biological pathway analysis indicated upregulation of processes such as stem cell proliferation, and replicative senescence.
D-gal treatment triggered pulmonary inflammation and fibrosis, elevated oxidative stress biomarkers, and increased infiltration of CD4+ lymphocytes and CD68+ macrophages, accompanied by upregulation of TGFβ and PPARγ expression. Combined D-gal administration and Calca-/- resulted in more severe alveolitis, the highest levels of oxidative stress, and AT2 subtype differentiation. Label-free proteomic analysis further demonstrated that αCGRP deficiency affects metabolic pathways and oxidative phosphorylation, confirming that αCGRP deficiency promotes AT2 senescence through modulation of PPAR signaling, metabolic dysfunction, and oxidative phosphorylation.
PF is known to be associated with senescence-related pathophysiological mechanisms, including genomic instability, epigenetic alterations, and mitochondrial dysfunction. DNA methylation, in particular, has emerged as a key epigenetic factor in senescence research [34]. The Calca gene consists of six exons and multiple sites for methylation and other epigenetic modifications [35]. A large CpG island and two CpG-rich regions are present in the 5’ promoter, which are typically unmethylated under normal conditions [36]. Abnormal CGRP expression has been associated with transcriptional regulation by DNA methylation, which is also implicated in cellular senescence [37]. Altered methylation in the Calca promoter has been shown to affect cellular stemness [38, 39]. Recent studies have also demonstrated that CGRP inhibits cardiac fibroblast differentiation and reduces collagen deposition [40, 41].
In the study, hypermethylation of the Calca promoter was observed in the peripheral blood in PF patients, alongside reduced αCGRP expression in lung tissue. Similarly, Calca-/- rats developed pulmonary inflammation and fibrotic remodeling, suggesting that Calca hypermethylation during fibrosis may suppress αCGRP expression and contribute to AT cell injury. However, the specific mechanism remains unclear.
PF is also characterized by high levels of ROS resulting from oxidative stress [42]. These ROS contribute to AEC injury and senescence via lipid peroxidation [4]. The regenerative AT2 cell subpopulation is capable of responding to regenerative cues following viral infections, bleomycin exposure, or pulmonary resection, enabling rapid alveolar expansion and regeneration [43,44,45]. Dysfunctional AT2 cells fail to maintain physiological lung regeneration and instead contribute to aberrant epithelial-mesenchymal crosstalk, thereby promoting fibrosis over repair [46].
CGRP has been shown to inhibit ROS production and exert antioxidant effects [47]. During early embryonic stages (E13-E15), CGRP is widely expressed in distal lung epithelial cells, but later becomes restricted to distinct epithelial lineages [48]. The transient expression in progenitor cells may reflect a role in stem cell regulation [48]. Prior studies have shown that CGRP protects AT2 cells from hyperoxia-induced injury [49], while inhibition of CGRP by CGRP8-37 leads to inflammation and alveolar remodeling [11].
Recent studies have indicated that accelerating aging, including the loss of epithelial progenitor cell function and/or numbers and senescence, is one of the key factors in IPF pathogenesis [50,51,52]. Senescence epithelial cells also arise in cystic remodeled areas of chronic autoimmune diseases (UIP/AuD) [53]. Comparison of IPF and SSc-ILD tissue identified a profound loss of alveolar type 1 (AT1) cells in both IPF and SSc-ILD with a distinct transcriptome signature defining each AT1 subset by disease [54]. In this study, alveolar inflammation and AT2 subtype differentiation were observed in both PF patients and Calca-/- rats. scRNA-seq revealed a significant reduction in AECs in the Calca-/- group compared to the WT group (4.21 vs 15.08%). Oxidized lipid metabolism was elevated in AT2 cells, indicating a potential link between oxidative lipid dysregulation and diminished regenerative capacity. AT2 senescence gene set scores and MDA levels were also elevated in Calca-/- rats. These findings support the hypothesis that αCGRP deficiency promotes AT2 senescence by inducing oxidative lipid metabolic dysregulation, ultimately impairing alveolar regeneration.
In the D-gal senescence model, activation of metabolic pathways in AT2 inflammatory senescence was also been observed in our study. Research has shown that the regeneration and differentiation of MSCs are jointly regulated by CGRP and PPARγ [55]. Activation of the PPARγ pathway following lung injury contributes to lipid metabolism disorders, which impair AT2 cell regeneration [56,57,58]. Since metabolic pathways are closely tied to cellular function and energy balance, elevated lipid metabolism has been negatively associated with AT2 proliferation, differentiation, and self-renewal [57]. Notably, AT2 cells must continue lipid production even under nutrient-deficient conditions such as starvation [59].
PPARγ disrupts lipid homeostasis by modulating peroxisomal activity, leading to excessive ROS generation [60]. In PF, PPARγ plays a dual role: in early inflammation, it promotes TGFβ expression and induces AEC apoptosis, thereby contributing to fibrosis; in later stages, it compensates for the suppression of TGFβ transcription [61]. Our previous research demonstrated that αCGRP deficiency promotes type 2 immune responses through PPARγ nuclear translocation, leading to increased TGFβ and AEC injury during fibrosis progression. UPLC-MS/MS metabolomics revealed elevated oxidative lipid metabolites, such as LTB4, PDX, and 12-HETE, and significant activation of the PPAR pathway in Calca-/- mice. Transcriptome and metabolomics data both highlighted the PPAR pathway as a shared mechanism [26]. Building on this, KEGG enrichment showed that Calca-/- AT2 cells upregulated linoleic acid, α-linoleic acid, and arachidonic acid metabolism, while HIF-1 and FoxO signaling were downregulated. GO enrichment further indicated increased stem cell proliferation and replicative senescence.
These findings suggest that αCGRP deficiency disrupts lipid metabolism and oxidative phosphorylation, impairing AT2 differentiation and regeneration. Elevated levels of MDA were found in the D-gal-induced senescence model, although there was no difference in oxidative phosphorylation between the Calca-/- and WT + D-gal groups by label-free proteomic profiling. Thus, αCGRP deficiency alters metabolic and oxidative pathways, promotes AT2 injury and senescence, and contributes to alveolitis development.
However, there are limitations. The low reproductive capacity of knockout mice restricted sample sizes, which may have amplified variability in scRNA-seq and proteomics analysis. Although fibrotic remodeling was limited in the Calca-/-+D-gal group, alveolitis was markedly severe, characterized by heightened infiltration of CD4+ lymphocytes and macrophages. These findings suggest that αCGRP deficiency, combined with D-gal exposure, exacerbates AEC damage and may accelerate fibrosis and impair AT2 regeneration over time. Further research is needed to understand the mechanisms driving this synergy.
In summary, αCGRP may improve oxidative lipid metabolism disorders by inhibiting PPARγ activation, thereby supporting AT2 regeneration, preserving alveolar structure, and reducing collagen deposition during fibrosis.
Conclusion
αCGRP deficiency disrupts lipid homeostasis by modulating the PPAR γ pathway in AT2 cells, inducing metabolic dysfunction, and oxidative phosphorylation. Moreover, αCGRP deficiency triggers AT2 inflammatory senescence, regeneration failure in cells, and persistent alveolar damage, ultimately promoting PF.
Data availability
Not applicable.
References
Wijsenbeek M, Cottin V. Spectrum of fibrotic lung diseases. N Engl J Med. 2020;383:958–68.
Raghu G, Remy-Jardin M, Richeldi L, Thomson CC, Inoue Y, Johkoh T, et al. Idiopathic pulmonary fibrosis (an Update) and progressive pulmonary fibrosis in adults: an official ATS/ERS/JRS/ALAT clinical practice guideline. Am J Respir Crit Care Med. 2022;205:e18–e47.
Parimon T, Yao C, Stripp BR, Noble PW, Chen P. Alveolar epithelial type II cells as drivers of lung fibrosis in idiopathic pulmonary fibrosis. Int J Mol Sci. 2020;21:2269.
Cui H, Xie N, Banerjee S, Dey T, Liu RM, Antony VB, et al. CD38 mediates lung fibrosis by promoting alveolar epithelial cell aging. Am J Respir Crit Care Med. 2022;206:459–75.
Santoro A, Bientinesi E, Monti D. Immunosenescence and inflammaging in the aging process: age-related diseases or longevity? Ageing Res Rev. 2021;71:101422.
Furman D, Campisi J, Verdin E, Carrera-Bastos P, Targ S, Franceschi C, et al. Chronic inflammation in the etiology of disease across the life span. Nat Med. 2019;25:1822–32.
Chaudhary MR, Chaudhary S, Sharma Y, Singh TA, Mishra AK, Sharma S, et al. Aging, oxidative stress and degenerative diseases: mechanisms, complications and emerging therapeutic strategies. Biogerontology. 2023;24:609–62.
Zhang J, Yu H, Man MQ, Hu L. Aging in the dermis: fibroblast senescence and its significance. Aging Cell. 2024;23:e14054.
Orgeig S, Bernhard W, Biswas SC, Daniels CB, Hall SB, Hetz SK, et al. The anatomy, physics, and physiology of gas exchange surfaces: is there a universal function for pulmonary surfactant in animal respiratory structures? Integr Comp Biol. 2007;47:610–27.
Yao C, Guan X, Carraro G, Parimon T, Liu X, Huang G, et al. Senescence of alveolar type 2 cells drives progressive pulmonary fibrosis. Am J Respir Crit Care Med. 2021;203:707–17.
Wang S, Dang H, Xu F, Deng J, Zheng X. The Wnt7b/β-catenin signaling pathway is involved in the protective action of calcitonin gene-related peptide on hyperoxia-induced lung injury in premature rats. Cell Mol Biol Lett. 2018;23:4.
Kuwano K, Kunitake R, Kawasaki M, Nomoto Y, Hagimoto N, Nakanishi Y, et al. P21Waf1/Cip1/Sdi1 and p53 expression in association with DNA strand breaks in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 1996;154:477–83.
Caporarello N, Lee J, Pham TX, Jones DL, Guan J, Link PA, et al. Dysfunctional ERG signaling drives pulmonary vascular aging and persistent fibrosis. Nat Commun. 2022;13:4170.
Caporarello N, Lee J, Pham TX, Jones DL, Guan J, Link PA, et al. Author correction: dysfunctional ERG signaling drives pulmonary vascular aging and persistent fibrosis. Nat Commun. 2022;13:5687.
Rahman I, Skwarska E, Henry M, Davis M, O’Connor CM, FitzGerald MX, et al. Systemic and pulmonary oxidative stress in idiopathic pulmonary fibrosis. Free Radic Biol Med. 1999;27:60–8.
Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91–100.
Russell FA, King R, Smillie SJ, Kodji X, Brain SD. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099–142.
Schou WS, Ashina S, Amin FM, Goadsby PJ, Ashina M. Calcitonin gene-related peptide and pain: a systematic review. J Headache Pain. 2017;18:34.
Schaeffer C, Vandroux D, Thomassin L, Athias P, Rochette L, Connat JL. Calcitonin gene-related peptide partly protects cultured smooth muscle cells from apoptosis induced by an oxidative stress via activation of ERK1/2 MAPK. Biochim Biophys Acta. 2003;1643:65–73.
Nagashima H, Mahlakõiv T, Shih HY, Davis FP, Meylan F, Huang Y, et al. Neuropeptide CGRP limits group 2 innate lymphoid cell responses and constrains type 2 inflammation. Immunity. 2019;51:682–95.e6.
Singh Y, Gupta G, Shrivastava B, Dahiya R, Tiwari J, Ashwathanarayana M, et al. Calcitonin gene-related peptide (CGRP): a novel target for Alzheimer’s disease. CNS Neurosci Ther. 2017;23:457–61.
Chen HF, Yu CY, Chen MJ, Chou SH, Chiang MS, Chou WH, et al. Characteristic expression of major histocompatibility complex and immune privilege genes in human pluripotent stem cells and their derivatives. Cell Transpl. 2015;24:845–64.
Watanabe N, Endo K, Komori K, Ozeki N, Mizuno M, Katano H, et al. Mesenchymal stem cells in synovial fluid increase in knees with degenerative meniscus injury after arthroscopic procedures through the endogenous effects of CGRP and HGF. Stem Cell Rev Rep. 2020;16:1305–15.
Li Z, Meyers CA, Chang L, Lee S, Li Z, Tomlinson R, et al. Fracture repair requires TrkA signaling by skeletal sensory nerves. J Clin Invest. 2019;129:5137–50.
Song H, Yao E, Lin C, Gacayan R, Chen MH, Chuang PT. Functional characterization of pulmonary neuroendocrine cells in lung development, injury, and tumorigenesis. Proc Natl Acad Sci USA. 2012;109:17531–6.
Lv X, Chen Q, Zhang Z, Du K, Huang Y, Li X, et al. αCGRP deficiency aggravates pulmonary fibrosis by activating the PPARγ signaling pathway. Genes Immun. 2023;24:139–48.
Lv X, Gao F, Zhang S, Zhang S, Zhou X, Ding F, et al. Maladjustment of β-CGRP/α-CGRP regulation of AQP5 promotes transition of alveolar epithelial cell apoptosis to pulmonary fibrosis. J Interferon Cytokine Res. 2020;40:377–88.
Raghu G, Remy-Jardin M, Myers JL, Richeldi L, Ryerson CJ, Lederer DJ, et al. Diagnosis of idiopathic pulmonary fibrosis. An Official ATS/ERS/JRS/ALAT clinical practice guideline. Am J Respir Crit Care Med. 2018;198:e44–e68.
Stuart T, Butler A, Hoffman P, Hafemeister C, Papalexi E, Mauck WM, et al. Comprehensive integration of single-cell data. Cell. 2019;177:1888–902.
Hafemeister C, Satija R. Normalization and variance stabilization of single-cell RNA-seq data using regularized negative binomial regression. Genome Biol. 2019;20:296.
Cable DM, Murray E, Zou LS, Goeva A, Macosko EZ, Chen F, et al. Robust decomposition of cell type mixtures in spatial transcriptomics. Nat Biotechnol. 2022;40:517–26.
Raredon MSB, Adams TS, Suhail Y, Schupp JC, Poli S, Neumark N, et al. Single-cell connectomic analysis of adult mammalian lungs. Sci Adv. 2019;5:eaaw3851.
Selman M, Pardo A. The leading role of epithelial cells in the pathogenesis of idiopathic pulmonary fibrosis. Cell Signal. 2020;66:109482.
Gulati S, Thannickal VJ. The aging lung and idiopathic pulmonary fibrosis. Am J Med Sci. 2019;357:384–9.
Bennett MM, Amara SG. Molecular mechanisms of cell-specific and regulated expression of the calcitonin/alpha-CGRP and beta-CGRP genes. Ann N. Y Acad Sci. 1992;657:36–49.
Broad PM, Symes AJ, Thakker RV, Craig RK. Structure and methylation of the human calcitonin/alpha-CGRP gene. Nucleic Acids Res. 1989;17:6999–7011.
Horvath S, Raj K. DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nat Rev Genet. 2018;19:371–84.
Wang L, Ding F, Shi S, Wang X, Zhang S, Song Y. Hypermethylation in calca promoter inhibited ASC osteogenic differentiation in rats with type 2 diabetic mellitus. Stem Cells Int. 2020;2020:5245294.
Sun R, Zhou G, Liu L, Ren L, Xi Y, Zhu J, et al. Fluoride exposure and CALCA methylation is associated with the bone mineral density of Chinese women. Chemosphere. 2020;253:126616.
Li W, Zhang Z, Li X, Cai J, Li D, Du J, et al. CGRP derived from cardiac fibroblasts is an endogenous suppressor of cardiac fibrosis. Cardiovasc Res. 2020;116:1335–48.
Li WQ, Tan SL, Li XH, Sun TL, Li D, Du J, et al. Calcitonin gene-related peptide inhibits the cardiac fibroblasts senescence in cardiac fibrosis via up-regulating klotho expression. Eur J Pharm. 2019;843:96–103.
Bocchino M, Agnese S, Fagone E, Svegliati S, Grieco D, Vancheri C, et al. Reactive oxygen species are required for maintenance and differentiation of primary lung fibroblasts in idiopathic pulmonary fibrosis. PLoS One. 2010;5:e14003.
Zacharias WJ, Frank DB, Zepp JA, Morley MP, Alkhaleel FA, Kong J, et al. Regeneration of the lung alveolus by an evolutionarily conserved epithelial progenitor. Nature. 2018;555:251–5.
Choi J, Park JE, Tsagkogeorga G, Yanagita M, Koo BK, Han N, et al. Inflammatory signals induce AT2 cell-derived damage-associated transient progenitors that mediate alveolar regeneration. Cell Stem Cell. 2020;27:366–82.
Ahmadvand N, Khosravi F, Lingampally A, Wasnick R, Vazquez-Armendariz AI, Carraro G, et al. Identification of a novel subset of alveolar type 2 cells enriched in PD-L1 and expanded following pneumonectomy. Eur Respir J. 2021;58:2004168.
Song L, Li K, Chen H, Xie L. Cell cross-talk in alveolar microenvironment: from lung injury to fibrosis. Am J Respir Cell Mol Biol. 2024;71:30–42.
Hajhashemy Z, Golpour-Hamedani S, Eshaghian N, Sadeghi O, Khorvash F, Askari G. Practical supplements for prevention and management of migraine attacks: a narrative review. Front Nutr. 2024;11:1433390.
Wuenschell CW, Sunday ME, Singh G, Minoo P, Slavkin HC, Warburton D. Embryonic mouse lung epithelial progenitor cells co-express immunohistochemical markers of diverse mature cell lineages. J Histochem Cytochem. 1996;44:113–23.
Dang HX, Li J, Liu C, Fu Y, Zhou F, Tang L, et al. CGRP attenuates hyperoxia-induced oxidative stress-related injury to alveolar epithelial type II cells via the activation of the Sonic hedgehog pathway. Int J Mol Med. 2017;40:209–16.
Demaria M, Ohtani N, Youssef SA, Rodier F, Toussaint W, Mitchell JR, et al. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev Cell. 2014;31:722–33.
Schafer MJ, White TA, Iijima K, Haak AJ, Ligresti G, Atkinson EJ, et al. Cellular senescence mediates fibrotic pulmonary disease. Nat Commun. 2017;8:14532.
Lehmann M, Korfei M, Mutze K, Klee S, Skronska-Wasek W, Alsafadi HN, et al. Senolytic drugs target alveolar epithelial cell function and attenuate experimental lung fibrosis ex vivo. Eur Respir J. 2017;50:1602367.
Gallob F, Brcic L, Eidenhammer S, Rumpp F, Nerlich A, Popper H. Senescence and autophagy in usual interstitial pneumonia of different etiology. Virchows Arch. 2021;478:497–506.
Valenzi E, Tabib T, Papazoglou A, Sembrat J, Trejo Bittar HE, Rojas M, et al. Disparate interferon signaling and shared aberrant basaloid cells in single-cell profiling of idiopathic pulmonary fibrosis and systemic sclerosis-associated interstitial lung disease. Front Immunol. 2021;12:595811.
Li J, Wang Y, Li Y, Sun J, Zhao G. The effect of combined regulation of the expression of peroxisome proliferator-activated receptor-γ and calcitonin gene-related peptide on alcohol-induced adipogenic differentiation of bone marrow mesenchymal stem cells. Mol Cell Biochem. 2014;392:39–48.
Ribeiro Filho HV, Guerra JV, Cagliari R, Batista FAH, Le Maire A, Oliveira PSL, et al. Exploring the mechanism of PPARγ phosphorylation mediated by CDK5. J Struct Biol. 2019;207:317–26.
Li X, Wu J, Sun X, Wu Q, Li Y, Li K, et al. Autophagy reprograms alveolar progenitor cell metabolism in response to lung injury. Stem Cell Rep. 2020;14:420–32.
Su S, Zhao Q, He C, Huang D, Liu J, Chen F, et al. miR-142-5p and miR-130a-3p are regulated by IL-4 and IL-13 and control profibrogenic macrophage program. Nat Commun. 2015;6:8523.
Liu G, Summer R. Cellular metabolism in lung health and disease. Annu Rev Physiol. 2019;81:403–28.
Chen H, Tan H, Wan J, Zeng Y, Wang J, Wang H, et al. PPAR-γ signaling in nonalcoholic fatty liver disease: pathogenesis and therapeutic targets. Pharmacol Ther. 2023;245:108391.
Yoon YS, Kim SY, Kim MJ, Lim JH, Cho MS, Kang JL. PPARγ activation following apoptotic cell instillation promotes resolution of lung inflammation and fibrosis via regulation of efferocytosis and proresolving cytokines. Mucosal Immunol. 2015;8:1031–46.
Acknowledgements
This work was financially supported by the Fujian Provincial Health Technology Project (No. 2023GGA030 to Xiaoting Lv), the Joint Funds for the Innovation of Science and Technology, Fujian Province of Fujian Province (No. 2023Y9043 to Xiaoting Lv), the National Natural Science Foundation of China (No. 81800070 to Xiaoting Lv), and Natural Science Fundation of Fujian Province (No. 2023L3012 to Yiming Zeng).
Author information
Authors and Affiliations
Contributions
Xiaoting Lv and Yiming Zeng planned the project. Xiaoting Lv, Qingquan Chen, Weijing Wu and Yiming Zeng conceived and designed the study. Ziying Zhou and Weijing Wu performed the sample collection. Qingquan Chen, XingLiang Yu and Xiaoting Lv prepared all figures. Weijing Wu and Ziying Zhou performed the expression analysis. Xiaoting Lv analyzed the data and drafted the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
We declare that we have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lv, X., Chen, Q., Zhou, Z. et al. αCGRP deficiency aggravates pulmonary fibrosis by promoting senescence in alveolar type 2 cells. Genes Immun 26, 589–598 (2025). https://doi.org/10.1038/s41435-025-00361-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41435-025-00361-3