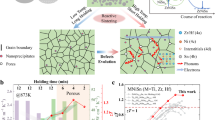
Abstract
In thermoelectric, phase interface engineering proves effective in reducing the lattice thermal conductivity via interface scattering and amplifying the density-of-states effective mass by energy filtering. However, the indiscriminate introduction of phase interfaces inevitably leads to diminished carrier mobility. Moreover, relying on a singular energy barrier is insufficient for comprehensive filtration of low-energy carriers throughout the entire temperature range. Addressing these challenges, we advocate the establishment of a composite phase interface using atomic layer deposition (ALD) technology. This design aims to effectively decouple the interrelated thermoelectric parameters in ZrNiSn. The engineered coherent dual-interface energy barriers substantially enhance the density-of-states effective mass across the entire temperature spectrum while preser carrier mobility. Simultaneously, the strong interface scattering on phonons is crucial for curtailing lattice thermal conductivity. Consequently, a 40-cycles TiO2 coating on ZrNi1.03Sn0.99Sb0.01 achieves an unprecedented zT value of 1.3 at 873 K. These findings deepen the understanding of coherent composite-phase interface engineering.
Similar content being viewed by others
Introduction
The usage of thermoelectric materials holds great promise for the direct conversion of waste heat into useful electricity, making them valuable candidates for energy conversion applications and sustainable energy development. The efficiency of thermoelectric device depends significantly on the material’s dimensionless Fig of merit, denoted as zT = σα2T/κ, where α, σ, κ, and T are the Seebeck coefficient, electrical conductivity, total thermal conductivity (including lattice contribution κL and carrier contribution κe), and the absolute temperature, respectively1. A more detailed evaluation of zT involves the quality factor \(\beta \propto {\mu }_{H}{m}^{\ast 3/2}/{\kappa }_{L}\), which reveals the utmost importance of maintaining high carrier mobility (µH) in materials with low κL2. Recent efforts toward high zT are in line with the quality factor, which boost the density-of-states effective mass (m*) by band structure manipulation3,4,5,6,7, as well as reducing κL via multiscale microstructures8,9,10,11,12,13,14,15,16.
As an emerging paradigm-shifting strategy, phase interface engineering plays a pivotal role in optimizing thermoelectric transport parameters. This strategy addresses two key aspects: enhancing m* via energy filtering effect and shrinking κL through dense phonon scattering at high-density interfaces17,18,19,20,21,22. However, leveraging phase interface engineering to achieve superior thermoelectric performance faces two significant challenges. Firstly, interface potential barriers between nanoinclusions and the matrix effectively filter out low-energy charge carriers, thereby augmenting the density of states (DOS) proximal to the Fermi level. Yet, this enhancement of m* tends to diminish at elevated temperatures due to the limitations imposed by a single energy potential barrier (ΔE)23,24,25. Secondly, arbitrarily introduced phase interfaces can scatter both phonons and carriers, leading to a detrimental effect on carrier mobility (μH) and hence the zT26,27. For example, the introduction of Al2O3 into ZrNiSn or the creation of amorphous ZrNiSn have both successfully led to a significant reduction in κL. However, these approaches did not effectively enhance PF due to the decrease in μH. Consequently, the coupling between electronic and phonon transport properties has limited the potential for improving the figure of merit zT28,29. To date, achieving a significant enhancement in \({\mu }_{H}{m}^{\ast 3/2}/{\kappa }_{L}\) over the entire temperature range of thermoelectric materials remains a challenging and elusive goal30. Hence, a judicious and rational phase interface design is crucial for implementing phase interface engineering in thermoelectrics.
In this context, the construction of a coherently composite-phase interface is proposed as a promising strategy for the synergistic optimization of \({\mu }_{{\rm H}}{m}^{\ast 3/2}/{\kappa }_{L}\). The connotation of a composite phase interface contains two fundamental principles. Initially, theoretical considerations suggest that the optimal energy barrier should lie between 1 to 10 kBT (kB and T stand for Boltzmann’s constant and absolute temperature)31, a range that varies with temperature. To ensure a consistent enhancement of m* across varying temperatures, the establishment of multiple energy barriers is crucial, ensuring the filtration of low-energy electrons throughout the entire temperature spectrum23. Additionally, an ideal phase interface should scatter phonons more effectively than electrons. Analysis of the phase interface structure reveals that both coherent and incoherent interfacial structures lead to an obvious decrease in κL. However, materials with coherent interfaces exhibit markedly higher µH than those with the incoherent interfaces32,33. Despite these insights, comprehensive studies that concurrently address these aspects remain scarce. The construction of coherent phase interfaces harboring multiple ΔE is pivotal in disentangling the intricately linked thermoelectric parameters18,23,34. However, the primary challenge lies in the systematic engineering of these coherent composite phase interfaces featuring multiple ΔE in experimental setups to optimize the zT. Historically, methods such as ball milling35, hydrothermal synthesis36, self-precipitation37,38, and melt spinning39 have been prevalent for introducing secondary phases into the matrix. Regrettably, these methods fall short in precisely constructing coherent phase interfaces with multiple ΔE. To surmount this challenge, employing atomic layer deposition (ALD) for the manipulation of coherent composite-phase interfaces emerges as a strategically effective pathway. ALD, renowned for its self-limiting and self-saturating characteristics, is ideal for the layer-by-layer growth of 2D films. Crucially, the unique capability of ALD technology to uniformly coat diverse 3D bulk materials enables meticulous controll over the thickness of the coating layer40. At elevated sintering temperatures, the amorphous nature of these coating enhances chemical reactivity, potentially initiating in-situ chemical reactions between the substrate and the ALD-coated layer. In this study, we present a unique methodology for constructing coherent composite-phase interfaces with multiple ΔE through the in-situ reflection between the matrix and the ALD coating. For this purpose, the half-Heusler (HH) ZrNiSn alloy was chosen as an exemplary template for implementing phase interface engineering due to its high sintering temperature. We employed an ALD coating of amorphous TiO2, chosen for its high chemical reactivity, as a precursor layer. The coherent composite-phase interfaces within the n-type ZrNi1.03Sn0.99Sb0.01 matrix (referred to ZNSS) is thereafter formed after high-temperature sintering. The initially amorphous TiO2 coating undergoes a comprehensive transformation, being replaced by a Ti-ZNSS layer (aka Zr1-xTixNi1.03Sn0.99Sb0.01) along with uniformly dispersed ZrO2 nanoparticles, as depicted in the schematic diagram in Fig. 1a.
a Schematic diagram of electron and phonon transport in the multi-scale phase interfaces. b Temperature-dependent zT for the TiO2 = 3.2 nm sample in this work, compared with those of other high-zT HH TE materials25,49,59,60,61,62,63,64,65,66. c Schematic diagram of energy filtering effect. d Schematic diagram of electron and phonon transport in the coherent phase interfaces.
The in-situ formation of the composite phase interfaces, consisting of the Ti-ZNSS layer and ZrO2 nanoparticles, impeccably orchestrates the simultaneous optimization of electrical and thermal transport through two synergistic mechanisms. Firstly, the augmentation of the energy filtering effect on m* facilitated by various ΔE strategically created between the matrix and composite phase, as illustrated in the schematic diagram in Fig. 1c. Secondly, the introduction of coherent phase interfaces substantially reduces the κL while maintaining µH unchanged, as depicted in the schematic diagram in Fig. 1d. Furthermore, we are pleasantly surprised to discover that there are a significant number of twins within the ZrO2 nanoparticles. This twin structure provides an additional scattering center for phonon transport at the nanoscale. Consequently, the state-of-the-art zT of 1.3 is attained in the ZNSS sample coated with 40 cycles of TiO2 (i.e., TiO2 = 3.2 nm) (Fig. 1b). These findings underscore the pivotal role of coherent composite-phase interface engineering in achieving cutting-edge zT in ZrNiSn-based and other thermoelectric materials.
Results
Design principle
Designing composite phase structures with coherent phase interfaces and multiple ΔE is aimed at enhancing m* and phonon scattering while minimally impacting the µH. Given the necessity for high-temperature sintering to facilitate the interface chemical reaction between matrix and amorphous ALD coating layer, ZrNiSn-based materials, known for their high melting point, are selected as an ideal platform for constructing the composite phase interfaces. The initial step involves the synthesis of a high purity ZNSS matrix. To this end, the ZNSS matrix is fabricated using levitation melting, a technique chosen for its efficiency in producing high-purity materials. The room temperature powder X-ray diffraction (PXRD) patterns, as illustrated in Fig S1, reveal the diffraction peaks of the ZNSS matrix that are exclusively indexed to the cubic MgAgAs-type crystal structure without any detectable second phases. A critical prerequisite for the construction of coherent composite-phase interfaces is the preparation of ZNSS powders uniformly coated with amorphous TiO2 layers. The dynamic formation process of depositing ultrathin TiO2 layers onto the ZNSS powders through ALD is meticulously captured in Fig. 2a. During the ALD process, conducted at 423 K, tetrakis (dimethylamido) titanium (TDMAT) and H2O serve as precursors for TiO2 deposition41,42. Notably, the PXRD pattern of the ALD-coated samples (Fig. S2) reveals the absence of crystalline TiO2 diffraction peaks, as the TiO2 layer obtained is actually amorphous and less than 10 nm, which is beyond the detection limit of XRD. Further validation of the successful coating of the ultrathin TiO2 layer on the surface of ZNSS powder is obtained through X-ray photoelectron spectroscopy (XPS). The XPS analysis, particularly for the coated 40 cycles TiO2 sample, demonstrates a distinct Ti 2p peak, unequivocally indicating the presence of the amorphous TiO2 layers on the ZNSS surfaces (Fig S3)43.
a Schematic diagram illustrating the synthesis of the TiO2 layer through ALD coating. b HRTEM images of ZNSS powders coated with 40 cycles of TiO2 (TiO2 = 3.2 nm). c Schematic representation of the interfacial reaction between the ZNSS matrix and ALD coating. d ABF images and corrsponding (Zr, O, Ti element) EDS mapping of the bulk ZNSS sample coated with 40 cycles of TiO2.
The ALD boasts significant advantages, particularly in its capability to homogeneously coat complex 3D structures and precisely control phase thickness. The quality of the ALD coating layer is validated through high-resolution transmission electron microscopy (HRTEM) images of a representative coated TiO2 powder coated for 40 cycles, as illustrated in Fig. 2b. It is evident that TiO2 with 40 cycles is uniformly enveloped by an ultrathin amorphous TiO2 layer, approximately 3 nm thick. The calculated growth rate of TiO2, is around 0.8 Å per cycle, which aligns with previous literatures40,44. The resulting samples, differentiated by the number of ALD-coated TiO2 layers, are designated as uncoated, TiO2 = 0.8 nm (10 cycles), TiO2 = 1.6 nm (20 cycles), TiO2 = 3.2 nm (40 cycles), TiO2 = 4.8 nm (60 cycles) and TiO2 = 6.4 nm (80 cycles). Such meticulous characterizations underscore the successful deposition of high-quality, ultrathin amorphous TiO2 layers on the surface of ZNSS powder, laying a robust foundation for the construction of composite phase interfaces.
The choice of amorphous TiO2 for the ALD coating is strategic, considering high chemical activity and susceptibility to oxygen deficiency at elevated temperatures, which are conducive to fostering the desired interfacial reactions. To expedite the chemical reaction at the interface between the TiO2 coating layer and ZNSS powder, a densification process is undertaken using spark plasma sintering (SPS) at 1373 K for 10 min. It is worth noting that no significant second phase was observed in the XRD patterns after sintering (Figs. S4 and S5). A comprehensive schematic depicting this interface chemical reaction is presented in Fig. 2c. In the initial stage, the O atoms within the amorphous TiO2 layer bond with the Zr atoms from the matrix, resulting in the formation of ZrO2 nanoparticles. Simultaneously, the residual Ti atoms occupy vacant Zr sites, creating a Ti-ZNSS layer. This sophisticated interfacial reaction mechanism is further substantiated by scanning transmission electron microscopy (STEM) images, including both annular bright field (ABF and corresponding EDS mapping) and atomically-resolved high angle annular dark field (HAADF) images, as shown in Fig. 2d. There images validate the uniform distribution of nanoparticles at the grain boundaries of the substrate, consistent perfectly with the hypothesized interface design. This intricate interplay at the nanoscale level highlights the ingenuity and precision of the phase interface engineering approach employed in this study.
Microscopic characterization of composite phase interfaces
In order to achieve a more precise elucidation of the reaction product of the SPSed TiO2 = 3.2 nm sample, we employed scanning electron microscope (SEM) and electron probe micro analysis (EPMA) to unveil the intricate composite phase compositions. Firstly, the X-ray energy dispersive spectrum (EDS) mapping and spot scanning results (Tables S1–S4) disclosed a noteworthy observation: Ti atoms did not enter into the ZNSS matrix. Instead, they replaced the pristine amorphous TiO2 layer, culminating in a uniform Ti-ZNSS layer, formally identified as Zr1-xTixNi1.03Sn0.99Sb0.01, as depicted in Fig. 3a. This uniform Ti-ZNSS layer encapsulates the ZNSS matrix. Further scrutiny, as shown in Figs. S6–S10, Supporting Information, revealed a profusion of nanoparticles uniformly dispersed around the ZNSS matrix. The excess of Ni in both the matrix and the Ti-ZNSS layer is a result of two factors: firstly, it is a consequence of our experimental design, and secondly, it reflects an inherent characteristic of ZrNiSn itself 45,46. The EDS mapping images corroborate that these nanoparticles predominantly consist of ZrO2. To cross-check these findings, we also conducted a thorough TEM-EDS analysis, focusing on the size and composition of these nanoparticles (Fig. 3b), which affirmed their ZrO2 constitution.
a EPMA backscattered electron image of TiO2 = 3.2 nm sample and the corresponding EDS element of O, Zr, Ni, Sn, Ti maps (the color from blue to red indicates the content of elements from less to more). b Low magnification TEM image and corresponding EDS mapping images of the ZrO2 nanoparticle.
From these observations, we infer the formation of multiscale composite phase structures, which encompasses ZrO2 nanoparticles, ranging from 50–100 nm in size, and the Ti-ZNSS layer, with a thickness of 50 to 500 nm (Fig. S8). These features are predominantly derived from the chemical interaction between the highly reactive amorphous TiO2 layer and the ZNSS matrix during the SPS sintering process. Analogous phenomena have been observed in Al2O3-coated ZrNiSn0.99Sb0.01 and TiO2-coated Bi2Te2.7Se0.3 systems25,44. The underlying mechanism of this interface chemical reaction can be understood in terms of electronegativity and thermodynamic considerations. The electronegativity difference between Zr and O (2.11) is greater than that between Ti and O (1.9), hence favoring the bond formation between Zr and O over Ti and O. The formation energies of various oxides during high-temperature processes are critical determinants of their stability and presence in the final product. ZrO2 exhibits a formation energy of −3.801 eV, which is lower than that of TiO2 (−3.3 eV), SnO2 (−2.123 eV), NiO2 (−1.761 eV), and Sb2O3 (−1.728 eV). This indicates that ZrO2 is thermodynamically the most favorable oxide under the experimental conditions. The detailed chemical reaction formula is delineated as follows:
Atomic scale characterization of the composite phase interface (ZNSS/ZrO2)
The microstructural intricacies at the phase interfaces play a critical role in disentangling the electrical and thermal transport properties. To this end, a comprehensive microstructural examination of two key interfaces of the SPSed TiO2 = 3.2 nm sample was conducted. Figure 4a–e illustrate the interface between the ZNSS matrix and ZrO2 nanoparticles. Employing aberration-corrected STEM with atomic resolution, we discerned a pristine coherent interface between the ZrO2 nanoparticles [100] and the ZNSS matrix [100]. The clarity of this interface is further validated by the inverse fast Fourier transform (IFFT) images, which exhibit a defect-free phase boundary. This immaculately coherent interface is crucial for sustaining a high carrier mobility μH, thereby leading to excellent electrical properties. Furthermore, detailed examination of high-resolution ZrO2 atomic images, alongside Fast Fourier Transform (FFT) data (Fig. S13), confirms the crystalline structure of ZrO2 as monoclinic. This finding lays a solid theoretical foundation for investigating the interface potential barriers. A deeper analysis, integrating data from Fig. 4c and Fig. 4d, suggests that the impeccable coherence of the interface can be attributed to the closely matched lattice parameters of ZNSS (a = 3.105 Å) and the monoclinic ZrO2 (a = 3.005 Å).
The detailed features of the composite phase interface in the TiO2 = 3.2 nm sample are as follows: A Low-magnification STEM-HADDF image (a) reveals ZrO2 nanoparticles attached to grain boundaries. In the HADDF image of aera 1 (b) the atomic structure between ZrO2 nanoparticles [100] and the ZNSS matrix [100] is distinctly visible. Further HADDF magnified images are presented in (c) with (d) and (e) showing their respective FFT image and GPA result. f STEM-HADDF image showing ZrO2 precipitates. HADDF (g) and ABF (h) images respectively focus on the details of the ZrO2 nanoparticles, with corresponding GPA results shown in i. Further HADDF magnified twin boundary images are presented in (j) and k.
Combining geometric phase analysis (GPA) with ZNSS as the reference matrix, we were able to spatially capture the high εxy strain within the ZrO2 nanoparticles (Fig. 4e). Notably, the strain distribution in this context deviates from the norm observed in traditional nanocomposites, where strain typically accumulates around the phase boundary. Instead, in our sample, the strain is distributed throughout the entire particle. Further exploration through STEM HADDF (Fig. 4g) and ABF (Fig. 4h) phase images revealed abundant twin boundaries within the second-phase ZrO2. Twinning within the ZrO2 structure was confirmed by lattice splitting observed in the FFT analysis. Concurrently, GPA analysis demonstrated substantial strain in the εxx direction at these twin boundary positions (Fig. 4i). This strain effectively impedes phonon propagation, resulting in a significant reduction in κL47. Remarkably, the presence of two distinct coherent twin boundaries within the ZrO2, each characterized by different rotation angles, as shown in Fig. 4j and k, underscores the sophisticated microstructural engineering. This intricate design is instrumental in achieving the decoupling of carrier and phonon transport.
Atomic scale characterization of the composite phase interface (ZNSS/Ti-ZNSS)
Figure 5a–d presents a detailed examination of the interface between the ZNSS matrix and the Ti-ZNSS layer. Elemental line scanning (Fig. 5a) indicates a heightened concentration of Ti around the grain boundaries, consistent with previous EDS results from EPMA. In regions with diminishing Ti concentration along the line scan (i.e., the interface between the Ti-ZNSS layer and the ZNSS matrix), a meticulous Z-contrast (Fig. 5b) discerns differences in mass between atoms, showcasing a flawlessly coherent interface established between the Ti-ZNSS layer and the ZNSS matrix, as confirmed by IFFT information48. As shown in Fig. 5c, d, advanced Z-contrast analysis of the HADDF images highlights pronounced darker features on the left side, conspicuously absent on the right. This phenomenon is attributed to the smaller atomic contrast exhibited by lighter Ti atoms compared to Zr. Consequently, through meticulous Z-contrast examination, a well-defined boundary delineates the Ti-ZNSS layer on the left from the ZNSS matrix on the right. To rigorously corroborate the accuracy of the data in Fig. 5d, a meticulous Z-contrast line scan is performed on the corresponding region in Fig. 5c. The observed trend reveals a sequential increase in atomic contrast from left to right, aligning precisely with the contrast patterns depicted in Fig. 5d. This meticulous analysis serves as a stringent cross-verification, ensuring the reliability and consistency of the presented results.
The yellow line in the (a) represents Ti element line scanning information, while (b) is an enlarged HADDF region within the pink box, and the inset displays the Z- contrast map of the yellow line. Further magnification of the HADDF atomic structure image is presented in (c) corresponding to the [100] HADDF image and normalized intensity of Ti columns in d.
Electrical and thermal transport properties
In the context of the formation of coherent composite phase interfaces in TiO2-coated ZNSS samples, a pertinent question emerges: Can these specialized phase interfaces facilitate the decoupling of carrier and phonon transport? Fig. 6 elucidates the electrical and thermal transport properties of these as-fabricated samples. A striking observation from Fig. 6a is the substantial enhancement of the Seebeck coefficient (α) in ALD-coated ZNSS samples, compared to the initial ZNSS sample (Fig. 6a). In typical thermoelectric materials, an increase in the α is commonly accompanied by a decrease in the carrier concentration (nH)49. However, as indicated by the Pisarenko curve of α - nH at room temperature (Fig. S16), the nearly constant nH before and after ALD coating indicates that the increase in α can only be attributed to the improvement in m* via the energy filtering effect50,51. Moreover, under the influence of coherent composite phase interfaces, the electrical conductivity (σ) of ALD-coated ZNSS samples does not show significant variation from the pristine uncoated sample (Fig. 6b). This stability in σ can be predominantly attributed to the coherent phase interfaces, which are integral in maintaining µH. In the case of TiO2 = 4.8 nm and TiO2 = 6.4 nm samples, the slight increase in the σ is primarily due to the elevated nH. Benefiting from the enhanced α and retained σ via the coherent composite phase interfaces with multiple energy barrier, the power factor (PF) has been significantly enhanced after ALD coating (Fig. 6c). Taking the TiO2 = 3.2 nm sample as an example, the simultaneously achieved high α of 204.1 μVK−1 and σ of 1448 Scm−1 at 873 K give rise to a large PF of 60.24 μWcm−1K−2. This attests to the efficacy of our phase interface engineering to improve the electrical properties of ZrNiSn-based materials.
The electrical and thermal properties of the ZNSS samples before and after different ALD cycles, where uncoated, TiO2 = 0.8 nm, TiO2 = 1.6 nm, TiO2 = 3.2 nm, TiO2 = 4.8 nm, TiO2 = 6.4 nm. a α, b σ, c PF, d κ, e κL and f zT. Error bars were estimated from the repeatability of the experimental result; three measurements were carried out for each material.
Figure 6d depicts the temperature dependence of the thermal conductivity (κ) and lattice thermal conductivity (κL) for all the samples. A marked reduction in κ is observed with increasing ALD coating layers. For instance, the room temperature κ quickly drops from 5.41 Wm−1K−1 for C = 0 sample to 4.78 Wm−1K−1 for TiO2 = 6.4 nm sample. Since the rational coherent phase interface design results in negligible changes in σ across all the samples, this decline in κ as increasing ALD coating layers can be largely ascribed to the diminished κL. Herein, κe is evaluated by the Wiedemann-Franz law κe = LσT and κL is obtained by subtracting the κe from κ, where the Lorenz number L is estimated via L = 1.5 + exp(-|α|/116)52. As expected, the κL substantially declines with increasing ALD coating layers over the entire temperature range (Fig. 6e). In particular, the lowest κL of 1.72 Wm−1K−1 is attained in the TiO2 = 6.4 nm sample, which is 20% lower than that of the pure ZNSS matrix. Ultimately, all ALD-treated samples exhibit superior zT values compared to the pristine ZNSS sample. Notably, the TiO2 = 3.2 nm sample achieves an advanced peak zT value of approximately 1.3 (Fig. 6f).
Analysis of electrical and thermal transport mechanisms
To elucidate the synergistic mechanism underlying the observed enhancement of thermoelectric performance, we employed the single parabolic band model (SPB) to generate Pisarenko curves for samples with different numbers of ALD cycles (Fig. 7a), with experimental data at 300 K and 800 K distinctly annotated. Initially, an analysis of the variation in m*, as depicted in Fig. 7b, reveals a crucial insight. The intrinsic band structure of the ZNSS matrix remains highly stable across temperatures, with m* being 2.7 me at both 300 K and 800 K. This finding aligns with the prior research conducted by Zhu et al.53. Furthermore, an intriguing trend is observed: regardless of at the temperature (300 K or 800 K), the m* demonstrates a gradual increase with an escalating number of ALD cycles. Most intriguingly, at 800 K, the m* for all ALD-treated samples is significantly elevated compared to their 300 K counterparts. According to the research conducted by Snyder et al.50, the interface potential barriers between nanoinclusions and the matrix can selectively filter out low-energy charge carriers, thereby enhancing the DOS near the Fermi level (i.e., m*) and consequently improving the α. Moreover, Zhang et al. proposed that the enhancement in m* resulting from a single interface potential barrier tends to diminish at elevated temperatures23. The theoretically optimal estimation of ΔE ~ 1-10 kBT supports notion that constructing hierarchical potential barriers could effectively mitigate this issue31. Therefore, the construction and optimization of potential energy barriers at the interfaces between the composite phases and the ZNSS matrix are critical to the enhancement of the electrical transport properties.
Temperature dependence of (a) Pisarenko relationship of α and component-dependent m*. b at 300 and 800 K for the ZNSS samples before and after different ALD cycles, where uncoated, TiO2 = 0.8 nm, TiO2 = 1.6 nm, TiO2 = 3.2 nm, TiO2 = 4.8 nm, TiO2 = 6.4 nm. c Schematic diagram of the energy filter effect of composite phase interface on electron transport. d The ratio of m* (with ΔE) to m* (without ΔE) at 300 K and 800 K for cases no barrier, with a single energy barrier, and with dual energy barriers54. e Component-dependent PF at 300 and 873 K for the ZNSS samples before and after different ALD cycles, where uncoated, TiO2 = 0.8 nm, TiO2 = 1.6 nm, TiO2 = 3.2 nm, TiO2 = 4.8 nm, TiO2 = 6.4 nm. f The µH and κL at room temperature for TiO2-coated ZNSS samples.
Given the composite phase structures prepared by ALD, it is imperative to further investigate the details of the two distinct types of ΔE at the Ti-ZNSS/ZNSS and ZrO2/ZNSS interfaces. After calibrating the work function for each band structure, an interface ΔE of approximately 0.2 eV was observed between the conduction band minimum (CBM) at the X-point between ZrNiSn, Zr0.67Ti0.33NiSn and TiNiSn (Fig. S13). This observation aligns with the the chemical reaction dynamics outlined in Equation 1, revealing that with an increasing number of ALD coating layers, there is a corresponding rise inTi content in the Ti-ZNSS layer. Consequently, this leads to a tunable ΔE between the Ti-ZNSS layer and the ZNSS matrix, ranging approximately from 0 to 0.20 eV. In contrast, the band structure analysis of the monoclinic ZrO2 indicated a higher ΔE, approximately 0.6 eV (Fig. S14). This differential in ΔE values for the two interfaces, at approximately 0.2 eV and 0.6 eV respectively (Fig. 7c), fits well within the theoretically optimal range of ΔE ~ 1-10 kBT (0.25 eV/300 K – 0.75 eV/873 K)31.
Furthermore, Fig. 7d distinctly demonstrates the remarkable elevation in the m* for the ALD-treated samples, observable at both ambient and elevated temperatures. The enhancement is more pronounced at higher temperatures. The underlying rationale for this phenomenon becomes apparent when juxtaposed with the semiconducting behavior of single ΔE-induced Half-Heusler (HH)/Full Heusler (FH) samples54. Particularly at 800 K, the ratio of m* (with ΔE) to m* (without ΔE) displays a diminishing trend relative to 300 K, highlighting a limitation inherent in a singular energy barrier system. Hence, the PF (Fig. 7e) reveals a clear and consistent enhancement across all ALD-treated samples than that in pristine ZNSS sample, evident at both 300 K and 873 K. Such a phenomenon is a testament to the strategic construction of interfaces through ALD, offering a compelling insight into the intricate dynamics governing the thermoelectric enhancement observed in our experimental samples.
From the aspect of defect physics, the introduction of a second phase with a distinct interface from the matrix is conventionally perceived as a two-dimensional defect, imparting scattering interactions on both phonons and carriers55,56. However, coherent phase interfaces are recognized as a distinctive subclass of two-dimensional defects57,58. Notably, these interfaces are remarkable for their ability to scatter phonons while simultaneously preserving high µH, rendering them a specialized and nuanced facet in the landscape of material defects. Therefore, the coherent composite phase interfaces constructed via ALD exhibit a comprehensive integration of this particular attribute. As depicted in Fig. 7f, a discernible pattern is observed with the continual increase in ALD cycles. At 300 K, µH remarkably consistent, while there is a notable successive reduction in κL. This observation underscores the meticulous orchestration of these interfaces in simultaneously upholding µH and modulating κL, highlighting the efficacy of ALD-driven fabrication of coherent composite phases.
Thermoelectric performance comparison
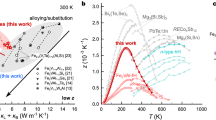
The remarkable advancements in elevating m*, preserving µH, and attenuating κL through coherent composite phase interface engineering have unequivocally established this methodology as a cornerstone in advancing thermoelectric performance. Figure 8a, b vividly illustrate that the PF achieved via the coherent composite interfaces significantly outperforms those observed in n-type ZrNiSn systems. Concurrently, the multidimensional defects meticulously constructed via composite phase interface engineering exert a profound constraint on phonon propagation, resulting in a marked reduction in κL. Consequently, this sample manifests a remarkable average zT value of 0.73 within the temperature range of 300 to 873 K (Fig. 8c). The relevance of this finding is amplified when considering the critical benchmarks for assessing thermoelectric materials: the average zT and average power factor. The average zT dictates the device’s conversion efficiency, whereas the average power factor governs the device’s output power. In this context, as illustrated in Fig. 8d, the TiO2 = 3.2 nm sample not only demonstrates a significant improvement, almost twofold, compared to the original ZrNiSn sample, but it also stands out remarkably against other high-performance n-type half-Heusler thermoelectric materials reported in contemporary research. These results demonstrate the effectiveness of coherent composite phase engineering in decoupling carrier and phonon transport in thermoelectric materials.
Comparison PF (a) and κL (b) between TiO2 = 3.2 nm sample and other advanced HH alloys59,60,61,62,63,64. c Comparison on zTavg, d zTavg and PFavg between TiO2 = 3.2 nm sample and other advanced HH alloys, where the hot-side Thot and cold-side Tcold temperature is 873 K and 300 K, respectively59,60,61,62,63,64,65,67.
Discussion
In the context of coherent composite phase interface engineering, a Ti-ZNSS layer (aka Zr1-xTixNi1.03Sn0.99Sb0.01) with uniformly distributed ZrO2 nanoparticles has been successfully constructed based on the ALD technique to decouple the interrelated thermoelectric parameters in n-type ZrNiSn. The composite phase structures including the Ti-ZNSS layer and ZrO2 nanoparticles are achieved through an interfacial chemical reaction between the ZNSS matrix and amorphous TiO2 during SPS process, where amorphous TiO2 is deposited on the ZNSS substrate surface in advance via ALD technique adopting with TDMAT and H2O as precursors. The constructed composite interfaces encompass energy barriers localized at the Ti-ZNSS layer/ZNSS and ZrO2/ZNSS interfaces. This strategic arrangement facilitates effective scattering of low-energy electrons across distinct temperature regimes, thereby elevating the density-of-states effective mass at both ambient and elevated temperatures. Furthermore, the imposition of the flawless coherent phase interfaces between Ti doped layer/ZNSS and ZrO2/ZNSS adeptly scatter phonons at the composite phase interface, while concurrently preserving decent carrier mobility. Combining with the ultrahigh PF ~ 60.24 μWcm−1K−2 and ultralow κL ~ 1.72 Wm−1K−1, a record high zT ~ 1.3 and average zTavg of ~ 0.73 between 300 and 873 K is attained in TiO2 = 3.2 nm sample. These results not only enrich our comprehension of phase interface dynamics, but also significantly contribute to the emerging paradigm of “coherent composite phase interface engineering” in thermoelectric and functional materials. Another potential application worth our attention is: By employing ALD technology to precisely modify the interfaces, it is not only possible to prevent Ostwald ripening, but also promising to significantly enhance the stability of thermoelectric materials under long-term operational conditions.
Methods
Polycrystalline samples ZrNi1.03Sn0.99Sb0.01 matrix was prepared by the levitation melting-ballmilling-spark plasma sintering (SPS). Alloys were first prepared by levitation melting the stoichiometric amounts of Zr (piece, 99.99%), Ni (rods, 99.99%), Sn (block, 99,99%), and Sb (block, 99.99%) under an argon atmosphere for 2 min, and the melt was quenched in a water-cooled copper crucible. The ingots were remelted six times to ensure homogeneity. The ballmilling process for 1 h at 800 rpm under an argon atmosphere. The fine powders were to use as the matrix of ALD.
A TiO2 layer was coated on the surface of the ZrNi1.03Sn0.99Sb0.01 matrix in a homemade continuous-flow ALD reactor operated under a base pressure of ~1 Torr. Typically, the 8 g as-prepared ZrNi1.03Sn0.99Sb0.01 matrix was transferred into an ALD chamber equipped with a vertical stainless rotating sample chamber. The TiO2 layer was deposited using tetrakis(dimethylamido)titanium (TDMAT) and H2O as ALD precursors at 423 K. The bubbler containing TDMA was heated to 323 K and the delivery line was heated to 343 K. The precursor dose and purge time were 30 seconds and 60 seconds, respectively. Ar gas served as both a carrier and a purging gas. Then, the coated fine powders were sintered by Spark Plasma Sintering (SPS) at 1373 K under 60 MPa in vacuum for 10 min. The as-sintered samples, of which the relative density was >98%, were cut for thermoelectric property measurement and characterization.
The phase structure of coated powders and sintered samples were measured by X-ray diffraction (XRD) on a Rigaku Smartlab 9kw (tube voltage: 45 kV, tube current: 200 mA) diffractometer using Cu K a radiation (λ = 1.5406 Å) and X-rayphotoelectron spectra (XPS) were acquired using a Microlab 350 surface analysis system equipped with a monochromatized Al anode X-ray source, pass energy is 20.0 eV. The chemical composition was obtained during electron probe microanalysis (EPMA, JOEL, JXA-8100) using wavelength dispersive spectroscopy (WDS), acceleration voltage of 300 kV and test beam current: 50 nA for surface analysis, 20 nA for quantitative testing. The TEM specimens of the sintered bulk materials were meticulously prepared utilizing a state-of-the-art focused ion beam system (FIB; JIB 4601 F, JEOL) to ensure precise cross-sectioning. Subsequently, the surface morphology and detailed crystallographic orientation were elucidated through high-resolution field-emission transmission electron microscopy (HR-TEM; JEM-ARM300F), operated at an acceleration voltage of 300 kV to achieve optimal imaging conditions.
The sintered specimen was carefully sectioned into two distinct shapes: a rectangular prism measuring 3 mm by 3 mm by 11 mm, and a square prism with dimensions of 9.8 mm by 9.8 mm by 2 mm. The rectangular prisms underwent meticulous polishing to facilitate the measurement of the Seebeck coefficient (α) and electrical conductivity (σ) using the ULVAC ZEM-3 apparatus, within a temperature span of 300 to 873 K. Subsequently, the overall thermal conductivity (κ) was deduced through the formula κ = DCpρ, where D denotes the thermal diffusivity, Cp is the specific heat at constant pressure, and ρ is the material density. The thermal diffusivity coefficient (D) was ascertained using a Netzsch LFA 467-HT laser flash analyzer, across the temperature gradient of 300 to 873 Kelvin. Concurrently, the specific heat capacity (Cp) was determined employing a differential scanning calorimeter (DSC 404 F3), with a controlled heating rate of 10 K per minute. Sapphire comparison method, calibration procedure: blank test, sapphire test and sample test, each DSC curve is subtracted from the blank curve. The carrier density of sintered samples were measured using a physical property measurement system (PPMS, Quantum Design).
Electronic structure calculations of ZrNiSn, TiNiSn and ZrO2 were carried out using the density functional theory (DFT) with the projector-augmented-wave (PAW) method as implemented in the Vienna Ab initio simulation package (VASP). The computational framework for our study is based on the Generalized Gradient Approximation (GGA), specifically incorporating the exchange-correlation functional formulated by Perdew, Burke, and Ernzerhof (PBE). The structural optimization of the models was performed using the conjugate gradient (CG) method, which was iterated until the maximum forces acting on the Hellmann-Feynman theorem and the total energy reached a threshold of 10−4 eV, indicating a stable configuration. The simulation parameters were carefully chosen, with a plane-wave cutoff energy set at 450 electron volts to ensure the accuracy of the calculations. For the integration over the Brillouin zone (BZ) of the primitive unit cell, we employed a Monkhorst–Pack grid with a dense k-point sampling of 11 × 11 × 11, which provides a comprehensive representation of the reciprocal space.
Data availability
All data generated or analyzed during this study are included in the published article and its Supporting Information. The data that support the findings of this study are available from the corresponding author (wuhaijunnavy@xjtu.edu.cn) upon reasonable request.
References
Jian, H. & Tritt, T. M. Advances in thermoelectric materials research: Looking back and moving forward. Science 357, 1367 (2017).
Qin, B. et al. Power generation and thermoelectric cooling enabled by momentum and energy multiband alignments. Science 373, 556–561 (2021).
Tang, J. et al. Manipulation of band structure and interstitial defects for improving thermoelectric SnTe. Adv. Funct. Mater. 28, 1803586 (2018).
Shi, X.-L., Zou, J. & Chen, Z.-G. Advanced thermoelectric design: from materials and structures to devices. Chem. Rev. 120, 7399–7515 (2020).
Yao, W. et al. Two-step phase manipulation by tailoring chemical bonds results in high-performance GeSe thermoelectrics. Innovation 4, 100522 (2023).
Zhong, J. et al. Nuanced dilute doping strategy enables high-performance GeTe thermoelectrics. Sci. Bull. 69, 1037–1049 (2024).
Wang, S. et al. High thermoelectric performance in Te-free (Bi, Sb)2Se3 via structural transition induced band convergence and chemical bond softening. Energy Environ. Sci. 9, 3436–3447 (2016).
Kim, S. I. et al. Dense dislocation arrays embedded in grain boundaries for high-performance bulk thermoelectrics. Science 348, 109–114 (2015).
Hu, L. et al. Tuning multiscale microstructures to enhance thermoelectric performance of n‐type Bismuth‐Telluride‐based solid solutions. Adv. Energy Mater. 5, 1500411 (2015).
Poudel, B. et al. High-thermoelectric performance of nanostructured bismuth antimony telluride bulk alloys. Science 320, 634–638 (2008).
Biswas, K. et al. High-performance bulk thermoelectrics with all-scale hierarchical architectures. Nature 489, 414–418 (2012).
Hu, L. et al. Entropy engineering of SnTe: multi‐principal‐element alloying leading to ultralow lattice thermal conductivity and state‐of‐the‐art thermoelectric performance. Adv. Energy Mater. 8, 1802116 (2018).
Zhao, L.-D. et al. Ultralow thermal conductivity and high thermoelectric figure of merit in SnSe crystals. Nature 508, 373–377 (2014).
Toprak, M. S. et al. The Impact of Nanostructuring on the Thermal Conductivity of Thermoelectric CoSb3. Adv. Funct. Mater. 14, 1189–1196 (2004).
Hu, L., Zhu, T., Liu, X. & Zhao, X. Point defect engineering of high‐performance bismuth‐telluride‐based thermoelectric materials. Adv. Funct. Mater. 24, 5211–5218 (2014).
Zhao, H. et al. Engineering the Thermoelectric Transport in Half‐Heusler Materials through a Bottom‐Up Nanostructure Synthesis. Adv. Energy Mater. 7, 1700446 (2017).
Gayner, C. & Amouyal, Y. Energy filtering of charge carriers: current trends, challenges, and prospects for thermoelectric materials. Adv. Funct. Mater. 30, 1901789 (2020).
He, S., Lehmann, S., Bahrami, A. & Nielsch, K. Current State‐of‐the‐Art in the Interface/Surface Modification of Thermoelectric Materials. Adv. Energy Mater. 11, 2170144 (2021).
Li, S. et al. Optimized hetero-interfaces by tuning 2D SnS2 thickness in Bi2Te2.7Se0.3/SnS2 nanocomposites to enhance thermoelectric performance. Nano Energy 39, 297–305 (2017).
Zeier, W. G. et al. Thinking like a chemist: intuition in thermoelectric materials. Angew. Chem. Int. Ed. 55, 6826–6841 (2016).
Ghosh, T., Dutta, M., Sarkar, D. & Biswas, K. Insights into low thermal conductivity in inorganic materials for thermoelectrics. J. Am. Chem. Soc. 144, 10099–10118 (2022).
Moyzhes, B. & Nemchinsky, V. Thermoelectric figure of merit of metal–semiconductor barrier structure based on energy relaxation length. Appl. Phys. Lett. 73, 1895–1897 (1998).
Zhang, J. et al. Extraordinary thermoelectric performance realized in n‐type PbTe through multiphase nanostructure engineering. Adv. Mater. 29, 1703148 (2017).
He, M. et al. Thermopower enhancement in conducting polymer nanocomposites via carrier energy scattering at the organic–inorganic semiconductor interface. Energy Environ. Sci. 5, 8351–8358 (2012).
Zhang, Y. et al. Zr vacancy interfaces: An effective strategy for collaborative optimization of ZrNiSn-based. Thermoelectr. Perform. J. Mater. Chem. A 7, 26053–26061 (2019).
Minnich, A., Dresselhaus, M. S., Ren, Z. & Chen, G. Bulk nanostructured thermoelectric materials: current research and future prospects. Energy Environ. Sci. 2, 466–479 (2009).
Zhou, C. et al. Polycrystalline SnSe with a thermoelectric figure of merit greater than the single crystal. Nat. Mater. 20, 1378–1384 (2021).
Huang, X. Y., Xu, Z., Chen, L. D. & Tang, X. F. Effect of γ-Al2O3 content on the thermoelectric performance of ZrNiSn/γ- Al2O3 composites. Key Eng. Mater. 249, 79–82 (2003).
Music, D., Khayyamifar, S. & Hektor, J. Conflicting primary and secondary properties of thermoelectric devices–A case study on the thermomechanical behavior of ZrNiSn. Comput. Mater. Sci. 230, 112530 (2023).
Li, S. et al. Recent advances in atomic layer deposition-based interface modification engineering in thermoelectric materials. Mater. Today Phys. 39, 101287 (2023).
Dehkordi, A. M., Zebarjadi, M., He, J. & Tritt, T. M. Thermoelectric power factor: Enhancement mechanisms and strategies for higher performance thermoelectric materials. Mater. Sci. Eng.: R: Rep. 97, 1–22 (2015).
Jabar, B. et al. Homo-composition and hetero-structure nanocomposite Pnma Bi2SeS2-Pnnm Bi2SeS2 with high thermoelectric performance. Nat. Commun. 12, 7192 (2021).
Liu, N. et al. Improved Thermoelectric Performance of Eco‐Friendly β‐FeSi2–SiGe Nanocomposite via Synergistic Hierarchical Structuring, Phase Percolation, and Selective Doping. Adv. Funct. Mater. 29, 1903157 (2019).
Li, J. et al. BiSbTe‐based nanocomposites with high ZT: the effect of SiC nanodispersion on thermoelectric properties. Adv. Funct. Mater. 23, 4317–4323 (2013).
Zhao, X. et al. Synthesis of YbyCo4Sb12/ Yb2O3 composites and their thermoelectric properties. Appl. Phys. Lett. 89, 092121–092124 (2006).
Shi, K. et al. Toward high performance n-type thermoelectric materials by rational modification of BDPPV backbones. J. Am. Chem. Soc. 137, 6979–6982 (2015).
Pei, Y., May, A. F. & Snyder, G. J. Self‐tuning the carrier concentration of PbTe/Ag2Te composites with excess Ag for high thermoelectric performance. Adv. Energy Mater. 1, 291–296 (2011).
Guo, B. et al. Thermocells driven by phase transition of hydrogel nanoparticles.J. Am. Chem. Soc. 142, 17318–17322 (2020).
Deng, R. et al. High thermoelectric performance in Bi0.46Sb1.54Te3 nanostructured with ZnTe. Energy Environ. Sci. 11, 1520–1535 (2018).
George, S. M. Atomic layer deposition: an overview. Chem. Rev. 110, 111–131 (2010).
Hackler, R. A., Kang, G., Schatz, G. C., Stair, P. C. & Van Duyne, R. P. Analysis of TiO2 atomic layer deposition surface chemistry and evidence of propene oligomerization using surface-enhanced Raman spectroscopy. J. Am. Chem. Soc. 141, 414–422 (2018).
Xie, Q. et al. Atomic layer deposition of TiO2 from tetrakis-dimethyl-amido titanium or Ti isopropoxide precursors and H2O. J.Appl. Phys. 102, 083521 (2007).
Xu, F. et al. Unique S-scheme heterojunctions in self-assembled TiO2/CsPbBr3 hybrids for CO2 photoreduction. Nat. Commun. 11, 4613 (2020).
Li, S. et al. Atomic-scale tuning of oxygen-doped Bi2Te2.7 Se0.3 to simultaneously enhance the Seebeck coefficient and electrical conductivity. Nanoscale 12, 1580–1588 (2020).
Douglas, J. E., Chater, P. A., Brown, C. M., Pollock, T. M. & Seshadri, R. Nanoscale structural heterogeneity in Ni-rich half-Heusler TiNiSn. J. Appl. Phys. 116, 163514 (2014).
Downie, R. A., Barczak, S., Smith, R. & Bos, J.-W. G. Compositions and thermoelectric properties of XNiSn (X= Ti, Zr, Hf) half-Heusler. Alloy. J. Mater. Chem. C. 3, 10534–10542 (2015).
Jiang, B. et al. High-entropy-stabilized chalcogenides with high thermoelectric performance. Science 371, 830–834 (2021).
Wu, H. et al. Alkali-deficiency driven charged out-of-phase boundaries for giant electromechanical response. Nat. Commun. 12, 2841 (2021).
Zhu, T. et al. Compromise and synergy in high‐efficiency thermoelectric materials. Adv. Mater. 29, 1605884 (2017).
Lin, Y. et al. Expression of interfacial Seebeck coefficient through grain boundary engineering with multi-layer graphene nanoplatelets. Energy Environ. Sci. 13, 4114–4121 (2020).
Li, Y. et al. Enhanced thermoelectric performance of Cu2Se/Bi0.4Sb1.6Te3 nanocomposites at elevated temperatures. Appl. Phys. Lett. 108, 062104 (2016).
Kim, H.-S., Gibbs, Z. M., Tang, Y., Wang, H. & Snyder, G. J. Characterization of Lorenz number with Seebeck coefficient measurement. APL Mater. 3, 041506 (2015).
Zhu, T., Fu, C., Xie, H., Liu, Y. & Zhao, X. High efficiency half‐Heusler thermoelectric materials for energy harvesting. Adv. Energy Mater. 5, 1500588 (2015).
Liu, Y. et al. Large enhancements of thermopower and carrier mobility in quantum dot engineered bulk semiconductors.J. Am. Chem. Soc. 135, 7486–7495 (2013).
Zhou, J. et al. Large thermoelectric power factor from crystal symmetry-protected non-bonding orbital in half-Heuslers. Nat. Commun. 9, 1721 (2018).
Sharp, J., Poon, S. & Goldsmid, H. Boundary scattering and the thermoelectric figure of merit. Phys. Status Solidi 187, 507–516 (2001).
Mao, J., Liu, Z. & Ren, Z. Size effect in thermoelectric materials. npj Quantum Mater. 1, 1–9 (2016).
Peng, G. et al. Structural‐functional unit ordering for high‐performance electron‐correlated materials. Interdiscip. Mater. 2, 30–52 (2023).
Chen, S. et al. Effect of Hf concentration on thermoelectric properties of nanostructured n‐type half‐heusler materials HfxZr1–xNiSn0.99Sb0.01. Adv. Energy Mater. 3, 1210–1214 (2013).
Yang, X. et al. Enhancement in thermoelectric properties of ZrNiSn-based alloys by Ta doping and Hf substitution. Acta Mater. 233, 117976 (2022).
Xie, H. et al. Beneficial contribution of alloy disorder to electron and phonon transport in half‐heusler thermoelectric materials. Adv. Funct. Mater. 23, 5123–5130 (2013).
Min, R. et al. Enhancement in the thermoelectric performance of ZrNiSn-based alloys through extra Zr-rich nanoprecipitates with superstructures. Chem. Eng. J. 464, 142531 (2023).
Min, R. et al. Significantly improved thermoelectric properties of Nb-doped ZrNiSn half-Heusler compounds. Chem. Eng. J. 449, 137898 (2022).
Gong, B. et al. Continuously enhanced structural disorder to suppress the lattice thermal conductivity of ZrNiSn-based half-Heusler alloys by multielement and multisite alloying with very low Hf content. ACS Appl. Mater. Interfaces 11, 13397–13404 (2019).
Liu, Y. et al. Lanthanide contraction as a design factor for high‐performance half‐Heusler thermoelectric materials. Adv. Mater. 30, 1800881 (2018).
Xia, K. et al. Short-range order in defective half-Heusler thermoelectric crystals. Energy Environ. Sci. 12, 1568–1574 (2019).
Liu, Y. et al. Demonstration of a phonon-glass electron-crystal strategy in (Hf, Zr)NiSn half-Heusler thermoelectric materials by alloying. J. Mater. Chem. A 3, 22716–22722 (2015).
Acknowledgements
The work is supported by the National Key R&D Program of China (2021YFB3201100), the National Natural Science Foundation of China (52071218, 52172128), the Shenzhen Science and Technology Innovation Commission (20200731215211001, 20200814110413001), the Guangdong Basic and Applied Basic Research Foundation (2022A1515012492). The authors also appreciate the Instrumental Analysis Center of Shenzhen University. The authors would like to thank the strong support from Instrumental Analysis Center of Xi’an Jiaotong University, with special thanks to Chenyu Liang for his support in the XRD tests.
Author information
Authors and Affiliations
Contributions
The paper was prepared though the contribution of all authors. L.H., H.W. and F.L. designed the work. Y.Z., S.L., K.C., J.W. prepared the sample and measured the thermoelectric transport properties. Y.Z., G.P., Z.Z., C.M., and S.G. performed structural nanocomposite characterization. G.P. and Z.Z. performed simulation. L.H., H.W., Y.Y, T.L., C. Z., X.D. and J.S. planed and supervised the work. Y.Z. and H.W. wrote the paper. All the authors edited the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Wenjie Xie and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, Y., Peng, G., Li, S. et al. Phase interface engineering enables state-of-the-art half-Heusler thermoelectrics. Nat Commun 15, 5978 (2024). https://doi.org/10.1038/s41467-024-50371-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-024-50371-4
This article is cited by
-
High-performance ZrNiSn-based half-Heusler thermoelectrics with hierarchical architectures enabled by reactive sintering
Nature Communications (2025)
-
Effect of Annealing and Preparation Conditions on Electrodeposited Bi2Te3−ySey Films
Journal of Materials Engineering and Performance (2025)