Abstract
Biocrude derived from biomass via hydrothermal liquefaction (HTL) is a sustainable substitute for petroleum to obtain energy and biochemicals. Upgrading biocrude inevitably faces the trade-off between consuming large amounts of hydrogen via hydrotreating and high yield of solid residue without additional hydrogen. In this work, we report a non-hydrogenated refinery paradigm for nearly complete valorization (~90%), via co-generating green coal and bio-lubricant. The obtained green coal has higher heating values comparable to commercial coals, with a lower fuel ratio and reduced ash content. Viscosity of upgraded vacuum distillate is comparable to that of a lubricant oil. A life cycle assessment confirms 28% reduction in greenhouse gas emission and 35% reduction in energy input of this paradigm compared with conventional hydrotreating biorefinery. This approach presents an environmentally friendly, safe and convenient paradigm for biocrude refining from huge biowaste towards carbon-neutral society.
Similar content being viewed by others
Introduction
As the world shifts towards lower carbon alternative energy sources, sustainable biomass is encouraged to substitute various fuels and chemicals derived from petroleum1,2. Potentially 5,760 Mt of oil equivalent per year of energy can be delivered from various biomass by 20603,4. Conversion of biomass to sustainable energy and chemicals is the key for achieving the petroleum substitution, where biomass serves as crucial raw materials for manufacturing renewable liquid fuels, materials and chemicals5. However, many types of wet biomass and biowaste, such as manure and sewage sludge, due to the presence of a high amount of water, are not yet to be properly utilized, causing environmental damage and significant greenhouse gas emission issues6.
Hydrothermal liquefaction (HTL) is a promising sustainable technology for producing renewable substitutes for fossil fuels and bio-based chemicals from biomass and biowaste7,8,9,10. It can efficiently convert biomass into biocrude11. However, biocrude exhibits undesirable characteristics, such as high oxygen (O) content and high nitrogen content and high viscosity, making it unsuitable for directly commercial use12.
Technologies are essential to fully realize the substitution potential of biocrude12,13. The most common technique for upgrading is hydrogenation, which effectively reduces heteroatom content and stabilizes the thermal reactive compounds, thereby enhancing the fuel quality8,14. Nevertheless, a significant amount of hydrogen is required due to the higher heteroatom contents (10-20 wt% oxygen (O), 2-7 wt% nitrogen (N) content) in biocrude13. Additionally, the use of catalysts and hydrogenation-supporting facilities involves technical complexity, safety considerations, regulatory compliance and high capital costs14.
Distillation is an effective technique for separating mixtures with different boiling points, and is critical for refining multiple biocrude-derived products. It matches well with hydrogenation for obtaining high quality fuels15. Existing research primarily focuses on biocrude distillates that can be utilized as drop-in fuels, heavily depending on feedstocks characteristics, especially lipid content. The yield of drop-in fuels from biocrude is below 30% without hydrogenation16,17. As a consequence, heavy components with boiling points above 350 °C, constituting approximately 40-60% of the total biocrude, remain largely underdeveloped without hydrogen treatment18. These heavy component, referred to solid residue and vacuum distillate, mainly consist mainly of long chain carbon, aromatic carbon and various oxygen/nitrogen-containing compounds19. Some of these fractions are thermally unstable and reactive under higher distillation temperature (>350 °C), posing control challenges. The polarity of these mixtures makes them suitable for directly conventional petroleum refinery method, unless refined together at a lower blending ratio20. Previous efforts have focused on diesel distillate cuts without added hydrogen, which depend heavily on feedstock properties16. Research on the heavy distillate applications is seldom reported in the open literature.
Therefore, the refinery of HTL biocrude heavily relies on hydrogenation to obtain high-quality fuels, constrained by operational and construction costs. Liquid fuel yields are limited by the biocrude characteristics without hydrogenation, resulting in significant amounts of heavy distillate during distillation. A big challenge still exists to impede the realization of the significant potential of HTL biocrude refinery. Hence, developing a general biocrude refinery strategy based on the characteristics of each distillate cut could significantly overcome hydrogenation limitations and address biocrude drawbacks.
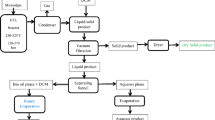
In this study, we developed a hydrogen-free biocrude refinery paradigm for multi-products co-generation. The utility of this paradigm focuses on the utilization of heavy distillates from biocrude (Fig. 1). HTL biocrude from low-lipid algae biomass Spirulina. sp. (lipid content = 11.19 wt%) was distillated into three distillates. The distillate residue (above 500 °C), referred to as green coal, demonstrated a comparable HHV to traditional coal with a lower fuel ratio and ash content. Vacuum distillate (350–500 °C) can be upgraded to bio-lubricant, exhibiting a comparable viscosity with axle oil, and improved tribological characteristics owing to higher saturation and lower polarity. Combined with HTL diesel distillate results obtained from our previous study20, a life cycle assessment (LCA) confirmed this paradigm could achieve a 28% reduction in greenhouse gas emission and a 35% reduction in energy input compared to a hydrotreating biorefinery. We believe these findings will advance the biocrude refinery to substitute petroleum products rather than petroleum energy.
The biocrude refinery process typically involves two key steps. The first step is the HTL process, which converts biomass and biowaste into biocrude. This step also produces valuable by-products that can be utilized as carbon materials, pesticides, or as the feedstock for anaerobic digestion. The second step traditionally involves distillation to produce diesel blendstocks or further refining to high-performance fuels using cost-intensive hydrogen. This work introduces a non-hydrogenated refinery paradigm that achieves nearly complete valorization (~90%) of biocrude. Instead of relying on hydrogenation, this approach co-generates green coal and bio-lubricants, offering a cost-effective and sustainable alternative to conventional refining methods. High-performance carbon materials, modified lubricants and fine chemicals might be further developed in the future.
Results
Physicochemical properties of distillates
The distillated products from Spirulina sp. were prepared via continuous HTL system and vacuum distillation facility (Supplementary Fig. 1 and Supplementary Fig. 2). The physicochemical properties of distillates in five temperature ranges are presented in Fig. 2a and Supplementary Table 3. In simulated distillation results, F3 comprised less than 10 wt% in atmospheric distillation but increased to 13.10 wt% under vacuum distillation. F5 had the highest yield at 61.90 wt%, while F4 was the highest liquid yield of 28.33 wt% in vacuum distillation. This result indicates that the 275–350 °C distillation range facilitated the decomposition of biocrude. The elemental distribution results show that the carbon (C) of distillated fractions gradually increased with increasing distillation temperature (Fig. 2b and Supplementary Fig. 4). F1 contained the lowest concentration of C, while F2-F5 exhibited progressively higher indicating that primary oxygen functional groups were concentrated in F1, making them suitable for modification into fine chemicals. N element was concentrated in F4 and F5, with the highest concentration of N in F5. Notably, the HHV of F1 to F4 increased from 24.33 to 38.48 MJ kg-1. Previous study inferred that the distillate for diesel blendstocks had similar HHV to diesel (approximate 45–47 MJ kg-1), which was depended on the feedstocks16.
a Distributions of simulated distillation and vacuum distillation results (n = 3 experimental repeats) and the corresponding pictures of distillated fractions. b Elements (n = 2 experimental repeats), HHVs distributions. c FTIR spectra. d Acidity (n = 3 experimental repeats). e Viscosity (n = 3 experimental repeats). f Density (n = 3 experimental repeats) of F1 to F4. g 1H NMR spectra of F4 and biocrude (% of the integral values for each range of ppm with respect to the total integral). h Molecular weight of distillates (the normalized response is from 0 to 1). i Chemical compounds distribution identified via GCMS. Source data are provided as a Source Data file.
The carbon number distribution of the biocrude (Supplementary Fig. 3), predominantly falls within the C18-C28, indicating potential suitability for lubricant and diesel21. FT-IR show F3 and F2 exhibited strong vibrational peaks at 3600–3000 cm−1 (Fig. 2c), while F1 and F2 showed stronger vibrational peaks at 1640-1720 cm−1, indicating compounds with more oxygen functional groups. F5 only exhibited vibrational peaks at 3600–3200 cm−1 and 1660–1580 cm−1, indicating the presence of nitrogen-containing functional groups and cyclic hydrocarbon.
The biocrude had an acid value of 17.73 mg KOH g-1 (Fig. 2d), in comparison to F1-F3 below 1 mg KOH g−1 and F4 with 8.45 mg KOH g-1, indicating that acidic compounds were concentrated in F4. Viscosity (Fig. 2e), which can reflect the carbon chain length and chain structure, significantly decreased after distillation due to the concentration of colloidal substances and heavy components in F522. F5 displayed a solid-like nature, mainly comprising long-chain carbon components. Unlike the fractions of F1 to F4, it is not amenable to the same testing procedures. A detailed analysis of F5 is provided in the section of “Distillated Residue for Green Coal”. F3 exhibited the highest density value at 0.96 g·cm-3, followed by F4 (0.92 g cm-3) (Fig. 2f), implying that F4 showed a lower molecular weight and denser packing between molecules. The low intensity of Har from Nuclear magnetic resonance (NMR) results suggest low contents of methoxy/ carbonyl and aromatic hydrocarbons (Fig. 2g and Supplementary Table 8)23.
The Gel permeation chromatography (GPC) results show that F1 and F3 had a significant proportion in the large molecular range (Fig. 2h and Supplementary Table 7), probably due to the long heating time of vacuum rectifying apparatus. During distillation, a small portion of large molecules would be polymerized and decomposed, resulting in a distribution of larger molecular weights of F3. In contrast, F4 exhibited a lower molecular weight distribution, indicating that F4 was decomposed and F3 was polymerized. F5 was untestable, causing the unbalanced results.
The primary hydrocarbon substances were concentrated in F3 and F4, accounting for more than 50% by Gas chromatography-mass spectrometry (GCMS) analysis (Fig. 2i and Supplementary Table 6). The main hydrocarbon substances in F3 were cyclic hydrocarbons and aromatic hydrocarbons, suggesting aromatic reactions during distillation. F4 mainly consisted of aliphatic hydrocarbon substances. Ketones were mainly distributed in F2, whereas F3 had a drastically low contents of ketone and ester compounds20.
In conclusion, vacuum distillation effectively prevents decomposition reactions, increases liquid fuels yield, and avoid secondary reactions during distillation process. Considering the high yields, the specific characteristics, F5 was selected to demonstrate its combustion characteristics as green coal, and F4 was selected to be upgraded for bio-lubricant.
Distillated residue for green coal
Green coal was produced by combining HTL and distillation, with HTL temperatures at 260 °C, 280 °C, 300 °C, and 320 °C for atmospheric distillation, and at 300 °C with vacuum distillation (VD300 °C). The characterization of green coal is depicted in Fig. 3. Notably, VD300 °C exhibited the highest green coal yield (61.90 wt%) (Fig. 3a). Yields of F5 increased from 260 °C to 280 °C but decreased from 280 °C to 320 °C. This trend can be attributed to two primary factors: First, higher HTL temperatures (å 300 °C) are considered to promote biomass depolymerization during HTL24; Second, pyrolysis reactions and secondary reactions occurred during the high distillation temperature (above 500 °C), leading to the cracking of biocrude20. The decreased yield of F4, accompanied by the increased yields of F1, F2 and F3 further support this trend. Previous studies found that F2 and F3 are suitable for blending with commercial fuel16,20. The green coal obtained at 260 °C has the highest HHV (31.72 MJ kg-1) (Fig. 3b). Increasing the HTL temperature reduced HHV slightly to 30.32 MJ kg-1. VD300 °C resulted in a slightly higher HHV than 300 °C, while lower than 320 °C.
a Green coal yields (n = 3 experimental repeats). b Element (n = 2 experimental repeats) and HHV analysis. c TG and DTG (inset figure) results. d Proximate analysis (n = 3 experimental repeats) (The fuel ratio is defined as the ratio of fixed carbon to volatile matter). e Comparison of green coal with other catalysts reported in literature: Energy density comparisons of several energy carriers and green coal (n = 5 for green coal, n = 7 for traditional coal, n = 12 for pyrolysis char, n = 7 for pyrolysis bio-coal, n = 11 for torrefaction bio-coal, n = 12 for hydrochar. data reported in literature). f Fuel ratio and ash content of green coal and other solid energy carriers. Red dots represented green coals in this study, dark blue dots represented the hydrothermal carbonization (HTC) hydrochar, light green dot represented the compressed bio-coals, yellow dots represented TO (torrefaction) bio-coals, light blue dots represented PY (pyrolysis) bio-coals, grey dots represented traditional coals. Data in e and f correspond to the data in Supplementary Table 4 and Supplementary Table 5. Green coal was prepared via integrating hydrothermal liquefaction (HTL) and distillation. The reaction conditions included HTL at various temperatures for atmospheric distillation: 260°C, 280°C, 300 °C, and 320°C; as well as HTL at 300 °C combined with vacuum distillation (VD300 °C). Source data are provided as a Source Data file.
The Thermal gravimetric analysis (TG) curves (Fig. 3c) indicate a minor mass loss between 160 °C to 400 °C. The ignition point of each sample was around 350 °C, higher than lignite (310 °C)25. Thermally decomposition occurred when the TG temperature exceeded 350 °C26. Green coal produced at 260 °C and 320 °C had comparable burnout temperatures, which were higher than those obtained at 280 °C and 300 °C. Notably, VD320 °C shows comparable burnout point temperature with 300 °C. The residue primarily consisted of fixed carbon with minimal ash content After the oxidation process.
The ash content of green coals was remained below 1.5 wt% (Fig. 3d). The majority of ash in the feedstock was retained in solid residue after HTL, resulting in low ash contents in both biocrude and its distillates27. Conversely, volatile contents ranged from 89 wt% to 95 wt%, with the increase of HTL temperature, peaking at 95.4% at 300 °C. The highest fixed carbon content was obtained at 320 °C. The fuel ratio, defined as the ratio of fixed carbon to volatile matter, is an indicator of coalification. The fuel ratio ranged from 0.05 to 0.1, via atmospheric distillation, whereas it rose to 0.16 via vacuum distillation.
Green coal exhibited comparable HHVs with solid energy carriers, including traditional coal and bio-coals produced through pyrolysis (PY), torrefaction (TO), and hydrothermal carbonization (HTC) (Fig. 3e and Supplementary Table 4)28,29,30,31. HHVs of bio-coals largely depend on feedstock characteristics and thermal processes, for instance, PY, TO and HTC. These so-called bio-coals exhibited lower HHVs due to their higher ash and O content29. The green coal obtained in this study had a similar energy content to pyrolysis bio-coke, demonstrating its potential for providing a high-quality sustainable coal in energy applications.
Green coal achieved comparable fuel ratio and ash content to typical solid fuels (Fig. 3f and Supplementary Table 5)19,29,30. In contrast, PY bio-coal, containing a high ash content, was hence similar to lignite coal. TO bio-coal showed a lower ash content, owing to the low ash content of the precursor feedstock. Green coal exhibited the lowest ash content and fuel ratio. A PY bio-coal with a HHV of 25 MJ kg-1 was reported via pyrolysis and distillation from lignocellulosic biomass, which had a higher oxygen content than green coal19. Hence, green coal showed comparable properties to high-grade commercial coals such as HHV, and further exhibited superior performance in combustion owing to its lower fuel ratio, O content and ash content. Cost-effective methods can be developed to reduce heteroatoms (N and O), e.g., by choosing low-N feedstocks for co-HTL and improving the HTL process using catalysts. In addition, the green coal might be distillated or upgraded by catalytic cracking into light fraction, suitable for Sustainable Aviation Fuel (SAF) production owing to its high volatile content.
Formation mechanism of green coal
Scanning Electron Microscopy (SEM) images are presented in Fig. 4b. The green coal, produced at an HTL temperature of 260 °C, displays a compact bulk morphology. In contrast, samples prepared at 280 °C, 300 °C and 320 °C exhibit a smooth, skirt-like morphology with burst bubble surfaces, indicative of boiling during the biocrude formation process (red circles). All the samples exhibited smooth surface (yellow rectangles). The boiled bubbling surface gradually settled when stopping distillation, due to uneven inner pressure, resulting in skirt-like morphology after cooling. Conversely, the VD300 °C exhibited a smooth bulk structure similar to the green coal at 260 °C, indicating a lack of pore structure and a lower specific surface area (Supplementary Table 9).
a Proposed formation mechanism of green coal. b SEM image of green coal preparing at various HTL temperatures and distillation conditions (red circles represent the form appearances, yellow rectangles represent the surface morphologies). XPS spectra of (c) C1s, (d) O1s and (e) N1s. f FTIR spectra of green coal prepared in different HTL temperatures. Source data are provided as a Source Data file.
The C1s XPS spectra (Fig. 4c) reveal distinct peaks corresponding to C-C, C-O-C and O-C = O bonds. The consistently strong C-C peak reflects the high concentration of hydrocarbons. Conversely, the intensities of C-O-C and O-C = O peaks increased with rising HTL temperature. A similar trend is observed in the O1s (Fig. 4d) and N1s (Fig. 4e) spectra with peaks corresponding to pyridinic-N and Graphitic-N32.
FTIR spectra (Fig. 4f) indicate that the intensities of C = O, Aro C-C, Aro C = C and Aro C-H IR peak increase with HTL temperature. Intriguingly, the VD300 °C sample shows similar XPS peaks to those of the green coal produced at 280 °C, but significantly lower IR peak intensities for the -CH3, -CH, Aro C = C and O-H functional groups. These findings reveal the existing of oxidation during the distillation at higher temperature.
A formation mechanism for green coal is proposed based on these characterization results (Fig. 4a). Macromolecular substances (F5) decompose into micromolecular substances at HTL temperatures above 280 °C, increasing the fixed carbon content while reducing the green coal yield. Conversely, HTL temperatures below 280 °C promote the formation of lower molecular weight substances (F4), further reducing the green coal yield. Higher distillation temperatures accelerate the decomposition of macromolecular substances into micro-molecules, thereby increasing the volatile content in green coal33. Moreover, elevated distillation temperatures enhance surface oxidation reactions, while reduced distillation temperatures minimize the intensity of thermochemical reactions, leading to higher fixed carbon content.
Physicochemical characteristics of bio-lubricant
We chose F4 as the feedstock for bio-lubricant due to its carbon chain length, content of hydrocarbons and viscosity. F4 was upgraded to produce bio-lubricant via two methods: (1) esterification with ethanol, isopropanol and n-butyl butanol, producing esterified products denoted as AL, IS and NB, respectively; (2) epoxidation combined with ethanol-based esterification (EE). The characteristics of the upgraded F4 are presented in Fig. 5 and Supplementary Table 10. Esterification with AL resulted in a significant decrease in acid value (4.19 mg KOH g-1), compared with those of IS and NB (above 10 mg KOH g-1) (Fig. 5a). The catalytic activity of Amberlite 732 ion-exchange resin was tested. The acid value of blank is 10.23 mg KOH g-1, similar to the value of F4. Notably, EE has a similar acid value (4.72 mg KOH g-1) to that of the AL-esterified product, suggesting that ethanol is an effective biocrude esterification agent.
a Acidity (n = 3 experimental repeats). b Elemental composition and HHV analysis (n = 2 experimental repeats). c Chemical compounds distribution by GCMS. d Density (n = 3 experimental repeats) of F4 and EE. e NMR results of 1H (% of the integral values for each range of ppm with respect to the total integral). F4 was esterified with ethanol, isopropanol and n-butyl butanol, producing esterified products denoted as AL, IS and NB, respectively; F4 epoxidated combining with ethanol-based esterification denoted as EE. Source data are provided as a Source Data file.
The elemental distribution of upgraded F4 is illustrated in Fig. 5b and Supplementary Fig. 5. An increase in the carbon chain length of the esterification additives slightly decreased the C content of the product. The elevated O content in EE indicated the introduction of oxygen-containing functional groups due to increased unsaturation function of F4. GCMS results (Fig. 5c and Supplementary Table 6) and FTIR spectrum (Supplementary Fig. 6) reveal that hydrocarbon content in AL decreased after esterification, indicating the epoxidation process effectively introduced oxygen-containing functional groups, providing more structures sites for esterification. The nitrogen-containing compound was highest in AL (7.29%), lowest in EE (6.23%). The content of alcohol group significantly decreased in all esterified products, and no phenolic compounds were detected. The alcohol content in all samples decreased after upgrading, except AL. Additionally, the acid and ester contents both decreased, Notably, the esters and acids increased significantly in EE, suggesting that side reactions during esterification may reduce its efficiency34. The density of products follows a decreasing trend (Fig. 5d), indicating the reactions extended the carbon chain length while introducing substantial amounts of oxygen-containing groups. NMR results (Fig. 5e and Supplementary Table 8) showed the contents of C = C (Hβ and Hα) decreased, C-H-O increased, demonstrating the incorporation of oxygen-containing groups35.
In summary, esterification and epoxidation effectively modified the physicochemical characteristics of F4. Esterification increased oxygen-containing functional groups, while epoxidation enhanced the saturation and ester content. Short-chain alcohols proved more effective for product modification, as they control acid value, extended molecular chain length, and improve viscosity. Nevertheless, the complex composition of the products, even after distillation, poses a challenge for broader applications.
Tribological properties of bio-lubricant
Friction coefficient (COF) is a crucial parameter for lubricant application, driven by the adhesion forces between contacting surfaces atoms (Fig. 6b). Typically, lubricants COF value range from 0.02 to 0.14 for various automotive applications36. The COF of F4 ranged from 0.12 to 0.16. Among the upgraded samples, the AL exhibited lower COF than IS, EE showed the lowest COF. The average COF values (Fig. 6c) highlight significant improvements after upgrading, with COF increasing alongside the carbon chain length of esterification agents. Corrected wear scar diameter reflects the extent of wear on metal surfaces under specific conditions (Fig. 6d and Supplementary Fig. 7). IS exhibited the largest wear scar diameter (0.93 mm). EE (0.77 mm) performed slightly better than NB. The values of volume loss showed similar trend with corrected wear scar. Viscosity results (Fig. 6e) demonstrate that the viscosity of the esterified products continuously increases as the carbon chain length of the esterification agent rises. At 80 °C, viscosity stabilized at 25 mm2 s-1 except F4 and AL. At 40 °C, EE showed a slightly higher viscosity than AL, indicating that interactions between saturated groups and ester groups contributed its viscosity34. The viscosity index indicates the viscosity-temperature characteristics. EE obtained the highest viscosity index of 92 after upgrading, owing to the increase length of carbon chain and epoxidation. The esters and other active molecular compounds would cause lower viscosity index than vegetable oil derived lubricant37. EE showed comparable viscosity and viscosity index (å 90) with axle oil (summer use (70 ~ 80 mm2 s-1 at 40 °C), meeting the required range of ISO VG 68 oil38.
a The tribological mechanism of upgraded distillate. b Friction coefficients (COF) waves. c Average COF (Error bars represent standard deviations of repeated tests). d Corrected wear scar of upgraded distillates (n = 3 experimental repeats). e Viscosities (n = 3 experimental repeats). f SEM and 3D imagines of wear surface. DBE distribution of (g) F4 of ESI-, (h) EE of ESI-, (i) F4 of ESI+ and (j) EE of ESI + . Source data are provided as a Source Data file.
3D and SEM images of the friction scars (Fig. 6f) reveal plow marks on the wear scar surfaces of AL, IS and EE. That of IS and F4 shows uneven wear scars. AL, NB and EE exhibited relatively uniform scratched surfaces, EE and AL showed pit-like features similar to the center of a circle. One possible reason is that the adsorption capacity of ester compounds to the metal surface increased after esterification39. The results of the friction volume loss corresponded to the wear scar diameter results. The worn surface of F4 exhibited relatively smooth edges, while AL and EE displayed noticeable signs of oxidation. Iron metal came into contact with oxygen, resulting in the formation of black iron oxide. This black oxide formed a thin film that covered the metal surface, causing it to appear black40. Energy dispersive spectrometer (EDS) results (Supplementary Fig. 9) identified O element as a key element affecting the friction surface. Interestingly, EE, which showed a low volume loss in friction, had a decrease in N vanish. Fourier transform ion cyclotron resonance mass spectrometry (FTICR MS) was employed for analysis the complex heavy compounds of F4 and EE (Fig. 6g–j). The results reveal compounds were mainly composed of acidic compounds. The Double Bond Equivalents (DBE) value reflects the number of cyclic functional groups and double-bonded carbon atoms. Higher DBE values indicate higher degrees of unsaturation41. Upgrading reduced the DBE, particularly in O2 and N2 groups of EE, increasing saturation levels. In the Ox series compounds of ESI- of EE, a noticeable decrease in the predominant DBE values (especially N2 series compounds, N1O4 and N1O5) was observed, while higher distribution DBE values (Ox, N1O1) exhibited a slightly increase, suggesting the alteration of unsaturated compounds in carbon structure. Overall, the upgrading can increase the saturated compounds. The major compounds of the C20-C24 class contain predominantly branch-chain grafting nitrogen-containing functional groups. The variations in nitrogen-containing compounds may be attributed to process involving deacylation, dehydration and amidation during upgrading42. ESI+ of F4 and EE, which characterizes alkaline compounds, displays distinct DBE values corresponding to various functional groups. The alkaline compounds in F4 exhibited a relatively low content, primarily dominated by N2. The mass spectra highlight four predominant compounds, namely C18H35N2O1, C19H23N2O1, C19H39N2 and C20H27N2 (Supplementary Fig. 8), with dominant N2 group. EE experienced a significant increase in the content of alkaline compounds. Notably, the DBE value of 2 within the N1O1 family showed a conspicuous increase. The alkaline additives in lubricating oil serve as both detergents and stabilizers43. Therefore, the stability and cleanliness of F4 can be improved after upgrading.
The proposed tribological mechanism (Fig. 6a) indicates that F4, primarily consists of compounds with N and O within the range of C22-C24, along with acidic functionality, was oxidized occurs during the friction process. The polarity and unsaturation of F4 result in the retention of N and O on the friction metal surface. C19H23N2O compounds provide a function of endogenous nitrogen additives. The acid value decreased and ester functional groups increased after esterification, further affecting the friction performance. Epoxidation and esterification significantly increased the saturation and alkaline groups content of F4. Therefore, the lubricating oil film of EE exhibited reduced oxidation reactivity, resulting in the formation of an inert film during the friction process. The nitrogen-containing compounds in EE in a certain concentration could have positive effects by physical adsorption on the friction surface, thus minimizing damage to the metal surface.
Life-cycle assessment of the biocrude refinery paradigm
The sustainability assessment for the biocrude refinery paradigm is presented in Fig. 7 and Supplementary Table 11. Three biocrude refineries route was evaluated. Route 1 yields bio-lubricant, diesel blendstocks and green coal; Route 2 produces only diesel blendstocks and green coal; Route 3 refines the biocrude into diesel and light naphtha via hydrotreating (Fig. 7a). Route 3 consumes the most energy and H2, resulting the highest Greenhouse gas (GHG) emission (-1.08 kgCO2eq MJ-1). Route 2 shows the lowest GHG emission (-1.69 kgCO2eq MJ-1) among these three routes, relying solely on distillation for upgrading (Fig. 7b). Route 1, which includes bio-lubricant production, has intermediate GHG emissions (-1.38 kg CO2eq MJ-1), approximately 27.78% lower than Route 3. All routes show high energy efficient (exceeding 75%) (Fig. 7c), Route 1 and Route 2 show energy efficient of 79.83% and 80.01%, respectively, about 5% higher than Route 3. Additionally, Route 1 reduced energy input by 35.12% compared to Route 3. Sensitivity Analysis in Supplementary Fig. 14 and Supplementary Table 17. reveals that all three routes exhibit sensitivity to the five indicator types: Global warming potential, Terrestrial ecotoxicity potential, Human carcinogenic toxicity, Human non-carcinogenic toxicity and Fossil resource scarcity. Therefore, a comparison was made of the contributions of these three routes to the aforementioned five indicators. Route 2 performs best across these indicators, followed by Route 1, with Route 3 being the least favorable. Furthermore, wastewater and flue gas treatment during hydrogenation need extra treatment facilities. The bio-lubricant preparation can be performed at mild conditions, which is thus a safe, environmental-friendly and facile process. In brief, Route 1 and 2 show the portability and safety of the operation with the environmental benefits. Further improvement of energy efficient is expectable since some of facilities and energy calculating methods in Route 1 are based on the experimental scale. Additionally, hydrogen cannot fully eliminate heavy compounds during hydrocracking, limiting utilization rates of Route 3.
a The system boundary of three biocrude refinery routes for the LCA. b Global warming potential (GWP). c Energy efficiency of three biocrude refinery routes. Three biocrude refinery routes were evaluated. Route 1 yields bio-lubricant, diesel blendstocks and green coal; Route 2 produces only diesel blendstocks and green coal; Route 3 refines the biocrude into diesel and light naphtha via hydrotreating. Source data are provided as a Source Data file.
In 2020, the domestic supply of biomass was 575 EJ globally by Global Bioenergy Statistics Report44. One could replace 58.5% of the total lubricating oil consumed and reduce coal consumption by 11%, given a full utilization of biomass via the proposed paradigm in this study (Route 1) (Supplementary Tables 12–16 and Supplementary Note 4). Considering the growing share of electric energy in transportation and the potential for diverse products, producing renewable products rather than only biofuel from HTL biocrude offers a promising and foreseeable economic benefits way in future.
Discussion
The biocrude refinery paradigm without using hydrogen has demonstrated to be feasible by valorizing multiple distillation cuts. In many places, organic waste such as waste plastics, manure and straw need to be disposed and utilized simultaneously. Hence, co-HTL can be a promising approach to improving the economics of the overall process, increasing the yield and quality of the biocrude products, owing to the synergistic interactions between various components of different feedstocks45. The characteristics of feedstocks, energy input and the yields of products are the main factors for the biocrude biorefinery. Therefore, there is a trade-off between HTL operating production conditions and refinery processes.
The use of commercial coals is often associated with serious environmental concerns due to their relatively high ash contents and the release of pollutants and greenhouse gas. In contrast, green coal possesses carbon-neutral characteristics. A commercial coal typically contains a lower volatile content, suggesting that the green coal is more combustible, implying that it will have better combustion performance than commercial coals31. Some nitrogen-rich feedstock such as microalgae, livestock manure and sewage sludge, will lead to the presence of N-containing heterocyclic compounds in biocrude products after HTL46. If the nitrogen content is too high, it can be adjusted through co-liquefaction with low-N feedstock such as woody biomass or by denitrification. Further investigations are required regarding the impacts of N-containing heterocyclic compounds on the performance of bio-lubricants. The diesel blendstock (275-350 °C) has a suitable acid value, HHV, wear scar diameter and calculated cetane index (CCI), so it can be used as diesel blends at the ratio up to 10% (Supplementary table 1, Supplementary Figs. 11–13).
A LCA confirms 28% reduction in greenhouse gas emission and 35% reduction in energy input of this paradigm compared with hydrotreating biorefinery. However, water management is indeed a great challenge for HTL technology since it handles wet biomass. HTL water contains nutrients and complex organics12. Several strategies have been reported including recovering nutrients47,48 or energy49 via algae and anaerobic digestion, extracting value-added chemicals50, treating it as wastewater51, recycling it to HTL52 and valorizing it for pathogen control in human53 or agriculture54. By valorizing products and by-products of the HTL process, including biocrude, hydrochar and HTL-aqueous phase, the economic feasibility of the proposed non-hydrogenated biocrude refinery paradigm can be greatly enhanced8. The purpose of proposing this concept is to maximize the utilization of biocrude.
Microalgae is just the starting point as typical biomass to inspire further exploration. This work supports further investigations on using biocrude made from different biomass, since biocrude varies depending on biomass types and operational conditions. This paradigm is more suitable for the application scenarios such as: (1) Large-scale livestock and poultry farms; (2) On-board self-powered ocean cleanup operations55; (3) Economic development of islands with limited land; (4) For special areas that require immediate treatment and harmless disposal of domestic waste and feces and the need for off-grid or emergency energy supply, such as military camps or national park. In addition, SAF has attracted increasing attentions worldwide to achieve the carbon neutrality goals. HTL of Biomass generates biocrude that can substitute for petroleum to produce SAF56. Co-HTL can promote the yield of distillated fractions for fuels as discussed preciously, making this paradigm promising for SAF production.
Overall, our work overcomes the dilemma of nonhydrogen biocrude refinery and giving a broader perspective in the productization of HTL biocrude cuts. The heavy distillates (with a boiling point above 500 °C) are effectively used as green coal after refinery, having a comparable HHV (31.72 ± 0.92 MJ kg-1) to that of conventional coal, and a superior fuel ratio (0.07-0.16) and lower ash content (below 1.5 wt%). Lower HTL temperature and distillation temperature can increase the fixed carbon content in green coal. A bio-lubricant product can be produced from the vacuum distillate (350-500 °C) after upgrading/modification (epoxidation + esterification). The viscosity of bio-lubricant product meets the axle oil standard and has superior tribological properties owing to its composition (more saturation and less acid function groups). LCAs confirm that this paradigm can reduce the energy consumption and GHG emission, suggesting the development of petroleum substitute via the use of wet biowaste to production of fungible fuels and chemicals in a convenient and sustainable way.
Methods
Chemicals and Materials
The feedstock of Spirulina sp. powder was purchased from Fuqing King Dnarmsa Spirulina Co., LTD, (Fuqing, China) (Supplementary Fig. 10). We chose microalgae as feedstocks because it contains three main components (carbohydrates, proteins and lipids), with fast growth rate and high photosynthetic efficiency, which is highly compatible with batch and continuous HTL. Moreover, Spirulina sp., a type of microalgae, has been extensively studied and achieved industrialization in active substance extraction. On the other hand, microalgae are highlighted as energy feedstocks, and considered one of Ten CO2 utilization and removal pathways57. Detailed physical properties of the feedstock are shown in Supplementary Table 2. Ethanol (≥99.7%), isopropanol (≥99.7%) and butanol (≥99.5%) were purchased from Sigma-Aldrich and used for biocrude distillate modification. Acetic acid (≥99.5%) and hydrogen peroxide (30%) were purchased from Sinopharm Chemical Reagent Co., Ltd and used for epoxidation. All remaining reagents were utilized in their received condition, except where noted differently. Amberlite 732 was purchased from Shanghai Macklin Biochemical Co., Ltd as catalysts.
Vacuum Distillation of Biocrude
Biocrude was obtained via a continuous HTL system as showing in Supplementary Fig. 158. The distillation curves were measured after fractional distillation of the biocrude: Fraction 1 (F1) (initial boiling point (IBP)-175 °C), Fraction 2 (F2) (175-275 °C), Fraction 3 (F3) (275-350 °C), Fraction 4 (F4) (350-500 °C) and Fraction 5 (F5) (above 500 °C)32. For each distillation test, 200 g biocrude was loaded into a 500 mL three-necked flask. atmospheric distillation was used to obtain F1, F2 and F3 below 350 °C, while vacuum distillation was used to obtain F4 and F5 in a pressure of -0.09 kPa and a distillation temperature of 350-500 °C. To avoid quick distillations that could cause ineffective separation and safety issues, the heating rate was set at about 1 °C min−1. To reduce the heat loss, glass wool was wrapped around the distillation equipment. The concept of vacuum distillation unit is shown in Supplementary Fig. 2. The reactor was cooled down to room temperature after reactions. Gaseous products were collected in gas bags. Mixed products from continuous HTL system were separated into biocrude, aqueous phase and solid residue through gravity settling. Biocrude and solid residue were dried in a vacuum drying chamber20. Each experiment was conducted at least three times. Simulated distillation (ASTM D 7169-2011) was applied to determine the simulated true boiling point and final boiling point distribution of crude oil and petroleum refining fractions by the GC analysis. The analysis of acidity, viscosity and density was tested only for F1 to F4. A detailed analysis of F5 is provided in the section of “Distillated Residue for Green Coal”. The yields of biocrude and distillated fractions were presented in Supplementary Table 3. The distillation fractions yields were calculated using the following formulas:
where MFn represents the mass of the distillated fraction.
Preparation of Green Coal
F5 (above 500 °C) was defined as green coal. For further study the discrepancy of HTL temperatures and distillation conditions, experiments were performed in a 1.8 L stainless Parr reactor (Model 4578, Parr Instrument Company, Moline, Illinois, USA). Each experiment was conducted with 200 g dry Spirulina sp. powder and 800 g deionized water at 260 °C, 280 °C, 300 °C and 320 °C, respectively. Atmospheric distillation cut biocrude strictly according to temperature cuts20. Each experiment was conducted at least three times. Green coal yield was calculated by the following formulas:
where MGreen coal and Mbiomass are the mass of biocrude and the mass of feedstock.
Preparation of Bio-lubricant
Esterification
F4 was chosen as the precursor for preparing the bio-lubricant by esterification. Ethanol, isopropanol and butanol were used to upgrade F4 (vacuum distillate). In a typical process, a certain amount of vacuum fraction of biocrude was added into a three-necked flask, and 10 g alcohol additive and 4 g F4 were loaded into the flask. At the same time, 0.12 g Amberlite 732 ion-exchange resin was added as the catalyst for esterification. The reaction was carried out at 65 °C for 2 h. At the end of the treatment, the solid catalyst was first recovered through filtration along with alcohol washing. The washing solution was then transferred to a three-necked flask for vacuum distillation and the resulting oil was subject to drying in a vacuum drying box at 70 °C for 12 h, obtaining the bio-lubricant59. The catalytic activity of Amberlite 732 ion-exchange resin was examined also in a blank test with ethanol only. Each experiment was conducted at least three times.
Epoxidation + Esterification
4 g F4, 4.4 mL H2O2, 0.72 mL acetic acid, 0.06 g Amberlite 732 ion-exchange resin (catalyst) were added into the three-necked flask for ring-opening epoxidation. The epoxidation operation was carried out at 60 °C for 8 h36,39. After the reaction, the catalyst was separated via filtration, the filtered mixture was distillated at 100 °C to remove the excess acetic acid. The epoxide vacuum fraction completed was put in to the flask with 10 g ethanol and 4 g esterified F4. At the same time, 0.12 g Amberlite 732 ion-exchange resin was added as the catalyst for esterification. The reaction was carried out at 65 °C for 2 h. Catalyst was then collected via filtration, and cleaned using alcohol. The cleaning solution was transferred to a three-necked flask, which was subjected to vacuum distillation, and then to a vacuum drying oven. The bio-lubricant was obtained after drying at 70 °C for 12 h. The catalytic activity of Amberlite 732 ion-exchange resin was examined also in a blank test with only ethanol. Each experiment was conducted at least three times.
Analysis Methods
The distribution of boiling points for distribution of biocrudes with residues were analyzed by high temperature GC according to ASTM D 7169-2011.
The distribution of boiling points for distribution of biocrudes with residues were analyzed by Simulated distillation, for determining the simulated true boiling point and final boiling point distribution of biocrude and refining fractions via the high temperature GC analysis. The American Society for Testing and Materials (ASTM) D 7169-2011 was applied for the wide boiling range from initial boiling point (IBP) to 720 °C and corresponded with the elution of n-C100.
The number-averaged molecular weight (Mn), weight-averaged molecular weight (Mw) and polydispersity index (PDI = Mw/Mn) were determined using a Waters Breeze GPC-UV instrument coupled with a Waters Styragel HR1 column (Shimadzu, Tokyo, Japan).
Elemental components (i.e., carbon, hydrogen and nitrogen) of Spirulina sp. and biocrude were determined using an elemental analyzer (Vario MICRO Cube, Element analysis system, Germany). Oxygen content was calculated by difference. The HHV of biocrude was calculated according to the Dulong formula60.
where, C, H and O are weight percentages of carbon, hydrogen and oxygen in biocrude, respectively.
The XPS was performed in an ESCALAB 250Xi instrument. The samples were irradiated with Al Kα X-rays (1486.7 eV), and the photoelectrons were analyzed using an HSA type analyzer. The C–C component of C1s (285.0 eV) was used for the calibration.
The organic compounds in biocrude were identified using GCMS (QP2010, Shimadzu Co., Tokyo, Japan) equipped with a Varian DB-5 column (30 m × 0.25 mm × 0.25 μm), Helium was used as the carrier gas at a flow rate of 1 mL min-1 at the pressure of 100 kPa. The injection temperature and interface temperature were set at 250 °C and 320 °C. The ion source was adjusted to 230 °C. The mass spectrometer was operated in positive electron impact mode (EI) at 70 eV. Scan range of mass spectrum was in m z-1 of 30-800. All chromatogram peaks in spectra were compared with electron impact mass spectrum from NIST Database (NIST11). Samples were directly diluted with acetone and filtered through a 0.45 μm filter. The column temperature was set at 60 °C for 2 min, then ramped up at a rate of 5 °C min-1 to 120 °C and maintained for 1 min, afterwards increased to 200 °C at the same heating rate and maintained for 1 min. Finally, the temperature was ramped up at a rate of 10 °C min-1 to 280 °C and maintained for 1 min20.
The NMR spectra of the samples were detected using deuterated methanol as a solvent on the Bruker Advance 700 M instrument. To simplify the relative complexity, the 1H NMR spectrum of the samples was divided into six ppm ranges. Each range was integrated and the integral values are reported in Supplementary Table 8.
The proximate analysis of green coal, the ash content was measured using the gravimetric method prescribed in ASTM D 3174-04, the content of volatile matter was determined using an on-isothermal thermogravimetric (TG) method, and the content of fixed carbon was calculated by the difference. In the TG method, 3.0 to 8.0 mg of samples were heated in a TG analyzer (NETZSCH STA 449 F3 Jupiter, Netzsch Co., Germany) under an atmosphere of 25 ml min−1 of N2. The temperature was programmed from room temperature to 110 °C at a rate of 10 °C min−1 and held for 10 min before ramping to 1200 °C at a rate of 20 °C min−1. Apart from the contents of volatile matter, the thermal characteristics of green coal were also obtained by the TG method.
The acid values were measured according to GBT 4945 -2002.
The density was measured according to ASTM D-4052 at 25 °C.
The viscosity of biocrude was measured at 40 °C, 80 °C and 100 °C using the NDJ-8S rotational viscometer (Shanghai Changji Geological Instrument Co., Ltd., Shanghai, China). Viscosity index was calculated according to GB 1995-80.
The organic functional groups in biocrude were determined via FT-IR (PerkinElmer Spectrum 100, Waltham, USA) in the spectral range of 4000-450 cm−1.
The Fourier transform-ion cyclotron resonance mass spectrometry (FT-ICR MS) analysis was carried out using a 9.4 T Bruker Apex ultra FT-ICR mass spectrometer. The electrospray ionization (ESI) was operated in positive-ion mode (emitter voltage 5.0 kV; capillary column front end voltage 4.5 kV; capillary column end voltage 320 V). A 10 mg portion of oil sample was first mixed with 1 mL of toluene. Then, 20 mL of the solution mixture was diluted with 1 mL of a toluene/methanol (1:1) solution. ICR was operated at 15 db attenuation, 150 − 800 Da mass range and 4 M acquired data size, time-domain data sets were coded from 128 data acquisitions. Regarding the aqueous analysis, 1 mL of the sample was diluted with redistilled methanol to a total volume of 10 mL and then filtered with a fiber filter membrane. A 10 mL portion of water-soluble organic matter was washed with redistilled methanol and then diluted 10 times with methanol. The extracts were then processed for ESI FT-ICR MS analysis41.
Friction-wear test experiments were performed using a Bruker UMT series multi-functional friction and wear testing machines (Bruker UMT-3, Germany). A liquid sample of approximately 1-5 mg was placed in a steel plate, and a GCr15 steel ball of 10 mm was chosen for the reciprocating motion test. The procedure involved a reciprocating motion in a 50-300 N load, a friction time of 70 min, a friction stroke of 1 mm and a friction rate of 50 Hz at room temperature. The values of coefficient friction, friction curve and volume loss were obtained during the friction-wear testing. An accompanying optical profilometer was used to obtain 3D images data and volume loss data.
Following the tribological experiments, the wear scars on the steel balls and the wear tracks on the respective flat surfaces were documented using a laser profilometer (VK-X100, Keyence, Japan) and scanning electron microscope (SEM, Quanta 200 F, FEI Thermo Scientific Apreo 2 C, USA), respectively. Energy Dispersive Spectroscopy (EDS, OXFORD ULTIM Max65, UK) was used in the elemental analysis of surface of bearing specimens.
Life cycle assessment (LCA)
The study was conducted using LCA based on the conceptual framework of ISO 14040 and guidelines of ISO 14044. A simple LCA was presented here. The functional unit is 1 kg biocrude. Deterministic LCA model of the HTL pathway based on the SOT model design is available by a previous study61. ReCiPe2016 (H) was applied in the life cycle impact assessment (Supplementary Table 11) where six environmental impact categories were assessed (Supplementary Table 17). GHG emissions is represented in grams of CO2-equivalent (CO2eq). This LCA model focuses on the wet waste HTL pathway. The feedstock to the process is treated as an unburdened waste and therefore its associated GHG emissions are assumed to be zero. The supply chain GHG emissions reduction is calculated by comparing with the supply chain GHG emissions of petroleum diesel (91 g CO2eq MJ-1)61.
Statistics & reproducibility
The experiments in this study were repeated three times independently with similar results unless otherwise noted.
Data availability
All relevant data that support the findings of this study are presented in the manuscript and supplementary materials. Source data are provided with this paper.
References
Aragon, N. Z. U. et al. Sustainable land use and viability of biojet fuels. Nat. Sustain. 6, 158 (2023).
Fan, Z. Y. & Friedmann, S. J. Low-carbon production of iron and steel: Technology options, economic assessment, and policy. Joule 5, 829–862 (2021).
Lee, K., Jing, Y., Wang, Y. & Yan, N. A unified view on catalytic conversion of biomass and waste plastics. Nat. Rev. Chem. 6, 635–652 (2022).
Staples, M. D., Malina, R. & Barrett, S. R. H. The limits of bioenergy for mitigating global life-cycle greenhouse gas emissions from fossil fuels. Nat. Energy 2, 16202 (2017).
Geissler, C. H. & Maravelias, C. T. Analysis of alternative bioenergy with carbon capture strategies: present and future. Energ. Environ. Sci. 15, 2679–2689 (2022).
Feng, J. A., Li, Y. L., Strathmann, T. J. & Guest, J. S. Characterizing the Opportunity Space for Sustainable Hydrothermal Valorization of Wet Organic Wastes. Environ. Sci. Technol. 58, 2528–2541 (2024).
Savage, P. E. Chemistry. Algae under pressure and in hot water. Science 338, 1039–1040 (2012).
Zhang, B., Biswal, B. K., Zhang, J. J. & Balasubramanian, R. Hydrothermal Treatment of Biomass Feedstocks for Sustainable Production of Chemicals, Fuels, and Materials: Progress and Perspectives. Chem. Rev. 123, 7193–7294 (2023).
Siskin, M. & Katritzky, A. R. Reactivity of organic compounds in superheated water: General background. Chem. Rev. 101, 825–835 (2001).
Tian, C. Y., Li, B. M., Liu, Z. D., Zhang, Y. H. & Lu, H. F. Hydrothermal liquefaction for algal biorefinery: A critical review. Renew. Sust. Energ. Rev. 38, 933–950 (2014).
Xu, C. Roles of Bioenergy and Green Hydrogen in Large Scale Energy Storage for Carbon Neutrality. Engineering 29, 32–34 (2023).
Watson, J. et al. Valorization of hydrothermal liquefaction aqueous phase: pathways towards commercial viability. Prog. Energ. Combust. Sci. 77, 100819 (2020).
Shahbeik, H. et al. Biomass to biofuels using hydrothermal liquefaction: A comprehensive review. Renew. Sust. Energ. Rev. 189, 113976 (2024).
Stone, M. L. et al. Continuous hydrodeoxygenation of lignin to jet-range aromatic hydrocarbons. Joule 6, 2324–2337 (2022).
Taghipour, A., Ramirez, J. A., Brown, R. J. & Rainey, T. J. A review of fractional distillation to improve hydrothermal liquefaction biocrude characteristics; future outlook and prospects. Renew. Sust. Energ. Rev. 115, 109355 (2019).
Chen, W.-T. et al. Renewable diesel blendstocks produced by hydrothermal liquefaction of wet biowaste. Nat. Sustain. 1, 702–710 (2018).
Bouzouita, D. et al. Co-processing bio-liquids with vacuum gasoil through hydrocracking. Appl. Catal. B-Environ. 304, 120911 (2022).
Eboibi, B. E. O., Lewis, D. M., Ashman, P. J. & Chinnasamy, S. Hydrothermal liquefaction of microalgae for biocrude production: Improving the biocrude properties with vacuum distillation. Bioresour. Technol. 174, 212–221 (2014).
Cheng, B. H. et al. Bio-coal: A renewable and massively producible fuel from lignocellulosic biomass. Sci. Adv. 6, eaay748 (2020).
Cao, M. J. et al. Renewable biocrude blends boost tribological properties and maintain fuel performance of diesel. Chem. Eng. J. 474, 145696 (2023).
Chan, C. H. et al. Tribological behavior of biolubricant base stocks and additives. Renew. Sust. Energ. Rev. 93, 145–157 (2018).
Magomedov, R., Pripakhaylo, A. & Maryutina, T. Fractionation of a vacuum residue with a mixture of CO-toluene as a method for characterizing heavy petroleum feedstocks. Sep. Sci. Technol. 56, 2626–2633 (2021).
Biswas, B., Fernandes, A. C., Kumar, J., Muraleedharan, U. D. & Bhaskar, T. Valorization of Value addition using hydrothermal liquefaction. Fuel 222, 394–401 (2018).
Basar, I. A., Liu, H., Carrere, H., Trably, E. & Eskicioglu, C. A review on key design and operational parameters to optimize and develop hydrothermal liquefaction of biomass for biorefinery applications. Green. Chem. 23, 1404–1446 (2021).
Wang, K., Han, T., Deng, J. & Zhang, Y. N. Comparison of combustion characteristics and kinetics of Jurassic and Carboniferous-Permian coals in China. Energy 254, 124315 (2022).
Zhang, Y. N. et al. Analysis of oxidation pathways for characteristic groups in coal spontaneous combustion. Energy 254, 124211 (2022).
Shah, A. A., Toor, S. S., Conti, F., Nielsen, A. H. & Rosendahl, L. A. Hydrothermal liquefaction of high ash containing sewage sludge at sub and supercritical conditions. Biomass-. Bioenerg. 135, 105504 (2020).
Shih, C. F., Zhang, T., Li, J. H. & Bai, C. L. Powering the Future with Liquid Sunshine. Joule 2, 1925–1949 (2018).
Ahmad, K. A., Ahmad, E., Al Mesfer, M. K. & Nigam, K. D. P. Bio-coal and bio-coke production from agro residues. Chem. Eng. J. 473, 145340 (2023).
Agar, D. & Wihersaari, M. Bio-coal, torrefied lignocellulosic resources - Key properties for its use in co-firing with fossil coal - Their status. Biomass-. Bioenerg. 44, 107–111 (2012).
Kurose, R., Ikeda, M., Makino, H., Kimoto, M. & Miyazaki, T. Pulverized coal combustion characteristics of high-fuel-ratio coals. Fuel 83, 1777–1785 (2004).
Taghipour, A., Hornung, U., Ramirez, J. A., Brown, R. J. & Rainey, T. J. Fractional distillation of algae based hydrothermal liquefaction biocrude for co-processing: changes in the properties, storage stability, and miscibility with diesel. Energ. Convers. Manag. 236, 114005 (2021).
Hu, X. & Gholizadeh, M. Progress of the applications of bio-oil. Renew. Sust. Energ. Rev. 134, 110124 (2020).
Cheng, Y. Y. et al. Upgrading of Bio-oil by Catalytic Esterification. Chem. J. Chin. U. 39, 2746–2751 (2018).
Xu, D. H. & Savage, P. E. Characterization of biocrudes recovered with and without solvent after hydrothermal liquefaction of algae. Algal Res 6, 1–7 (2014).
Joshi, J. R., Bhanderi, K. K., Karve, M., Patel, J. V. Chemical modification of waste cooking oil for the bio lubricant production through epoxidation process. Biomass Convers. Bior. (2023).
Hamnas, A. & Unnikrishnan, G. Bio-lubricants from vegetable oils: Characterization, modifications, applications and challenges - Review. Renew. Sust. Energ. Rev. 182, 113413 (2023).
SH/T 0139-1995 Alxe oil. China Petroleum and Chemical Industry Federation (1995).
Panchal, T. M., Patel, A., Chauhan, D. D., Thomas, M. & Patel, J. V. A methodological review on bio-lubricants from vegetable oil based resources. Renew. Sust. Energ. Rev. 70, 65–70 (2017).
Edla, S., Thampi, A. D., Prasannakumar, P. & Rani, S. Evaluation of physicochemical, tribological and oxidative stability properties of chemically modified rice bran and karanja oils as viable lubricant base stocks for industrial applications. Tribol. Int. 173, 107631 (2022).
Lu, J. W., Li, H. G., Zhang, Y. H. & Liu, Z. D. Nitrogen Migration and Transformation during Hydrothermal Liquefaction of Livestock Manures. Acs Sustain. Chem. Eng. 6, 13570–13578 (2018).
Cheng, F. et al. Hydrothermal liquefaction of high- and low-lipid algae: Bio-crude oil chemistry. Appl. Energ. 206, 278–292 (2017).
McNutt, J. & He, Q. Development of biolubricants from vegetable oils via chemical modification. J. Ind. Eng. Chem. 36, 1–12 (2016).
Association, W. B. Global Bioenergy Statistics Report. (ed Association WB). 10 edn (2022).
Wu, X. Y. et al. Co-liquefaction of microalgae and polypropylene in sub-/super-critical water. Rsc Adv. 7, 13768–13776 (2017).
Yin, S. Y., Shao, Y. Y., Bao, T. Y. & Zhu, J. S. Review on Nitrogen Transformation during Microalgae Thermochemical Liquefaction: Recent Advances and Future Perspectives. Energ. Fuel. 37, 1525–1544 (2023).
Zhou, Y., Schideman, L., Yu, G. & Zhang, Y. H. A synergistic combination of algal wastewater treatment and hydrothermal biofuel production maximized by nutrient and carbon recycling. Energ. Environ. Sci. 6, 3765–3779 (2013).
Leng, L., Li, J., Wen, Z. & Zhou, W. Use of microalgae to recycle nutrients in aqueous phase derived from hydrothermal liquefaction process. Bioresour. Technol. 256, 529–542 (2018).
Si, B. C. et al. Anaerobic conversion of the hydrothermal liquefaction aqueous phase: fate of organics and intensification with granule activated carbon/ozone pretreatment. Green. Chem. 21, 1305–1318 (2019).
Lyu, H. et al. Two-stage nanofiltration process for high-value chemical production from hydrolysates of lignocellulosic biomass through hydrothermal liquefaction. Sep. Purif. Technol. 147, 276–283 (2015).
Shen, R. X. et al. Microbial electrolysis cell to treat hydrothermal liquefied wastewater from cornstalk and recover hydrogen: Degradation of organic compounds and characterization of microbial community. Int J. Hydrog. Energ. 41, 4132–4142 (2016).
Hu, Y. L., Feng, S. H., Yuan, Z. S., Xu, C. B. & Bassi, A. Investigation of aqueous phase recycling for improving bio-crude oil yield in hydrothermal liquefaction of algae. Bioresour. Technol. 239, 151–159 (2017).
Yu, F. B. et al. Biosafety of human environments can be supported by effective use of renewable biomass. P. Natl Acad. Sci. Usa. 119, 3 (2022).
Xu, Y. D. et al. Understand the antibacterial behavior and mechanism of hydrothermal wastewater. Water Res 226, 119318 (2022).
Belden, E. R. et al. Thermodynamic feasibility of shipboard conversion of marine plastics to blue diesel for self-powered ocean cleanup. P. Natl Acad. Sci. Usa. 118, 46 (2021).
Alherbawi, M., McKay, G. & Al-Ansari, T. Development of a hybrid biorefinery for jet biofuel production. Energ. Convers. Manag. 276, 116569 (2023).
Hepburn, C. et al. The technological and economic prospects for CO2 utilization and removal. Nature 575, 87–97 (2019).
Li, H. et al. Establishment and performance of a plug-flow continuous hydrothermal reactor for biocrude oil production. Fuel 280, 118605 (2020).
Wang, J. J., Chang, J. & Fan, J. A. Upgrading of Bio-oil by Catalytic Esterification and Determination of Acid Number for Evaluating Esterification Degree. Energ. Fuels 24, 3251–3255 (2010).
Rathsack, P., Wollmerstaedt, H., Kuchling, T. & Kureti, S. Analysis of hydrogenation products of biocrude obtained from hydrothermally liquefied algal biomass by comprehensive gas chromatography mass spectrometry (GC×GC-MS). Fuel 248, 178–188 (2019).
Jiang, Y. et al. Uncertainty analysis for techno-economic and life-cycle assessment of wet waste hydrothermal liquefaction with centralized upgrading to produce fuel blendstocks. J. Environ. Chem. Eng. 11, 109706 (2023).
Acknowledgements
This work was supported by National Natural Science Foundation of China (52261145701, U21A20162), the National Key Research and Development Program of China (2016YFD0501402) and the 2115 Talent Development Program of China Agricultural University, China. We acknowledge Dr. Zhang Jiaren (Sinopec research institute of petroleum processing CO.LTD., China) for kind suggestions on biocrude distillation and valorization of products. We thank Jia Jixiu (Institute of environment and sustainable development in agriculture, CAAS, China) for the assistance on the LCA datasets.
Author information
Authors and Affiliations
Contributions
Z.L. and M.C. designed the research. M.C. performed the experiments of biocrude refinery, the data extraction and analysis, wrote the draft of the manuscript in close consultation with L.A.R., Y.Z., C.C.X., Y.W. and J.L. R.E., C.Y. and D.K., participated in the manuscript writing. Z.L. initiated and supervised the project and paper writing. All authors contributed to the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cao, M., E, R., Yuan, C. et al. Green coal and lubricant via hydrogen-free hydrothermal liquefaction of biomass. Nat Commun 16, 722 (2025). https://doi.org/10.1038/s41467-025-56037-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-56037-z
This article is cited by
-
Evaluation of the Products from Hydrothermal Liquefaction of Palm Empty Fruit Bunch
BioEnergy Research (2025)