Abstract
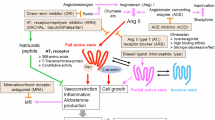
Angiotensin II type 1 receptor (AT1R) is a vital therapeutic target for hypertension. Sorting nexin 1 (SNX1) participates in the sorting and trafficking of the renal dopamine D5 receptor, while angiotensin and dopamine are counterregulatory factors in the regulation of blood pressure. The effect of SNX1 on AT1R is not known. We hypothesized that SNX1, through arterial AT1R sorting and trafficking, is involved in blood pressure regulation. CRISPR/Cas9 system-generated SNX1−/− mice showed dramatic elevations in blood pressure compared to their wild-type littermates. The angiotensin II-mediated contractile reactivity of the mesenteric arteries and AT1R expression in the aortas were also increased. Moreover, immunofluorescence and immunoprecipitation analyses revealed that SNX1 and AT1R were colocalized and interacted in the aortas of wild-type mice. In vitro studies revealed that AT1R protein levels and downstream calcium signaling were upregulated in A10 cells treated with SNX1 siRNA. This may have resulted from decreased AT1R protein degradation since the AT1R mRNA levels showed no changes. AT1R protein was less degraded when SNX1 was downregulated, as reflected by a cycloheximide chase assay. Furthermore, proteasomal rather than lysosomal inhibition increased AT1R protein content, and this effect was accompanied by decayed binding of ubiquitin and AT1R after SNX1 knockdown. Confocal microscopy revealed that AT1R colocalized with PSMD6, a proteasomal marker, and the colocalization was reduced after SNX1 knockdown. These findings suggest that SNX1 sorts AT1R for proteasomal degradation and that SNX1 impairment increases arterial AT1R expression, leading to increased vasoconstriction and blood pressure.
Similar content being viewed by others
Introduction
Hypertension, one of the most common chronic diseases worldwide, has been identified as the leading risk factor for cardiovascular events such as heart attack, heart failure, aneurysm, chronic kidney disease, stroke, cognitive impairment, and dementia [1]. Despite substantial progress has been made in understanding the epidemiology, pathophysiology, and risks associated with hypertension, hypertension and its complications remain heavy burdens on global medical care [2, 3].
Pathophysiological changes in the vasculature have been considered to play a critical role in the development of hypertension [4,5,6]. Blood vessels not only act as mechanical channels through which the blood flows but also exert physiological functions in modulating blood pressure due to a variety of receptors distributed in the vessels. Angiotensin II (Ang II), a crucial effector hormone, causes vasoconstriction and increases blood pressure, while Ang II type 1 receptor (AT1R), a G protein-coupled receptor (GPCR) mediating most of the effects of Ang II, is well known to participate in the process of hypertension. Both AT1R expression and its functions are enhanced in hypertension [7, 8]. Furthermore, AT1R is not only involved in the contraction of vascular smooth muscle cells (VSMCs) [9] but also related to other pathophysiological alterations, such as increased oxidative stress [10, 11], chronic inflammation [12, 13], and cardiovascular remodeling [14, 15].
The balance of GPCR recycling and degradation is pivotal for cellular homeostasis, and its perturbation may lead to impaired homeostatic responses and morbid states such as coronary artery disease and hypertension [16,17,18]. Sorting nexins (SNXs) are a diverse group of cellular sorting proteins characterized by the phox homology domain that can bind to phosphoinositide [19] and are key in orchestrating the processes of protein sorting and trafficking, including endocytosis, endosomal sorting, and signaling [16]. Studies in human renal proximal tubule cells (hRPTCs) and in mice have shown that SNXs exert their physiological effects by regulating the expression and function of GPCRs [20,21,22], and SNX impairment-induced GPCR dysfunction is associated with cardiovascular diseases [16, 23]. Among SNXs, SNX1 was the first mammalian sorting nexin to be characterized and is the ortholog of yeast vacuolar protein-sorting (Vps)5p, a protein involved in trans-Golgi network trafficking [24]. Previous studies have reported that renal SNX1 is involved in blood pressure modulation via sorting and recycling of the dopamine D5 receptor (D5R) back to the cell membrane in hRPTCs [20]. However, whether arterial SNX1 exerts physiological effects on blood pressure regulation is still unknown. Ang II and dopamine are counterregulatory hormones participating in blood pressure control [25, 26]. Thus, we speculate that arterial SNX1 may take part in the sorting and trafficking of AT1R in VSMCs. The aim of the present study was to investigate the regulatory effects of SNX1 on the expression and function of arterial AT1R and its underlying mechanisms.
Methods
Transgenic mice
SNX1-deficient mice on a C57BL/6 background were generated via CRISPR/Cas9-mediated genome engineering by Cyagen (Cyagen Biosciences, Guangzhou, China). The mouse SNX1 gene (GenBank accession number: NM_019727.2) is located on mouse chromosome 9, and exon 1 was selected as the target site. Cas9 mRNA and guide RNAs (gRNAs) (gRNA1: 5′-CCCTGGCATGGATCCGGAGTCGG-3′; gRNA2: 5′-GGATATTTTCACTGGCGCCGCGG-3′) generated by in vitro transcription were injected into fertilized eggs for knockout mouse production. The founders were genotyped by polymerase chain reaction (PCR) followed by DNA sequencing analysis. The founders were bred to the next generation and genotyped by PCR and agarose gel electrophoresis.
Genotyping of Snx1 −/− mice
Genomic DNA was extracted from mouse tail tissue using a mammalian genomic DNA extraction kit (Beyotime, Jiangsu, China). The target DNA was amplified by PCR using specific primers (forward: 5′-CAGCCTTGCGGTTCAGTGCTT-3′; reverse: 5′-AAATGCCCGCTGAATCCTTGG-3′), and PCR was carried out under the following conditions: 1 μL of template at a concentration of 100 ng/μL, 10 μL of GoTaq Green Master Mix (Promega, Madison, WI, USA), 1 μL of forward primer (10 μmol/L, Sangon Biotech, Shanghai, China), 1 μL of reverse primer (10 μmol/L, Sangon Biotech), and nuclease-free water (Promega) were combined in a final volume of 20 μL per reaction.
The PCR protocol consisted of an initial denaturing step at 95 °C for 3 min; 30 cycles of 95 °C for 30 s, 59 °C for 30 s, and 72 °C for 1 min; and a final extension at 72 °C for 5 min in a T100 thermal cycler (Bio-Rad, Hemel Hempstead, UK). The PCR products were then analyzed by 1% agarose gel electrophoresis.
Measurement of blood pressure
The blood pressure of mice was measured by the tail–cuff plethysmography method using a Softron BP-2010A programmed electronic sphygmomanometer (Softron Biotechnology, Beijing, China) as described previously [27,28,29]. Briefly, 10-week-old mice were trained for 1 h to adapt to the environment before blood pressure measurement. The mice were gently placed in a small soft cage without being anesthetized, and their behavior and heart rates were allowed to stabilize for 15 min. The systolic, diastolic, and mean blood pressures were recorded, and the average of ten recorded values was used for further statistical analysis.
Preparation and study of small resistance arteries
Mesenteric artery ring studies were performed according to previous reports [30,31,32]. In brief, second-order mesenteric artery branches were cut into 2 mm rings, and each segment was suspended between two tungsten wires (25 μm in diameter) in the chambers of a Multi Myograph System (DMT-620, Aarhus, Denmark) to measure isometric tension. The chambers were filled with physiological saline solution (PSS) and continuously bubbled with carbogen (95% O2, 5% CO2) at 37 °C [33]. After a 15-min equilibration period, the mesenteric arterial rings were stretched to the optimal luminal diameter for active tension development. In the first set of experiments, rings were contracted with phenylephrine HCl (PHE; 10 μmol/L) and high-potassium PSS (125 mmol/L) to reach the maximum tension and were then rinsed with PSS. Then, different drugs were administered to test the reactivity of the arterial rings, and the response curves were measured with a cumulative concentration-dependent protocol.
Immunohistochemical and immunofluorescence staining of the aortas
Samples of aortas were fixed in 4% paraformaldehyde, dehydrated in increasing concentrations of ethanol, cleared in xylene, and embedded in paraffin. The samples were cut into 4 μm thick sections for further experimentation. Immunohistochemical staining was carried out using a rabbit anti-AT1R antibody (1:100, Proteintech, Wuhan, China). The reactions were detected with horseradish peroxidase-conjugated goat anti-rabbit IgG, the color was developed with 3,3′-diaminobenzidine tetrahydrochloride (Solarbio, Beijing, China), and the reaction was stopped by rinsing in deionized water.
For immunofluorescence staining of aortic sections, we followed a protocol described previously [34]. In brief, the sections were permeabilized and blocked with Immunostaining Blocking/Primary Antibody Dilution Buffer (Sangon Biotech). Primary antibodies (rabbit anti-AT1R, 1:100, Proteintech; goat anti-SNX1, 1:100, Sigma-Aldrich, St. Louis, MO, USA) were added, and the samples were incubated overnight at 4 °C. After rinsing, secondary antibodies (Alexa Fluor 488-labeled goat anti-rabbit IgG, 1:100, Zsbio, Beijing, China; Cy3-conjugated AffiniPure donkey anti-goat IgG, 1:100, Proteintech) were applied for 1 h at room temperature. For negative controls, the samples were incubated with isotype-matched control primary antibodies. In addition, 4′,6-diamidino-2-phenylindole (DAPI) was used to stain the nuclei. Colocalization analysis was performed with the ImageJ Colocalization Finder plugin, and Pearson’s correlation coefficient (Rr) and the overlap coefficient (R) were calculated [35,36,37].
Cell culture
Embryonic thoracic aortic smooth muscle cells (passages 10~20) from normotensive Berlin-Druckrey IX rats (A10; CRL 1476, ATCC) were cultured at 37 °C in 95% air and 5% CO2 in Dulbecco’s modified Eagle’s medium (HyClone, South Logan, UT, USA) supplemented with 10% v/v fetal bovine serum (Invitrogen, Karlsruhe, Germany) and 1% v/v penicillin/streptomycin (Invitrogen).
SNX1 knockdown via small interfering RNA
Small interfering RNA (siRNA) against SNX1 mRNA and its control scrambled siRNA were purchased from Ruibo Biotechnology (Guangzhou, China). The target sequence of Snx1 siRNA (siSNX1) was 5′-GCCTAATAGGAATGACAAA-3′, the sense strand sequence was 5′-GCCUAAUAGGAAUGACAAAdTdT-3′, and the antisense strand sequence was 5′-UUUGUCAUUCCUAUUAGGCdTdT-3′. RNA interference experiments were performed using Lipofectamine RNAiMAX Reagent (Invitrogen) according to the manufacturer’s instructions. Briefly, cells were cultured in six-well plates until 60% confluence, and then 30 pmol siSNX1, scrambled siRNA and negative control were mixed with 9 μL of RNAiMAX in 150 μL of Opti-MEM (Invitrogen). The cells were then incubated for 24 h for mRNA extraction or 48 h for protein extraction.
Immunoblotting
Immunoblotting studies were performed according to procedures described in our previous reports [38, 39]. Briefly, proteins were extracted from mouse aortas or A10 cells with ice-cold Tissue Extraction Reagent (Thermo Fisher Scientific, Waltham, MA, USA) containing a protease inhibitor cocktail (Roche, Indianapolis, IN, USA). Protein samples (50 μg) were separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis and then electrotransferred to nitrocellulose membranes. Ponceau S staining was used as a quality control for transfer. Then, the blocked membranes were incubated at 4 °C overnight with primary antibodies, including a rabbit anti-AT1R antibody (1:500, Proteintech), a rabbit anti-AT2R antibody (1:300, Beyotime), a rabbit anti-D5R antibody (1:500, Proteintech), a rabbit anti-SNX1 antibody (1:1000, Thermo Fisher Scientific), and a mouse anti-GAPDH antibody (1:5000, Proteintech). The blotted membranes were then washed and incubated with an IRDye 800-labeled goat anti-rabbit secondary antibody (1:15000, Li-Cor Biosciences, NE, USA) or an IRDye 680-labeled goat anti-mouse secondary antibody (1:15000, Li-Cor Biosciences) at room temperature for 1 h. The membranes were washed three times in Tris-buffered saline/Tween, and the bands were detected using an Odyssey Infrared Imaging System (Li-Cor Biosciences). The images were analyzed using ImageJ Software to obtain the integrated intensities.
Enzyme-linked immunosorbent assay
The supernatants of cultured A10 cells were collected and centrifuged at 1000 × g at 4 °C for 20 min, and then the levels of Ang II were assessed with an enzyme-linked immunosorbent assay kit (Cloud-Clone Corp., Wuhan, China) according to the manufacturer’s instructions. The absorbance was measured at 450 nm, and the concentration of Ang II was calibrated with the standard curve.
Real-time quantitative polymerase chain reaction
SNX1 and AT1R mRNA levels in mouse aortas or A10 cells were quantified by real-time quantitative polymerase chain reaction (qPCR) with forward and reverse primers (Table 1) following the protocol in our previous study [38]. The following PCR conditions were applied: 95 °C for 3 min; 40 cycles of 95 °C for 10 s and 62 °C for 30 s; and 62 °C for 10 s. Since there are several different transcriptional variants of mouse GAPDH, mouse β-actin was used instead as the internal control.
Immunoprecipitation
Equal amounts of cell lysates (1000 μg total protein) were incubated with an anti-SNX1 antibody (5 μg, Proteintech; SNX1-AT1R immunoprecipitation), an anti-AT1R antibody (5 μg, Proteintech; AT1R-SNX1 immunoprecipitation), or an anti-ubiquitin antibody (5 μg, Proteintech; ubiquitin-AT1R immunoprecipitation) overnight at 4 °C. Then, 50 μL of protein G Plus-agarose beads (Santa Cruz, CA, USA) were added, and the lysates were incubated for another 4 h at 4 °C. The immunoprecipitates were subjected to immunoblotting (with an anti-AT1R antibody or an anti-SNX1 antibody). In addition, a negative control (rabbit IgG) and a positive control (an anti-SNX1 antibody or anti-AT1R antibody) were used as the immunoprecipitants to test the specificity of the bands on the immunoblots.
Confocal microscopy
A10 cells (1 × 104 cells/well) were seeded in 20 mm glass-bottom culture dishes (Nest, Wuxi, China). After incubation with siSNX1 for 48 h, the cells were fixed with 4% paraformaldehyde for 30 min at 4 °C and then permeabilized and blocked with immunostaining blocking buffer (Sangon Biotech). The cells were then incubated overnight at 4 °C with primary antibodies, including a mouse anti-AT1R antibody (1:50, Santa Cruz), a rabbit anti-SNX1 antibody (1:100, Thermo Fisher Scientific), a rabbit anti-PSMD6 antibody (1:100, Sigma-Aldrich), and a rabbit anti-LAMP1 antibody (1:100, Sigma-Aldrich). To check for the colocalization of AT1R and Rab5, a rabbit anti-AT1R antibody (1:100, Proteintech) and a mouse anti-Rab5 (1:50, Santa Cruz) were added. After incubation with the secondary antibody (Alexa Fluor 488-labeled goat anti-rabbit IgG, 1:100, Zsbio; Cy3-conjugated AffiniPure goat anti-mouse IgG, 1:100, Proteintech) and DAPI, the samples were observed under a confocal microscope (Olympus Corporation, Tokyo, Japan). Colocalization analysis was performed with the ImageJ Colocalization Finder plugin. The index for colocalization corresponds to the mean ± standard deviation (SD) of the overlap coefficient (R) × 100 obtained for at least ten cells for each colabeling experiment [37].
Intracellular calcium measurement
Intracellular calcium was labeled with Fluo-4 AM (Beyotime) following the manufacturer’s instructions. Briefly, cells were plated in six-well plates, loaded with 5 µM Fluo-4 AM for 30 min at 37 °C, and then incubated in the dark for another 30 min. The cells were subjected to Ang II (10–7 mol/L) treatment with or without 15 min of losartan (10–5 mol/L) pretreatment [7]. Images were captured and analyzed as described previously [40, 41], and quantifications were performed using ImageJ software.
Statistical analysis
The data are expressed as the mean ± SD values. Statistical significance was tested via ANOVA followed by a post hoc (Dunnett’s) test for multigroup (>2) comparisons, and Student’s t-test was used for two-group comparisons. For assays involving mesenteric arterial rings, comparisons were performed with repeated-measures ANOVA (or paired t-tests when only two groups were compared) [32]. A value of P < 0.05 was considered to indicate statistical significance.
Results
Generation and identification of Snx1 −/− mice
Snx1−/− mice on a C57BL/6 background were generated using CRISPR/Cas9-mediated genome engineering, as illustrated by the schematic diagram in Fig. 1a. The offspring were genotyped by PCR and agarose gel electrophoresis. The homozygous genotype (Snx1−/−) showed a single band of 565 base pairs (bp), the wild-type (WT) genotype (Snx1+/+) showed a single band of 645 bp, and the heterozygous genotype (Snx1+/−) showed bands of both 565 and 645 bp (Fig. 1b). Furthermore, the qPCR and immunoblotting results showed SNX1 mRNA and protein expression in aortas from WT mice but not in aortas from Snx1−/− mice (Fig. 1c, d). The above results suggested that the generation of Snx1−/− mice was successful.
Generation and identification of Snx1−/− mice. a Schematic diagram showing the generation of Snx1−/− mice based on CRISPR/Cas9. b Agarose gel electrophoresis demonstrating genotype determinations using DNA from three individuals representing Snx1−/−, Snx1+/−, and WT mice. c Representative images of agarose gel electrophoresis of qPCR products indicating the expression of SNX1 mRNA in aortas from WT mice but not in aortas from Snx1−/− mice. d Immunoblots showing the expression of SNX1 protein in aortas from WT mice but not in aortas from Snx1−/− mice
Elevated blood pressure and enhanced reactivity of the mesenteric artery to Ang II in Snx1 −/− mice
The Snx1−/− mice showed elevated systolic blood pressure (Fig. 2a), diastolic blood pressure (Fig. 2b), and mean blood pressure (Fig. 2c) compared to their WT littermates, as measured by the tail–cuff method. Due to the important role of vascular contraction or dilation in the regulation of blood pressure, vascular reactivity was next measured. The artery ring studies indicated that there were no obvious differences between Snx1−/− and WT mice in PHE-induced contraction (Fig. 3a) and sodium nitroprusside (SNP)-mediated relaxation (Fig. 3b). However, Ang II produced the most remarkable changes in contraction among the applied drugs, and the Ang II-induced contraction of mesenteric arteries was much stronger in Snx1−/− mice than in WT littermates (Fig. 3c). These results confirmed that both blood pressure and vascular reactivity were elevated in Snx1−/− mice.
Elevated blood pressure in Snx1−/− mice measured using tail–cuff plethysmography. a Systolic blood pressure was higher in Snx1−/− mice than in their wild-type (WT) littermates (122.0 ± 4.2 vs. 99.8 ± 3.3 mm Hg, *P < 0.05 vs. WT, n = 5). b Increased diastolic blood pressure in Snx1−/− mice compared to their WT littermates (69.6 ± 2.8 vs. 59.4 ± 3.1 mm Hg, *P < 0.05 vs. WT, n = 5). c Elevated mean blood pressure in Snx1−/− mice compared to their WT littermates (80.8 ± 3.1 vs. 67.8 ± 2.2 mm Hg, *P < 0.05 vs. WT, n = 5)
Enhanced reactivity of mesenteric arteries to Ang II in Snx1−/− mice. a Phenylephrine (PHE)-induced contractions of mouse mesenteric arteries were not changed in Snx1−/− mice; there was no significant difference between WT and Snx1−/− mice (n = 5). b Sodium nitroprusside (SNP)-induced relaxation of mouse mesenteric arteries was not remarkably changed in Snx1−/− mice (n = 5). c Ang II-induced contractions of mouse mesenteric arteries were increased in Snx1−/− mice (*P < 0.05 vs. WT, n = 5)
Increased AT1R expression in aortas from Snx1 −/− mice
We next investigated the expression of the main receptors of Ang II and found that AT1R protein levels were significantly elevated in the aortas from Snx1−/− mice, as reflected by the immunoblotting results (Fig. 4a). Similarly, immunohistochemical staining of AT1R also suggested that AT1R expression was higher in the aortas of Snx1−/− mice than in those of their WT littermates (Fig. 4b). However, the expression of AT2R, another Ang II receptor, was not different between the Snx1−/− and WT mice (Fig. 4c). In addition, the expression of D5R, another GPCR that has been shown to promote AT1R degradation [42], was not changed in aortas from Snx1−/− mice (Fig. 4d). These results indicate that the expression of AT1R, not AT2R or D5R, is increased in the aortas of Snx1−/− mice. Furthermore, we tested whether SNX1 and AT1R were colocalized in aortas from WT mice via immunofluorescence laser confocal microscopy, and the quantification results indicated that SNX1 and AT1R were colocalized in the tunica media (Fig. 4e). The immunoprecipitation results also suggested an interaction between SNX1 and AT1R in the aortas (Fig. 4f).
Increased AT1R expression in aortas from Snx1−/− mice. a AT1R protein levels were upregulated in aortas from Snx1−/− mice (*P < 0.05 vs. WT, n = 5). b Immunohistochemical staining of AT1R revealed higher expression of AT1R in Snx1−/− mice than in WT mice (these studies were repeated three times; NC negative control; scale bar = 50 μm). c AT2R expression was not changed in the aortas from Snx1−/− mice, as analyzed by immunoblotting (n = 5). d D5R expression was not changed in the aortas from Snx1−/− mice, as analyzed by immunoblotting (n = 3). e Colocalization of AT1R and SNX1 detected by immunofluorescence laser confocal microscopy. Representative images suggesting the colocalization of AT1R and SNX1 in aortic sections from WT mice (scale bar = 50 μm) (e1). Colocalization was quantitatively analyzed with the ImageJ Colocalization Finder plugin, and Pearson’s correlation coefficient (Rr) and the overlap coefficient (R) were calculated (e2). f Immunoprecipitation assay results indicating the interaction between AT1R and SNX1 in the aortas from WT mice (these studies were repeated three times; NC negative control; PC positive control)
Upregulated AT1R protein levels in SNX1-knockdown A10 cells
In vitro studies in A10 cells also confirmed that the protein expression of AT1R was upregulated after SNX1 knockdown via specific siRNA (Fig. 5a). Moreover, the calcium signal, which can be activated by stimulation of AT1R that then causes VSMC contraction [9, 43], was elevated in siSNX1-treated A10 cells in the basal state and increased more significantly after Ang II treatment. However, under conditions of pretreatment with losartan, an AT1R antagonist, the elevation in calcium concentration was abolished (Fig. 5b). Furthermore, to effectively demonstrate the role of SNX1 in the regulation of intracellular AT1R trafficking, the endosomal marker Rab5 was used. We assessed the colocalization between Rab5 and AT1R, and the quantified results from confocal microscopy revealed an increase in the AT1R mean fluorescence intensity but a decrease in the colocalization of Rab5 and AT1R in siSNX1-treated A10 cells (Fig. 5c). In addition, to determine whether the changed AT1R expression and its mediated functions were associated with the ligand Ang II, we assessed the content of Ang II in supernatants and found that it was not altered after SNX1 knockdown (Fig. 5d). Moreover, immunoprecipitation indicated the interaction between AT1R and SNX1 in A10 cells (Fig. 5e). These results suggest that SNX1 has marked effects on AT1R expression, AT1R trafficking, and AT1R-mediated signaling but no effect on Ang II production.
Upregulated AT1R protein levels in SNX1-knockdown A10 cells. a AT1R protein levels were upregulated after SNX1 knockdown, as indicated by immunoblotting studies (*P < 0.05 vs. control, n = 3). b Intracellular calcium concentrations were increased in SNX1-knockdown A10 cells, as revealed by representative images of Fluo-4 AM fluorescence (b1) and the statistical results (b2) (scale bar = 20 μm, *P < 0.05 vs. control, n = 3). c Colocalization of AT1R and Rab5, as detected by immunofluorescence laser confocal microscopy. Representative images revealing the colocalization of AT1R and Rab5 in control and siSNX1-treated A10 cells (scale bar = 20 μm) (c1). The AT1R mean fluorescence intensity was quantified with ImageJ (*P < 0.05 vs. control, n = 10 cells) (c2). Colocalization was quantitatively analyzed with the ImageJ Colocalization Finder plugin, and the index of colocalization corresponds to the mean ± standard deviation (SD) of the overlap coefficient (R) × 100 (*P < 0.05 vs. control, n = 10 cells) (c3). d Ang II concentrations in the supernatants of cultured A10 cells were not changed after SNX1 knockdown, as determined by enzyme-linked immunosorbent assay (ELISA) (n = 3). e Immunoprecipitation analysis of the interaction between SNX1 and AT1R in A10 cells (these studies were repeated three times; NC negative control; PC positive control)
Involvement of the proteasome pathway in SNX1-mediated AT1R protein degradation
To further explore the underlying mechanism by which AT1R protein levels were increased, qPCR was performed, and the results showed that the AT1R mRNA level was not changed when SNX1 was knocked down (Fig. 6a). However, AT1R protein was less degraded in siSNX1-treated A10 cells than in control cells at different time points in the presence of cycloheximide (500 μmol/L) [44], which inhibited de novo protein synthesis (Fig. 6b). This indicated that the upregulation of AT1R protein levels resulted from reduced degradation rather than increased mRNA expression. To explore the pathway of SNX1-mediated AT1R protein degradation, we added chloroquine (100 μmol/L) [45] or clasto-lactacystin beta-lactone (CLBL, 10 μmol/L) [46] to inhibit the activity of lysosomes or proteasomes, respectively. We found no change in the AT1R content after chloroquine and cycloheximide cotreatment, but the AT1R content was markedly increased after CLBL and cycloheximide cotreatment, and the difference was more pronounced in siSNX1-treated A10 cells than in control cells (Fig. 6c). In addition, immunoprecipitation assays showed that there was an interaction between ubiquitin and AT1R, which was decreased after SNX1 knockdown (Fig. 6d). Moreover, the results from immunofluorescence laser confocal microscopy revealed that AT1R mainly colocalized with PSMD6, a proteasomal marker, rather than LAMP1, a lysosomal marker, in the cytoplasm of A10 cells, and the colocalization of AT1R and PSMD6 was reduced after SNX1 knockdown (Fig. 6e). These results suggest that SNX1 knockdown decelerates AT1R protein degradation and that the mechanism may be, at least in part, associated with the proteasome pathway.
Involvement of the proteasome pathway in SNX1-mediated AT1R protein degradation. a AT1R mRNA levels, as determined by qPCR, were not changed after SNX1 knockdown (*P < 0.05 vs. control, n = 3). b Degradation of AT1R protein was reduced when SNX1 was knocked down, as reflected by immunoblotting. The cells were incubated with cycloheximide (500 μmol/L) for the indicated times. The results are expressed as the percentage change in each group from the control group value (*P < 0.05 vs. n = 3). c Proteasome inhibition, but not lysosome inhibition, increased AT1R protein expression, and the increase was more pronounced in siSNX1-treated A10 cells than in control cells (500 μmol/L cycloheximide was coadministered; CHX cycloheximide; Chl chloroquine, a lysosomal inhibitor, 100 μmol/L; CLBL clasto-lactacystin beta-lactone, a proteasomal inhibitor, 10 μmol/L; *P < 0.05 vs. control, n = 3). d Immunoprecipitation analysis indicated a decayed interaction between AT1R and ubiquitin when SNX1 was knocked down. e Representative images from immunofluorescence laser confocal microscopy and the quantification of colocalization indicated that AT1R mainly colocalized with PSMD6 (a proteasomal marker) rather than LAMP1 (a lysosomal marker), and the colocalization of AT1R and PSMD6 was reduced after SNX1 knockdown. Colocalization was quantitatively analyzed with the ImageJ Colocalization Finder plugin, and the index of colocalization corresponds to the mean ± standard deviation (SD) of the overlap coefficient (R) × 100 (scale bar = 20 μm, *P < 0.05 vs. control, n = 10 cells)
Discussion
The present study suggests that SNX1 plays an important role in the regulation of arterial AT1R expression and function, as deficiency of SNX1 in mice leads to increased AT1R protein expression and enhanced AT1R protein function in arteries. Mechanistically, SNX1 interacts with AT1R and participates in AT1R sorting and trafficking to the proteasome for its degradation in A10 cells. Thus, SNX1 is involved in the regulation of blood pressure.
SNX1 was originally identified as a sorting protein that interacts with cytoplasmic sequences, including the tyrosine kinase domain and the adjacent lysosomal targeting signal, of epidermal growth factor receptor (EGFR) and is vital in the degradation of EGFR [47, 48]. More importantly, in hRPTCs, SNX1 initiates the sorting of ligand-activated D5R at the plasma membrane by tagging the receptor for endocytosis, which has a crucial effect on D5R trafficking; further studies in mice have found that renal-specific SNX1 depletion via renal subcapsular infusion of siSNX1 results in a blunted natriuretic response to D5R stimulation and elevated blood pressure [20]. Our present study shows that compared with their WT littermates, Snx1−/− mice exhibit elevations in systolic blood pressure, diastolic blood pressure, and mean blood pressure. It is a limitation that blood pressure was measured by the noninvasive tail–cuff method. However, the results were consistent with those of our previous study, in which blood pressure was assessed by telemetry [49], a more accurate and reliable but invasive technique [50]. These findings all suggest that SNX1 plays a vital role in the regulation of blood pressure. However, our previous study revealed increased renal oxidative stress and impaired natriuretic effects in the kidneys of Snx1−/− mice [49] but did not investigate the role of SNX1 in the regulation of blood vessels, which also contributes to hypertension. Thus, in our present study, we explored the effects of SNX1 on vascular tone and its intrinsic mechanisms.
There are multiple humoral systems related to high blood pressure, among which the renin-angiotensin system (RAS) has attracted the most attention. Ang II is classically considered the main mediator of RAS and exerts its effects via two major distinct GPCRs: AT1R and AT2R. Most Ang II actions are mediated by AT1R, which is involved in the development of hypertension via increases in vasoconstriction [9, 51], renal sodium reabsorption [52], induction of vasopressin release [53], and facilitation of sympathetic nerve activity [54]. Thus, the regulation of AT1R expression and function is of great significance for the prevention and treatment of hypertension. However, little attention has been paid to the sorting and trafficking of AT1R and their effects on blood pressure. Our current study focused on the pivotal role of SNX1 in AT1R sorting and explored the underlying mechanisms, and the findings might indicate a novel aspect of blood pressure regulation. Our studies in small resistance arteries revealed that among the applied drugs, Ang II strongly induced contraction in Snx1−/− mice. This might be ascribed to upregulated AT1R expression because the AT1R protein levels were increased in both aortas from Snx1−/− mice and SNX1-knockdown A10 cells. Similar results have been found in previous studies, which have shown elevated AT1R expression in siSNX1-treated mouse kidneys [20], kidneys from Snx1−/− mice [49], and siSNX1-transfected hRPTCs [20]. Furthermore, to evaluate the function of AT1R in siSNX1-treated A10 cells, we tested the concentrations of intracellular calcium, which could be increased by stimulation of AT1R and then cause VSMC contraction [43, 55], as described in our previous investigation [7]. We found that the concentrations were significantly elevated in siSNX1-treated A10 cells in the basal state and that the difference was more pronounced after Ang II treatment. More importantly, the elevation could be blocked by pretreatment with losartan, an inhibitor of AT1R. These results uncover a crucial role of SNX1 in the regulation of AT1R expression and function in VSMCs. In addition, SNX1 depletion led to increased AT1R protein levels and elevated Ang II-mediated mesenteric artery contractility, which further contributed to hypertension. However, one of the limitations of our present study was lack of losartan-blocking experiments in vivo or ex vivo, which needs to be addressed in future studies.
We next investigated the mechanisms by which SNX1 regulates AT1R expression and found that AT1R protein but not mRNA levels were elevated after SNX1 knockdown in A10 cells, suggesting that SNX1 may be involved in the process of AT1R protein degradation. The cycloheximide chase assay, a classical assay used to analyze protein degradation [56, 57], further revealed that AT1R protein expression was increased in siSNX1-treated A10 cells after incubation with cycloheximide [44], indicating that AT1R protein degradation is reduced after SNX1 knockdown. Moreover, immunofluorescence laser confocal microscopy and immunoprecipitation assays indicated spatial colocalization and physical interactions in aortas and A10 cells, respectively. Since colocalization analysis has often been conducted in a rather ad hoc fashion and since the results could be subject to misinterpretation and inconsistencies [58,59,60], we tried to quantify colocalization via ImageJ as indicated in previous reports [35,36,37]. It should be noted that Förster resonance energy transfer and proximity ligation assay are more reliable methods than ImageJ analysis to investigate protein–protein interactions, so more attention should be paid to these methods in future investigations. In vitro studies also showed that the colocalization of AT1R and Rab5 was reduced in siSNX1-treated A10 cells, which indicated the role of SNX1 in AT1R sorting. SNX1 has also been found to modulate the functions of renal D5R, which mediates renal tubular AT1R degradation [20, 42]. Thus, we further explored the expression of aortic D5R in Snx1−/− mice but found no obvious changes. Therefore, we propose that the regulation of aortic AT1R by SNX1 might be independent of D5R.
Proteasomes and lysosomes represent the two most important proteolytic machineries in cells [61, 62]. The activity of GPCRs can be tightly controlled by GPCR endocytosis, which drives the receptors into divergent lysosomal and recycling pathways [63]; the signaling proteins can also undergo regulated ubiquitination in response to GPCR activation, and the regulated ubiquitination can cause proteasomal degradation [64]. AT1R has been reported to be degraded through the ubiquitin-proteasome pathway mediated by activation of D5R in hRPTCs [42]. This has also been confirmed by another study showing that stimulation of D5R, not D1R, leads to degradation of AT1R in RPTCs and that this effect can be completely blocked by CLBL, a proteasome inhibitor [46]. Studies have shown that SNXs are involved in protein degradation; for example, SNX1 has been suggested to sort protease-activated receptor-1 from early endosomes to lysosomes for degradation [65]. In our study, proteasomal inhibition, rather than lysosomal inhibition, increased AT1R expression in A10 cells; the increase was more pronounced after siSNX1 knockdown and was accompanied by decreased interaction between ubiquitin and AT1R. We also found that AT1R was mainly colocalized with PSMD6, a proteasomal marker, rather than LAMP1, a lysosomal marker, in the cytoplasm of A10 cells, and that the colocalization of AT1R and PSMD6 decayed after SNX1 knockdown. These results indicate that SNX1-mediated AT1R degradation occurs mainly through the proteasomal pathway.
In conclusion, our data reveal a crucial role of SNX1 in AT1R sorting and degradation in VSMCs. Depletion of SNX1 results in increased AT1R-mediated mesenteric artery contractility, which may represent a novel mechanism for the regulation of blood pressure.
References
Zhou B, Danaei G, Stevens GA, Bixby H, Taddei C, Carrillo-Larco RM, et al. Long- term and recent trends in hypertension awareness, treatment, and control in 12 high- income countries: an analysis of 123 nationally representative surveys. Lancet. 2019;394:639–51.
Ettehad D, Emdin CA, Kiran A, Anderson SG, Callender T, Emberson J, et al. Blood pressure lowering for prevention of cardiovascular disease and death: a systematic review and meta-analysis. Lancet. 2016;387:957–67.
Forouzanfar MH, Liu P, Roth GA, Ng M, Biryukov S, Marczak L, et al. Global burden of hypertension and systolic blood pressure of at least 110 to 115 mm Hg, 1990-2015. JAMA. 2017;317:165–82.
Bohr DF, Dominiczak A, Webb RC. Pathophysiology of the vasculature in hypertension. Hypertension. 1991;18:III69.
Brown IAM, Diederich L, Good ME, DeLalio LJ, Murphy SA, Cortese-Krott MM, et al. Vascular smooth muscle remodeling in conductive and resistance arteries in hypertension. Arterioscler Thromb Vasc Biol. 2018;38:1969–85.
Laurent S, Boutouyrie P. The structural factor of hypertension: large and small artery alterations. Circ Res. 2015;116:1007–21.
Chen K, Fu C, Chen C, Liu L, Ren H, Han Y, et al. Role of GRK4 in the regulation of arterial AT1 receptor in hypertension. Hypertension. 2014;63:289–96.
Wang Z, Zeng C, Villar VAM, Chen S-Y, Konkalmatt P, Wang X, et al. Human GRK4γ 142V variant promotes angiotensin II type I receptor–mediated hypertension via renal histone deacetylase type 1 inhibition. Hypertension. 2016;67:325–34.
Wynne BM, Chiao C-W, Webb RC. Vascular smooth muscle cell signaling mechanisms for contraction to angiotensin II and endothelin-1. J Am Soc Hypertens. 2009;3:84–95.
Nickenig G, Harrison DG. The AT1-type angiotensin receptor in oxidative stress and atherogenesis: part I: oxidative stress and atherogenesis. Circulation. 2002;105:393–6.
Vaziri ND, Bai Y, Ni Z, Quiroz Y, Pandian R, Rodriguez-Iturbe B. Intra-renal angiotensin II/AT1 receptor, oxidative stress, inflammation, and progressive injury in renal mass reduction. J Pharm Exp Ther. 2007;323:85–93.
Sanz-Rosa D, Oubina MP, Cediel E, de las Heras N, Vegazo O, Jiménez J, et al. Effect of AT1 receptor antagonism on vascular and circulating inflammatory mediators in SHR: role of NF-κB/IκB system. Am J Physiol-Heart Circulatory Physiol. 2005;288:H111–5.
Smith GR, Missailidis S. Cancer, inflammation and the AT1 and AT2 receptors. J Inflamm. 2004;1:3.
Williams B. Angiotensin II and the pathophysiology of cardiovascular remodeling. Am J Cardiol. 2001;87:10–17.
Takemoto M, Egashira K, Tomita H, Usui M, Okamoto H, Kitabatake A, et al. Chronic angiotensin-converting enzyme inhibition and angiotensin II type 1 receptor blockade: effects on cardiovascular remodeling in rats induced by the long-term blockade of nitric oxide synthesis. Hypertension. 1997;30:1621–7.
Yang J, Van Anthony MV, Rozyyev S, Jose PA, Zeng C. The emerging role of sorting nexins in cardiovascular diseases. Clin Sci. 2019;133:723–37.
Zhang H, Huang T, Hong Y, Yang W, Zhang X, Luo H, et al. The retromer complex and sorting nexins in neurodegenerative diseases. Front aging Neurosci. 2018;10:79.
Teasdale RD, Collins BM. Insights into the PX (phox-homology) domain and SNX (sorting nexin) protein families: structures, functions and roles in disease. Biochem J. 2012;441:39–59.
Worby CA, Dixon JE. Sorting out the cellular functions of sorting nexins. Nat Rev Mol Cell Biol. 2002;3:919.
Van Anthony MV, Jones JE, Armando I, Asico LD, Escano CS, Lee H, et al. Sorting nexin 1 loss results in D5 dopamine receptor dysfunction in human renal proximal tubule cells and hypertension in mice. J Biol Chem. 2013;288:152–63.
Villar VAM, Armando I, Sanada H, Frazer LC, Russo CM, Notario PM, et al. Novel role of sorting nexin 5 in renal D1 dopamine receptor trafficking and function: implications for hypertension. FASEB J. 2013;27:1808–19.
Li F, Yang J, Jones JE, Villar VAM, Yu P, Armando I, et al. Sorting nexin 5 and dopamine D1 receptor regulate the expression of the insulin receptor in human renal proximal tubule cells. Endocrinology. 2015;156:2211–21.
Drake MT, Shenoy SK, Lefkowitz RJ. Trafficking of G protein–coupled receptors. Circ Res. 2006;99:570–82.
Carlton J, Bujny M, Peter BJ, Oorschot VM, Rutherford A, Mellor H, et al. Sorting nexin-1 mediates tubular endosome-to-TGN transport through coincidence sensing of high-curvature membranes and 3-phosphoinositides. Curr Biol. 2004;14:1791–1800.
Zeng C, Jose PA. Dopamine receptors: important antihypertensive counterbalance against hypertensive factors. Hypertension. 2011;57:11–17.
Gildea JJ. Dopamine and angiotensin as renal counter regulatory systems controlling sodium balance. Curr Opin Nephrol Hypertens. 2009;18:28.
Xiao J, Liu J, Lio I, Yang C, Chen X, Zhang H, et al. All-trans retinoic acid attenuates the progression of Ang II-induced abdominal aortic aneurysms in ApoE−/−mice. J Cardiothorac Surg. 2020;15:1–9.
Lin P, Wu M, Qin J, Yang J, Ye C, Wang C. Magnesium lithospermate B improves renal hemodynamics and reduces renal oxygen consumption in 5/6th renal ablation/infarction rats. BMC Nephrol. 2019;20:49.
Cheng Z, Zhang M, Hu J, Lin J, Feng X, Wang S, et al. Cardiac specific Mst1 deficiency inhibits ROS mediated JNK signalling to alleviate Ang II induced cardiomyocyte apoptosis. J Cell Mol Med. 2019;23:543–55.
Mulvany MJ, Halpern W. Contractile properties of small arterial resistance vessels in spontaneously hypertensive and normotensive rats. Circ Res. 1977;41:19–26.
Yao Y, Wang W, Li M, Ren H, Chen C, Wang J, et al. Curcumin exerts its anti-hypertensive effect by down-regulating the AT 1 receptor in vascular smooth muscle cells. Sci Rep. 2016;6:25579.
Fu J, Han Y, Wang J, Liu Y, Zheng S, Zhou L, et al. Irisin lowers blood pressure by improvement of endothelial dysfunction via AMPK Akt eNOS NO pathway in the spontaneously hypertensive Rat. J Am Heart Assoc. 2016;5:e003433.
Fu J, Han Y, Wang H, Wang Z, Liu Y, Chen X, et al. Impaired dopamine D 1 receptor- mediated vasorelaxation of mesenteric arteries in obese Zucker rats. Cardiovasc Diabetol. 2014;13:50.
Wu LP, Gong ZF, Wang H, Zhou ZS, Zhang MM, Liu C, et al. TSPO ligands prevent the proliferation of vascular smooth muscle cells and attenuate neointima formation through AMPK activation. Acta Pharm Sin. 2020;41:34–46.
Xu H, Zhang H, Liu G, Kong L, Zhu X, Tian X, et al. Coumarin-based fluorescent probes for super-resolution and dynamic tracking of lipid droplets. Anal Chem. 2019;91:977–82.
Bigarella CL, Borges L, Costa FF, Saad ST. ARHGAP21 modulates FAK activity and impairs glioblastoma cell migration. Biochim Biophys Acta. 2009;1793:806–16.
Han JM, Jeong SJ, Park MC, Kim G, Kwon NH, Kim HK, et al. Leucyl-tRNA synthetase is an intracellular leucine sensor for the mTORC1-signaling pathway. Cell. 2012;149:410–24.
Liu C, Hu YH, Han Y, Wang YB, Zhang Y, Zhang XQ, et al. MG53 protects against contrast-induced acute kidney injury by reducing cell membrane damage and apoptosis. Acta Pharmacol Sin. 2020. https://doi.org/10.1038/s41401-020-0420-8.
Liu C, Chen K, Wang H, Zhang Y, Duan X, Wang H, et al. Gastrin attenuates renal ischemia/reperfusion injury by a PI3K/Akt/Bad-mediated anti-apoptosis signaling. Front Pharmacol. 2020;11:1672.
Gavet O, Pines J. Progressive activation of CyclinB1-Cdk1 coordinates entry to mitosis. Dev Cell. 2010;18:533–43.
Jin SW, Choi CY, Hwang YP, Kim HG, Kim SJ, Chung YC, et al. Betulinic acid increases eNOS phosphorylation and no synthesis via the calcium-signaling pathway. J Agric Food Chem. 2016;64:785–91.
Li H, Armando I, Yu P, Escano C, Mueller SC, Asico L, et al. Dopamine 5 receptor mediates Ang II type 1 receptor degradation via a ubiquitin-proteasome pathway in mice and human cells. J Clin Investig. 2008;118:2180–9.
Do KH, Kim MS, Kim JH, Rhim B-Y, Lee WS, Kim CD, et al. Angiotensin II-induced aortic ring constriction is mediated by phosphatidylinositol 3-kinase/L-type calcium channel signaling pathway. Exp Mol Med. 2009;41:569.
Wang X, Zhao Y, Zhang X, Badie H, Zhou Y, Mu Y, et al. Loss of sorting nexin 27 contributes to excitatory synaptic dysfunction by modulating glutamate receptor recycling in Down’s syndrome. Nat Med. 2013;19:473.
Mangieri LR, Mader BJ, Thomas CE, Taylor CA, Luker AM, Tse TE, et al. ATP6V0C knockdown in neuroblastoma cells alters autophagy-lysosome pathway function and metabolism of proteins that accumulate in neurodegenerative disease. PLoS ONE. 2014;9:e93257.
Gildea JJ, Wang X, Jose PA, Felder RA. Differential D1 and D5 receptor regulation and degradation of the angiotensin type 1 receptor. Hypertension. 2008;51:360–6.
Kurten RC, Cadena DL, Gill GN. Enhanced degradation of EGF receptors by a sorting nexin, SNX1. Science. 1996;272:1008–10.
Kurten RC, Eddington AD, Chowdhury P, Smith RD, Davidson AD, Shank BB. Self- assembly and binding of a sorting nexin to sorting endosomes. J Cell Sci. 2001;114:1743–56.
Yang J, Asico LD, Beitelshees AL, Feranil JB, Wang X, Jones JE, et al. Sorting nexin 1 loss results in increased oxidative stress and hypertension. FASEB J. 2020. https://doi.org/10.1096/fj.201902448R.
Wilde E, Aubdool AA, Thakore P, Baldissera L Jr, Alawi KM, Keeble J, et al. Tail-cuff technique and its influence on central blood pressure in the mouse. J Am Heart Assoc. 2017;6:e005204.
Arun K, Kaul C, Ramarao P. High glucose concentration augments angiotensin II mediated contraction via AT1 receptors in rat thoracic aorta. Pharm Res. 2004;50:561–8.
Navar LG, Harrison-Bernard LM, Imig JD, Wang C-T, Cervenka L, Mitchell KD. Intrarenal angiotensin II generation and renal effects of AT1 receptor blockade. J Am Soc Nephrology. 1999;10:S266–72.
Qadri F, Culman J, Veltmar A, Maas K, Rascher W, Unger T. Angiotensin II-induced vasopressin release is mediated through alpha-1 adrenoceptors and angiotensin II AT1 receptors in the supraoptic nucleus. J Pharm Exp Ther. 1993;267:567–74.
Zheng H, Li Y-F, Wang W, Patel KP. Enhanced angiotensin-mediated excitation of renal sympathetic nerve activity within the paraventricular nucleus of anesthetized rats with heart failure. Am J Physiol Regul Integr Comp Physiol. 2009;297:R1364–74.
Zhang Z, Li M, Lu R, Alioua A, Stefani E, Toro L. The angiotensin II type 1 receptor (AT1R) closely interacts with large conductance voltage- and Ca2+-activated K+ (BK) channels and inhibits their activity independent of G-protein activation. J Biol Chem. 2014;289:25678–89.
Kao SH, Wang WL, Chen CY, Chang YL, Wu YY, Wang YT, et al. Analysis of protein stability by the cycloheximide chase assay. Bio Protoc. 2015;5:e1374.
Buchanan BW, Lloyd ME, Engle SM, Rubenstein EM. Cycloheximide chase analysis of protein degradation in Saccharomyces cerevisiae. J Vis Exp. 2016. https://doi.org/10.3791/53975.
Comeau JW, Costantino S, Wiseman PW. A guide to accurate fluorescence microscopy colocalization measurements. Biophys J. 2006;91:4611–22.
Shulei W, Arena ET, Eliceiri KW, Ming Y. Automated and robust quantification of colocalization in dual-color fluorescence microscopy: a nonparametric statistical approach. IEEE Trans Image Process. 2018;27:622–36.
Bolte S, Cordelières FP. A guided tour into subcellular colocalization analysis in light microscopy. J Microsc. 2006;224:213–32.
Wang X, Robbins J. Proteasomal and lysosomal protein degradation and heart disease. J Mol Cell Cardiol. 2014;71:16–24.
Ciechanover A. The ubiquitin-proteasome proteolytic pathway. Cell. 1994;79:13–21.
Hanyaloglu AC, Zastrow MV. Regulation of GPCRs by endocytic membrane trafficking and its potential implications. Annu Rev Pharm Toxicol. 2008;48:537–68.
Wojcikiewicz RJ. Regulated ubiquitination of proteins in GPCR-initiated signaling pathways. Trends Pharm Sci. 2004;25:35–41.
Wang Y, Zhou Y, Szabo K, Haft CR, Trejo J. Down-regulation of protease-activated receptor-1 is regulated by sorting nexin 1. Mol Biol Cell. 2002;13:1965–76.
Acknowledgements
This study was supported in part by grants from the National Natural Science Foundation of China (31730043, 81570379), the National Key R&D Program of China (2018YFC1312700), the Program of Innovative Research Team by the National Natural Science Foundation (81721001), the Program for Changjiang Scholars and Innovative Research Team in University (IRT1216), The Third Affiliated Hospital of Chongqing Medical University (KY19024), and the National Institutes of Health (R01-DK039308, P01-HL074940).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, C., Li, X., Fu, J. et al. Increased AT1 receptor expression mediates vasoconstriction leading to hypertension in Snx1−/− mice. Hypertens Res 44, 906–917 (2021). https://doi.org/10.1038/s41440-021-00661-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41440-021-00661-x
Keywords
This article is cited by
-
SNX16 aggravates AngII-induced cardiac hypertrophy in mice via EGFR transactivation
Communications Biology (2025)
-
Role of the renin-angiotensin pathway in epilepsy: a strategy for its management by drug repurposing
Molecular Biology Reports (2025)
-
Update on Hypertension Research in 2021
Hypertension Research (2022)
-
ATRAP, a receptor-interacting modulator of kidney physiology, as a novel player in blood pressure and beyond
Hypertension Research (2022)
-
New insight into increased angiotensin II type 1 receptor expression in Snx1−/− mice
Hypertension Research (2021)