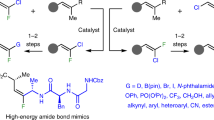
Abstract
Monofluoroalkenes serve as nonhydrolyzable mimetics of amides and are frequently encountered in drug candidates. Herein we report a regio-, enantio-, and stereoselective NiH-catalyzed ipso- and migratory defluorinative olefin cross-coupling employing readily available olefins and gem-difluoroalkenes under mild conditions. This approach enables the efficient synthesis of a broad array of structurally diverse monofluoroalkenes bearing a tertiary allylic stereogenic center. Mechanistically, the challenging migratory defluorinative olefin cross-coupling process is successfully realized through a ligand relay catalytic strategy, enabling the formal C(sp3)–H/C(sp2)–F activation with high levels of regio-, stereo-, and enantiocontrol.
Similar content being viewed by others
Introduction
Owing to the unique properties of fluorine atom, the selective introduction of fluorine-containing motifs is an often-used strategy to modulate the properties of target molecules in pharmaceutical, agrochemical, and material science1,2,3. In modern drug discovery, the monofluoroalkene moiety is considered an ideal mimic of the amide bond (−NH–CO−) due to similar steric and electronic properties but higher metabolic stability and improved biological activity (Fig. 1a)4,5. Therefore, efficient and selective methods to access various densely functionalized monofluoroalkenes will undoubtedly facilitate the development of novel drugs and functional materials. Complementary to direct C–F bond formation, the selective activation of C–F bonds and subsequent defluorinative functionalization of readily accessible poly- or perfluorinated chemicals provides an appealing alternative for accessing fluorinated molecules6,7,8,9,10. In particular, the judicious use of easily accessible gem-difluoroalkenes has recently been broadly utilized in various defluorinative functionalization processes to construct densely functionalized monofluoroalkenes (Fig. 1b)11,12,13,14,15,16,17,18,19,20,21. Although effective, the requirement for a conventional coupling partner restricts broader applicability, frequently resulting in a mixture of E/Z stereoisomers. Furthermore, only three groups investigated the enantioselective defluorinative alkylation of gem-difluoroalkenes, allowing access to pharmaceutically relevant monofluoroalkenes bearing an allylic stereogenic center19,20,21.
a Monofluoroalkenes as amide bioisosteres. b Prior work: defluorinative functionalization of gem-difluoroalkenes. c Concept: ipso- & migratory defluorinative olefin cross-coupling. d Demo: migratory defluorinative olefin cross-coupling via ligand relay catalysis.
Recently, the synergistic combination of chain-walking and cross-coupling chemistry has enabled the judicious use of abundant and easily accessible olefinic starting materials as latent carbanion equivalents22,23,24,25,26, allowing the rapid assembly of structurally complex molecules27,28,29,30,31. In particular, NiH-catalyzed ipso- and migratory hydrofunctionalization of olefins with a wide variety of cross-coupling partners has enabled the discovery of a large and varied range of unique and valuable transformations32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57. In this context, our laboratory has previously realized a migratory defluorinative coupling between two electronically differentiated olefins to form gem-difluoroalkenes56. Continuing this theme, we recently questioned whether this strategy could be used to access to highly functionalized monofluoroalkenes when gem-difluoroalkenes are used as cross-coupling partners (Fig. 1c). Given the intrinsic utility of monofluoroalkenes along with the widespread availability of both olefins and gem-difluoroalkenes, we envisioned that this practical migratory defluorinative olefin cross-coupling would accelerate the discovery of novel monofluoroalkene-containing therapeutics. To realize such an attractive process in a regio-, stereo- and enantioselective fashion, numerous potential challenges must be addressed. First, the initial hydronickelation step must proceed with significantly different reactivities due to the presence of C = C double bonds in the alkene, the gem-difluoroalkene, and the monofluoroalkene product. Second, the subsequent coupling step must be regioselective to prevent the formation of isomeric products via chain-walking. Third, the chemoselective C − F bond activation must occur through more challenging selective oxidative addition, as the competitive alkyl radical addition–β-F elimination pathway would result in racemic product. Moreover, both chain-walking and subsequent asymmetric coupling must be effectively proceeded. Inspired by the ligand relay catalytic strategy58,59,60,61,62,63,64,65,66,67 as well as its recent application in nickel catalysis to enhance both catalytic efficiency and selectivity62,63,64,65, we hypothesized that a simple combination of an achiral chain-walking ligand and a chiral asymmetric coupling ligand would well facilitate both stages of alkene isomerization and asymmetric hydroalkenylation of styrenes, (Fig. 1d). In terms of the subsequent chiral ligand promoted asymmetric hydroalkenylation process, an enantiodifferentiating syn-hydronickelation with styrene would produce an enantioenriched benzylnickel intermediate, subsequent an ester assisted selective oxidative addition followed by reductive elimination would produce the desired monofluoroalkene with high levels of regio-, stereo-, and enantiocontrol.
In this work, we describe a highly regio-, stereo-, and enantioselective Ni-catalyzed ipso- and migratory defluorinative olefin cross-coupling between readily available olefins and gem-difluoroalkenes under mild conditions. A wide variety of densely functionalized monofluoroalkenes bearing a tertiary allylic stereogenic center were directly obtained in high yields with high levels of regio-, stereo-, and enantiocontrol. The challenging migratory defluorinative olefin cross-coupling process, the formal C(sp3)–H/C(sp2)–F cross-coupling, was successfully realized through a ligand relay catalytic strategy.
Results and discussions
Reaction design and optimisation
Our initial studies focused on the asymmetric defluorinative cross-coupling of styrenes (1a) with gem-difluoroalkenes (2a) (Table 1). After systematic examination of numerous reaction parameters, it was found that NiI2 · xH2O and the chiral N,N-bisoxazolines (BOX) ligand (L1*) with diethoxymethylsilane (DEMS) could successfully produce the desired product (3a) in 83% isolated yield with 92% ee (entry 1). The use of other nickel(II) sources such as NiBr2·dimethoxyethane (dme) gave lower yields (entry 2). Ni(cod)2 failed to yield the desired product (entry 3), as the gem-difluoroalkene (2a) underwent defluorination to produce a monofluoroalkene, while styrene (1a) remained intact. Other ligands (L1a*, L1b*, L2*) led to significantly lower yields and ee (entries 4–6). The use of a different silane source (dimethoxymethylsilane, DMMS) resulted in a slightly lower yield (entry 7). The addition of KI as an iodide additive slightly improved the yield (entry 8), but NaI was ineffective (entry 9). Control experiments revealed that a catalytic amount of tetrabutylammonium fluoride (TBAF) as a base was critical for initiating the reaction (entry 10), and the use of Na3PO4 or NaF was comparably effective (entries 11, 12). Interestingly, a deleterious effect on both reactivity and enantioselectivity was observed when using THF as a solvent (entry 13). Conducting the reaction at 0 °C also led to somewhat lower yield (entry 14).
Substrate scope
Under the optimal conditions, the scope in terms of the asymmetric hydroalkenylation of styrenes is shown in Fig. 2. As expected, the substrate scope of the styrene component was found to be quite broad (Fig. 2a). Styrenes bearing electron-donating (3a–3 f) or electron-withdrawing (3h–3o) substituents were competent substrates, and the substituent could be placed at ortho (3k), meta (3 f, 3j, 3 h), or para position (3a–3e, 3i, 3l–3o). A variety of functional groups were tolerated, including ethers (3a, 3c, 3 f, 3 m), esters (3b, 3o), aryl fluorides (3j, 3k), an aryl chloride (3 l), a trifluoromethyl group (3n), and an alkyl chloride (3 l). A series of heterocycles, including a furan (3p), a thiophene (3q) and a pyridine (3r) were also compatible. Notably, the E,Z-configuration of the styrenes had no significant effect on the yield and ee of the products ((E)-1g vs (Z)-1g). Moreover, β-unsubstituted styrenes were also suitable substrates (3 u, 3 v). Further interrogation of the reaction scope demonstrated the broad scope of gem-difluoroalkene partner (Fig. 2b). As anticipated, gem-difluoroalkenes bearing a wide variety of aryl (4b–4e), heteroaryl (4 f), or alkyl (4g–4 m) substituents were competent substrates. In terms of electronic properties, various neutral (4b), electron-donating (4c) and electron-withdrawing (4d, 4e) substituents on the aryl ring couple efficiently. As anticipated, a variety of ester groups, including a benzyl ester (4j), an ethyl ester (4k), an isopropyl ester (4 l), and a tert-butyl ester (4 m), exhibited no substantial influence on the reaction outcome. Generally, good yields and excellent enantioselectivities were achieved. Unfortunately, when gem-dicholoroalkenes (2o) or gem-difluoroalkenes lacking the ester group (2p) were used, no desired dehalogenative olefin cross-coupling product was observed under the current conditions.
Yield under each product refers to the isolated yield of purified product (0.20 mmol scale, average of two runs), enantioselectivities were determined by chiral HPLC analysis. *24 h, 0 °C. †PhNH2 (10 mol%) and NaF (1 equiv) were used instead of TBAF (6 mol%, 1 M in THF).
The more challenging asymmetric migratory defluorinative olefin cross-coupling was next explored (Fig. 3a). However, only low reactivity and moderate regioselectivity were observed when a single ligand L1* was used (7a, see Supplementary Table 2 in SI for conditions optimization). It was found that the addition of an achiral chain-walking ligand could enhance both catalytic efficiency and regioselectivity with no significant erosion in enantioselectivity (7a). With this ligand relay catalytic strategy, a wide variety of unactivated terminal (7a–7k) and internal (7l–7o) alkenes could undergo alkene isomerization–hydroalkenylation smoothly, providing the corresponding migratory defluorinative olefin cross-coupling products in moderate to excellent yields and with excellent regio- and enantioselectivities (Fig. 3a, i). Similarly, a wide array of electronic and positional differentiated substituents on the remote aromatic ring were all well-tolerated (7a–7o). In general, the substituents could be placed at the ortho-, meta- or para-position. Moreover, the E/Z alkene mixtures could be utilized as competent substrates. As before, a wide variety of gem-difluoroalkenes with various functional groups was equally applicable (Fig. 3a, ii, 8b–8k). With a similar ligand relay catalytic strategy, asymmetric migratory reductive cross-electrophile coupling was also successfully achieved (Fig. 3b). A range of alkyl iodides served as suitable substrates, yielding the desired migratory products (10a–10c) with high regioselectivity and excellent enantiomeric excess (ee).
a Asymmetric migratory defluorinative olefin cross-coupling of remote olefins with gem-difluoroalkenes. b Asymmetric migratory reductive alkenylation of alkyl halides with gem-difluoroalkenes. Yield under each product denotes the isolated yield (0.20 mmol scale, average of two independent runs), regioisomeric ratio (rr) was determined by GC and GC-MS analysis, enantiomeric excess (ee) was determined by chiral-phase HPLC analysis. *12 mol% I2 used. †7 mol% 6,6’-di-sec-butyl-2,2’-bipyridine used. ‡4 mol% 2,9-dimethyl-1,10-phenanthroline was used. §10 h.
Mechanistic investigation
Under the standard dual-ligand conditions, the migratory defluorinative olefin cross-coupling leads to accumulation of styrene isomer (Fig. 4a). This observation suggests that alkene isomerization occurs more rapidly than the subsequent asymmetric hydroalkenylation. To figure out whether the achiral chain-walking ligand L1 participates in the subsequent stage of styrene hydroalkenylation, isotopic labeling experiments of ipso-hydroalkenylation of styrene intermediates were conducted using Ph2SiD2 as the hydride source (Fig. 4b). When the chiral ligand L1* was solely employed, the deuteroalkenylation products (4m-D) were obtained with excellent but opposing diastereoselectivities for E- and Z-configurations of styrenes, suggesting that syn-hydronickelation is the enantio-determining step. In contrast, when the chain-walking ligand L1 was solely employed, the desired deuteroalkenylation products (4m-D) were obtained as a 1:1 diastereomeric mixture under similar conditions, indicating that the enantio-determining step may vary with the choice of ligand. Notably, under the dual-ligand conditions, the contribution of the chain-walking ligand plays a minimal role in the stage of styrene hydroalkenylation, as comparable dr and ee were observed with or without the addition of 4 mol% chain-walking ligand. These obversions lead us to the conclusion that the achiral chain-walking ligand (L1) only participates in the stage of alkene isomerization but not in the stage of styrene hydroalkenylation. Additionally, when deuterated styrene (1u-d8) and undeuterated styrene (1 v) were used together in a crossover reaction, no H/D scrambled crossover products were obtained, suggesting that the chiral ligand L1* promoted hydronickelation step occurs most likely in an irreversible manner (Fig. 4c). Moreover, a competition experiment between styrenes with different configurations indicates that hydroalkenylation of (E)-styrene is faster than that of (Z)-styrene due to the lower through-space ligand–substrate interaction energy68 (Fig. 4d). We also investigated the reactivities of various olefins, including styrenes, unactivated terminal alkenes, and unactivated internal alkenes. Relative rates from competition experiments indicate the order: styrene > unactivated terminal alkene > unactivated internal alkene (Fig. 4e).
a Monitoring migratory reaction progress: isomerization is faster than coupling. b NiD experiment: hydronickelation process of styrenes. c Crossover experiment: no intermolecular H/D scrambled crossover product. d Competition experiment: (E)-styrene is more reactive than (Z)-styrene. e Competition experiment for olefin reactivity: styrene > unactivated terminal alkene > unactivated internal alkene. f No desired product w/ Ni(0) catalyst: rule out the Ni0/NiII pathway. g Competition experiment with an electron-deficient CF3-substituted alkene as a radical acceptor. h Monofluoroalkene as substrate.
Another possible pathway for chemoselective C − F bond activation through oxidative addition is illustrated in Fig. 4f. In this pathway, the key oxidative addition of gem-difluoroalkenes occurs via a Ni(0) intermediate rather than alkylnickel(I) species. However, the use of a Ni(0) catalyst did not produce any desired product 3a, thereby ruling out this Ni(0)/Ni(II) pathway. In a competition experiment, the addition of electron-deficient CF3-substituted alkene (11) as a radical acceptor did not yield the gem-difluoroalkene product (12) through the potential radical trapping–β-F elimination pathway. This observation remained consistent even in the absence of gem-difluoroalkene 2a, indicating no involvment of a benzylic radical (Fig. 4g). Notably, not limited to gem-difluoroalkenes, (E)-monofluoroalkene (13) was also identified as a suitable coupling partner, albeit with a slight erosion in enantioselectivity (Fig. 4h). In this case, the oxidative addition of C − F bond could proceed chemoselectively without the assistance of an ester directing group, without observation of any isomerized product (Z)-14a.
In conclusion, we have developed a regio-, stereo-, and enantioselective NiH-catalyzed ipso- and migratory defluorinative olefin cross-coupling between readily available olefins and gem-difluoroalkenes under mild conditions. In this protocol, both styrenes and unactivated remote alkenes can be directly used in place of traditional alkylation reagents, providing general access to a broad array of densely functionalized monofluoroalkenes bearing a tertiary allylic stereogenic center. Furthermore, mechanistic studies suggest that the ipso-defluorinative olefin cross-coupling proceeds through an enantiodifferentiating syn-hydronickelation followed by an ester assisted chemoselective C(sp2)–F activation. By combining chain-walking ligand promoted alkene isomerization with chiral ligand promoted asymmetric hydroalkenylation, a more challenging migratory defluorinative olefin cross-coupling process was successfully realized, offering a complementary technique to enable formal C(sp3)–H/C(sp2)–F activation with high levels of regio-, stereo-, and enantiocontrol. We anticipate that this protocol will be of value to the medicinal chemistry community by accelerating access to complex monofluoroalkene-containing therapeutics.
Methods
General procedure (A) for NiH-catalyszed asymmetric defluorinative cross-coupling of styrenes with gem-difluoroalkenes
In a nitrogen-filled glove box, to an oven-dried 8 mL screw-cap vial equipped with a magnetic stir bar was added NiI2·xH2O (7.6 mg, 10 mol%), L1* (8.6 mg, 12 mol%), KI (33.6 mg, 0.20 mmol, 1.0 equiv), TBAF (1.0 M in THF, 12 μL, 6.0 mol%) and anhydrous DMA (0.20 M, 1.0 mL). The mixture was stirred for 5 min at room temperature, at which time styrene (1, 0.30 mmol, 1.5 equiv), gem-difluoroalkene (2, 0.20 mmol, 1.0 equiv), and DEMS (60 μL, 0.40 mmol, 2.0 equiv) were added to the resulting mixture in this order. The tube was sealed with a teflon-lined screw cap, removed from the glove box and the reaction was stirred at 25 °C for up to 10 h (the mixture was stirred at 800 rpm). After the reaction was complete, the reaction was quenched upon the addition of H2O, and the mixture was extracted with EtOAc. n-Dodecane (20 μL) was added as an internal standard for GC analysis. The product was purified by chromatography on silica gel (petroleum ether/EtOAc) for each substrate. The yields reported are the average of at least two experiments, unless otherwise indicated. The enantiomeric excesses (% ee) were determined by HPLC analysis using chiral stationary phases.
Data availability
The authors declare that the main data supporting the findings of this study, including experimental procedures and compound characterization, are available within the article and its supplementary information files, and also are available from the corresponding authors. The NMR data in Mnova format can be downloaded from figshare Dataset: https://doi.org/10.6084/m9.figshare.26820607.
References
Meanwell, N. A. Fluorine and fluorinated motifs in the design and application of bioisosteres for drug design. J. Med. Chem. 61, 5822–5880 (2018).
Kumari, S., Carmona, A. V., Tiwari, A. K. & Trippier, P. C. Amide bond bioisosteres: strategies, synthesis, and successes. J. Med. Chem. 63, 12290–12358 (2020).
Zhang, C. et al. Biological utility of fluorinated compounds: from materials design to molecular imaging, therapeutics and environmental remediation. Chem. Rev. 122, 167–208 (2022).
Drouin, M. & Paquin, J.-F. Recent progress in the racemic and enantioselective synthesis of monofluoroalkene-based dipeptide isosteres. Beilstein J. Org. Chem. 13, 2637–2658 (2017).
Morand, S., Jubault, P., Bouillon, J.-P. & Couve-Bonnaire, S. gem-Heteroatom-substituted fluoroalkenes as mimics of amide derivatives or phosphates: a comprehensive review. Chem. – Eur. J. 27, 17273–17292 (2021).
Fujita, T., Fuchibe, K. & Ichikawa, J. Transition-metal-mediated and -catalyzed C–F bond activation by fluorine elimination. Angew. Chem. Int. Ed. 58, 390–402 (2019).
Wang, J. et al. Recent advances in radical-based C–F bond activation of polyfluoroarenes and gem-difluoroalkenes. Chem. Commun. 57, 12203–12217 (2021).
Ai, H.-J., Ma, X., Song, Q. & Wu, X.-F. C–F Bond activation under transition-metal-free conditions. Sci. China: Chem. 64, 1630–1659 (2021).
Lu, M.-Z. et al. Recent advances in alkenyl sp2 C–H and C–F bond functionalizations: scope, mechanism, and applications. Chem. Rev. 122, 17479–17646 (2022).
Ge, D. et al. Transition-metal-catalyzed asymmetric defluorinative reactions. Org. Chem. Front. 10, 3909–3928 (2023).
Tian, P., Feng, C. & Loh, T.-P. Rhodium-catalysed C(sp2)–C(sp2) bond formation via C–H/C–F activation. Nat. Commun. 6, 7472–7478 (2015).
Dai, W., Shi, H., Zhao, X. & Cao, S. Sterically controlled Cu-catalyzed or transition-metal-free cross-coupling of gem-difluoroalkenes with tertiary, secondary, and primary alkyl Grignard reagents. Org. Lett. 18, 4284–4287 (2016).
Lu, X. et al. Nickel-catalyzed defluorinative reductive cross-coupling of gem-difluoroalkenes with unactivated secondary and tertiary alkyl halides. J. Am. Chem. Soc. 139, 12632–12637 (2017).
Sakaguchi, H. et al. Copper-catalyzed regioselective monodefluoroborylation of polyfluoroalkenes en route to diverse fluoroalkenes. J. Am. Chem. Soc. 139, 12855–12862 (2017).
Yang, L. et al. Synthesis of alkylated monofluoroalkenes via Fe-catalyzed defluorinative cross-coupling of donor alkenes with gem-difluoroalkenes. Org. Lett. 20, 1924–1927 (2018).
Ma, Q., Wang, Y. & Tsui, G. C. Stereoselective palladium-catalyzed C–F bond alkynylation of tetrasubstituted gem-difluoroalkenes. Angew. Chem. Int. Ed. 59, 11293–11297 (2020).
Wang, K. & Kong, W. Synthesis of fluorinated compounds by nickel-catalyzed defluorinative cross-coupling reactions. ACS Catal. 13, 12238–12268 (2023).
Xu, W.-Y., Xu, Z.-Y., Zhang, Z.-K., Gong, T.-J. & Fu, Y. Tunable synthesis of monofluoroalkenes and gem-difluoroalkenes via solvent-controlled rhodium-catalyzed arylation of 1-bromo-2,2-difluoroethylene. Angew. Chem. Int. Ed. 62, e202310125 (2023).
Zhu, Z., Xiao, J. & Shi, Z. Nickel-catalyzed stereo- and enantioselective cross-coupling of gem-difluoroalkenes with carbon electrophiles by C−F bond activation. Angew. Chem. Int. Ed. 62, e202113209 (2023).
Lin, H. et al. Enantioselective Cu-catalyzed nucleophilic addition of fluorinated reagents: C–C bond formation for the synthesis of chiral vicinal difluorides. Org. Lett. 24, 2197–2202 (2022).
Chen, Z., Huang, X., Liao, J. & Wang, M. Copper-catalyzed enantioselective fluoroalkenylation of cyclic imino esters. Org. Chem. Front. 10, 163–168 (2023).
Crossley, S. W. M., Obradors, C., Martinez, R. M. & Shenvi, R. A. Mn-, Fe-, and Co-catalyzed radical hydrofunctionalizations of olefins. Chem. Rev. 116, 8912–9000 (2016).
Liu, R. Y. & Buchwald, S. L. CuH-catalyzed olefin functionalization: from hydroamination to carbonyl addition. Acc. Chem. Res. 53, 1229–1243 (2020).
Chen, J., Guo, J. & Lu, Z. Recent advances in hydrometallation of alkenes and alkynes via the first row transition metal catalysis. Chin. J. Chem. 36, 1075–1109 (2018).
Wang, X.-X., Lu, X., Li, Y., Wang, J.-W. & Fu, Y. Recent advances in nickel-catalyzed reductive hydroalkylation and hydroarylation of electronically unbiased alkenes. Sci. China Chem. 63, 1586–1600 (2020).
He, Y., Chen, J., Jiang, X. & Zhu, S. Enantioselective NiH-catalyzed reductive hydrofunctionalization of alkenes. Chin. J. Chem. 40, 651–661 (2022).
Larionov, E., Li, H. & Mazet, C. Well-defined transition metal hydrides in catalytic isomerizations. Chem. Commun. 50, 9816–9826 (2014).
Vasseur, A., Bruffaerts, J. & Marek, I. Remote functionalization through alkene isomerization. Nat. Chem. 8, 209–219 (2016).
Sommer, H., Juliá-Hernández, F., Martin, R. & Marek, I. Walking metals for remote functionalization. ACS Cent. Sci. 4, 153–165 (2018).
Zhang, M., Ji, Y. & Zhang, C. Transition metal catalyzed enantioselective migratory functionalization reactions of alkenes through chain-walking. Chin. J. Chem. 40, 1608–1622 (2022).
Wang, Y., He, Y. & Zhu, S. NiH-catalyzed functionalization of remote and proximal olefins: new reactions and innovative strategies. Acc. Chem. Res. 55, 3519–3536 (2022).
Lu, X. et al. Practical carbon–carbon bond formation from olefins through nickel-catalyzed reductive olefin hydrocarbonation. Nat. Commun. 7, 11129 (2016).
He, Y., Cai, Y. & Zhu, S. Mild and regioselective benzylic C–H functionalization: Ni-catalyzed reductive arylation of remote and proximal olefins. J. Am. Chem. Soc. 139, 1061–1064 (2017).
Juliá-Hernández, F., Moragas, T., Cornell, J. & Martin, R. Remote carboxylation of halogenated aliphatic hydrocarbons with carbon dioxide. Nature 545, 84–88 (2017).
Chen, F. et al. Remote migratory cross-electrophile coupling and olefin hydroarylation reactions enabled by in situ generation of NiH. J. Am. Chem. Soc. 139, 13929–13935 (2017).
Xiao, J., He, Y., Ye, F. & Zhu, S. Remote sp3 C–H amination of alkenes with nitroarenes. Chem. 4, 1645–1657 (2018).
Zhang, Y., Xu, X. & Zhu, S. Nickel-catalysed selective migratory hydrothiolation of alkenes and alkynes with thiols. Nat. Commun. 10, 1752 (2019).
Wang, Z., Yin, H. & Fu, G. C. Catalytic enantioconvergent coupling of secondary and tertiary electrophiles with olefins. Nature 563, 379–383 (2018).
Zhou, F., Zhang, Y., Xu, X. & Zhu, S. NiH-catalyzed remote asymmetric hydroalkylation of alkenes with racemic α-bromo amides. Angew. Chem. Int. Ed. 58, 1754–1758 (2019).
He, S.-J. et al. Nickel-catalyzed enantioconvergent reductive hydroalkylation of olefins with α-heteroatom phosphorus or sulfur alkyl electrophiles. J. Am. Chem. Soc. 142, 214–221 (2020).
He, Y., Liu, C., Yu, L. & Zhu, S. Enantio- and regioselective NiH-catalyzed reductive hydroarylation of vinylarenes with aryl iodides. Angew. Chem. Int. Ed. 59, 21530–21534 (2020).
Bera, S., Mao, R. & Hu, X. Enantioselective C(sp3)–C(sp3) cross-coupling of non-activated alkyl electrophiles via nickel hydride catalysis. Nat. Chem. 13, 270–277 (2021).
Shi, L., Xing, L.-L., Hu, W.-B. & Shu, W. Regio- and enantioselective Ni-catalyzed formal hydroalkylation, hydrobenzylation, and hydropropargylation of acrylamides to α-tertiary amides. Angew. Chem. Int. Ed. 60, 1599–1604 (2021).
Meng, L., Yang, J., Duan, M., Wang, Y. & Zhu, S. Facile synthesis of chiral arylamines, alkylamines and amides by enantioselective NiH-catalyzed hydroamination. Angew. Chem. Int. Ed. 60, 23584–23589 (2021).
Cuesta-Galisteo, S., Schörgenhumer, J., Wei, X., Merino, E. & Nevado, C. Nickel-catalyzed asymmetric synthesis of α-arylbenzamides. Angew. Chem. Int. Ed. 60, 1605–1609 (2021).
Liu, J., Gong, H. & Zhu, S. Nickel-catalyzed, regio- and enantioselective benzylic alkenylation of olefins with alkenyl bromide. Angew. Chem. Int. Ed. 60, 4060–4064 (2021).
Cheng, Y., Gui, Z., Tao, R., Wang, Y. & Zhu, S. NiH-catalyzed asymmetric hydroalkynylation of α,β-unsaturated amides. Green Synth. Catal. 3, 377–379 (2022).
Zhao, L. et al. Ligand-controlled NiH-catalyzed regiodivergent chain-walking hydroalkylation of alkenes. Angew. Chem. Int. Ed. 61, e202204716 (2022).
Wang, J.-W. et al. Nickel-catalyzed switchable site-selective alkene hydroalkylation by temperature regulation. Angew. Chem. Int. Ed. 61, e202205537 (2022).
Yang, J.-S. et al. NiH-catalyzed regio- and enantioselective hydroalkylation for the synthesis of β- or γ-branched chiral aromatic N-heterocycles. J. Am. Chem. Soc. 145, 22122–22134 (2023).
Bera, S., Fan, C. & Hu, X. Enantio- and diastereoselective construction of vicinal C(sp3) centres via nickel-catalysed hydroalkylation of alkenes. Nat. Catal. 5, 1180–1187 (2022).
Chen, J., Wu, L., Zhao, Y. & Zhu, S. Enantio- and diastereoselective NiH-catalyzed hydroalkylation of enamides or enecarbamates with racemic α-bromoamides. Angew. Chem. Int. Ed. 62, e202311094 (2023).
Lee, C., Kang, H.-J., Seo, H. & Hong, S. Nickel-catalyzed regio- and enantioselective hydroamination of unactivated alkenes using carbonyl directing groups. J. Am. Chem. Soc. 144, 9091–9100 (2022).
Lyu, X. et al. Intramolecular hydroamidation of alkenes enabling asymmetric synthesis of β-lactams via transposed NiH catalysis. Nat. Catal. 6, 784–795 (2023).
Bai, D. et al. Highly regio- and enantioselective hydrosilylation of gem-difluoroalkenes by nickel catalysis. Angew. Chem. Int. Ed. 61, e202114918 (2022).
Chen, F., Xu, X., He, Y., Huang, G. & Zhu, S. NiH-catalyzed migratory defluorinative cross olefin-coupling: trifluoromethyl-substituted alkenes as acceptor olefins to form gem-difluoroalkenes. Angew. Chem. Int. Ed. 59, 5398–5402 (2020).
Zhou, L., Zhu, C., Bi, P. & Feng, C. Ni-catalyzed migratory fluoro-alkenylation of unactivated alkyl bromides with gem-difluoroalkenes. Chem. Sci. 10, 1144–1149 (2019).
Chen, M. S., Prabagaran, N., Labenz, N. A. & White, M. C. Serial ligand catalysis: a highly selective allylic C−H oxidation. J. Am. Chem. Soc. 127, 6970–6971 (2005).
Fors, B. P. & Buchwald, S. L. A multiligand based Pd catalyst for C−N cross-coupling reactions. J. Am. Chem. Soc. 132, 15914–15917 (2010).
Chen, C., Peters, J. C. & Fu, G. C. Photoinduced copper-catalysed asymmetric amidation via ligand cooperativity. Nature 596, 250–256 (2021).
Kim-Lee, S.-H., Mauleón, P., Arrayás, R. G. & Carretero, J. C. Dynamic multiligand catalysis: a polar to radical crossover strategy expands alkyne carboboration to unactivated secondary alkyl halides. Chem 7, 2212–2226 (2021).
Zhang, Y. et al. A relay catalysis strategy for enantioselective nickel-catalyzed migratory hydroarylation forming chiral α-aryl alkylboronates. Chem 7, 3171–3188 (2021).
He, Y. et al. Regio- and enantioselective remote hydroarylation using a ligand-relay strategy. Nat. Commun. 13, 2471 (2022).
Jiang, X., Sheng, F.-T., Zhang, Y. & Zhu, S. Ligand relay catalysis enables asymmetric migratory reductive acylation of olefins or alkyl halides. J. Am. Chem. Soc. 144, 21448–21456 (2022).
Zhou, J., He, Y., Liu, Z., Wang, Y. & Zhu, S. Ligand relay catalysis enables asymmetric migratory hydroarylation for the concise synthesis of chiral α-(hetero)aryl-substituted amines. Adv. Sci. 11, 2306447 (2024).
Sun, Y., Guo, J., Shen, X. & Lu, Z. Ligand relay catalysis for cobalt-catalyzed sequential hydrosilylation and hydrohydrazidation of terminal alkynes. Nat. Commun. 13, 650 (2022).
Sun, Y., Wang, B. & Lu, Z. Ligand relay catalysis: a newly emerged synthetic strategy. Org. Chem. Front. 10, 4146–4160 (2023).
Lu, G. et al. Ligand-substrate dispersion facilitates the copper-catalyzed hydroamination of unactivated olefins. J. Am. Chem. Soc. 139, 16548–16555 (2017).
Acknowledgements
Support was provided by National Key R&D Program of China (2022YFA1503200, S.Z.), NSFC (92156004 and 22271143, S.Z.; 22271146, Y.W.), NSF of Jiangsu Province (BK20230078, Y.W.), Fundamental Research Funds for the Central Universities (491916003301, S.Z.), Open Research Fund of School of Chemistry and Chemical Engineering, Henan Normal University (S.Z.), and Open Research Fund of Shanghai Key Laboratory for Molecular Engineering of Chiral Drugs (S.Z.). G.H. thanks the Innovation Training Program of Nanjing University (202410284432X).
Author information
Authors and Affiliations
Contributions
S.Z. and Y.W. designed and supervised the project. D.Z., Z.L. and G.H. performed and analyzed the experiments. All authors co-wrote the manuscript, analyzed the data, discussed the results, commented on the manuscript, and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zeng, D., Liu, Z., Huang, G. et al. Regio-, stereo-, and enantioselective ipso- and migratory defluorinative olefin cross-coupling to access highly functionalized monofluoroalkenes. Nat Commun 15, 7645 (2024). https://doi.org/10.1038/s41467-024-52054-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-024-52054-6