Abstract
In Aotearoa New Zealand, zoster vaccine live is used for the prevention of zoster and associated complications in adults. This study assessed the risk of pre-specified serious adverse events following zoster vaccine live immunisation among adults in routine clinical practice. We conducted a self-controlled case series study using routinely collected national data. We compared the incidence of serious adverse events during the at-risk period with the control period. Rate ratios were estimated using Conditional Poisson regression models. Falsification outcomes analyses were used to evaluate biases in our study population. From April 2018 to July 2021, 278,375 received the vaccine. The rate ratio of serious adverse events following immunisation was 0·43 (95% confidence interval [CI]: 0·37–0·50). There was no significant increase in the risk of cerebrovascular accidents, acute myocardial infarction, acute pericarditis, acute myocarditis, and Ramsay–Hunt Syndrome. The herpes zoster vaccine is safe in adults in Aotearoa New Zealand.
Similar content being viewed by others
Introduction
Varicella zoster virus causes two distinct diseases, varicella (primary varicella-zoster virus infection) occurring predominantly in children1, and herpes zoster, commonly known as shingles, which occurs following reactivation of the latent varicella zoster virus2.
In Aotearoa New Zealand, the zoster vaccine live, a single dose live attenuated vaccine, was licenced on 1st April 2018 for use in adults ≥ 50 years and funded for those 65 years old (catchup cohort: 66–80 years old, up to 31st December 2021)3. The zoster vaccine lives to boost cell-mediated immunity and significantly reduces the risk and severity of herpes zoster and associated complications4. The efficacy and effectiveness of the zoster vaccine live against herpes zoster and associated complications have been demonstrated in meta-analyses of randomised control trials and observational studies5,6,7,8,9,10,11.
The safety of the herpes zoster vaccine has been demonstrated in prelicensure clinical trials and post-licensure observational studies12, including inoculation site and systemic adverse events8. The relationship between serious adverse events (cardiovascular and cerebrovascular diseases, meningitis, encephalopathy, severe autoimmune diseases and disseminated varicella-zoster virus infection) has been investigated in post-licensure observational studies13,14,15,16. However, none of these studies have assessed the risk of serious adverse events (requiring hospitalisation) using whole country electronic health record data. Only two studies have investigated the safety of the zoster vaccine live in people with immunosuppressive conditions (haematologic malignancy and haematopoietic cell transplant)17, and immunosuppressant drugs18. Also, to our knowledge, the safety of the zoster vaccine live has not been evaluated in Aotearoa New Zealand.
We assessed the risk of pre-specified serious adverse events among Aotearoa New Zealand adults who received the zoster vaccine live between 1st April 2018 and 24th July 2021. Post-licensure national observational studies offer a unique opportunity to assess rare adverse outcomes requiring a large sample size.
Results
Baseline characteristics of the study population
From April 2018 to July 2021, 278,375 adults were vaccinated with the zoster vaccine live. The mean age was 71.1 (standard deviation = 5.0). Of these, 161,217 (58.9%) were ≥70 years old, 145,360 (52.2%) were female, 225,707 (81.1%) were European, and 16,964 (6.1%) had immunocompromising conditions. During the study period, 16,081 (5.8%) Māori and 9170 (3.3%) Pacific people who received the zoster vaccine lived.
A total of 1082 (0.4%) vaccinated people were hospitalised for a pre-specified adverse event of interest during the at-risk (n = 321) and control (n = 761) periods and were included in the study population. The mean age of the study population with an adverse event was 73.4 (standard deviation = 5.0; range: 58–92), and 809 (74.8%) were ≥70 years old. Furthermore, 59.3% (n = 642) of these people were males, 72.0% (n = 779) were European, and 8.6% (n = 93) were immunocompromised (Table 1). Most of the study participants (24.5%) were from areas with the most deprived scores.
Pre-specified adverse events
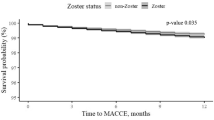
Overall, 1431 serious adverse events occurred during the study period (Fig. 1 and Table S6). After assessment against the inclusion criteria, 1130 pre-specified adverse events were identified during 124,741 person-days of follow-up. There were 320 and 810 serious adverse events in the at-risk (40,775 person-days) and control (64,204 person-days) periods, respectively. The rate ratio of serious adverse events following immunisation was 0.43 (0.37–0.50) (Table 2).
A total of 278,375 adults were vaccinated during the study period (April 2018 to July 2021). A total of 1082 people developed pre-specified serious adverse events during the follow-up period and were included in the analysis. Group 1: Stroke, Cerebrovascular diseases; Group 2: Cardiovascular events; Group 3: Meningitis, encephalitis and encephalopathy; Group 4: Ramsay–Hunt Syndrome, Bell’s Palsy; Group 5: Medically attended reactions.
There were 488 cases with a group one adverse event (cerebrovascular diseases) identified during the study period. The rate ratio was 0.46 (95% CI: 0.36–0.56). There was no increase in the risk of stroke (haemorrhage or infarction) during the at-risk period (134 events during 17,677 person days) compared to the control period (352 cases during 28,642 person-days of follow-up): rate ratio = 0.45 (95% CI: 0.36–0.56). The rate ratio for transient cerebral ischaemia was 2.07 (95% CI: 0.08–56.10).
Cardiovascular diseases were the most common pre-specified adverse events requiring hospitalisation (group two). Overall, 182 cardiovascular events occurred during 22,970 person-days of follow-up in the at-risk period compared to 454 events in 35,367 person-days (control period). The rate ratio was 0.41 (95% CI: 0.34–0.50). The rate ratios for acute myocardial infarction, acute pericarditis and acute myocarditis were 0.28 (95% CI: 0.16–0.47), 0.44 (95% CI: 0.12–1.33) and 0.21 (95% CI: 0.01–2.12), respectively.
No group three adverse events (meningitis, encephalitis and encephalopathy) were identified during the entire follow-up period, and the rate ratio was therefore not estimated.
There were six serious adverse events for group four (Ramsay–Hunt syndrome, Bell’s palsy and facial palsy) which occurred during the follow-up period (116 person-days). There was no difference in the risk of group four adverse events when comparing the at-risk and control periods (rate ratio = 0.79 (95% CI: 0.11–5.68).
There were no reported medically attended reactions (the group five serious adverse events—cellulitis and infections, and allergic reactions) during the follow-up period, and the rate ratio was not estimated.
Falsification outcomes
The incidence of negative controls in the at-risk period was compared to the control window (Table 3). These negative controls should not be associated with herpes zoster and zoster vaccine live vaccination (adverse events following immunisation). There was no difference in incidence between the at-risk and control periods for diverticulitis (rate ratio = 0.65; 0.08–3.78); femoral fractures (rate ratio = 1.51; 0.42–5.19); and cholecyst and pancreatic diseases (rate ratio = 4.38; 0.99–22.15).
Discussion
In this self-controlled case series study using routinely collected national data, we found no increased risk of serious adverse events (requiring in-patient hospitalisation) within the at-risk period following zoster vaccine live vaccination in adults in Aotearoa New Zealand.
These results are consistent with previous pre- and post-licensure safety studies of the zoster vaccine live. In a network meta-analysis of randomised controlled trials involving 103,899 adults 50 years and older, Tricco et al. found no increased risk (risk ratio = 1.08; 95% CI: 0.91–1.30) of serious adverse events (requiring hospitalisation) within 30 days of herpes zoster vaccination5. An updated Cochrane review (including 24 randomised controlled trials and 88,551 adults ≥ 60 years) found no statistically significant difference in the risk of serious adverse events between the vaccinated and placebo groups (risk ratio = 1.08; 95% CI: 0.95–1.21)8. A network meta-analysis of 21 randomised controlled trials by McGir et al., showed that there was no statistically significant difference in the risk of serious adverse events following vaccination between the vaccine and placebo groups for any formulation (subcutaneous. intramuscular and refrigerated) of the zoster vaccine live6. In a long-term follow-up study, the risk of serious adverse events, hospitalisation and death were similar in the vaccinated and placebo groups12. A 2019 GRADE analysis of 21 randomised controlled trials and seven observational studies confirmed that there were no severe adverse events associated with zoster vaccine live vaccination19. The rate of serious adverse events in the vaccinated and placebo groups was similar.
A cohort study involving 29,000 people ≥ 60 years who received the zoster vaccine live from July 2006 to November 2007 found no increased risk of serious adverse events (resulting in hospitalisation) within 42 days post-vaccination14. Serious pre-specified adverse events assessed using electronic diagnostic codes and review of medical records were coronary atherosclerosis and other heart diseases (relative risk = 1·79; 95% CI: 0.69–4.51), acute myocardial infarction (relative risk = 1.29; 95% CI: 0.66–2.43); and stroke (relative risk = 0.91; 95% CI: 0.43–1.81). Using a case-centred study and SCCS designs (electronic diagnostic code and review of medical records), Tseng et al., examined a large cohort of 193,083 adults ≥ 50 years old for increased risk of pre-specified adverse events following zoster vaccine live vaccination13. In both study designs, they observed no difference in risk of serious adverse events between the at-risk and control windows: cerebrovascular events (relative risk = 0.99; 95 CI: 0.83–1.19); meningitis, encephalitis and encephalopathy (relative risk = 0.78; 95% CI: 0.39–1.56); Ramsey–Hunt syndromes and Bell’s palsy (relative risk = 0.78; 95% CI: 0.29–2.09); and cardiovascular diseases (relative risk = 0.99; 95% CI: 0.86–1.14). In a recent SCCS study in Australia involving 150,054 adults 70–79 years old (vaccinated between November 2016 and July 2018), there was no increased risk of serious adverse events within 42 days following zoster vaccine live inoculation: stroke (relative incidence = 0.58; 95% CI: 0.44–0.78); myocardial infarction (relative incidence = 0.74; 95% CI: 0.41–1.33); and medical attendance (relative incidence = 0.94; 95% CI: 0.94–0.95). In the sensitivity analysis, burns (relative incidence = 1.23; 95% CI: 0.97–1.57) were used as a negative control to assess the risk of bias16. Active surveillance of 17,000 adults 70–79 years using a short message service (self-reported) found no increased risk of adverse events (rate ratio = 1.05; 95% CI: 0.93–1.18) and medical attendance (rate ratio = 0.98; 95% CI: 0.50–1.91) when zoster vaccine live was concomitantly administered with other vaccines20. In a case–control study using the Vaccine Adverse Event Reporting System, Lai and Yew found no significant association between autoimmune adverse events and zoster vaccine live vaccination except arthritis (odds ratio = 2·7; 95% CI: 1.7–4.3), and alopecia (odds ratio = 2.2; 95% CI: 1.2–4.3)15.
Evidence from epidemiological studies has shown the risk of myocardial infarction and stroke, and transient ischaemic attack significantly increased in the first month following the reactivation of the varicella-zoster virus and decreased with time since infection. The risk of stroke is two to three times higher than the baseline level when reactivation occurs in the ophthalmic branch of the fifth cranial (trigeminal) nerve21. A recent systematic review and meta-analysis and a nationwide retrospective matched observational study found that the zoster vaccine live effectively prevented herpes zoster hospitalisation in people with cerebrovascular disease and heart disease9,10. In the current study, we identified a large (>50%) protective effect of the zoster vaccine live for most of the outcomes (stroke, acute myocardial infarction and heart failure) in the first 42 days following zoster vaccine live vaccination. A significantly reduced risk of stroke (relative incidence = 0.58; 95% CI: 0.44–0.78) following the zoster vaccine live immunisation has been reported in a recent self-controlled case series study16. The main difference between our findings and numerous antecedent studies is that measure of association for the pre-specified adverse events was closer to one, or the confidence interval of the measure crossed one5,6,8,13,16,22. The apparent protective effect observed in our study could be attributed to the zoster vaccine’s live effectiveness against herpes zoster and associated complications, the healthy vaccine effect, delayed serious adverse events following vaccination, and healthy vaccine bias and healthy vaccinee time14,16. Although a previous study9 comparing the vaccinated (from which the current sample was selected) and the unvaccinated population found that both cohorts were comparable, further investigations are required to explore the significantly reduced risk of these outcomes following zoster vaccination.
Using representative national data, we have provided evidence that the zoster vaccine live was safe in the older New Zealand population (including immunocompromised). The SCCS method was used to assess the risk of pre-specified adverse severe events requiring hospitalisation. This study design controlled for measurable but not measured and unmeasurable confounding factors (e.g. sex, ethnicity, body mass index, smoking, genetics, frailty, level of immunosuppression and severity of co-morbidities) since comparisons were within the same individuals (each case was its control)23. The strength of this approach is that it minimises biases introduced when comparing vaccinated and unvaccinated cohorts and has, using nationally linked health record datasets, been previously used to assess the safety of COVID-19 vaccines24,25,26. We used a Conditional Poisson regression model with offset for the time at risk. We assumed that serious adverse events were independent, vaccine uptake was high, exposure was transient and the time at-risk post-vaccination was short23. Since events are independent, we were able to assess multiple outcomes with varying observation periods. In the falsification outcome analysis, multiple negative controls were used to assess bias in our study population16. In the selection process for negative controls, further consideration was made to try and identify controls which have a similar severity and prevalence to the pre-specified adverse events27. There was a possible borderline increase in the risk of cholecyst and pancreatic diseases. However, the confidence intervals were wide and crossed 1.0, so we are still satisfied that our study is not subject to bias. We used pre-existing Ministry of Health databases, and the data are considered accurate and complete. We excluded all adverse events registered on the index date except anaphylaxis, as these were likely to have begun prior to zoster vaccine live vaccination13.
We assumed the registration date in the National Immunisation Registry was the date the zoster vaccine live was administered (index date). Pre-specified serious adverse events were identified using the ICD-10-AM-iii diagnostic codes in electronic medical records. The positive predictive value of ICD codes compared to the review of the medical records is ≥83%28,29. This may have led to some underestimation of outcomes and a spurious association between adverse events and zoster vaccine live vaccination30. Misclassification and miscoding of adverse events and co-morbidities are inherent limitations of observational studies that use electronic medical record data16.
We also assumed the zoster vaccine live was administered independently. In Australia’s MedicineInsight programme, 92,857 adults 70–79 years received the zoster vaccine live from November 2016 to July 2018. Zoster vaccine live was administered independently (82%) and concomitantly with inactivated influenza vaccine (16%) and 23-valent pneumococcal vaccine (2%). There was no significant difference in risk of adverse events when the zoster vaccine live was modelled independently or with other vaccines16. The serious adverse events observed during the study period may be associated with other vaccines.
In New Zealand, the zoster vaccine live is approved for use in adults ≥ 50 years and used as part of the vaccination programme at age 65 from April 2018 to August 2022. The current guidelines preferentially recommend the recombinant zoster vaccine for the prevention of herpes zoster and postherpetic neuralgia in adults (≥50 years old) and immunocompromised people (≥18 years old). Two doses of the recombinant zoster vaccine were funded for adults 65 years old3.
In conclusion, we found that the zoster vaccine live vaccination was not associated with serious adverse events (requiring hospitalisation) in New Zealand adults (including people with immunocompromising conditions). Subsequent research should systematically assess all adverse events following immunisation by linking general practice to Ministry of Health databases. Comprehensive active surveillance should be used for monitoring participants post-vaccination to identify rare serious adverse events.
Methods
Ethics and permissions
An ethics exemption (21/NTB/118) was obtained from the Health and Disability Ethics Committee of the Ministry of Health, New Zealand. After review and discussion by the Health and Disability Ethics Committees, the reviewers were satisfied that despite the large datasets being linked, there would be no way of identifying individuals, and the risk was minimal.
Study setting
We used routinely collected national data available at the Ministry of Health (Table S1). The National Health Index was used to link data extracted from multiple datasets. Clinical, discharge and demographic information of hospitalised patients (in-patients and day patients in public and private hospitals) were obtained from the national minimum datasets31. Detailed clinical and pathological data (morphology code, basis of diagnosis, laboratory code and extent of disease) about cancer and demographic information of patients with malignancy were extracted from the New Zealand cancer registrations32. The national immunisation register (NIR) is an accurate electronic medical record that contains immunisation information on childhood and adult vaccines administered in New Zealand33. Detailed vaccine-related information (vaccine, event ID, event status description, vaccination date, indication, description and vaccine dose) was extracted from the NIR. The Pharmaceutical Collection is an electric record of all pharmaceuticals funded by the government and dispensed in the community. We extracted information on antiviral drugs and immunosuppressive drugs (Table S2)34. These datasets are exhaustive and have been used for research, public health surveillance and policy formulation31.
Study population
In step one, we identified all individuals who had completed herpes zoster vaccination (a single dose intramuscular injection of 0.65 mL Zostavax®) between 1st April 2018 and 24th July 2021 and were born in 1971 or earlier.
In step two, we identified all individuals who had a publicly funded hospital discharge where the date of admission fell within 42 days (inclusive) of the herpes zoster vaccination date (index date), and there was any diagnosis of any of the pre-specified outcomes in Table S4. We considered the date of admission as the day of onset of the adverse event. For each master NHI number identified in step two, we eliminated all publicly funded hospital discharges with an admission date that falls within one year of the admission date of the index event (except for group five adverse events). This was to ensure that the adverse event identified was the first event in 12 months (except for group five, where we considered the first event in 30 days)13.
For each master encrypted NHI number with a completed herpes zoster vaccination identified in step two, we extracted all publicly funded hospital discharges with an admission date that falls within 73 and 162 days (inclusive) of the vaccination date. For each master NHI number, we excluded all publicly funded hospital discharges with an admission date that fell within one year of the admission date of the index event (except group five adverse events). In scenarios where a person had more than one index event, each index event had its year of retrospective data included.
The final study population comprised individuals with the outcome of interest that occurred during the follow-up (predefined observation) period. The follow-up period is the time after vaccination when we monitored the vaccinated cohort for any pre-specified adverse events and was divided into the at-risk (1–42 days) and the control (73–162 days) periods. This study design has the advantage of controlling for all time–fixed individual-level confounding variables as comparisons are within the same individual rather than between vaccinated and unvaccinated cohorts23,35,36. For each outcome of interest, we noted the event ID, start date, clinical code and diagnosis type (primary or secondary diagnosis).
Study design
A self-controlled case series (SCCS) study design was used to estimate the risk of pre-specified serious adverse outcomes following herpes zoster vaccination (Table S3)23. SCCS is bidirectional; hence the control window can be pre or post-vaccination35. The control interval (background level) and post-vaccination period (risk interval) within each person were derived for every serious adverse event (requiring hospitalisation) and used to classify an individual as unexposed or exposed23. We assessed multiple independent pre-specified serious adverse events in the same individuals and acute adverse outcomes that occurred immediately post-vaccination37.
The at-risk period was a duration of 42 days (days 1–42) following receipt of the zoster vaccine live vaccination (index date) (Fig. 2). Pre-specified adverse events that occurred on the day of vaccination (index day; Day 0) were excluded from the analyses. The at-risk period of 42 days is consistent across pre- and post-licensure studies that assessed the safety of herpes zoster vaccines13,14,16,38. A 30-day washout interval (day 42 to day 72) was included to reduce the probability of spillover of vaccine-attributable risks into the control window35. The post-risk period (post-vaccination self-comparison window) was 90 days, beginning on day 73 following vaccination. The post-risk period was set at 90 days to increase the chance of capturing relevant adverse events. Also, 90 days of follow-up compensated for the absence of a pre-risk period. In a previous study conducted by Baxter et al. in 2012, the rate of all diagnoses of pre-specified adverse events of interest listed for hospitalisations and emergency department visits occurring in a 42-day risk period (days 1–42) following vaccination with herpes zoster live was compared to that in a 90-day postvaccination comparison period (days 91–180 post-vaccination), using vaccinees as their own controls14. Also, the control period of 90 days has been used in recent studies to assess adverse events associated with live attenuated and trivalent inactivated influenza vaccination36 and to evaluate the associations between exposure to ChAdOx1 or BNT162b2 vaccination and haematological and vascular outcomes using a nested incident-matched case-control study and a confirmatory self-controlled case series analysis25.
Each vaccinated person served as their own control, hence eliminating the need for a separate control group. The follow-up period was divided into the at-risk and control (postvaccination comparison or baseline) periods. We introduced a washout period of 30 days to eliminate the carryover effects hence the outcomes during the control period were not influenced by the previous exposure (vaccination).
Pre-specified adverse events
Serious adverse events associated with herpes zoster vaccination were defined using the International Statistical Classification of Diseases and Related Health Problems, Tenth Revision, Australian Modification (ICD-10-AM-iii)39 and extracted from the electronic medical records (NMDS). These outcomes (Table S4) were divided into five main clinically related groups: Group one: Cerebrovascular diseases (occlusion and stenosis, transient cerebral ischaemia, acute and another ill-defined cerebrovascular disease, and haemorrhage or Infarction); Group two: Cardiovascular diseases (acute myocardial infarction, acute pericarditis, acute myocarditis, cardiomyopathy and heart failure); Group three: meningitis, encephalitis and encephalopathy, Group four: Ramsay–Hunt Syndrome and Bell’s palsy (geniculate herpes zoster, herpetic geniculate ganglionitis, Bell’s palsy and facial palsy) and Group five: medically attended reactions (cellulitis and infection and allergic reactions)13. The selection and classification of these serious adverse events were based on previous studies and biological plausibility12,13,14,16,21.
Falsification outcomes
In the self-controlled case series, only vaccinated people who developed the pre-specified adverse event during the follow-up period were included in the analysis. The follow-up period was divided into the at-risk window and the control (baseline) period. Since the duration of follow-up is short, baseline characteristics such as co-morbidity, sex, prioritised ethnicity and index of deprivation were self-controlled40,41. Differences might exist between the at-risk and control windows in terms of time-varying residual confounders. We assessed for bias in our study population by comparing the incidence of possible causes of hospitalisation (not previously associated with herpes zoster and related complications, i.e., negative controls) in the at-risk window to the control period9,42,43. These negative controls (appendicitis, hernia, diverticulitis, femoral fracture, cholelithiasis and cholecystitis, pancreatic diseases, sepsis, haemorrhoids, renal calculi, burns, lipoma and epistaxis) were extracted from the electronic database (NMDS) (Table S5).
Statistical analysis
Proportions were used to describe the baseline characteristics of the study population. These baseline characteristics included age, sex, ethnicity, index of deprivation (Table S1), location (District Health Board) and immunosuppression (immune-compromising diseases and immunosuppressive therapy; Table S2). A single case contributed to exposed (at-risk period) and unexposed (control period) person-time. The incidence rate of an adverse event in the at-risk window (code = 1) was compared to that in the control interval (code = 0). The incidence in the at-risk window was estimated as the number of pre-specified adverse severe events occurring while individuals contributed to the respective at-risk period divided by the total person-days contributed to the at-risk period. The incidence in the control period was computed as the number of pre-specified adverse severe events occurring during the control period divided by the total person-days contributed to the control period (this was considered the baseline level).
Conditional Poisson and conditional logistic regression models were used to derive rate ratios and 95% CI for pre-specified adverse events with offset for person-time. Rate ratios were estimated at the level of groups (clinically related adverse events) because of the small number of serious adverse events per diagnostic code. This aggregation of diagnostic codes has been used in previous studies13. The analyses were carried out by J.M. and A.W. and independently checked by B.N. and S.B. All statistical analyses were carried out using R/R Studio (version R-4.0.5) and Python (version 3.7.12)44,45.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data used in this study are based on de-identified national clinical records and will not be made publicly available. Access is controlled for privacy, confidentiality and legal issues (under the provisions of the New Zealand Privacy Act 2020, the New Zealand Health Information Privacy Code 2020 and any other legislation specifically referred to by the Ministry in the supply of this information). These are, however, available by application via the Ministry of Health New Zealand. Details can be obtained by contacting the Analytical Services team (data-enquiries@health.govt.nz), with the specification of the information required. The team will acknowledge the request and respond within 3–5 working days. It will take 3–4 weeks from receiving written confirmation to fulfil the request. The data agreement stipulates that the information provided is to be used for the purposes set out in the proposal/documentation provided to the New Zealand Ministry of Health (the Ministry) and is for your internal use only. It should not be made available to any other party without the Ministry’s prior consent; this includes requests from academic journals for the supply of research datasets. The only exceptions to this would be the dissemination of data through research documents and/or statistical publications46.
Code availability
All code used in this study is publicly available at Vaccine-studies47.
References
Gershon A. A., Marin M. & Seward J. F. Varicella Vaccines. in (eds Plotkin S. A., Orenstein W. A., Offit P. A., Edwards K. M.). Plotkin’s Vaccines. 7th ed. (Elsevier; 2018). p. 1145–1180.e17.
Levin M. J. Zoster Vaccines. in (eds Stanley Plotkin, Walter Orenstein, Paul Offit, Kathryn M. Edwards). Plotkin’s Vaccines. 7th ed. 19103-2899: (Elsevier, Philadelphia, PA, 2018). p. 1268–1281.
Ministry of Health. Immunisation Handbook 2020 New Zealand: Ministry of Health. https://www.health.govt.nz/our-work/immunisation-handbook-2020/23-zoster-herpes-zoster-shingles (2022).
Oxman, M. N. et al. A vaccine to prevent herpes zoster and postherpetic neuralgia in older adults. N. Engl. J. Med. 352, 2271–2284 (2005).
Tricco, A. C. et al. Efficacy, effectiveness, and safety of herpes zoster vaccines in adults aged 50 and older: systematic review and network meta-analysis. Br. Med. J. 363, k4029 (2018).
McGirr, A. et al. The comparative efficacy and safety of herpes zoster vaccines: a network meta-analysis. Vaccine 37, 2896–909 (2019).
Mbinta, J. F., Nguyen, B. P., Awuni, P. M. A., Eme, P. E. & Simpson, C. R. Postlicensure herpes zoster vaccine effectiveness: systematic review protocol. BMJ Open 11, e040964 (2021).
Gagliardi, A. M. Z. et al. Vaccines for preventing herpes zoster in older adults. Cochrane Database Syst. Rev. 3, CD008858 (2019).
Mbinta, J. F. et al. Herpes zoster vaccine effectiveness against herpes zoster and postherpetic neuralgia in New Zealand: a retrospective cohort study. The Lancet 31, 100601 (2023).
Mbinta, J. F., Nguyen, B. P., Awuni, P. M. A., Paynter, J. & Simpson, C. R. Post-licensure zoster vaccine effectiveness against herpes zoster and postherpetic neuralgia in older adults: a systematic review and meta-analysis. The Lancet 3, e263–e75 (2022).
Mbinta, J. et al. Zoster vaccine effectiveness against zoster and postherpetic neuralgia in New Zealand: a retrospective cohort study. Aust. J. Prim. Health iii–lxxi 28 (2022).
Simberkoff, M. S. et al. Safety of herpes zoster vaccine in the shingles prevention study: a randomized trial. Ann. Intern. Med. 152, 545–554 (2010).
Tseng, H. F. et al. Safety of zoster vaccine in adults from a large managed-care cohort: a vaccine safety datalink study. J. Intern. Med. 271, 510–520 (2012).
Baxter, R. et al. Safety of Zostavax™—a cohort study in a managed care organization. Vaccine 30, 6636–6641 (2012).
Lai, Y. C. & Yew, Y. W. Severe autoimmune adverse events post herpes zoster vaccine: a case-control study of adverse events in a national database. J. Drugs Dermatol. 14, 681–684 (2015).
Totterdell, J. et al. Safety of live attenuated herpes zoster vaccine in adults 70–79 years: a self-controlled case series analysis using primary care data from Australia’s MedicineInsight program. Vaccine 38, 3968–79 (2020).
Naidus, E., Damon, L., Schwartz, B. S., Breed, C. & Liu, C. Experience with use of Zostavax® in patients with hematologic malignancy and hematopoietic cell transplant recipients. Am. J. Hematol. 87, 123–125 (2012).
Cheetham, T. C. et al. Risk of herpes zoster and disseminated varicella zoster in patients taking immunosuppressant drugs at the time of zoster vaccination. Mayo Clin. Proc. 90, 865–873 (2015).
Advisory Committee on Immunization Practices. Grading of recommendations, assessment, development, and evaluation (GRADE): recombinant zoster vaccine (RZV) and herpes zoster live-attenuated vaccine (ZVL). (US Department of Health and Human Services, CDC, Advisory Committee on Immunization Practices, Atlanta, GA, 2017) (2019).
Phillips, A. et al. Safety of live attenuated herpes zoster vaccine in Australian adults 70–79 years of age: an observational study using active surveillance. BMJ Open 11, e043880 (2021).
Wu, P. H., Chuang, Y. S. & Lin, Y. T. Does herpes zoster increase the risk of stroke and myocardial infarction? A comprehensive review. J. Clin. Med. 8, 547 (2019).
Hamad, M. A., Allam, H., Sulaiman, A., Murali, K. & Cheikh Hassan, H. I. Systematic review and meta-analysis of herpes zoster vaccine in patients with CKD. Kidney Int. Rep. 6, 1254–64 (2021).
Glanz, J. M. et al. Four different study designs to evaluate vaccine safety were equally validated with contrasting limitations. J. Clin. Epidemiol. 59, 808–818 (2006).
Farrington, C. P. Control without separate controls: evaluation of vaccine safety using case-only methods. Vaccine 22, 2064–2070 (2004).
Simpson, C. R. et al. First-dose ChAdOx1 and BNT162b2 COVID-19 vaccines and thrombocytopenic, thromboembolic and hemorrhagic events in Scotland. Nat. Med. 27, 1290–1297 (2021).
Simpson, C. R. et al. Second-dose ChAdOx1 and BNT162b2 COVID-19 vaccines and thrombocytopenic, thromboembolic and hemorrhagic events in Scotland. Nat. Commun. 13, 4800 (2022).
Schuemie, M. J. et al. Vaccine safety surveillance using routinely collected healthcare data—an empirical evaluation of epidemiological designs. Front. Pharmacol. 13, 893484 (2022).
Yawn, B. P., Wollan, P. & St. Sauver, J. Comparing shingles incidence and complication rates from medical record review and administrative database estimates: how close are they? Am. J. Epidemiol. 174, 1054–1061 (2011).
Tseng, H. F. & Sy, L. S. Use of real-world evidence to evaluate the effectiveness of herpes zoster vaccine. J. Infect. Dis. 218, S63–S7 (2018).
Nørgaard, M., Ehrenstein, V. & Vandenbroucke, J. P. Confounding in observational studies based on large health care databases: problems and potential solutions—a primer for the clinician. Clin. Epidemiol. 9, 185–93 (2017).
Ministry of Health. National Minimum Dataset (hospital events). https://www.health.govt.nz/nz-health-statistics/national-collections-and-surveys/collections/national-minimum-dataset-hospital-events (2021).
Ministry of Health. New Zealand Cancer Registry—what is collected. https://www.health.govt.nz/nz-health-statistics/national-collections-and-surveys/collections/new-zealand-cancer-registry-nzcr/new-zealand-cancer-registry-what-collected (2021).
Ministry of Health. National Immunisation Register. https://www.health.govt.nz/our-work/preventative-health-wellness/immunisation/national-immunisation-register (2021).
Ministry of Health. Pharmaceutical Collection. https://www.health.govt.nz/nz-health-statistics/national-collections-and-surveys/collections/pharmaceutical-collection (2021).
Baker, M. A. et al. A vaccine study design selection framework for the postlicensure rapid immunization safety monitoring program. Am. J. Epidemiol. 181, 608–618 (2015).
Simpson, C. R. et al. Vaccine effectiveness of live attenuated and trivalent inactivated influenza vaccination in 2010/11 to 2015/16: the SIVE II record linkage study. Health Technol. Assess. 24, 1–66 (2020).
Farrington, C. P. Relative incidence estimation from case series for vaccine safety evaluation. Biometrics 51, 228–235 (1995).
Oxman, M. N. Zoster vaccine: current status and future prospects. Clin. Infect. Dis. 51, 197–213 (2010).
Elsworthy, A. M. et al. ICD-10-AM: the international statistical classification of diseases and related health problems, 10th revision, Australian Modification: Tabular List of Diseases. (2013).
Petersen, I., Douglas, I. & Whitaker, H. Self controlled case series methods: an alternative to standard epidemiological study designs. Br. Med. J. 354, i4515 (2016).
Farrington, P., Whitaker, H. & Weldeselassie, Y. G. Self-controlled case series studies: a modelling guide with R. (Chapman and Hall/CRC, 2018).
Tseng, H. F. et al. Herpes zoster vaccine in older adults and the risk of subsequent herpes zoster disease. J. Am. Med. Assoc. 305, 160–166 (2011).
Tseng, H. F. et al. Herpes zoster vaccine and the incidence of recurrent herpes zoster in an immunocompetent elderly population. J. Infect. Dis. 206, 190–196 (2012).
R Core Team. R: A Language and Environment for Statistical Computing. (R Foundation for Statistical Computing, Vienna, Austria, 2019).
Rossum G. V. & Drake F. L. Python 3 Reference Manual. (CreateSpace, ScottsValley, CA, 2009).
Ministry of Health. Current Data Access Policy. https://www.health.govt.nz/publication/current-data-access-policy (2011).
Mbinta, J. et al. Safety of zoster vaccine in the older New Zealand population: a self-controlled case series. Zenodo https://zenodo.org/record/8022695 (2023).
Acknowledgements
A Wellington Doctoral Scholarship was awarded to J.F.M. The sponsors of the study had no role in the study design, data collection, data analysis, data interpretation or the writing of this report. There was no input to the methodology and results of this study by any commercial entity.
Author information
Authors and Affiliations
Contributions
J.F.M., B.P.N., J.P. and C.R.S. conceived the idea of the study and developed the protocol. All authors reviewed the protocol. J.F.M., A.W., S.B. and B.P.N. cleaned and analysed the data. J.F.M., B.P.N., R.P., S.B., A.A.S. and C.R.S. interpreted the data. J.F.M. wrote the first draft of the paper with input from J.P., B.P.N., P.M.A.A., A.A.S. and C.R.S. All authors read and commented critically on drafts of the paper. All authors approved the final version. C.R.S. supervised the entire work and is the guarantor.
Corresponding authors
Ethics declarations
Competing interests
We declare that one or more of the authors have a competing interest as defined by Nature Portfolio. Professor Simpson reports grants from the UK National Institute for Health Research, the UK Medical Research Council, the NZ Health Research Council and the Ministry of Business Innovation and Employment. Dr Nguyen reports grants from the NZ Ministry of Business Innovation and Employment. Dr Janine Paynter reports grants (2021) and consulting fees (2019) from GlaxoSmithKline (all grants and consulting fees were institutional), the NZ Health Research Council, the NZ Ministry of Health and a travel grant from GAVI, The Vaccine Alliance. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Kenneth McCall and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mbinta, J.F., Wang, A.X., Nguyen, B.P. et al. Herpes zoster vaccine safety in the Aotearoa New Zealand population: a self-controlled case series study. Nat Commun 14, 4330 (2023). https://doi.org/10.1038/s41467-023-39595-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-023-39595-y
This article is cited by
-
Impfen schützt Herz und Gefäße
MMW - Fortschritte der Medizin (2025)
-
Herpes zoster vaccine safety in the Aotearoa New Zealand population: a self-controlled case series study
Nature Communications (2023)